Abstract
Background and Objectives:
Patients’ movements often need to be restrained to obtain high-quality diagnostic images. Chloral hydrate is the primary agent for pediatric sedation prior to diagnostic studies in some countries. However, because of the agent’s long half-life, the need for augmentation must be minimized by ensuring the success of the initial dose. This study aimed to identify factors influencing sedation success rate and sedation duration.
Methods:
Pediatric patients’ age, sex, and dose according to body weight, type of clinic, duration of stay, and weight-for-age percentile were analyzed in univariate and multivariate regression analyses.
Results:
Of the total of 1590 patients, 1325 (82.7%) were successfully sedated. Predictors of successful sedation were a minimum dosage of 60 mg/kg and a younger age. Early sedation (mean − 1 standard deviation, <18 min) occurred in 10.9% of the pediatric patients who were successfully sedated. For this value, visits to emergency center B (adjusted odds ratio, 2.673; 95% confidence interval, 1.660–4.305) and visits during daytime hours appeared to be influencing factors.
Conclusion:
For safe and successful pediatric sedation, the child’s age and appropriate dose must be thoroughly reviewed, and a quiet and secluded environment must be ensured. Furthermore, taking into account that the patient’s age and developmental stage can affect induction time, care should be taken to avoid unnecessary augmentation
Introduction
Patients’ movements often need to be restrained to obtain high-quality diagnostic images. Particularly in young patients, anxiety caused by confined spaces, noise, and unfamiliar surroundings routinely requires sedation. 1 Ketamine, midazolam, and propofol are common procedural sedation drugs for interventions and diagnostic tests. 2 However, many emergency physicians prefer to use chloral hydrate as a pediatric sedation drug in South Korea because there is a reduced chance of an adverse event.3,4
The recommended dosage for chloral hydrate is 50–100 mg/kg, although it varies greatly depending on the literature consulted.1,5,6 If the initial dose fails to achieve adequate sedation, augmentation is necessary. Unfortunately, repeat doses increase the risk of adverse effects, as the long half-life of chloral hydrate can lead to its accumulation in the body,7,8 and high doses also increase the risk of adverse effects. 6 It can also increase the required patient monitoring time and the length of the hospital stay. Therefore, care must be taken to increase the success rate of the initial dose and minimize the need for augmentation by ensuring that an appropriate dosage is used initially.
This study aimed to identify factors influencing the induction time of chloral hydrate, an agent used widely in the emergency room (ER), to increase the sedation success rate and minimize the risk of adverse effects by helping to ensure an optimal initial dose.
Methods
The subjects of this study were children below the age of 12 years requiring sedation for diagnostic radiologic examination, for assessments of traumatic brain injuries or surgical abdomen, and patients sedated for painful procedures, such as sutures or fracture/dislocation reductions, were excluded. Patients with agitation/behavioral emergencies were not ruled out in principle. However, it is difficult to ensure oral intake of medicine without the cooperation of the patient; as such, sedation was carried out using other processes. Therefore, ultimately, behavioral emergency patients were not included.
A retrospective review was conducted of medical records of 1590 children who visited two emergency departments for a radiological study, from January 2013 to December 2014. A and B emergency medical centers are visited by approximately 80,000 patients and 50,000 patients each year, respectively. B emergency medical center has a separated space for pediatric sedation, whereas A emergency medical center is not equipped. Upon receiving the caregivers’ consent, sedation was induced in the pediatric patients with constant monitoring of oxygen saturation and pulse rate. Flumazenil was prepared as reversal agent. 9
The success of sedation was determined when a patient was sedated below the V level (response to verbal stimulus) on the AVPU scale after the first dose of chloral hydrate had been administered and the diagnostic tests had been completed. And we defined the failure of sedation as the inability to proceed the test or obtain the appropriate images by patients’ movement after the administration of initial dose of chloral hydrate. Sedation induction time was defined as the time taken until the physician decided to move patients to the testing rooms, as the patients were sedated below the V level (AVPU scale) after chloral hydrate was applied. The primary outcome of our study was the success of the initial dose of sedation.
For a descriptive analysis, the patients’ age, sex, emergency clinic, body weight, weight-for-age percentile, time of visit, duration of stay, chloral hydrate dose per body weight, sedation success status, sedation induction time, type of diagnostic test, test purpose, and adverse effects were recorded.
Subjects were categorized into two groups, namely, the successful group and the failure group. We compared the two groups with respect to hospital, sex, age, chloral hydrate dose per weight, visiting time, and weight-for-age percentile. To assess the weight-for-age percentile, the Korean pediatric standard growth chart (2007) was referenced. 10 In terms of chloral hydrate dosage, seven groups were created, at 10 mg/kg intervals. Three groups were created for the weight-for-age percentiles, which were <25th percentile, from the 25th percentile to <75th percentile, and ≥75th percentile. Two groups were created according to the time of visit: the daytime (6:00 a.m. to 6:00 p.m.) and nighttime (6:00 p.m. to 6:00 a.m.) groups.
Those who were adequately sedated within 1 standard deviation (SD) were defined as the early sedation group. Factors influencing sedation induction time were analyzed. Age, sex, hospital, visiting time, dose of chloral hydrate, and weight-for-age percentiles were entered into a backward stepwise logistic regression model.
Statistical analyses were conducted using SPSS version 22.0 (IBM, Armonk, NY, USA). Categorical variables are expressed as n (%) and continuous variables are expressed as the median and interquartile range (IQR). Categorical variables were analyzed using the χ2 or Fisher’s exact test. Continuous variables were analyzed with an independent t test or Mann–Whitney U test. Predictors of sedation success, early sedation, and delayed sedation were analyzed by multivariate logistic regression.
Results
Demographic characteristics
The total number of patients administered chloral hydrate according to the centers’ sedation protocols during the study period was 4050. Of these, 1708 patients were administered chloral hydrate for diagnostic testing. A total of 1590 patients were enrolled in this study after excluding those whose body weight and sedation status were unknown because of inadequate medical records.
The 1590 patients comprised 966 boys and 624 girls. Among the patients, 1337 visited emergency medical center A and 253 visited emergency medical center B. The median age was 23 months, and the median weight was 12 kg. The chloral hydrate dosage was 85.5 mg/kg. The weight-for-age percentiles of the patients administered chloral hydrate followed a regular distribution. In terms of visit time, 736 patients (46.3%) visited during the daytime (6:00 a.m. to 6:00 p.m.) and 854 patients (53.7%) visited during the nighttime (6:00 p.m. to 6:00 a.m. the following day). The average length of stay was 126 min.
Among the patients, 1587 (99.8%) were sedated for a computed tomography (CT) scan, and only 3 patients were sedated for magnetic resonance imaging (MRI) or an X-ray (0.2%). In all, 27 tests were performed to diagnose an internal disease, and 1541 (96.9%) were performed to obtain images from patients who sustained a traumatic injury.
In total, 104 subjects reported vomiting/spitting (6.5%), which was the most common adverse effect, with 31 patients showing hyperactivity (1.9%). There were no cases of cardiorespiratory depression (Table 1).
General characteristics of patients administered chloral hydrate.
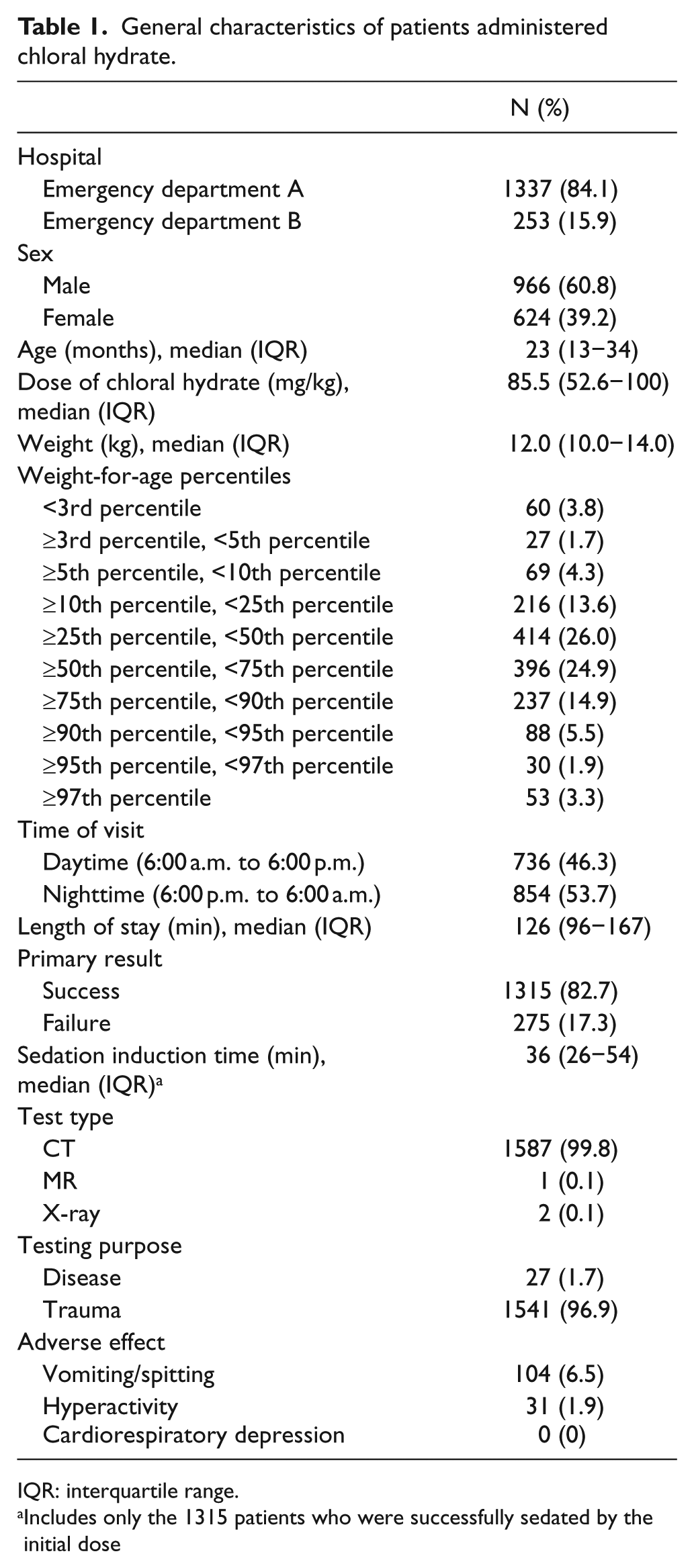
IQR: interquartile range.
Includes only the 1315 patients who were successfully sedated by the initial dose
Of the 1590 patients, 1315 patients (82.7%) were adequately sedated by the initial dose of chloral hydrate. Of the 275 patients who failed to be sedated adequately by the initial dose, 171 patients were administered the complete dose of the agent, but showed no effects, and 104 patients required an additional dose because of vomiting or spitting out the drug. Of the patients who were not sedated by the initial dose, 25 were discharged for monitoring, before a further decision regarding testing was made. A total of 250 patients were considered to require a second dose, 230 of whom (92.0%) were re-administered oral dose of chloral hydrate. The remaining 20 patients were administered a second dose via a different method or were administered a different agent altogether. Of the patients administered a second chloral hydrate dose, 217 (94.3%) were adequately sedated for testing, and 13 patients remained inadequately sedated (Figure 1).
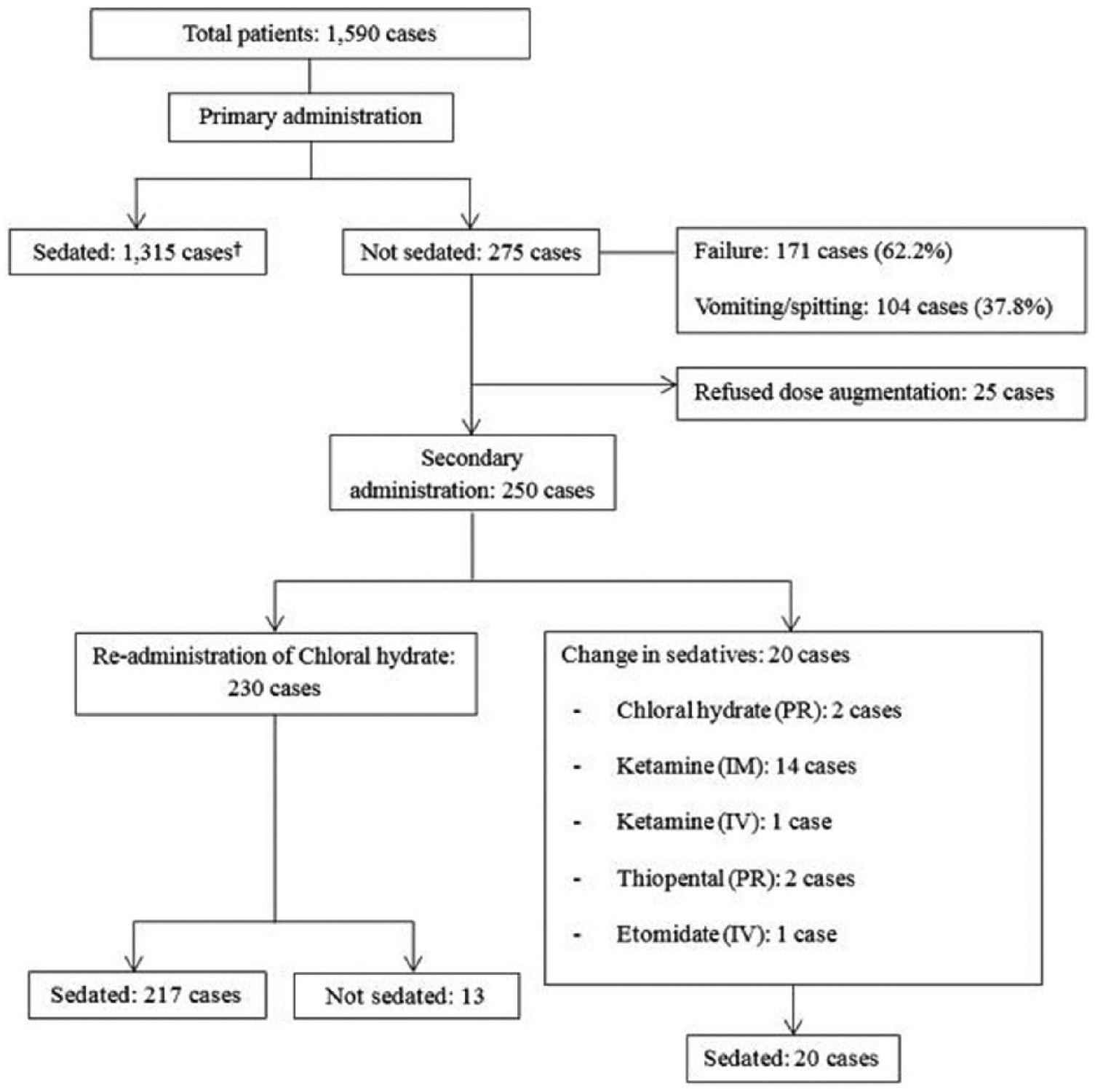
Chloral hydrate–induced sedation results.
Predictors of sedation success
Patients who were adequately sedated by the first dose, and those who were not, were compared in the analysis. The median age and body weight in the success group were 22 months and 12 kg, respectively, indicating that this group of patients was younger and lighter than the failure group (p = 0.017 and p = 0.018, respectively). The dose per body weight was 79.7 ± 21.8 mg/kg in the success group, which varied significantly from that of the failure group at 68.9 ± 22.6 mg/kg. A univariate analysis did not find a difference in terms of sex, visiting time, or body weight percentile, and the success rate of the B emergency center was found to be 2.32 times higher than that of the other. However, after adjusting for the confounding factors of age, dosage, sex, hospital, visiting time, and weight-for-age percentile, multivariate logistic regression analysis showed that independent predictors of successful sedation were a minimum dosage of 60 mg/kg and a younger age (Table 2).
Univariate and multivariate analyses of successful sedation of pediatric patients.
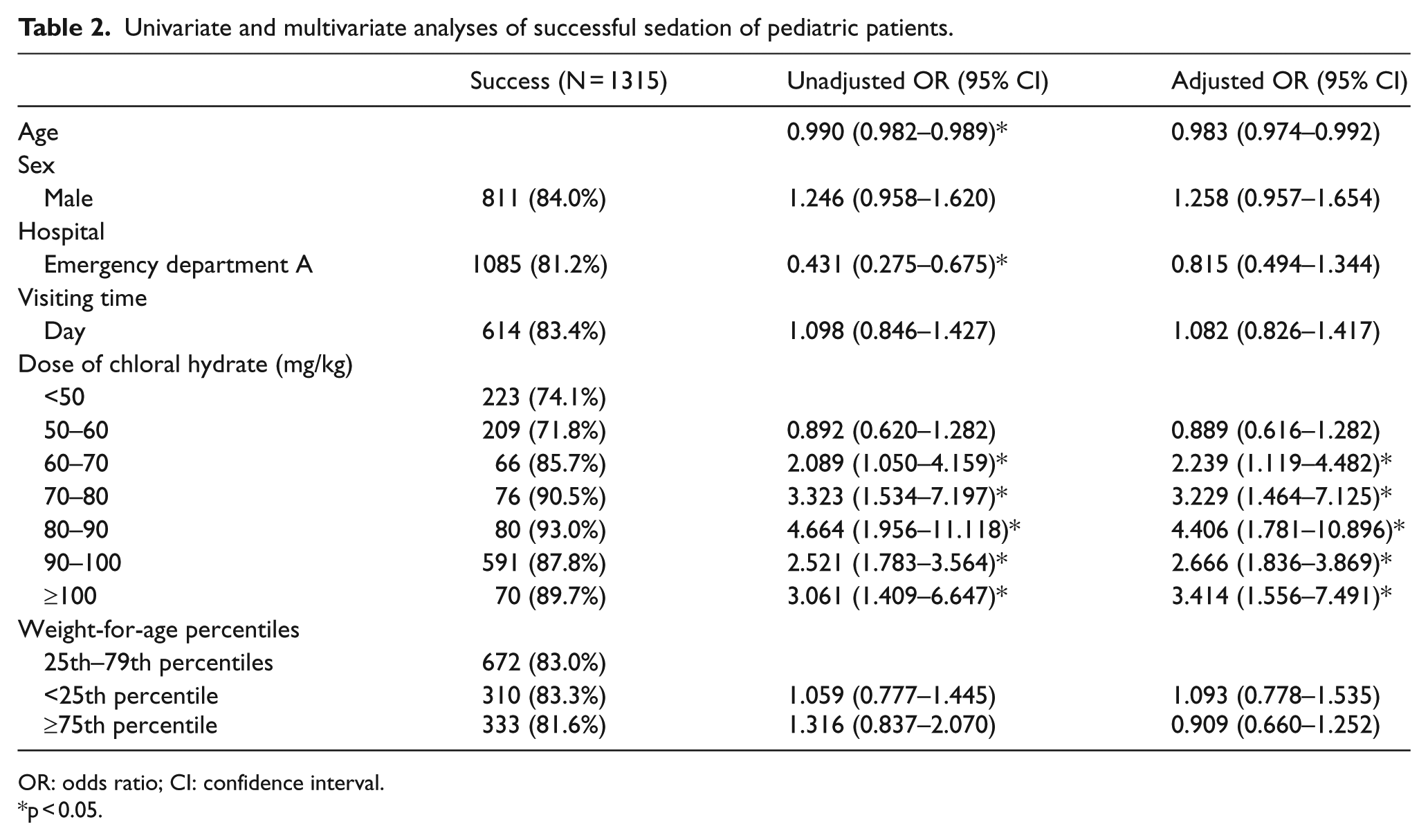
OR: odds ratio; CI: confidence interval.
p < 0.05.
Predictors of early sedation
A total of 143 pediatric patients were sedated early, which accounted for 10.9% of the total number of pediatric patients who were adequately sedated by the initial dose. The univariate analysis found that the early sedation rate was higher among younger patients, 1.67 times higher among those who visited emergency centers during the daytime hours and 2.35 times higher among those who visited the B emergency center. After controlling for age, dosage, sex, hospital, visiting time, and weight-for-age percentile, the B emergency center (adjusted odds ratio (aOR), 2.673; 95% confidence interval (CI), 1.661–4.310), a daytime visit (aOR, 1.786; 95% CI, 1.244–2.564), and younger age (aOR, 0.977; 95% CI, 0.963–0.991) were found to be predictors of successful early sedation. Age, dosage, and body weight percentile did not demonstrate a significant effect on early sedation (Table 3).
Univariate and multivariate analyses of the early sedation group and those who did not experience early sedation.
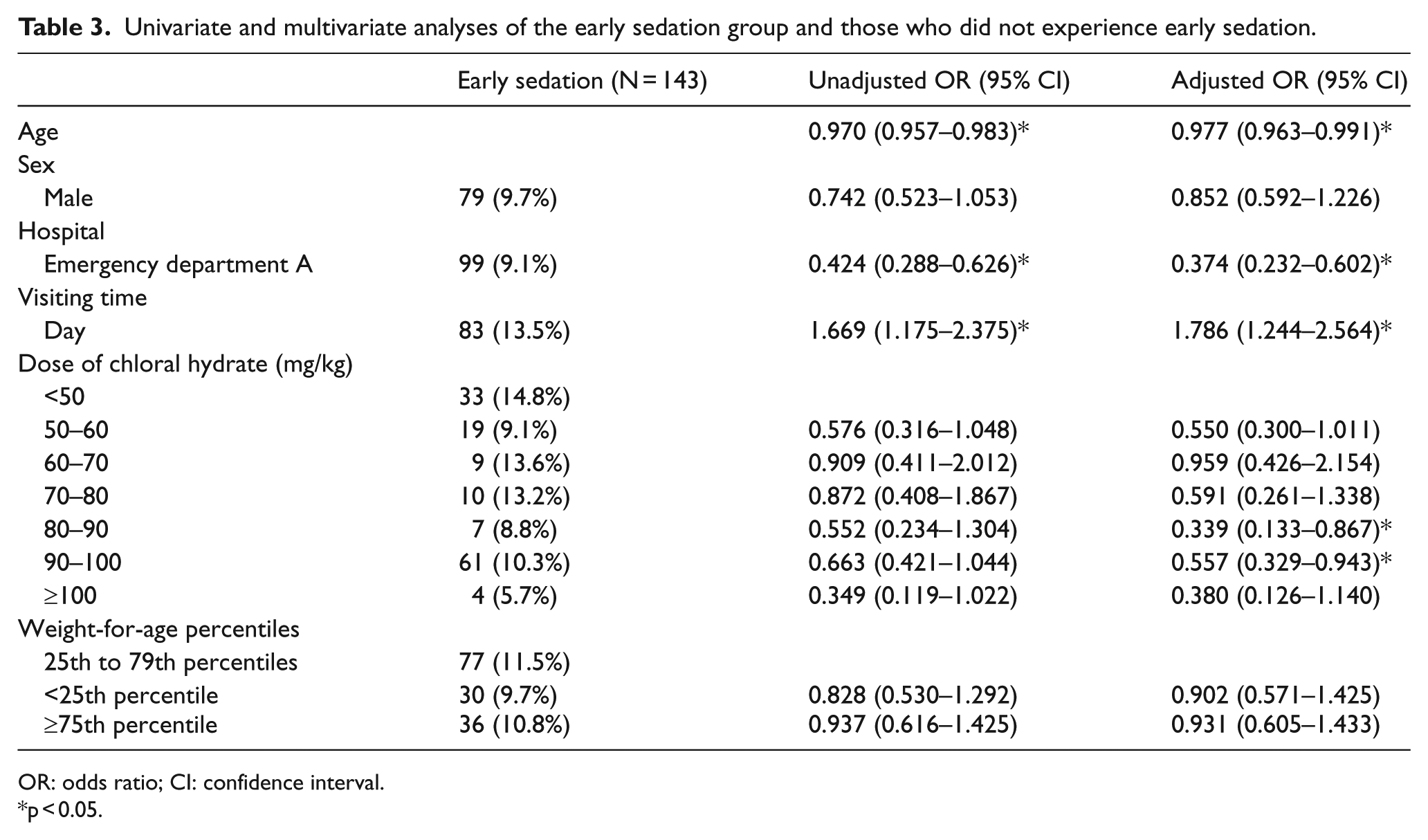
OR: odds ratio; CI: confidence interval.
p < 0.05.
Discussion
No previous studies have suggested factors contributing to successful chloral hydrate–induced sedation or those influencing the induction time until adequate sedation is achieved. Expanding on the findings of existing studies that had suggested associations between successful sedation and the patient age and dosage, this study examined the potential effects of environmental factors and developmental factors on successful sedation.
Pediatric sedation is performed when a patient’s movement must be restrained for an imaging test or when the testing or treatment procedure might be painful. Ideal sedation requires high efficacy, no adverse effects, ease of administration, and complete recovery. The most commonly used sedative for diagnostic imaging and painful procedures in Korea is chloral hydrate. 4 Many physicians are reluctant to use chloral hydrate because of its low efficacy and the risk of residual effects that can last for 24 h. Nevertheless, chloral hydrate remains widely used because it is inexpensive and easy to administer.11,12 Chloral hydrate is particularly more effective when it is administered orally. It is metabolized quickly into trichloroethanol via alcohol dehydrogenase in the liver and erythrocytes, after which it binds to glucuronic acid in the liver, and is excreted in the urine. Trichloroethanol has high plasma protein–binding capacity (70%–80%) and is rapidly distributed to all organs, including the brain. The recommended dosage of chloral hydrate ranges from 25 to 100 mg/kg, with a maximum dose of 2 g. In a typical clinical setting, 75–100 mg/kg is used. 13
Numerous studies have reported on the appropriate dosage of chloral hydrate and sedation success rates. Ronchera et al. 14 reported a success rate of 80% achieved with 55 mg/kg chloral hydrate, and of 94% with 65 mg/kg chloral hydrate. Greenberg et al. 15 reported that 100 mg/kg was able to achieve 91% adequate motion control for MRI, but in the case of children who were 4 years and older, the success rate dropped to 81%. Lee et al. 1 reported that a single or double dose of 50 mg/kg was able to achieve a success rate of 97.9% in infants 18 months and younger, and for children 18 months and older, this dose was able to achieve a success rate of 85.6%. A study by Delgado et al. 16 reported a 95% success rate. In this study, 50–100 mg/kg was administered according to the hospital sedation guidelines and yielded a success rate of 82.7%, which was lower than those reported in previous studies. This is thought to be because there were many cases where the lower limit of the recommended dosage (50 mg/kg) was administered and because cases where augmentation was required because of vomiting or spitting out the drug were excluded from the calculation, which narrowed the definition of sedation success. In this study, factors influencing successful sedation were a younger age and a minimum dose of chloral hydrate of 60 mg/kg, which were in agreement with the findings of existing studies. Chloral hydrate is occasionally administered concomitantly with other agents to increase the odds of successful sedation and to reduce the risk of adverse effects. In studies where hydroxyzine or midazolam were administered concomitantly with chloral hydrate, the success rates were 76.7% and 73.7%, respectively, which were lower than in other studies. 17
Chloral hydrate typically shows an onset of action within 30–60 min, and the duration of action varies widely from 2 to 8 h. 18 The elimination half-life typically ranges from 4 to 12 h and varies depending on the patient’s age; however, it is known to last up to 28 h in infants. 19 Because of the wide range of onset time and duration of action, extreme caution is required before administering a second dose when the initial dose fails to achieve adequate sedation. Because there are no studies currently available that suggest an optimal time for administering a second dose, this study suggests a variety of schedules, ranging from 15 to 60 min after the initial dose.
Early sedation accounted for 10.9% of the total sedations. This rate was higher among younger patients and among those who visited the centers during daytime hours. The mean − 1 SD of induction time was 18 min, which was shorter than the currently known chloral hydrate onset time. This may mean that early sedation is influenced by factors other than the agent itself. Emergency center A was not equipped with an isolated sedation space, and thus, the patients were exposed to the same environment as the other ER patients. In contrast, emergency center B transferred the patients to a dark and quiet location to induce sedation if the surroundings were not conducive to sedation. These differences in the environment are thought to have influenced outcomes. Patients who visited the centers during daytime hours had a success rate that was 1.789 times greater than that of those who visited the centers during nighttime hours. This finding directly challenged our hypothesis, which posited that the nighttime would be more conducive to successful sedation. In fact, the daytime in emergency centers tends to be quieter than the night because of reduced patient crowding. Such environmental characteristics appear to have had a positive effect on early sedation. In this sense, securing a quiet space conducive to sedation with patient monitoring capacity is expected to reduce the amount of sedatives required to sedate patients successfully.
Similar to sedation success, the time of the visit, emergency center, sex, and dosage did not have significant effects on delayed sedation (greater than or equal to mean + 1 SD, 67 min). The rate of delayed sedation increased as the patients’ ages increased. Based on this finding, it appears prudent to take an adequate amount of time to monitor older patients after the initial dose. Delayed sedation was significantly more prevalent in patients above the 25th percentile in body weight, which suggests that adequate sedation is more easily achieved in patients whose physical development lags behind that of their peers. In underweight patients, the decision for augmentation was made immediately in cases where the initial dose failed to achieve adequate sedation. However, no association was found among overweight patients in the 75th percentile.
This study had a few limitations. First, because it was a retrospective study, data regarding factors influencing success rate and induction time were limited. Incorporating additional factors, such as the patients’ fasting hours and sleep cycles, would be beneficial. 20 Although the level of pain could impact sedation, the subjects of this study were young children who had limitations in autonomously describing pain scores; in addition, this study was retrospective, and hence, information on pain could not be gathered. In order to calculate body surface area and the body mass index, which affects the onset time, duration of action, and half-life, the patient’s height is required. However, only body weight was available from the medical records. Second, the sedation protocol does not define the length of time required between the initial dose and the second dose. As such, attending physicians determined whether to wait until adequate sedation was achieved or to proceed with administering an additional dose. Because this study defined sedation failure as cases where augmentation was required, physicians’ decisions could have affected the success rate. Finally, adverse effects were not sufficiently evaluated in this study. Initially, the relationship between prolonged sedation and chloral hydrate dose was considered to influence the appropriate chloral hydrate dose; however, accurate recovery time could not be checked on hospital records. Length of stay was considered as a surrogate marker; however, patients visiting the hospital at nighttime tended to be discharged before their sedation had ended. As such, it would have been difficult to predict sedation duration from length of stay. Thus, we intend to conduct a follow-up study controlling for such influencing factors.
Conclusion
In order to ensure safe pediatric sedation, a thorough understanding of the patient’s age and dosage is required, as well as an environment conducive to sedation. Taking into account that increasing age and developmental stage can affect induction time, unnecessary augmentation should be avoided.
Footnotes
Acknowledgements
This retrospective study involves no more than minimal risk to the subject and the waiver of informed consent does not adversely affect the rights and welfare of the subjects. A waiver of consent was approved by the appropriate ethics review boards. All authors have approved the manuscript and have agreed to submit it to your esteemed journal.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was supported by Hallym University Research Fund 2014 (HURF-2014-47).
