Abstract
Introduction:
Arsenic is a traditional poison that has a history extending back to ancient times.
Case presentation:
A 43-year-old male presented to our Emergency Department 50 min after ingestion of 300 mL self-prepared arsenic trioxide solution (砒霜). He produced the solution from heating xionghuang (雄黃), an arsenic sulfide mineral (As4S4) which is a Chinese herb, for academic purposes a few years ago.
Discussion:
Supportive care including fluid replacement and decontamination with gastric lavage and chelation therapy with intravenous dimercaptopropanesulphonate sodium was given to patient. Spot urine arsenic to creatinine ratio significantly decreased from 343,124 nmol/mmol (day 3) to 7116 nmol/mmol (day 5). He had an uneventful course without any neurological deficit.
Conclusions:
Treatment of acute arsenic poisoning includes supportive care, decontamination, and chelation therapy. DMPS is an antidote for arsenic poisoning and its use should be directed by clinical status and urinary arsenic excretion.
Introduction
Arsenic is a traditional poison that has a history extending back to ancient times.1 –3 The name “arsenic” is derived from arsenikon, Greek for potent. Its resemblance to sugar and its tastelessness made it a popular assassination poison in the Middle Ages. 4 It was widely used to treat syphilis until the mid-20th century and is currently used as a chemotherapeutic agent to treat acute promyelocytic leukemia and other myeloproliferative disorders. 5 There are only a few case reports of arsenic poisoning in recent literature 6–10 that make treatment to be given empirically. It is believed that the primary treatment modalities are rapid and effective decontamination and early initiation of chelation therapy. We report a case of arsenic trioxide poisoning for illustration.
Case report
A 43-year-old man, with the history of depression, presented to our Emergency Department 50 min after ingesting 300 mL self-prepared arsenic trioxide (砒霜) solution together with wine. On arrival, he was alert and conscious but appeared dehydrated. He had abdominal pain with several episodes of vomiting. He had tachycardia with heart rate of 122 beats per minute, blood pressure of 107/62 mmHg, temperature of 36℃ and oxygen saturation of 98% on room air.
A plastic bottle containing brownish solution, alleged arsenic trioxide, was brought in by ambulance crews as shown in Figure 1. He claimed he produced the arsenic trioxide solution from heating xionghuang (雄黃), an arsenic sulfide mineral (As4S4) which is a Chinese herb, for academic purposes a few years ago.

A plastic bottle containing arsenic trioxide solution.
His cardiovascular and respiratory system was normal. Electrocardiogram revealed sinus tachycardia. QRS and QTc intervals were within normal ranges. Intravenous fluid replacement with 1 L normal saline was given to him for rehydration. Decontamination with gastric lavage was performed. An amount of 50 g of activated charcoal was subsequently given through the orogastric tube after consultation of the hospital toxicology team and the Hong Kong Poison Information Center. Abdominal X-ray studies did not reveal any radiopaque shadow. Chelation therapy with intravenous dimercaptopropanesulphonate sodium (DMPS) was given 90 min after arrival to hospital or 140 min post ingestion time. DMPS was administered intravenously 250 mg every 6 h for 13 doses, followed by every 12 h for 4 doses.
He was managed in the Emergency Medicine Ward of the hospital. Itchiness was noted over his arms 6 h after the administration of DMPS for which chlorpheniramine was administered and itchiness subsided. Symptoms of nausea and diarrhea persisted. Diarrhea was noted upon the first day of admission. He developed marked weakness over lower limbs of grade 0/5. Severe hypokalemia was noted (serum potassium level 2.0 mmol/L). Electrocardiogram revealed the presence of U wave. Replacement with oral potassium was given. He regained full power after correction of serum potassium (4.7 mmol/L) 2 days later.
There is no blood test of serum arsenic level available in laboratory in general public hospitals in Hong Kong. His 24 h urine arsenic level collected 3 days post ingestion was 120,295 nmol in a urine volume of 3.45 L. Upon clinical improvement, intravenous DMPS was switched to oral form as 100 mg eight times a day. Spot urine arsenic to creatinine ratio decreased from 343,124 nmol/mmol Cr (day 3) to 7116 nmol/mmol Cr (day 5) as shown in Figure 2. The normal reference level of As/Cr is <150 nmol/mmol Cr.
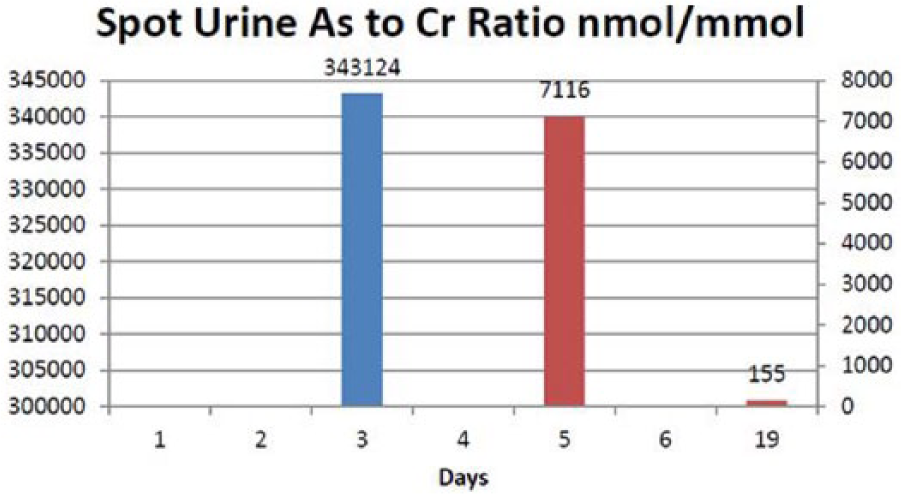
Spot urine arsenic to creatinine ratio over the patient’s clinical course.
The specimen, containing arsenic trioxide solution, was sent to the Toxicology Reference Laboratory in Princess of Margaret Hospital for further analysis. The arsenic (As) content was found out to be 12,047,380 µg/L. Based on the reported intake of 300 mL liquid taken by the patient, the total arsenic intake in this patient was 3,614,214 µg (or 3.6 g). This grossly exceeds its maximum permitted level in drinking water at <10 µg/L, defined by World Health Organization; 11 and the total intake at 1500 µg/day, defined by Chinese Medicine Council of Hong Kong. 12 This result was available 18 days after admission.
He was assessed by in-patient psychiatry with a diagnosis of recurrent depressive disorder. He could be discharged with acknowledgment of medical advice after 7 days of admission. He was prescribed outpatient oral DMPS 6 times a day for 10 days. Toxicology follow-up clinic was arranged to him. He had an uneventful course without any neurological deficits. Repeat complete blood count and liver and renal function were normal. The oral DMPS was by then stopped. A 24-h urine arsenic collected 19 days post ingestion revealed 2219 nmol arsenic in 2.5 L urine.
Discussion
Acute arsenic overdose is a rare poisoning that many clinical physicians may not be familiar with. The diagnosis is straightforward if there is no concealed deliberate self-harm.
Arsenic occurs in an inorganic or organic form. Its inorganic form is more toxic than its organic form. Its inorganic compounds include the arsenites, the arsenates, and elemental arsenic. In vivo organic-to-inorganic and inorganic-to-organic conversion may occur.1,2,13 Arsenic is primarily absorbed by ingestion, inhalation, or skin.1,13 It distributes rapidly into erythrocyte and binds to the hemoglobin. Redistribution to the liver, kidneys, spleen, lungs, and gastrointestinal tract occurs within 24 h. It impairs cellular respiration by inhibiting mitochondrial enzymes and uncoupling oxidative phosphorylation via inhibition of sulfhydryl group–containing cellular enzymes and substitution of phosphate with arsenate in “high-energy” compounds.1,3 These arsenate compounds are unstable and are rapidly hydrolyzed, termed arsenolysis. Arsenic also blocks steps in Krebs cycle.1,3 These inorganic arsenic is excreted in urine, the majority within the first 4–5 days after exposure.14,15
Acute arsenic poisoning is characterized by an initial gastrointestinal illness that develops in the first 30–60 min after ingestion and results in persistent vomiting and diarrhea. 4 This patient has persistent vomiting, and this could account for severe hypokalemia with potassium level of 2.0 mmol/L and subsequent muscle weakness. With massive ingestion, there is rapid progression to multi-organ failure, predominated by cardiac toxicity with tachycardia, hypotension, cardiac failure and pulmonary edema, QT prolongation and arrhythmia, and sensorimotor neuropathy with major disability at subacute phase.4,5 In sublethal doses, the gastrointestinal illness would resolve, and the most important delayed effect is the development of peripheral neuropathy. 4
The primary modalities of treatment are rapid and effective decontamination and early initiation of chelation therapy. There are only a few case reports6 –10 of arsenic poisoning in recent literature, but no controlled trials to demonstrate the effectiveness of different forms of decontamination and chelation therapy, making the use of these therapies empirical. Gastric lavage was used as the decontamination method in this patient as it may help to remove arsenic from the stomach. It was followed by activated charcoal, although it did not absorb significant amount of arsenic. 16 Whole bowel irrigation has been used in one previous report 9 of two cases with radio-opaque arsenic.
Chinese herbal medicine is classified into schedule 1 and 2 in Hong Kong. There are 31 schedule 1 toxic herbs and arsenic trioxide (砒霜) is one of them. Practice, trading, manufacture, and use are controlled under the Chinese Medicine Ordinance. 16 The dispensing of schedule 1 herb should be based on prescription by a registered Chinese Medicine practitioner. Xionghuang (雄黃), with another name of realgar (黃金石/雞金石), α-As4S4, is an arsenic sulfide mineral being used in Chinese Medicine.
The introduction of 2,3-dimercaptopropane-1-ol (dimercaprol or British anti-Lewisite (BAL)) as an arsenic antidote by Sir Rudolph Peters et al. 17 marked the beginning of a causative therapy of arsenic poisoning. BAL, however, is rather toxic by itself. It may cause hypertensive episodes and has been shown to redistribute arsenic into organs that are shielded by a blood-organ barrier, for example, the brain and the testes. There are no oral preparations available and it has an unpleasant odor. That is why we use dimercaptosuccinic acid (DMSA) in this patient because they cause less adverse effects and do not increase deposition of arsenic into the central nervous system due to its hydrophilic property.18,19 There are insufficient data to determine which chelating agent is the agent of choice, but limited evidence favors DMPS which has now been used successfully in a number of cases of arsenic poisoning.
DMPS is a level-1 antidote, which means that it is available in every acute hospital in Hong Kong. It works with non-enzymatic methylation of arsenic. Its side effects include nausea, dysguesia, leucopenia, fever, elevated liver enzymes, headache, fatigue, bronchospasm, and allergic reaction. Our patient could tolerate DMPS satisfactorily. There is a lack of conclusive data to compare the relative efficacy between DMPS and DMSA. In a human report of arsenic poisoning, DMSA failed to significantly increase the urine arsenic excretion and the symptom of neuropathy progressed despite under treatment. After starting intravenous DMPS, urine arsenic elimination increased during the first 24 h, and initial improvement of neuropathy was noted. 20 In mice, DMPS exhibited greater antidotal activity in reversing the inhibition of arsenite on pyruvate dehydrogenase. 21
Animal data support that the efficacy of chelation for arsenic poisoning is greater when it is administered earlier. 22 During chelation therapy, clinical and laboratory arsenic level response should be followed. Chelation is recommended to continue until a 24 h urinary arsenic level reaches less than 50 µg/L (or 0.65 µmol/L). 23
Conclusion
Treatment of acute arsenic poisoning includes supportive care, decontamination, and chelation therapy. DMPS is an antidote for arsenic poisoning and its use should be directed by clinical status and urinary arsenic excretion.
Footnotes
Acknowledgements
The authors thank the Hong Kong Poison Information Center for toxicology consultation of patient care.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of data and materials
Clinical data were retrieved from patient’s clinical files, including the resuscitation chart and electronic medical system (Clinical Management System).
Informed consent
Verbal consent from the patient was obtained.
Ethical approval
Ethical approval of this study was obtained from the Ethical Committee.
