Abstract
Background:
Iatrogenic injuries due to drug–drug interactions are particularly significant in critical care units because of the severely compromised state of the patient. The risk further increases with the use of multiple drugs, increasing age, and stay of the patient.
Objective:
The aim was to assess potential drug–drug interactions, evaluate clinically significant potential drug–drug interactions and their predictors in medical intensive care units of tertiary hospitals in Pakistan.
Methods:
Analysis of patient data collected from medical intensive care units of tertiary hospitals in Pakistan were carried out using Micromedex DrugReax. Various statistical tools were applied to identify the significance of associated predictors.
Results:
In a total of 830 patients, prevalence of potential drug–drug interactions was found to be 39%. These attributed to 190 drug combinations, of which 15.4% were clinically significant. A significant association of potential drug–drug interactions was present with number of prescribed drugs, age, and gender. In terms of clinically significant potential drug–drug interactions, the association was significant with increasing age. Moreover, one-way analysis of variance revealed a significant difference in the means of potential drug–drug interactions among the four hospitals.
Conclusion:
A prevalence of 39% potential drug–drug interactions was observed in patients of medical intensive care unit, with 22.8% being clinically significant. These attributed to nine drug pairs and could easily be avoided to reduce the risk of adverse effects from potential drug–drug interactions.
Introduction
Iatrogenic injuries in the health-care system are not an unknown phenomenon and may be caused by any drug-related problem, commonly due to drug–drug interactions. 1 Although readily accessible drug information sources are available for health-care professionals, drug–drug interactions remain to be a substantial problem in clinical practice. In fact, many reviews claim that drug–drug interactions are one of the most recurrent and easily preventable drug-related problems.2,3
Intensive care units (ICUs) support patients with severely compromised health and life-threatening conditions. In comparison to patients of a general ward, these patients are at a greater risk of experiencing drug–drug interactions due to multiple drug therapy. Even acute changes in drug effects, supporting and treating vital body functions may drastically deteriorate the patient’s condition. Therefore, keeping drug–drug interactions at bay for ICU patients is of prime importance. 4
Studies reporting prevalence of potential drug–drug interactions (PDDIs) in medical intensive care units (MICUs) are scarce. Many studies report PDDIs in general ICU settings; however, PDDIs in MICU may differ due to the change in the medical condition of the patient as well as the drugs used. Moreover, in Pakistan, no multicentered study has been conducted in MICU to provide a wider picture of the existing problem of PDDIs, particularly clinically significant PDDIs.
Methodology
The multicentered study was conducted in MICUs of the following four tertiary care hospitals in Peshawar, Pakistan: Lady Reading Hospital (LRH), Khyber Teaching Hospital (KTH), Hayatabad Medical Complex (HMC), and Northwest General Hospital and Research Center (NWGH & RC). These hospitals receive patients from the northwestern region of Pakistan and Afghanistan. A total of 1465 patients were admitted to the MICUs of the four hospitals in the study year (July 2013–July 2014). Sample size was calculated using the formula for known populations with finite population correction, which came out to be 830. So data of around 1000 patients were collected; of which, 830 met the inclusion criteria and were included in the study. Patient admitted to the MICU for at least 24 h and prescribed two or more drugs was set as the inclusion criteria.
Ethical approvals from the respective hospitals were obtained beforehand vide letter number 8075-79/HMC, 488/pharm (KTH), 010 (LRH), and NWGH/Research/01. Data were collected on a predesigned proforma which included patient demographics and treatment profile; patient identification was kept confidential and good clinical practice (GCP) guidelines under the Declaration of Helsinki by the International Conference on Harmonization and Nuremburg Code were followed. 5 Written informed consent was not necessary because no personal patient data have been included in the article.
Data were evaluated for PDDIs using Micromedex DrugReax, 6 which provided detailed information on the severity, documentation, onset, and mechanism of the PDDIs. On the basis of severity and documentation data provided by Micromedex DrugReax, clinical significance for all the drug combinations was derived. Only those drugs were evaluated for PDDIs which were administered concomitantly.
To evaluate the association of PDDIs and clinically significant PDDIs with predetermined predictors independently and in the presence of other predictors, univariate and multivariate logistic regression was used. One-way analysis of variance (ANOVA) was used to calculate the difference in means of PDDIs among the four hospitals. Statistical interpretation was performed using IBM SPSS Statistics for Windows, Version 20 (IBM Corp.; Armonk, NY). 7
Results
A total of 830 patients were admitted in the MICUs of the four hospitals, and 701 PDDIs (39%) were observed in these patients leading to a mean frequency of 0.8 PDDIs per patient (95% confidence interval (CI) = 0.7–0.9). Of the total patients, 323 experienced a minimum of one PDDI leading to a frequency of 2.2 PDDIs per patient (95% CI = 2.0–2.4) within this subgroup. These two outcomes for the total patients stratified for different patient characteristics such as gender, age, duration of stay, and number of prescribed drugs are listed under Table 1.
Main outcome parameters.
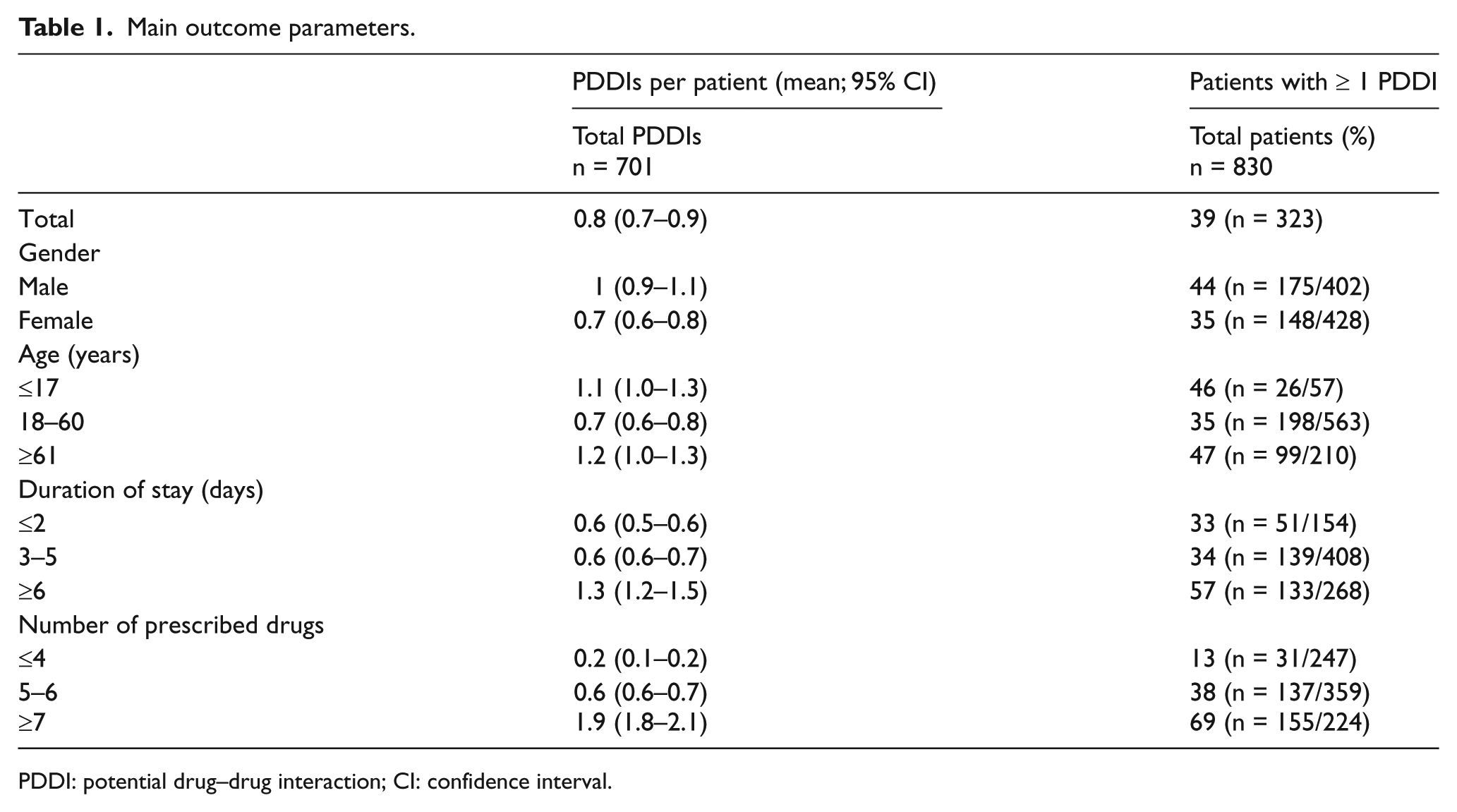
PDDI: potential drug–drug interaction; CI: confidence interval.
A total of 190 drug combinations were responsible for 701 PDDIs, of which 39 drug pairs were clinically significant and were observed in 22.8% patients. In total, 108 PDDIs were clinically significant. Of these, 66% clinically significant PDDIs attributed to nine drug combinations, which along with their potential outcomes are enlisted in Table 2. PDDIs with major severity and fair documentation were encountered by 47.7% and 45.6% patients, respectively. The frequencies of severity and documentation of all PDDIs are shown in Figure 1. Delayed onset was prevalent in clinically significant PDDIs while pharmacokinetics and metabolism were the prevalent type and mechanism in clinically significant and all the PDDIs, as shown in Figure 2.
Frequently observed clinically significant drug combinations.
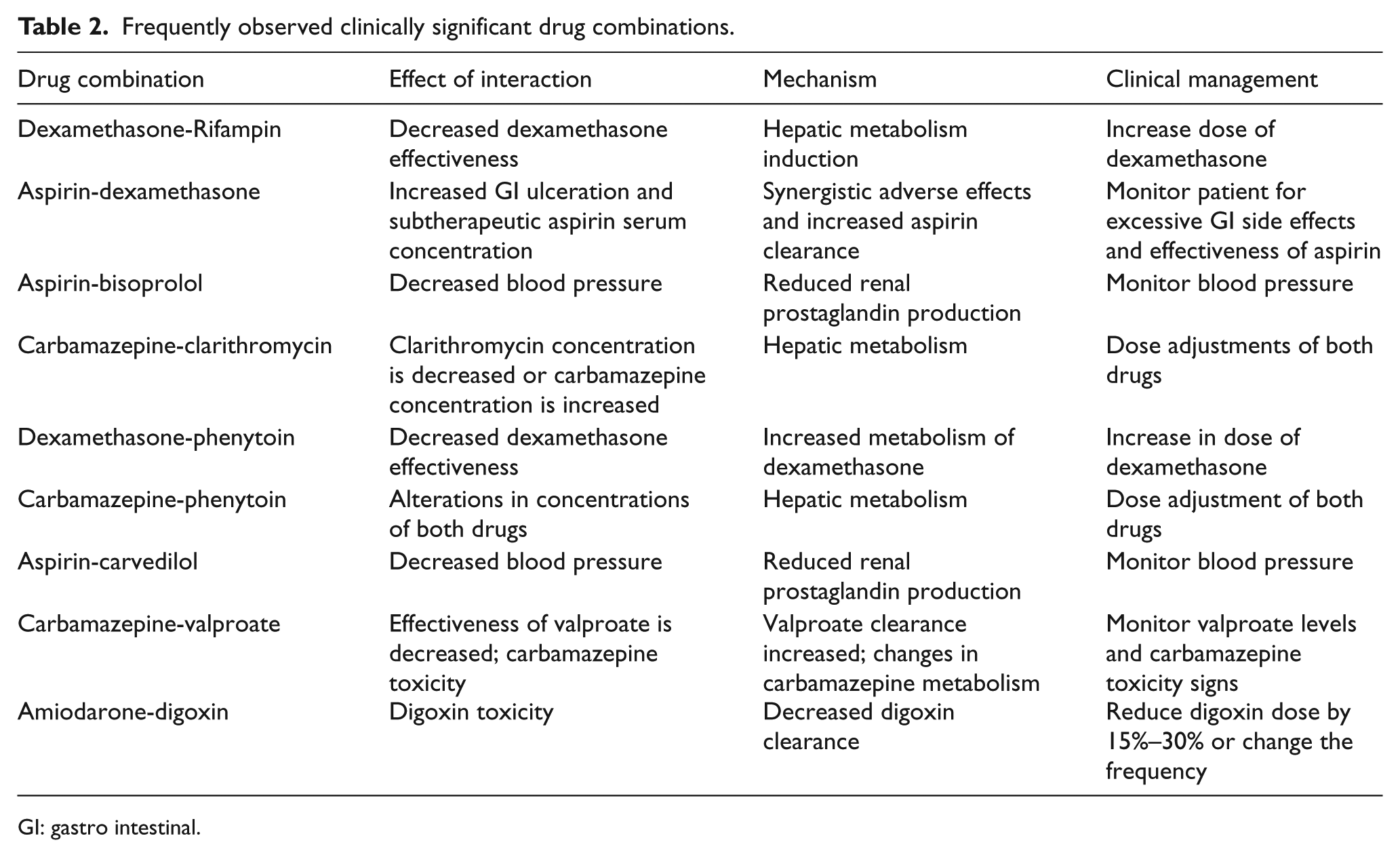
GI: gastro intestinal.
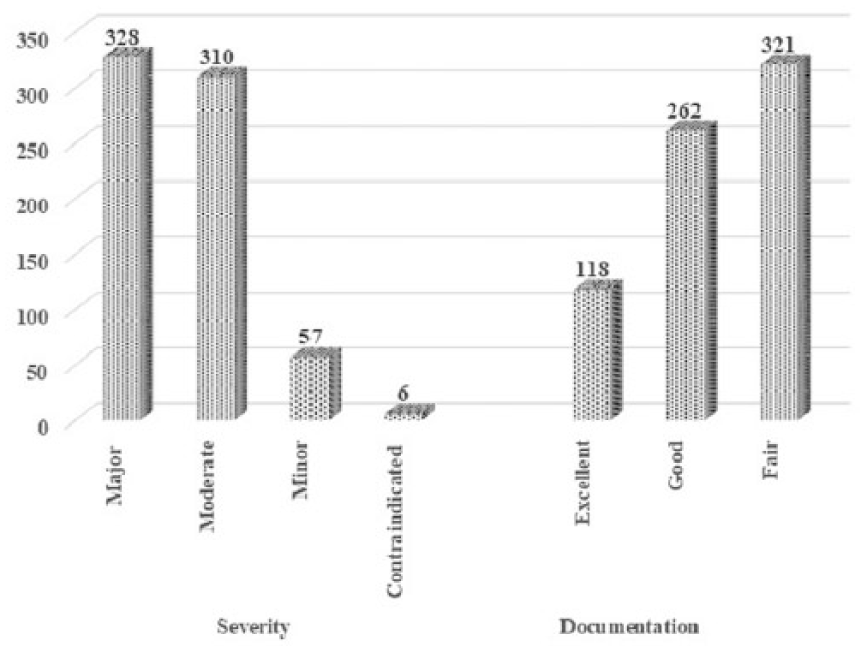
Frequency of PDDIs according to severity and documentation.
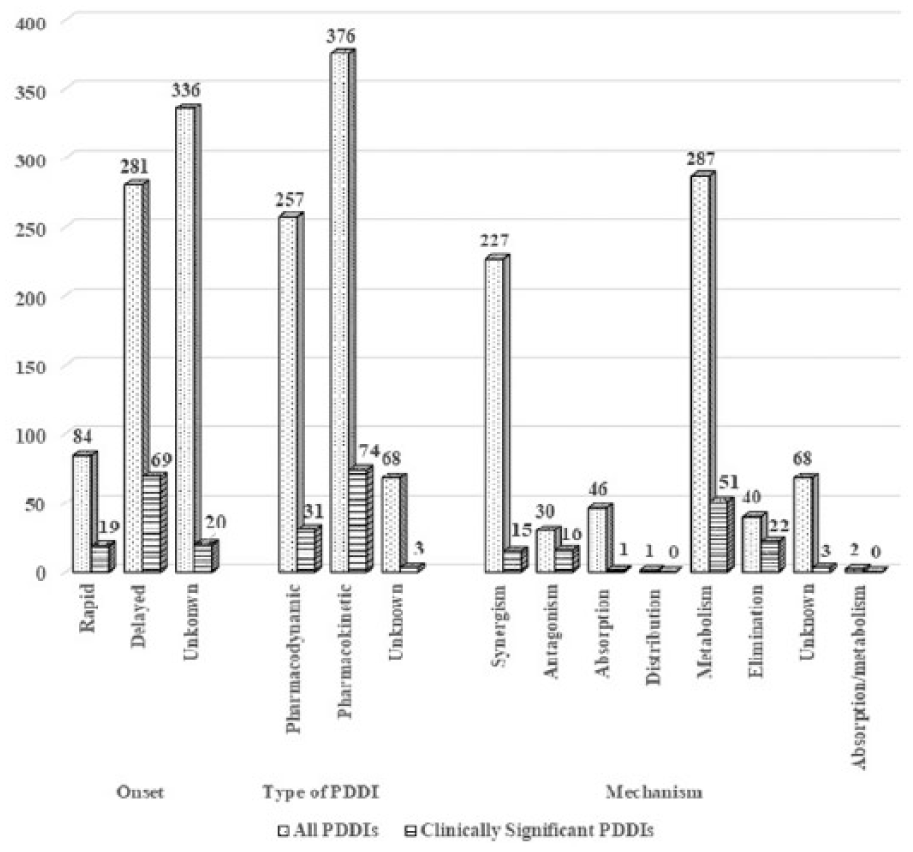
Comparison of PDDI levels between clinically significant and all PDDIs.
The association of prescribed drugs, duration of stay, and gender with PDDIs was determined using univariate and multivariate logistic regression to calculate the odds ratio (OR) with 95% confidence interval (CI) and significance (p)
In terms of exposure to all PDDIs, a significant association was present between PDDIs and with the patients prescribed five or more drugs (p = 0.000). A significant association of PDDIs was also observed with patient gender (p = 0.008) and stay duration of 4 days or more (p = 0.017). However, an insignificant association was found between the presence of PDDIs and patient age above 60 years (p = 0. 061).
In terms of exposure to clinically significant PDDIs, their association was significant with patient age above 60 years (p = 0.007) and patient gender (p = 0.032). An insignificant association was observed between clinically significant PDDIs and five or more drugs prescribed (p = 0.060) and patient stay of more than 4 days (p = 0.467). Table 3 illustrates results of univariate logistic regression.
Univariate and multivariate logistic regression based on exposure to all PDDIs and clinically significant PDDIs.
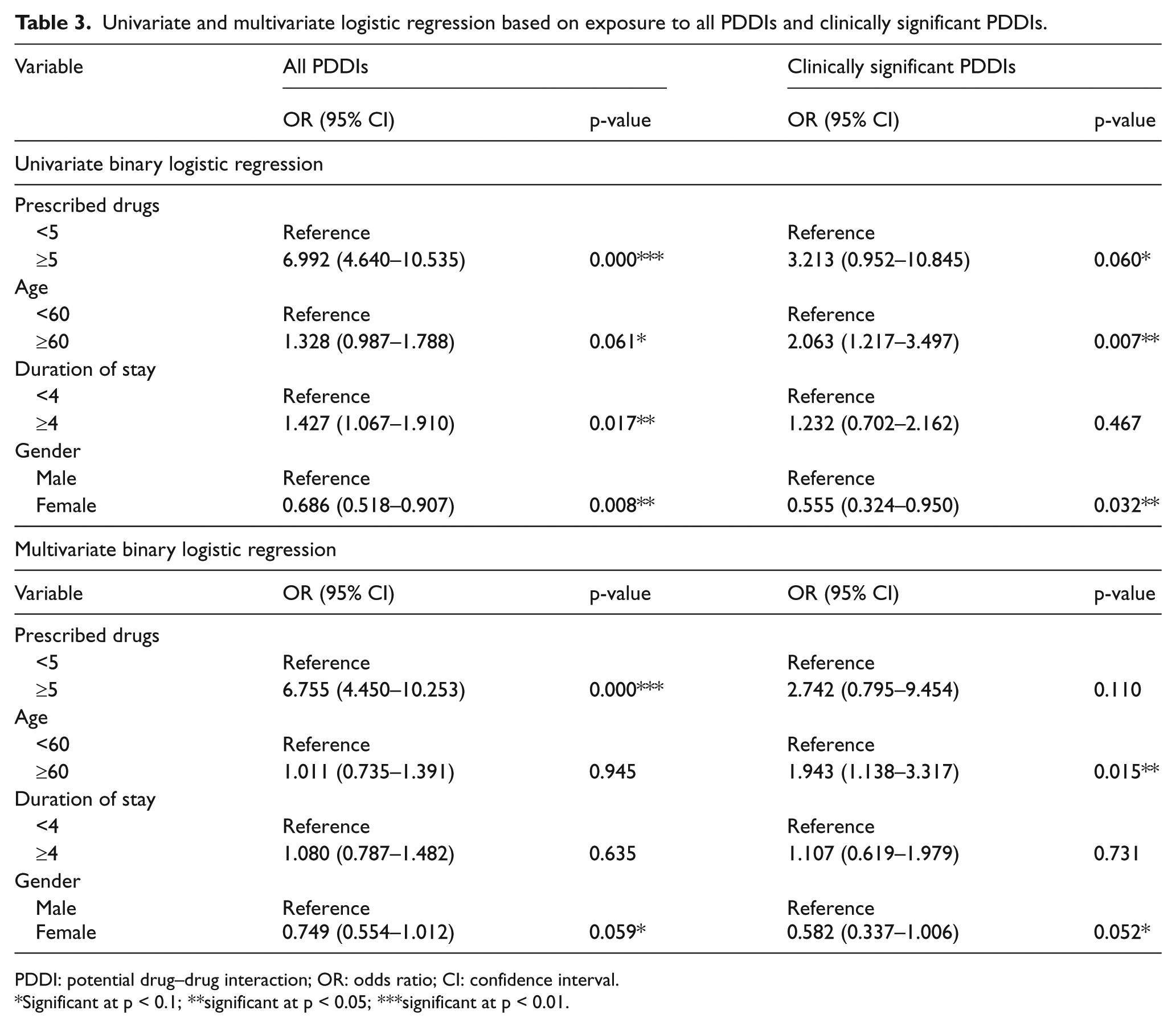
PDDI: potential drug–drug interaction; OR: odds ratio; CI: confidence interval.
Significant at p < 0.1; **significant at p < 0.05; ***significant at p < 0.01.
Multivariate logistic regression analysis in terms of exposure to all PDDIs revealed significant association of PDDIs with increasing number of prescribed drugs (p = 0.000). Insignificant associations were observed of PDDIs with gender (p = 0.052), increasing age over 60 years (p = 0.945) and prolonged stay of 4 days and above (p = 0.635).
While in terms of exposure to clinically significant PDDIs, their association with patient age of 60 years and above was observed to be significant (p = 0.015) while insignificant associations were found between clinically significant PDDIs and gender (p = 0.052), five or more prescribed drugs (p = 0.110) and patient stay of 4 or more days (p = 0.731). Table 3 illustrates results of multivariate logistic regression.
One-way ANOVA showed a significant difference (p = 0.000) in the means of PDDIs among the four hospitals. Post hoc test (least significant difference (LSD)) revealed that there was a significant difference (p = 0.042) of PDDIs between NWGH & RC and HMC. A significant difference (p = 0.000) was also present between KTH and LRH and HMC.
Discussion
PDDIs occur in every setting attributing mainly to polypharmacy and other minor factors. Developed countries have systems in place to routinely monitor these PDDIs, but in developing countries, identification of this problem is a recent phenomenon. Studies regarding PDDIs are being conducted in the developing world, each with its unique objective. Our study targets the MICU setting and also is concerned not only with PDDIs but with the ones which are clinically significant.
A prevalence of 39% PDDIs was observed, which is significant if the critical nature of the setting involved and patient critical condition is accounted. Moreover, 22.8% clinically significant PDDIs were observed of the total encountered PDDIs. A Dutch study conducted in the general ICU reported a PDDI prevalence of 54%. 8 The comparative higher prevalence of PDDIs in the Dutch study was due to the involvement of cardiac patients, in which PDDIs are known to be high as reported in many studies,9,10 while an Indian study conducted in the MICU reported the prevalence of PDDIs to be 90%. 11 However, the number of patients included in this study was very small. This suggests that the nature of PDDIs depend on factors like the drugs used in a hospital, prescribing behavior, and treatment patterns which varies from country to country and even varies among the hospitals in a country.
A high frequency of moderate and major severity PDDIs were observed while the prevalent documentation of PDDIs was good or fair, which complies with other studies. An Indian study reported PDDIs of moderate severity and of good documentation to be the most prevalent. 11 Moderate severity PDDIs were noted to be the most prevalent (61.2%) in an Ethiopian study. 12 Of the total PDDIs, 71.7% were of moderate severity and 60% were of fair documentation in an Iranian study. 13 A Brazilian study also reported that 86% of the PDDIs were of major and moderate severity. 14 A Swiss study conducted in the medical wards reported 70.1% PDDIs to be of moderate severity while 69.4% were of good documentation. 3
A total of 64% clinically significant PDDIs had a delayed onset while 68.5% were of pharmacokinetic type, and 47.2% had a mechanism of metabolism. The unique derivation of clinically significant PDDIs was performed for the first time; therefore, the levels of interactions are not comparable to any other study.
A significant association of number of PDDIs was found with number of prescribed drugs, gender, and duration of stay when evaluated under univariate model. However, in multivariate model, the association of number of PDDIs was significant with number of prescribed drugs and gender only, while the association with duration of stay became insignificant. In terms of clinically significant PDDIs, their association was significant with increasing age and gender when evaluated both under univariate and multivariate models. Various other studies have also reported a similar association. A significant association was reported in an American study between the number of PDDIs and increasing age and the number of drugs in univariate model. 15 A study conducted in Italy also reported a significant association of PDDIs with increasing number of drugs and age. 16 An Ethiopian study revealed a significant association of PDDIs with patients receiving three or more drugs and stay. 12 An Indian study reported a significant positive association between these two factors, 17 while various Iranian, Brazilian, and Swedish studies also reported a significant association between the number of interactions and the number of prescribed drugs.18–20 In a study conducted in an ICU of a university hospital, an insignificant association of PDDIs was found with duration of stay and the age of patients. 21
Limitations
The limitation of this study is that the adverse outcomes of the PDDIs could not be studied, as patient follow-up was not possible due to administrative issues.
Conclusion
A prevalence of 39% PDDIs was observed in the MICUs of four hospitals, with 22.8% being clinically significant. Most of the PDDIs had moderate and major severity and fair documentation. In terms of clinically significant PDDIs, delayed onset and pharmacokinetic type with metabolism as the mechanism were prevalent. The association of PDDIs with increasing drugs, age, and gender was significant while the association of clinically significant PDDIs with increasing age and gender was significant. Lack of drug interaction–monitoring systems in hospitals lead to undetected PDDIs in patients; thus, installation of such systems is recommended. Moreover, the clinically significant PDDIs described in this article can at least provide some guidance and lead to some control of this problem.
Footnotes
Acknowledgements
The authors thank the management, administrative, and clinical staff of all the hospitals involved in this study for their complete co-operation during the research.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of data and materials
Data can be provided upon request from the primary author.
Informed consent
Not required as no patient data were collected, and patient was not directly involved.
Ethical approval
Ethical approvals from the respective hospitals were obtained beforehand vide letter number 8075-79/HMC, 488/pharm (KTH), 010 (LRH), and NWGH/Research/01.
Human rights
Patient identification was kept confidential, and GCP guidelines under the Declaration of Helsinki 5 by the International Conference on Harmonization and Nuremburg Code were followed.
