Abstract
Introduction:
Low back pain or numbness of the lower extremities is quite common in the present population. Numerous conditions may cause these symptoms, including spinal vascular anomaly. Identifying and diagnosing the cause of these symptoms are crucial for treatment. Accurate diagnosis based on particular radiological findings in magnetic resonance imaging is crucial for administering adequate therapy to patients, especially in spinal dural arteriovenous fistula.
Case presentation:
We report a case presenting with low back pain and rapid deterioration in paraplegia. Magnetic resonance imaging provided the typical image presentations such as spinal cord edema with tortuous dilated perimedullary venous plexus. The patient was subsequently successfully treated with endovascular embolization.
Discussion:
Spinal dural arteriovenous fistula should be suspected in any patient who presents with myelopathy. Angiography remains the gold standard for confirmation of diagnosis.
Conclusion:
Prompt treatment with endovascular embolization or surgery could improve patients’ outcomes.
Introduction
Spinal dural arteriovenous fistula (SDAVF) is a rare and usually underdiagnosed disease, referred to as the fistula from the dural branch of the dorsospinal artery which drains into the perimedullary vein through the reticulospinal vein, and becomes symptomatic from the onset of intramedullary venous hypertension. 1 The symptoms include low back pain, weakness of the lower extremities, sexual dysfunction, incontinence, and urine retention. Because of variation in symptoms, differentiating SDAVF from other myelopathies, such as cord compression, myelitis, and intramedullary neoplasm, is difficult, impeding the accurate diagnosis of SDAVF.
Prompt diagnosis of SDAVF enables endovascular embolization with liquid polymers, such as N-butyl 2-cyanoacrylate or microsurgery ligation, if endovascular embolization fails and its prognosis is usually favorable. 2 Here, we report a case of a 57-year-old man who had received a diagnosis of SDAVF on the basis of spinal magnetic resonance imaging (MRI) findings and was subsequently successfully treated with endovascular embolization.
Case Presentation
A 57-year-old man, working as a porter of fuel gas pails, presented to our emergency department complaining of weakness of the bilateral lower extremities and unsteady gait. He had no significant medical history, except for a previous back injury due to a car accident during youth. Three days before the visit, he sprained his waist at work and had lower back pain, which could be partially relieved using oral analgesics. The pain was not aggravated by bending or extending the trunk. However, the pain aggravated the next day, associated with radiation numbness from the waist to the bilateral knees and burning sensation of the feet. He had no stool or urine incontinence; however, he experienced some difficulty in urinating because of back pain and from the burning sensation in the feet and legs. On examination, he had no fever, and his consciousness was clear and vital signs were stable. Other findings revealed no obvious wound and intact muscle power except in the bilateral lower extremities. The grade of muscle power of the bilateral lower extremities was decreased to 3 according to the Oxford scale. Acute urine retention was confirmed through bladder distention and catheterization with residual urine volume of 1 L. Neurological examination revealed saddle anesthesia and absent bulbocavernosus reflex with increased knee and ankle jerks. The straight leg raise test (SLRT) was negative for the bilateral lower extremities and Babinski sign was not observed.
X-ray revealed severe compression fracture at the T12 spine and mild compression fracture at L2. On the basis of the patient’s history, we ruled out spinal abscess owing to his acute symptoms without other signs of infection. Concurrently, we suspected conus medullaris or cauda equina syndrome because of spinal cord compression of the protruding bony fragment and disk herniation related to T12 and L2 compression fractures. Furthermore, MRI demonstrated mild spinal cord compression by the compression fracture at T12. In addition, the presence of a tortuous linear structure over the T12–L5 indicated venous hypertension (Figures 1 and 2). Progressive deterioration of symptoms, paresthesia of the lower extremities, and micturition and defecation problems are typical symptoms of SDAVF. On the basis of the typical image of beading or tubular engorged veins with spinal edema in the coronal view, it was consistent with SDAVF (Figure 1). The patient immediately underwent an endovascular intervention in which the final diagnosis revealed T12 SDAVF with the feeding artery from the left intercostal artery (Figure 3). Embolization to the dural fistula site was performed with successful interruption of the blood flow from the T12 intercostal artery to the dural venous plexus. The neurologic function of our patient had a dramatic improvement after endovascular embolization was performed. A few sequelae, such as buttock numbness and mild voiding difficulty, persisted for 1 year and were treated through medication. The patient eventually had spontaneous remission and has been symptom-free until now with no evidence of disease recurrence.
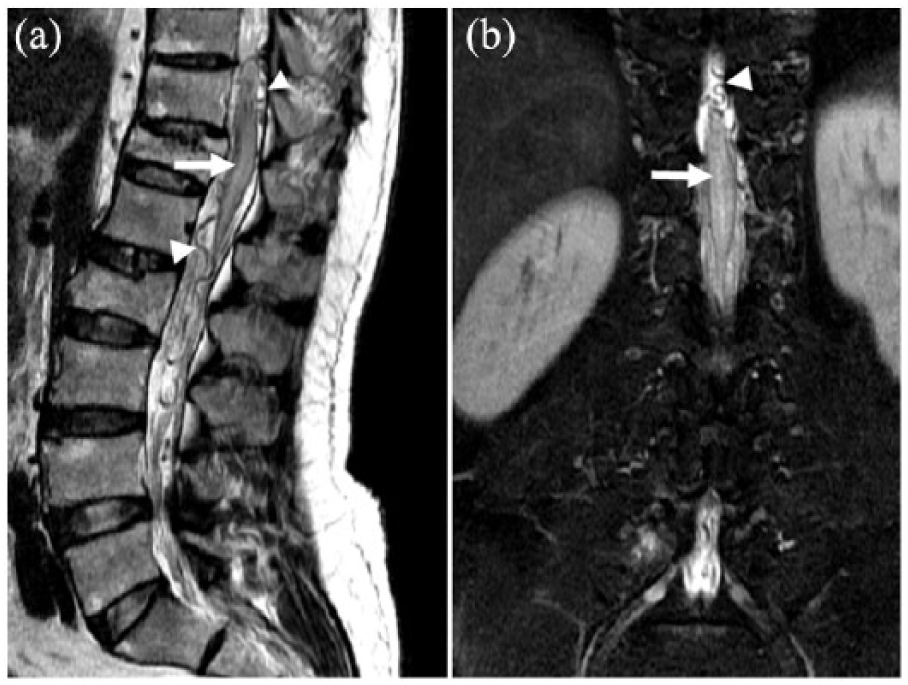
Lumbar sacral spinal MRI T2W (a) sagittal and (b) corona views displayed the conus with high signal (arrow); the most striking findings are the tortuous and dilated perimedullary vessels surrounding the conus medullaris and cauda equina arrowhead, consistent with venous hypertension.
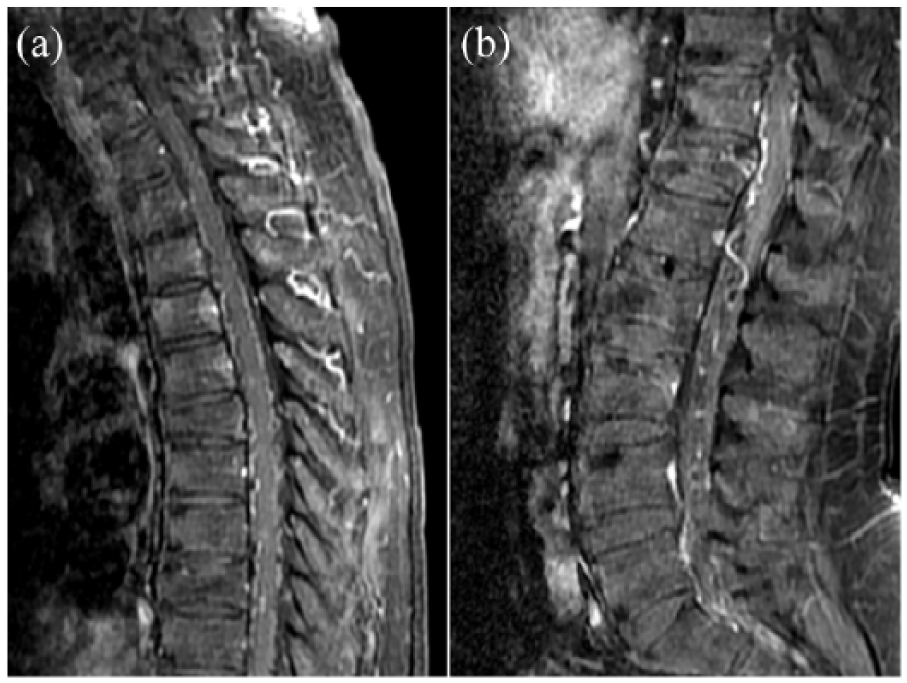
(a) Thoracic and (b) lumbar sacral spinal MRI T1 sagittal view with gadolinium enhancement showed patchy intramedullary enhancement (breakdown of the blood–brain barrier because of either chronic infarction or a capillary leak phenomenon secondary to venous hypertension) with serpentine enhancing veins on the cord surface.
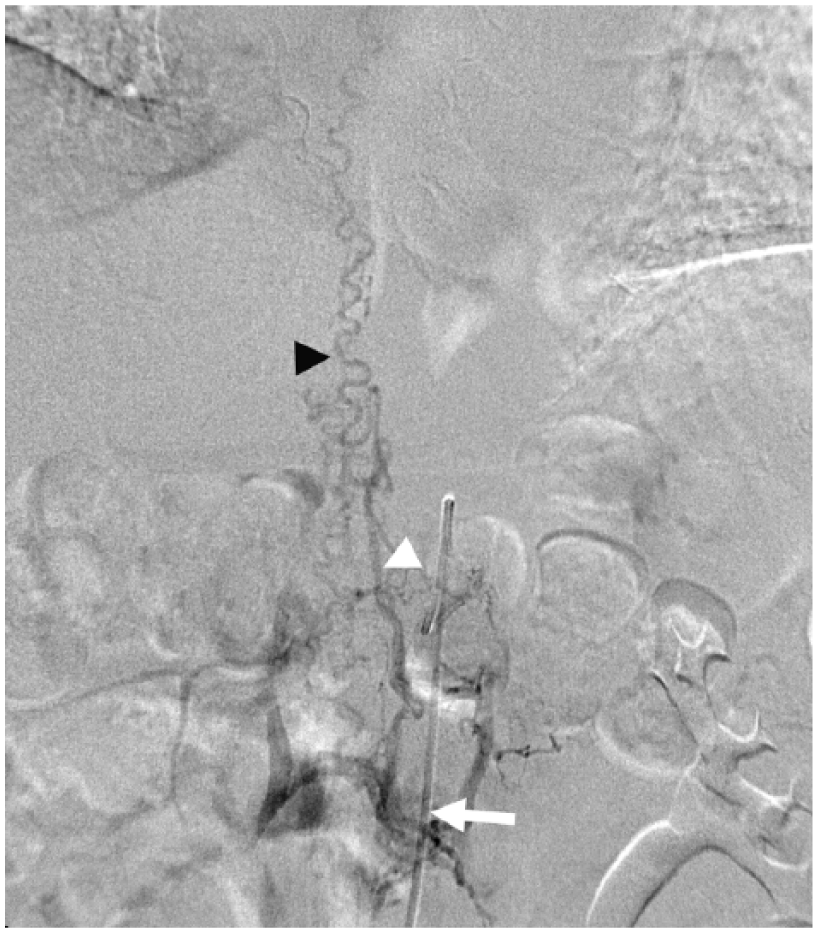
Angiography revealed T12 SDAVF. Afferent artery with catheter in situ (arrow), dural arteriovenous fistula (white arrowhead), and perimedullary serpentine drainage vein (black arrowhead).
Discussion
SDAVF was first described by Foix and Alajouanine in 1926 and efficient methods for its diagnosis and treatment were developed by 1974. 3 The pathophysiology of SDAVF indicates that the formation of a fistula between the radicular artery and the corresponding radicular vein within the dural root sleeve leads to congestion of the venous outflow of the spinal cord and, eventually, ischemia. 4 The resulting gray matter necrosis causes clinical features of progressive myelopathy. It had been reported that about 4% of SDAVF patients combined with vertebral trauma, which was once considered a cause of SDAVF, but trauma does not seem to play a major factor currently. 2 SDAVF is recognized as an unknown etiology of acquired disease at the present time.
The most common symptoms observed in the early stage are sensory disturbance, gait difficulties, motor disturbance, back pain, and radicular pain. Difficulty in micturition, defecation problems, and sexual dysfunction occur in the later stage. Because the initial symptoms are variable and nonspecific, early diagnosis of SDAVF is difficult. In 2003, Jellema et al.5,6 examined 80 cases of SDAVF and reported that the median interval between the onset of symptoms and date of diagnosis was 15 months, ranging from 7 days to 197 months. Approximately 60% of the patients received an accurate diagnosis after more than 1 year and, thus, were wheelchair dependent for entire life. In addition, the symptoms of SDAVF usually develop progressively and cannot be easily differentiated from sensory polyneuropathy, acute or chronic inflammation, demyelinating polyneuropathy, spinal muscular atrophy, and intramedullary tumors. Although the clinical manifestations of SDAVF and polyneuropathy are quite similar, there is still some scope for differentiation. First, paresthesia in polyneuropathy usually involves four extremities, whereas that in SDAVF involves a few upper extremities, except for C-spine localization. Second, in polyneuropathy, paresthesia is manifested as peripheral stocking-and-glove-like sensory loss. However, in SDAVF, paresthesia is manifested as a burning sensation extending proximally to the buttock and perineal region, and it is bilaterally less symmetric compared with that in polyneuropathy. Third, micturition difficulties, such as acute urine retention, are more common in patients with SDAVF, at approximately 80%.
MRI and angiography are valuable techniques for diagnosis of SDAVF. A typical image finding of “flow void phenomenon” indicates a tortuous and dilated medullary draining vein on the dorsal surface of the spinal cord, which was visible in 35%–91% patients.7,8 Other MRI findings include intramedullary edema with cord central hyperintensity; however, differentiating these findings from those of spinal artery infarction myelitis or spinal cord neoplasm is difficult. With the advancement of remodeling images, gadolinium-enhanced magnetic resonance angiography and 64-cut computerized tomography has been used for localization. 9 However, spinal angiography remains the gold standard in the diagnosis of SDAVF. 2 Our patient identically presented typical MRI features, including a slight hyperintense area in the conus medullaris, flow voids with subtle coronal venous plexus enhancement, and tortuous dilated perimedullary vessels. Angiography also confirmed the diagnosis of T12 SDAVF with feeding artery from left intercostal artery and tortuous dilated venous plexus. Failure to recognize the typical MRI findings may lead to misdiagnosis of other spinal cord diseases, such as intramedullary tumors, demyelination, inflammatory myelitis, and traumatic spinal cord edema, eventually resulting in poor outcomes. 10 On the other hand, some studies have suggested that myelography could be performed in patients who have incomplete angiography findings because of atherosclerosis of the lumbar arteries. 11
Current therapeutic interventions for SDAVF include endovascular embolization and surgical ligation of the fistula. The success rates of endovascular therapy, varying between 25% and 75%, have been reported in the literature; nevertheless, complete occlusion of the fistula following surgery is 98% in a recent meta-analysis.12,13 In addition to a higher recurrence rate postembolization, the two main contraindications of embolization were fistulas with segmental medullary artery feeding and dissection of feeding vessels. 14 With further understanding of this disease, use of decompression of multilevel laminectomy was discontinued and the most efficient surgical intervention was total removal of the draining vein. 15 After embolization or surgical intervention, gait disturbance and muscle power improved by approximately 64% and 56%, respectively. 6 However, compared with gait disturbance, micturition, pain, and muscle spasms did not often respond well to treatment. 16 These reported outcomes are consistent with those observed in our patient. Information about risk factors for SDAVF is not yet available; however, most researchers believe that the fewer the symptoms before treatment, the better are the outcomes. A study reported that a fistula at T9 and T12 had more favorable prognosis, indicating that the accurate diagnosis and treatment of a fistula in the lower thoracic region are easier. 17 Similarly, the outcome in our patient with T12 SDAVF was satisfactory with few sequelae. However, if symptoms persist after therapeutic intervention, angiography must be performed again to examine for possible recurrence of the disease. Our patient has remained symptom-free for more than 3 years until now, without evidence of recurrence. The possible mechanism underlying the recurrence of SDAVF may be closure of the first fistula, causing hypertensive changes in the venous drainage of the spinal cord and resulting in the formation of another fistula.
Conclusion
In conclusion, SDAVF should be suspected in any patient who presents with myelopathy. MRI provided the typical image presentations such as spinal cord edema with tortuous dilated perimedullary venous plexus. Angiography remains the gold standard for confirmation of diagnosis. Prompt treatment with endovascular embolization or surgery could improve patients’ outcomes.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of data and materials
All data generated during this study are included in this published article.
Blinding
The response letter is properly blinded of the authors’ identity.
Informed consent
We asked the patient to help us to publish the case report in an international journal for discussion, including disease symptoms, diagnosis, and image related content. The patient agreed us to use his medical records and signed the consent form.
Ethical approval
This article is a case report that does not include human experiment and any privacy information about patient. Based on the Institute Review Board policy and regulars, the Institute Review Board did not need to oversee manuscripts of a case report. The identity of our patient has been removed and deleted carefully in this article to ensure the patient’s privacy and rights.
Human rights
The informed consent was signed before the publication of this article, at the same time we promise the patient’s request to do everything possible to protect privacy and avoid the interference of the patient’s life.
