Abstract
Introduction:
Although liver support systems play a major role as a bridge to transplantation, they may sometimes serve as a lifesaving treatment eliminating this need.
Case presentation:
We report on a 4-year-old boy who developed acute liver failure due to an influenza A (H3N2) infection as confirmed by clinical and laboratory data (molecular typing, stage 3 encephalopathy, brain edema, increased levels of ammonia, bilirubin, and international normalized ratio 5.2). Testing for any possible underlying liver disease showed no congenital or acquired liver pathology. Oral oseltamivir treatment was initiated and liver support therapy with plasma exchange was performed as a bridge to transplantation. A total of three plasma exchange sessions every other day, with fresh frozen plasma 1.5 times the total blood volume for the first course and 1 times for the subsequent courses, were performed. After the first plasma exchange, encephalopathy improved to stage 2, accompanied by substantial decreases in the elevated liver function tests. At the end of three plasma exchange sessions, the patient’s clinical condition improved significantly. At 14 days after admission, deep tendon reflexes of the patient were normal and levels of alanine transaminase, aspartate transaminase, bilirubin, ammonia, and international normalized ratio returned to normal. Liver support treatment with plasma exchange resulted in complete recovery and the patient was discharged on the 17th day of admission.
Conclusion:
Acute supportive treatment with plasma exchange proved to be life-saving in our case.
Keywords
Introduction
Influenza viruses are single-stranded RNA viruses belonging to the family Orthomyxoviridae, with types A, B, and C. Epidemic outbreaks are caused by types A and B, for which several types of vaccines have been developed. Influenza A viruses are also subdivided according to the surface antigens having hemagglutinin (HA) or neuraminidase (NA) activity, such as H1N1 and H3N2. 1 Influenza causes outbreaks every year during the winter months in the northern and southern hemispheres. Influenza often presents as an acute, self-limited, and uncomplicated disease characterized by symptoms of fever, headache, myalgia, cough, rhinitis, and sore throat; however, it may lead to serious complications, even to death, especially in children below 5 years of age. 2 We report on a 4-year-old boy who developed acute liver failure (ALF) during the influenza (H3N2) outbreak in the winter of 2016–2017 in our country and was successfully treated with plasma exchange (PE) in the pediatric intensive care unit.
Case
A 4-year-old boy was brought to the pediatric emergency unit with vomiting, restlessness, and, within the past 2 days, drowsiness, and behavioral changes on January 2017. His core temperature was 37.8°C. He had not presented to any hospital during the period of past 4 days. On examination, his weight was 16 kg (10–25 percentiles), height was 102 cm (10–25 percentiles), respiratory rate was 24/min, heart rate was 65 beats/min, and blood pressure was 140/90 mm Hg. His general status was poor with stupor, isochoric pupils, the pupillary light reflex of +/+, the Glasgow Coma Score of 8 (E2, M4, V2), bilaterally increased deep tendon reflexes and clonus, the Babinski sign of +/+, and stage 3 hepatic encephalopathy. The patient was admitted to the pediatric intensive care unit. He had no previous history of drug use. He had not been vaccinated for seasonal influenza. At presentation, laboratory findings were as follows: leukocytes 9000/mm3 with a differential cell count of 25% neutrophils, 65% lymphocytes, 10% monocytes, hemoglobin 11.7 g/dL, thrombocytes 234,000/mm3, aspartate transaminase (AST) 8775 U/L, alanine transaminase (ALT) 7114 U/L, gamma-glutamyltransferase (GGT) 186 U/L, alkaline phosphatase (ALP) 324 U/L, ammonia 480 μmol/L, total/direct bilirubin 4.5/2.8 mg/dL, prothrombin time (PT) 50.5 s, partial thromboplastin time (PTT) 42 s, international normalized ratio (INR) 5.2, fibrinogen 120 mg/dL, C-reactive protein (CRP) 22.6 mg/dL, erythrocyte sedimentation rate 42 mm/h, procalcitonin 1.5 mg/dL, blood urea nitrogen (BUN) 30 mg/dL, and creatinine 0.8 mg/dL. Model for End-Stage Liver Disease (MELD) score was 32. In the blood gas pH: 7.45, pCO2: 32 mm Hg, pO2: 120 mm Hg, HCO3: 23.9 meq/L. Chest X-ray (CXR) findings were normal. On the basis of the findings remarkable for ALF, intracranial hypertension syndrome, and stage 3 encephalopathy, the patient was immediately intubated and initiated with intravenous cefotaxime 200 mg/kg/day, vitamin K, intravenous
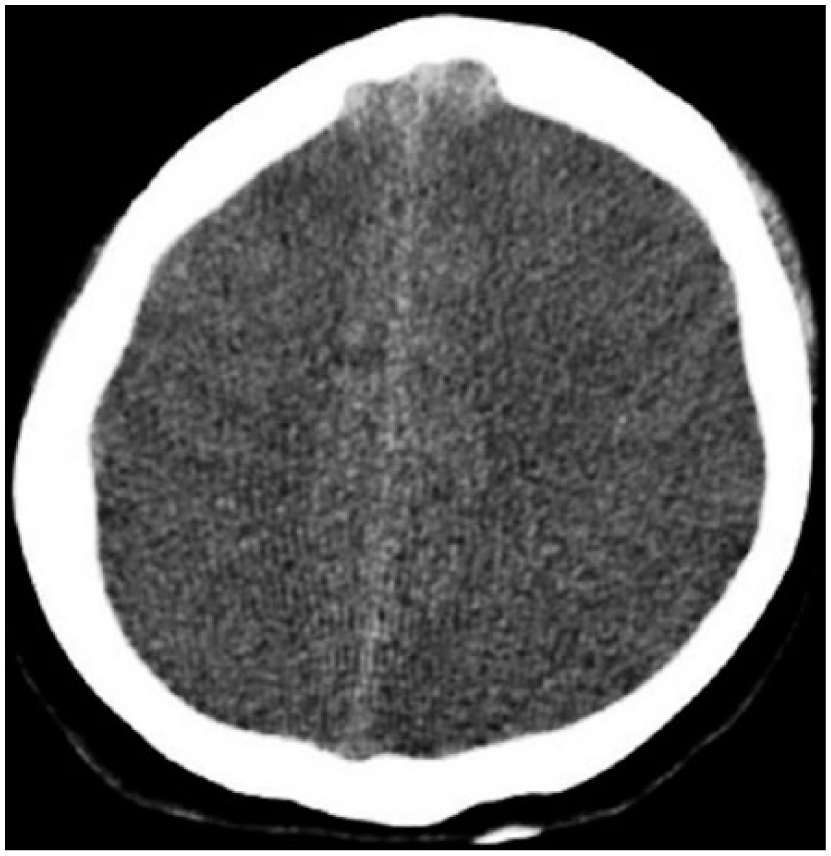
Cranial computed tomography revealed findings of brain edema.
Laboratory investigation into the presence of any underlying cause of liver disease in the patient.

HAV: hepatitis A virus; HCV: hepatitis C virus; EBV: Epstein-Barr virus viral capsid antigen; IgM: immunoglobulin M.
Taking into consideration stage 3 encephalopathy and increased levels of ammonia, bilirubin, and INR, liver support therapy with PE was planned as a bridge to transplantation. PE treatment was started at the fourth day of admission to intensive care unit. A total of three PE sessions every other day, with fresh frozen plasma 1.5 times the total blood volume for the first course and 1 times for the subsequent courses, were performed using the continuous filtration technique on a Prisma Machine (Gambro, Sweden). Each PE treatment lasted for 4 h and no complication was encountered. After the first PE, encephalopathy improved to stage 2, accompanied by substantial decreases in the elevated liver function tests. At the end of three PE sessions, the patient’s clinical condition improved significantly (Table 2). Control cranial computed tomography taken in this period revealed a decrease in cerebral edema (Figure 2). At 14 days after admission, deep tendon reflexes of the patient were normoactive with no abnormal reflexes, and minimal elevation of levels of ALT, AST, bilirubin, ammonia, and INR returned to normal. Despite the initial indication for liver transplantation, PE resulted in complete recovery and the patient was discharged on the 17th day of admission.
Clinical and laboratory data before and after PE a .
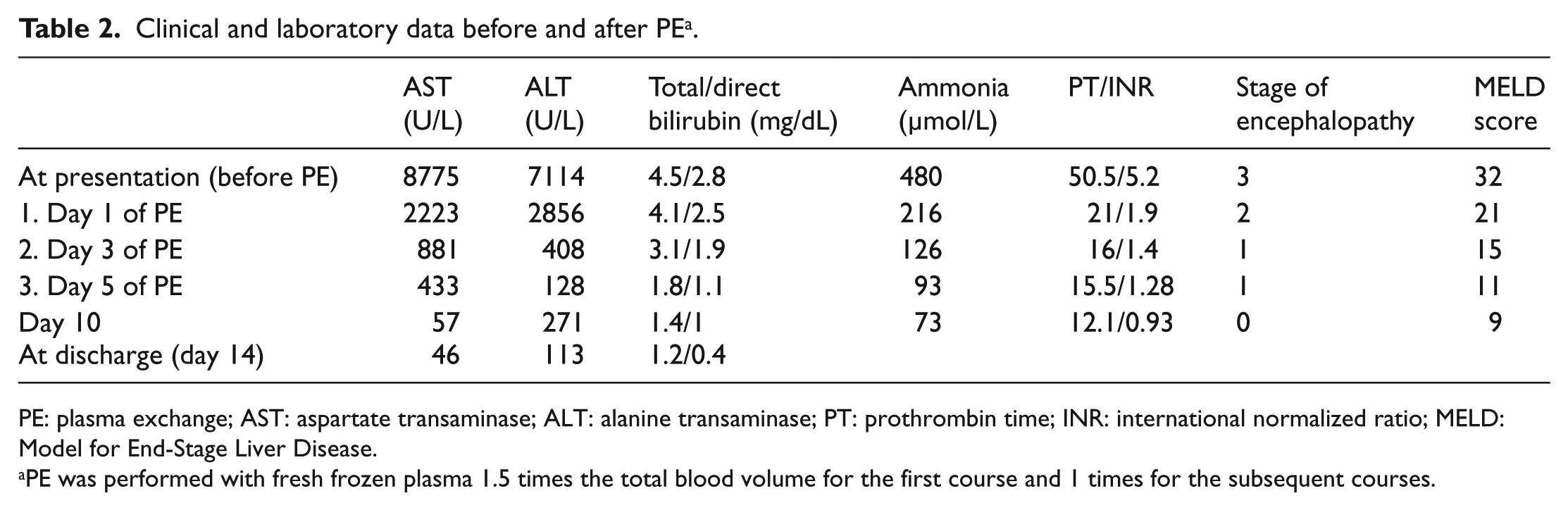
PE: plasma exchange; AST: aspartate transaminase; ALT: alanine transaminase; PT: prothrombin time; INR: international normalized ratio; MELD: Model for End-Stage Liver Disease.
PE was performed with fresh frozen plasma 1.5 times the total blood volume for the first course and 1 times for the subsequent courses.
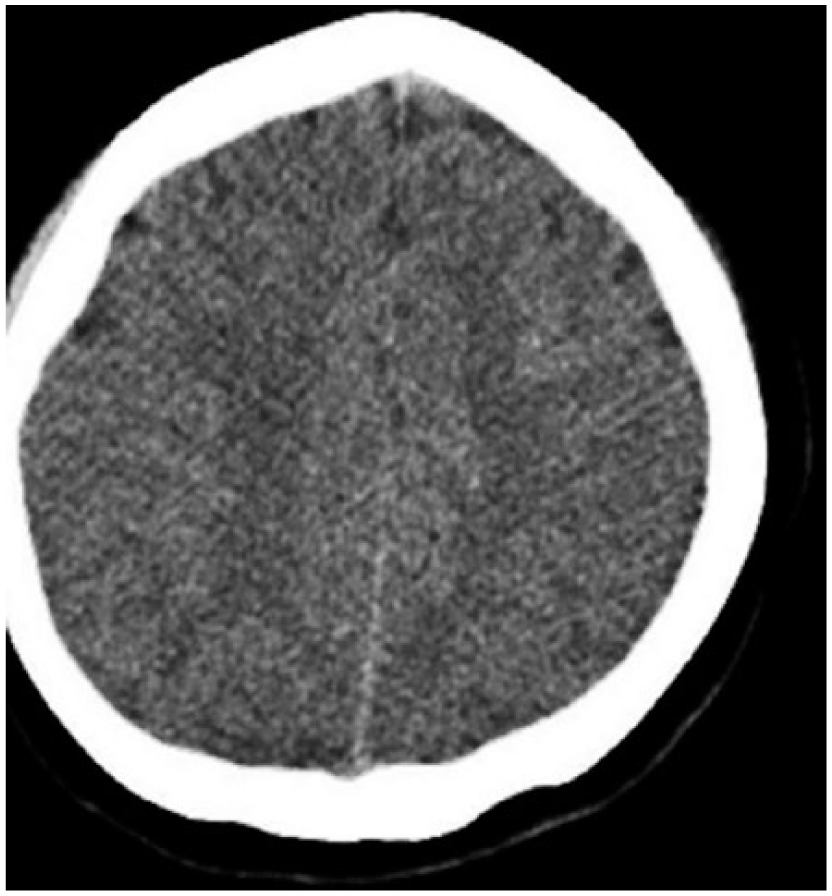
Control cranial computed tomography, decrease in cerebral edema.
Discussion
Extrapulmonary involvement is extremely rare for influenza viruses, which may present as severe gastrointestinal complications, such as pancreatitis, hemorrhagic gastritis, and Reye’s syndrome, and influenza-associated myocarditis and pericarditis.3–5
Influenza A H3N2 is associated with increased mortality in pediatric patients with underlying diseases, age less than 5 years (especially <6 months), and coinfections with Staphylococcus aureus or Streptococcus pneumonia. 6 Even though underlying diseases have been implicated as a cause of increased mortality, 40% of influenza-related deaths in children have been shown to have no association with such conditions. 7
Involvement of the liver tissue by influenza viruses is quite uncommon in humans, accounting for less than 3% of all cases. 8 However, hepatic involvement has been demonstrated in animal models, including induction of hepatitis in a murine model.8,9 A hepatotropic variant of influenza A was obtained in a mouse model, which in turn gave rise to necrotizing hepatitis. 10
Hepatic decompensation can be seen in adults with underlying liver diseases. Three adults with cirrhosis were reported to develop hepatic failure during infection with influenza A. 11 An adult hemodialysis patient also developed hepatic failure due to influenza A (H3N2). 12
Studies on children are limited to case reports, making it difficult to draw clear conclusions. Moreover, all reported cases were identified during influenza outbreaks. During the influenza epidemic in Colorado in 2003–2004, four cases were reported to have acute hepatitis and hepatic failure predominantly due to the type H3N2.13,14 Likewise, the present case was also identified during a seasonal emergence of the influenza virus and was documented to be H3N2. Screening for possible underlying liver diseases in our case yielded no other explanation for hepatic involvement (Table 2).
During the 2010 influenza A (H1N1) pandemic in Kuwait, a 9-year-old boy was infected with H1N1 and developed ALF, for which no underlying hepatic cause was present as well as history of drug use. 15 As the severity of the condition was not so serious, he received fresh frozen plasma and other supportive treatments without plasmapheresis, which sufficed for clinical and laboratory resolution. 15 In a retrospective analysis, Cantez et al. examined seven pediatric cases who had undergone liver transplantation with a positive polymerase chain reaction (PCR) test for the 2009 influenza A (H1N1). The authors reported that all seven cases survived the transplant surgery and the infection without sequela and attributed this favorable outcome to oseltamivir treatment. 16
The stage of encephalopathy, bilirubin levels, and INR are timely evaluated for monitoring of the condition. An INR above 4 is considered to justify an urgent organ request. 17 In our case, a combination of the admission PT >50 s, INR 5.2, stage 3 encephalopathy, and brain edema prompted a notification to the transplant center about liver transplantation, during which PE therapy along with other supportive treatments was continued as a bridge to transplantation. In PE or plasmapheresis, the patient’s plasma is filtered to be replaced by fresh frozen plasma or albumin in order to remove various toxins, including protein-bound toxins, as well as to improve coagulopathy. 18
Despite our meticulous inquiry into our case as well as into reported cases in the literature, we could not elucidate the exact mechanism by which influenza A (H3N2) causes acute organ failure such as liver failure. Nonetheless, acute supportive treatment with PE proved to be life-saving and obviated the need for transplantation in our case.
The NA inhibitors are the drugs of choice for treatment. The Centers for Disease Control and Prevention (CDC) recommend that either oseltamivir (Tamiflu®) or inhaled zanamivir (Relenza®) should be given to all hospitalized patients with confirmed, probable, or suspected influenza A infection. A higher dose of oseltamivir can be considered in critically ill influenza A patients. The recommended duration of treatment is 5 days. Hospitalized patients with severe infections (such as those with prolonged infection or who require intensive care unit admission) might require longer treatment courses. Zanamivir formulated for oral inhalation is approved by Food and Drug Administration (FDA) for the treatment of influenza in patients 7 years of age and older who, similar to approved uses for oseltamivir, have uncomplicated illness and have been symptomatic for no more than 2 days. 19 Our patient was administered oral oseltamivir at 45 mg twice a day for 10 days. His viral infection was poorly responsive to the antiviral therapy and his general medical condition gradually deteriorated. Veno-venous extracorporeal membrane oxygenation (ECMO) is an effective way to support patients with refractory hypoxaemia who fail to respond to conventional ventilatory strategies. 20
Vaccination with seasonal influenza vaccine is the most effective way of avoiding influenza and its complications. The recommendation of Advisory Committee on Immunization Practices (ACIP) that everyone ≥6 months old should be vaccinated still maintains its validity.
Footnotes
Acknowledgements
The authors thank the patient’s parent for allowing us to share details of his case. They also thank the doctors and nurses involved in the management of the patient in the intensive care unit.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of data and materials
Data sharing is not applicable to this article as no data sets were generated or analyzed during this study.
Informed consent
Informed consent was obtained from the patient’s parent.
Human rights
Authors state that they are committed to human rights.
