Abstract
Fat embolism syndrome is a life-threatening condition that may develop after orthopedic trauma and surgery. Fat embolism is caused by lipid droplets in the systemic circulation inducing mechanical or biochemical injury. Diagnosis is clinical and treatment is supportive. Clinical features including respiratory changes, neurological abnormalities, and petechial rashes generally present 12–72 h after the initial injury. However, respiratory failure accompanying diffuse alveolar hemorrhage in fat embolism syndrome is rare. This report describes a 44-year-old male patient suffering from fat embolism syndrome with diffuse alveolar hemorrhage caused by traumatic long bone fracture, in which mechanical ventilation was required to provide respiratory support.
Introduction
Fat embolism syndrome (FES) is usually associated with surgery for large bone fractures, which occur in 0.5–3.5% of cases. 1 Although FES remains relatively rare, it is a severe complication with an overall mortality rate of 5–15%. 2 The pathophysiology of FES has yet to be fully understood, but generally end-organ pathophysiology is thought to be mediated by mechanical obstruction and biochemical injury. 3 The diagnosis of FES is made clinically, and the main clinical features including respiratory changes, neurological disturbances, and petechial rash generally present 12–72 h after traumatic injury. 4 Currently, there are no disease-specific treatments for FES. Therefore, early diagnosis and adequate symptomatic treatment including mechanical ventilation or extracorporeal membrane oxygenation in special situations is very important.5–7 The occurrence of intra-alveolar hemorrhages in fat embolisms is rare. 8 In this report, we describe a patient with FES complicated by diffuse alveolar hemorrhage and was treated by mechanical ventilation.
Case report
A previously healthy man in his forties was transferred to the emergency center after injury in a pedestrian traffic accident 1 day before. He had fractures of the left femur, tibia, and fibula and was admitted to a local orthopedic hospital for surgery (Figure 1). Prior to admission to the emergency center, the patient developed respiratory distress of sudden onset with hemoptysis and altered consciousness. Brain magnetic resonance imaging (MRI) performed at the emergency center showed multiple tiny diffusion restrictions in both frontal lobes, both parietal lobes, and the left thalamus indicating acute embolic infarction (Figure 2). The patient remained conscious without definite neurological symptoms such as seizure. Spiral chest computed tomography (CT) and plain chest radiographic studies showed bilateral, pre-dominantly peripheral, asymmetrical consolidation with small amounts of bilateral pleural effusion consistent with acute respiratory distress syndrome, but no evidence of pulmonary thromboembolism. We performed emergent bedside bronchoscopy in order to distinguish the causes of hemoptysis, and bronchoscopic findings overall indicated hemorrhage from the distal trachea to both bronchi with no evidence of damage, infection, or mucous membrane injuries (Figure 3). After transfer to the intensive care unit (ICU), the patient was alert and his oxygen saturation according to pulse oximetry was maintained at 88%–93% by high-flow nasal cannula with 80% oxygen and 40 L/min flow. We promptly decided to apply mechanical ventilation for the worry of the deterioration of pulmonary hemorrhage.
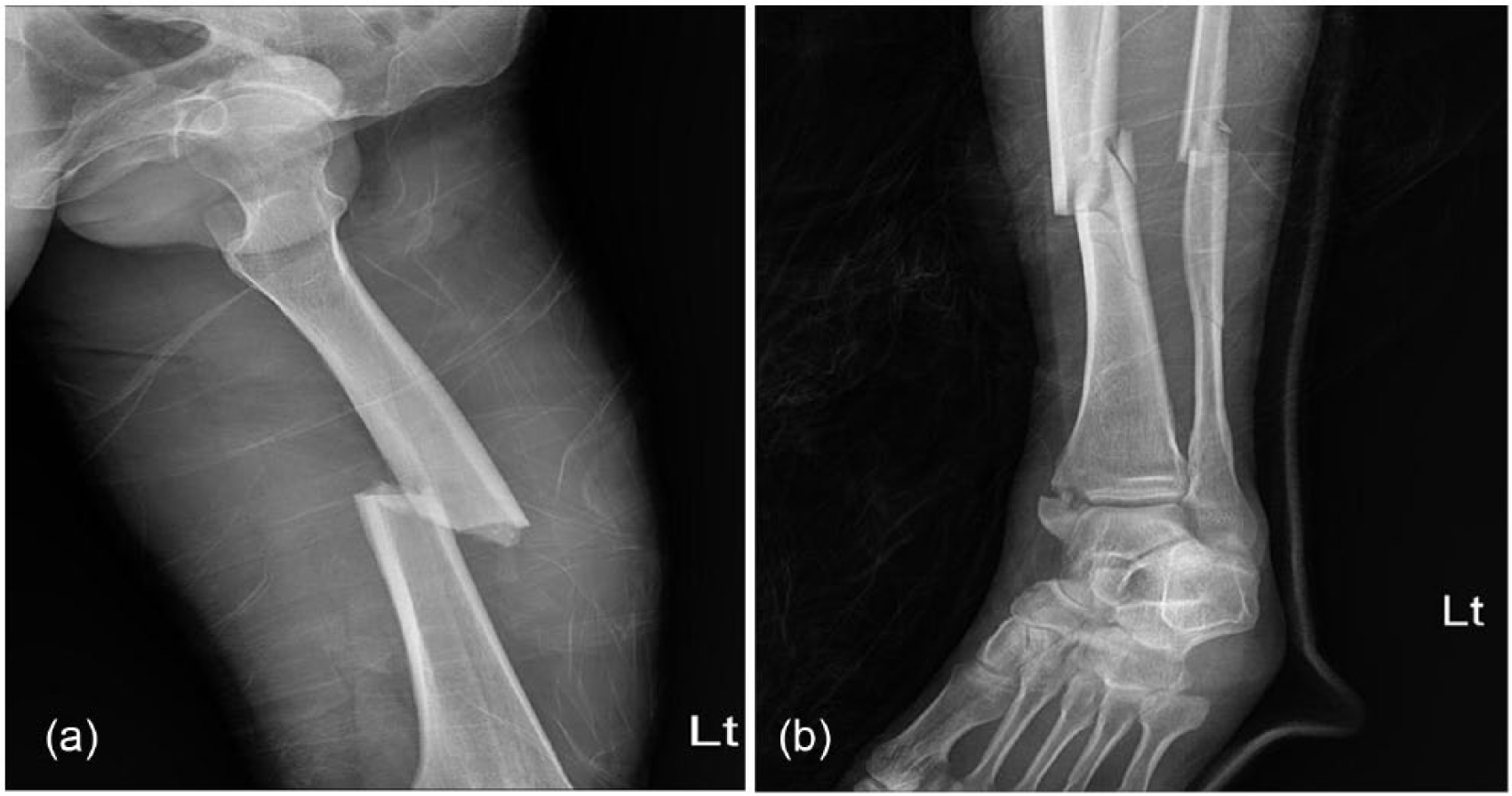
Initial imaging. (a) X-ray of the left lower extremity shows a closed, oblique fracture of the left femur mid-shaft. (b) X-ray of the left lower extremity shows an open fracture of the tibio-fibular mid-shaft and fracture of medial malleolus ankle, left.
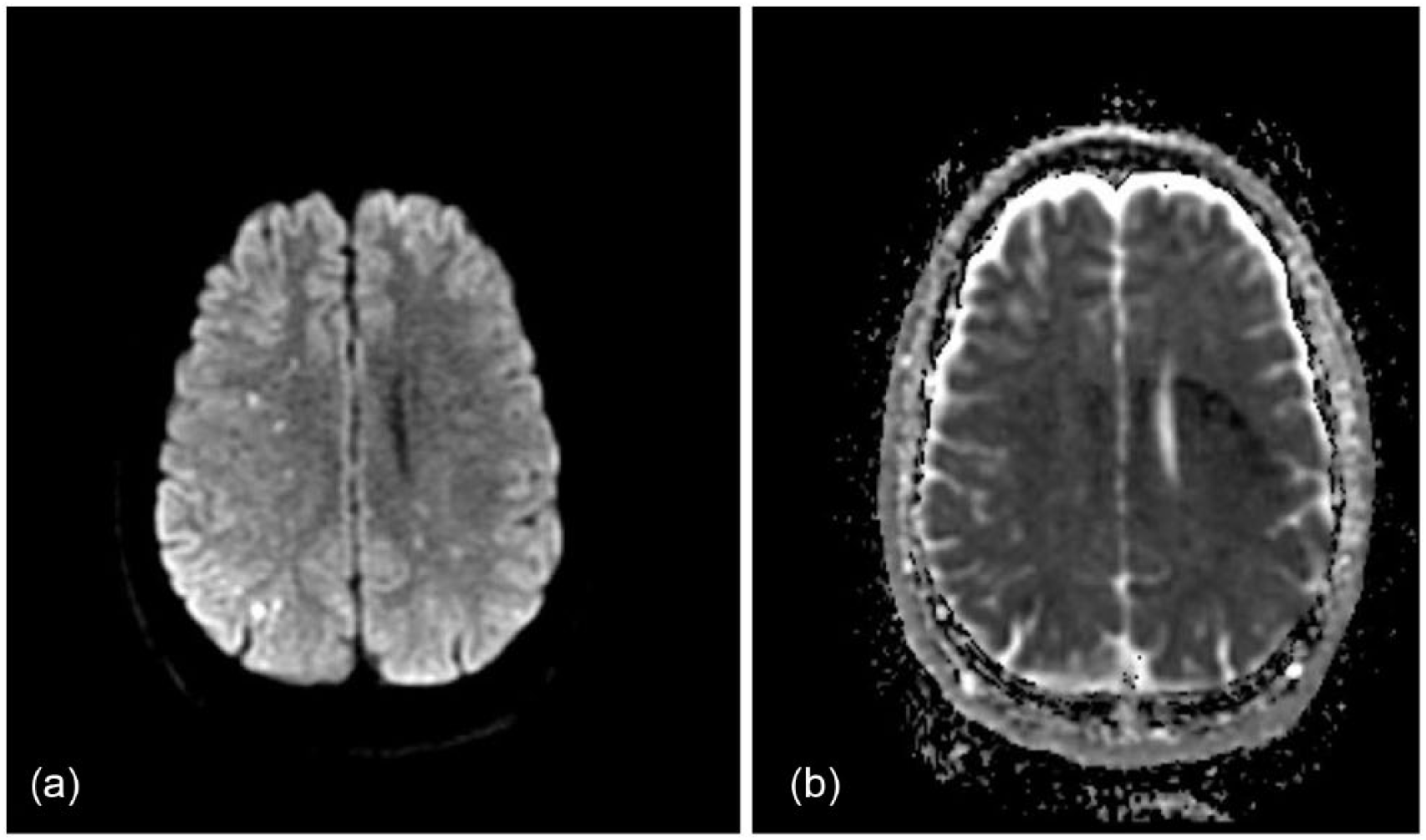
Diffusion MRI of the brain shows multiple tiny diffusion restriction in frontal lobes, parietal lobes, the occipital lobe, and the left thalamus: (a) diffusion-weighted imaging and (b) apparent diffusion coefficient.
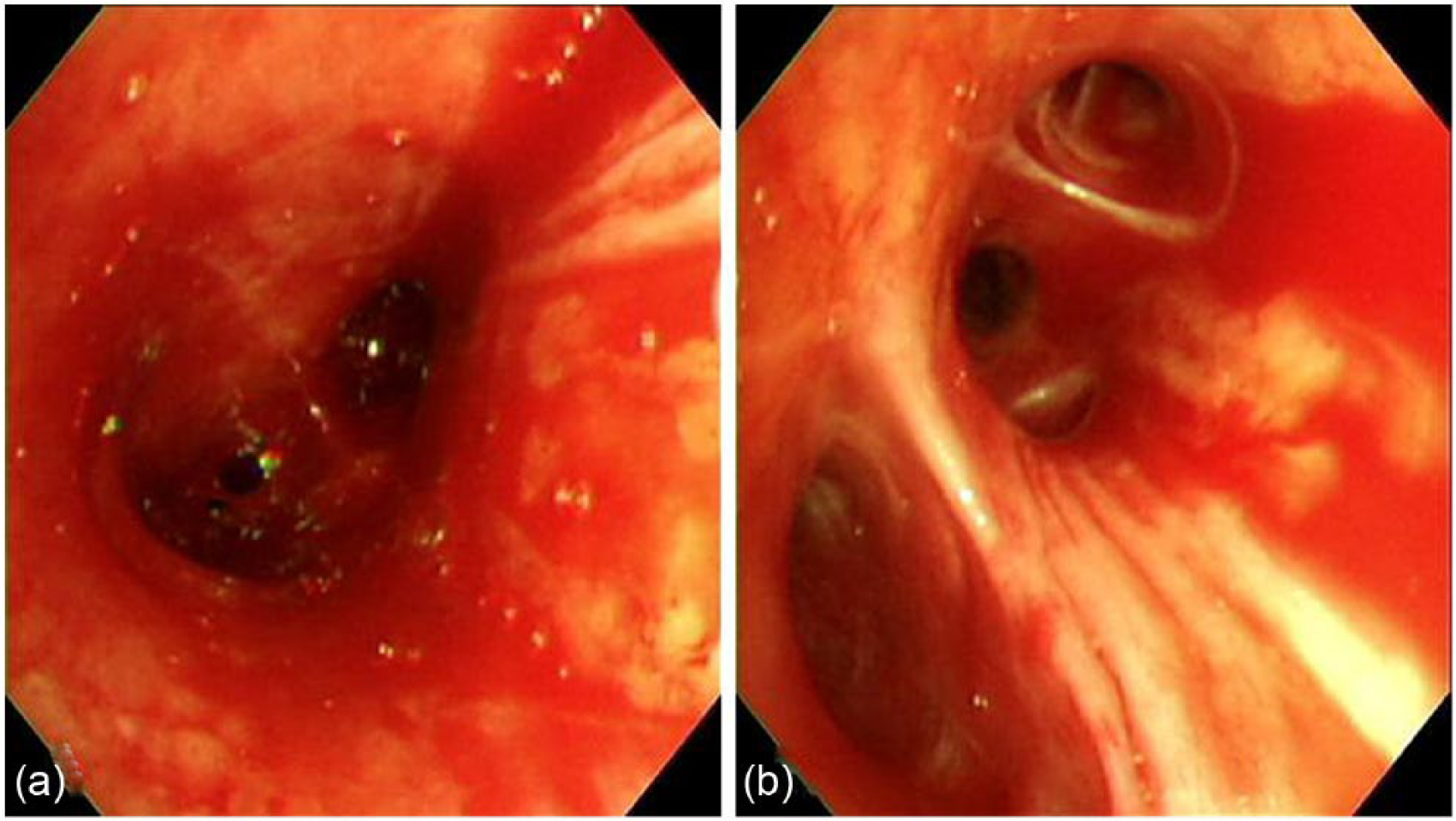
Bedside bronchoscopy on ICU day 1 shows (a) large amounts of diffuse and fresh bleeding at the carina and (b) a moderate amount of diffuse fresh bleeding at the right intermedius bronchus.
On the third ICU day, bloody secretion via endotracheal suction decreased after supportive care. Infiltration shown on chest radiographs also improved. Demands for oxygen and ventilator support decreased and after withholding sedatives, the patient regained consciousness. On the fifth ICU day, follow-up bronchoscopy showed nearly normalized endobronchial findings. On the seventh ICU day, open reduction and internal fixation of the femur and closed reduction of fractures and internal fixation of the tibia and fibula were performed. Surgery was successful and there were no complications such as bleeding or hypoxemia. On the eighth ICU day, oxygen saturation was maintained at 95% on mechanical ventilation with the following settings: positive end-expiratory pressure (PEEP) 5 mmHg, FiO2 30% of pressure support mode. The patient’s vital signs were stable. On the ninth ICU day, the patient was transferred to the general ward after mechanical ventilator weaning and extubation. He fully recovered without neurologic deficits.
Discussion
Fat embolism may occur after long bone fractures. It is caused by fat particles entering the systemic circulation. In most patients, fat embolism is benign and has few clinical consequences. But a small number of patients progress to FES. The major clinical manifestations of FES include respiratory insufficiency, neurologic symptoms, and a petechial skin rash. Pulmonary symptoms including dyspnea and tachypnea are the most common manifestations, and in severe cases, progression to respiratory failure and acute respiratory distress syndrome may occur. However, hemoptysis by diffuse alveolar hemorrhage is rarely reported. FES is diagnosed on the basis of clinical features and the exclusion of other diseases. Schonfeld et al. 9 and Gurd 10 proposed a classification system to aid the diagnosis of FES, but the pathogenesis of FES has yet to be clearly elucidated. Mechanical obstruction and biochemical injuries are hypothesized to be the two main processes of end-organ pathology. In the mechanical obstruction phase, the fat emboli in the bloodstream reach pulmonary capillaries and other distal organs causing mechanical obstructions in areas such as the central nervous system. Embolized fat is degraded in plasma to free fatty acids in the biochemical phase. Free fatty acids cause lung injuries and cardiac contractile dysfunction. Because FES is associated with high mortality rates of 5%–15%, early diagnosis and adequate treatment are essential to prevent severe complications. There is no specific treatment for FES. If necessary, supportive measures such as oxygen therapy, fluid therapy, vasopressor use, and mechanical ventilation are important in conjunction with appropriate hemodynamic status monitoring in the ICU. 11 In the present case, the patient’s oxygenation was maintained after use of high-flow nasal cannulas, so it is not necessary to apply early mechanical ventilation. Because we suspected hemoptysis, we immediately performed bronchoscopy and diagnosed the patient of diffuse alveolar hemorrhage combined with FES, which is very rare. Therefore, by applying aggressive and early mechanical ventilation, clinicians may prevent complications that may arise in connection with unexpected intubation and hypoxemia. We treated a patient with FES successfully without adverse effects.
Footnotes
Informed consent
Every effort was made to obtain informed consent from the patient with no success so the case report has therefore been anonymised as far as possible.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
