Abstract
Purpose
Posterior medial meniscus root tears often cause persistent extrusion and altered joint mechanics despite repair. This study compared standard transtibial pull-out repair with repair plus a second-tunnel centralization, hypothesizing better patient-reported outcomes without major MRI extrusion changes.
Methods
This retrospective two-center cohort (2019–2024) included adults with MRI-confirmed root tears treated with anatomic repair alone or with an additional centralization tunnel. Propensity matching yielded 96 patients (54 vs 42). The primary endpoint was 24-months change in IKDC score. Secondary outcomes included KOOS subscales, Lysholm, visual analogue scale pain, and Tegner activity. Structural outcomes—medial meniscus extrusion and Meniscal Extrusion Index—were assessed on coronal MRI at 12 ± 4 months. Analyses used ANCOVA adjusted for baseline values, supported by inverse-probability weighting and mixed-effects checks.
Results
Baseline characteristics were balanced. Extrusion changes were small and similar; residual pathologic extrusion rates were comparable (65% vs 62%). Centralization showed greater IKDC improvement (+5.7 points; p = 0.008) and higher KOOS-Quality of Life. Knees with ≥3 varus demonstrated additional benefit (interaction p = 0.048). Complications were infrequent.
Conclusions
Second-tunnel centralization significantly increased the probability of achieving
Keywords
Introduction
Posterior meniscal root tears (PMRTs), which most commonly involve the posterior horn root of the medial meniscus, disrupt the circumferential (“hoop”) fibers that convert axial loads into tangential stresses, thereby approximating the contact mechanics of subtotal meniscectomy. 1 In cadaveric models, a medial root tear increases the peak medial compartment contact pressure to levels comparable to those after total meniscectomy, whereas anatomic repair restores the pressure to normal levels. 2 Clinically, untreated or debrided (meniscectomized) PMRTs are associated with pain, rapid joint space narrowing, and high rates of radiographic osteoarthritis (OA) progression and conversion to total knee arthroplasty (TKA), in contrast to repair.3,4 Contemporary meta-analyses and matched cohorts consistently show that repair yields superior joint preservation compared to meniscectomy or nonoperative care at mid-to long-term follow-up.5,6
Although the lateral root can fail in the setting of ligamentous injury, the prototypical medial PMRT arises in middle age, frequently in women with degenerative cartilage changes and coronal malalignment. 7 Risk factors repeatedly linked to medial root pathology, poorer healing biology, or worse mechanics include varus alignment and elevated body mass index (BMI). 8 These demographic and morphological features help explain the characteristic association between PMRTs and meniscal extrusion, which is the radial displacement of the meniscus beyond the tibial margin that diminishes the meniscus’ load-sharing function and correlates with pain and OA progression.9,10
Anatomic root fixation (most commonly a transtibial “pullout” repair or, less commonly, suture anchor fixation to the tibial footprint) is the current standard of care for a repairable PMRT in an otherwise salvageable knee. 11 Repair reliably improves symptoms and function and reduces downstream OA/TKA risk compared with debridement or observation.3,12 Nevertheless, persistent or even progressive medial meniscal extrusion after technically successful root repair is common and is increasingly recognized as a plausible mechanism for ongoing cartilage overload and clinical failure in some patients.13,14 The situation is exacerbated by varus alignment and insufficiency of the posteromedial peripheral stabilizers (meniscotibial and meniscocapsular attachments), which increase the forces transmitted across the root fixation under physiologic loading.15,16
To address extrusion—and to unload the repaired root—surgeons have adopted “meniscal centralization,” a family of techniques designed to tether the peripheral or midbody meniscus back to the tibial rim. Centralization can be performed with knotless suture anchors along the capsulomeniscal junction or by creating a
Early clinical series of
This article therefore undertakes a focused comparative analysis of these two strategies. First, we synthesize the biomechanical and clinical rationale for anatomic posterior root fixation and for centralization via a second transtibial tunnel with a midbody suture, emphasizing how each targets distinct failure modes (loss of hoop tension vs persistent extrusion). Second, we critically appraise laboratory and clinical evidence bearing on patient-reported outcomes, radiographic extrusion, cartilage status, and joint-preservation endpoints (OA progression, TKA conversion). Finally, we propose a decision framework integrating alignment, cartilage grade, and posteromedial soft-tissue integrity to guide selection between isolated root fixation and second-tunnel centralization—and to identify scenarios in which a combined construct may be preferable. Our a priori hypothesis is that, in repairable PMRTs, root fixation remains essential for hoop restoration, whereas a second-tunnel centralization suture offers incremental benefit primarily by reducing extrusion and unloading the repair in varus or peripheral-attachment insufficiency; by contrast, centralization alone is unlikely to fully replicate the biomechanical functions of anatomic root reattachment.1,15,19
While recent studies, such as
In this study, “posterior root fixation” is defined as the anatomical reattachment of the posterior horn root to its tibial footprint, achieved through either transtibial pullout or anchor techniques. “Centralization (second tunnel)” refers to a transosseous construct involving a separate tibial tunnel near the medial plateau rim, which is utilized to tension a horizontal mattress suture passed through the midbody (meniscocapsular junction) to re-center the meniscus. This approach is distinct from anchor-based centralization, which is referenced for context but analyzed separately. By explicitly contrasting these strategies, this research seeks to elucidate the indications, anticipated benefits, and trade-offs for surgeons managing extruded posterior meniscal root tears (PMRTs)—a domain characterized by rapid technical advancements yet limited comparative evidence.
Methods
Study design and setting
We conducted a retrospective, two-center comparative cohort study across two tertiary referral hospitals. Consecutive patients who underwent arthroscopic management of
Eligibility criteria were prespecified. Inclusion criteria comprised adults aged 18–75 years with MRI-confirmed posterior medial meniscus root tear who underwent arthroscopic treatment with either posterior tunnel fixation or double-tunnel centralization, had a preoperative MRI obtained within 3 months before surgery and a postoperative MRI at 12 ± 4 months, and completed at least 24 months of clinical follow-up including patient-reported outcomes (PROs). Exclusion criteria were the presence of lateral or anterior root tears, discoid meniscus, or prior meniscal transplantation; concomitant realignment osteotomy or cartilage restoration in the medial compartment; radiographic osteoarthritis of Kellgren–Lawrence grade ≥3 or focal Outerbridge grade 4 chondral loss in the medial weight-bearing zone; coronal malalignment exceeding 5 of varus on long-leg alignment radiographs; inflammatory arthropathy; ipsilateral knee arthroplasty or ligament reconstruction within the preceding 12 months; and incomplete imaging or PRO data. These parameters were selected to minimize confounding and to isolate the effects of root fixation and centralization on meniscal extrusion and clinical function, consistent with established associations among extrusion, degenerative change, and malalignment.
All operations used standard anteromedial/anterolateral portals. For posterior tunnel fixation, the tibial root footprint was refreshed, two UHMWPE sutures (simple or cinch) were passed through the root stump, and a single transtibial tunnel was drilled to the anatomic footprint for cortical-button (Artrotek, Adana, Turkey) fixation. For double-tunnel centralization, after completing the root repair, a second transtibial tunnel was created at the medial plateau rim adjacent to the meniscocapsular junction; a horizontal mattress suture was passed through the midbody and tied over a cortical button (Artrotek, Adana, Turkey) to re-centre the meniscus (Figures 1–3). Technique details mirror published centralization descriptions (anchor-based variants exist but were not used in this cohort). Postoperatively, both groups followed the same rehabilitation: protected weightbearing for 4–6 weeks with 0–90 motion, progressive closed-chain strengthening from week 6, return to light jogging by ∼ 12 weeks, and pivoting activities after ∼ 6 months as tolerated. Schematic illustration of the surgical construct. Arthroscopic steps of posterior root footprint preparation and repair. (a) Debridement and refreshment of the posterior medial meniscus root footprint. (b) Passage of the UHMWPE sutures through the posterior root stump. (c) Final arthroscopic view after fixation. Red arrow: completed posterior root repair; green arrow: added second-tunnel centralization suture. Arthroscopic views demonstrating posterior root repair and the second-tunnel centralization technique (images from a different patient than Figure 2). (a) Completed posterior medial meniscus root repair at the anatomic footprint. (b) Arthroscopic view of the second transtibial centralization tunnel, showing passage of the horizontal-mattress suture through the meniscus midbody.


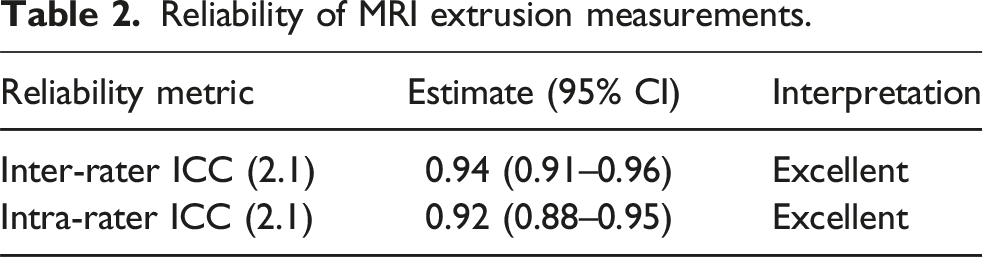
Imaging served as the structural assessment. MRIs (1.5-T or 3-T) included coronal and sagittal proton-density fat-suppressed and T2-weighted sequences (3–4-mm slices; in-plane resolution ≤0.5 mm). The primary structural variable was medial meniscus extrusion (MME, mm) on coronal MRI at the level of the medial collateral ligament (MCL), measured as the perpendicular distance from a tangent to the medial tibial cartilage edge to the outer meniscal margin; values were averaged across three contiguous slices centred on the MCL. MME >3 mm was recorded as pathologic. In addition, we computed a size-normalized Meniscal Extrusion Index (MEI) (extrusion/meniscal width on the same slice) to complement absolute MME, in line with literature supporting either a 3-mm rule or a ratio-based threshold for pathology.9,25,26 Two musculoskeletal radiologists, blinded to treatment and time point, independently measured MME/MEI on pre- and postoperative MRIs (Figures 4 and 5). Inter- and intra-rater reliability were quantified using ICC (2. 1) with 95% CIs and interpreted per Koo & Li (poor <0.50, moderate 0.50–0.75, good 0.75–0.90, excellent >0.90). Discrepancies >0.5 mm were resolved by consensus.
27
MRI measurement of medial meniscus extrusion. (a) Standard MME assessment on coronal imaging at the MCL level (green arrow). (b) Meniscal Extrusion Index (MEI) calculation, demonstrating measurements of meniscal width and radial extrusion. Postoperative MRI demonstrating tunnel positions. (a) Second transtibial centralization tunnel at the medial tibial rim. (b) Posterior root tunnel located at the anatomic footprint. (c) Coronal MRI showing the cortical buttons of the centralization construct (green arrow) and the posterior root repair (orange arrow).
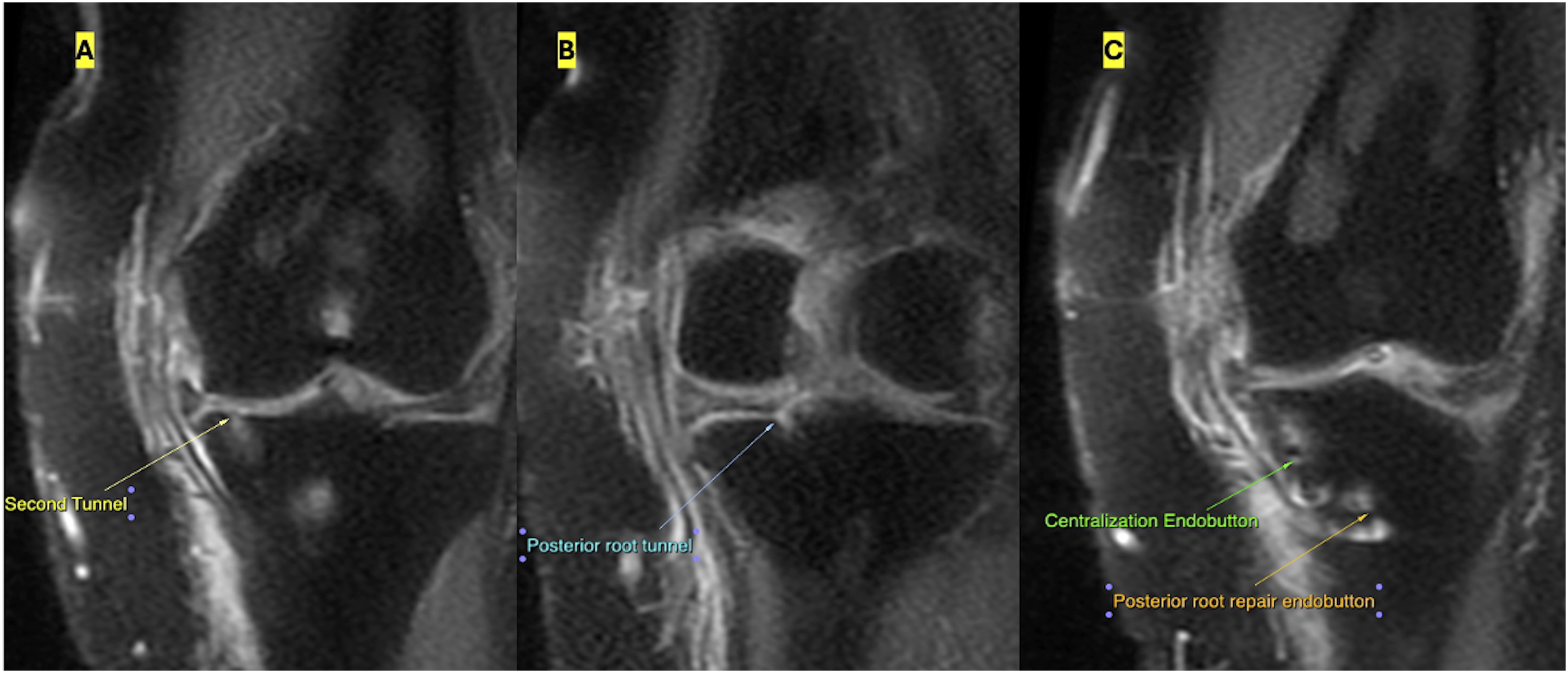
Clinical assessment provided the functional evaluation. The primary clinical endpoint was the change in IKDC (International Knee Documentation Committee) Subjective Knee Form from baseline to 24 months. Secondary PROs included KOOS (Knee Injury and Osteoarthritis Outcome Score) subscales (Pain, ADL, QoL), Lysholm, VAS pain (0–10), and Tegner activity. We classified responders using published MCID (Minimal Clinically Important Difference) and PASS (Patient Acceptable Symptom State) thresholds for meniscal surgery: we prespecified an IKDC MCID around 10–11 points and applied contemporary KOOS MIC (Minimal important change)/PASS (patient acceptable symptom state) references specific to meniscus populations. Radiographic secondary outcomes were preoperative to postoperative changes in MME and MEI, and the presence of residual pathologic extrusion (MME >3 mm) at follow-up. Potential confounders recorded a priori included age, sex, BMI, hip–knee–ankle (HKA) angle (varus degrees), time to surgery, baseline MME/MEI, Outerbridge grade, and qualitative posteromedial peripheral-attachment quality (meniscotibial/meniscocapsular) from operative notes, as these factors influence extrusion and repair loading and underpin published indications for centralization (Figures 6 and 7). Preoperative long-leg standing radiographs. (a) Hip–knee–ankle (Hka) angle measurement with mechanical axis deviation. (B–C) Varus alignment assessment on anteroposterior and lateral views. Postoperative radiographs after posterior root repair and double-tunnel centralization. (a) Anteroposterior radiograph showing tibial tunnel positions and cortical button fixation. (b) Lateral radiograph demonstrating appropriate tunnel direction and fixation hardware.

Sample size was guided by the literature and powered for clinically meaningful differences. For the primary clinical endpoint, we targeted detection of the IKDC MCID (∼10–11 points) with SD 16–18, α = 0.05 (two-sided), and 80% power, yielding ∼ 34–42 patients per group; allowing 10–15% attrition/missingness, the planned target was ∼45–50 per group. For the primary structural endpoint, prior MRI series report MME SD ≈ 1.0–1.5 mm; detecting a 1.0-mm between-group difference requires ∼32–45 per group for 80–90% power at α = 0.05. These targets are consistent with contemporary series establishing MCID/PASS after meniscal repair and with MRI variability for extrusion.
Statistical analysis followed an analysis plan designed to identify small structural and small-to-moderate functional effects, consistent with the hypothesis that adding centralization primarily refines meniscal position/load sharing rather than dramatically changing extrusion when both groups receive anatomic root fixation. Continuous variables were assessed for normality (visual inspection, Shapiro–Wilk) and summarized as mean ± SD or median (IQR); categoricals as n (%). Between-group comparisons used independent-samples t-tests (or Mann–Whitney U) and χ2 (or Fisher’s exact) tests. Within-group pre-to postoperative changes used paired t-tests (or Wilcoxon signed-rank). Because treatment allocation was nonrandom, we implemented propensity-score matching (PSM) using a
Primary endpoints were analysed using
Finally, we specified
Results
Baseline characteristics after matching.

BMI, body mass index; HKA, hip–knee–ankle; MME, medial meniscus extrusion; MEI, Meniscal Extrusion Index; IKDC, International Knee Documentation Committee.
Note. All baseline covariates were balanced (standardized mean differences <0.10).
Reliability of MRI extrusion measurements.
MRI extrusion outcomes.

aWithin-group paired p values: posterior tunnel p = 0.29; centralization p = 0.21.
MME, medial meniscus extrusion; MEI, Meniscal Extrusion Index; CI, confidence interval; OR, odds ratio.
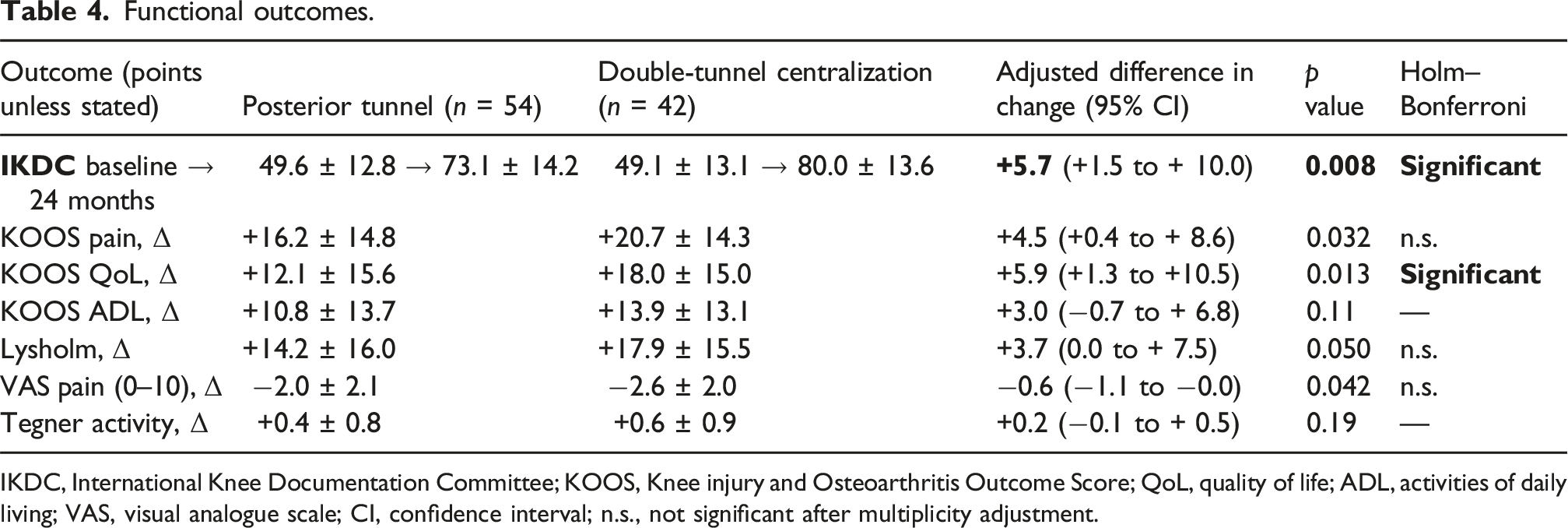
Functional outcomes.
IKDC, International Knee Documentation Committee; KOOS, Knee injury and Osteoarthritis Outcome Score; QoL, quality of life; ADL, activities of daily living; VAS, visual analogue scale; CI, confidence interval; n.s., not significant after multiplicity adjustment.
Responder analysis at 24 months.

MCID, minimal clinically important difference; PASS, patient acceptable symptom state; OR, odds ratio.
Secondary PROs showed a similar pattern. KOOS Pain improved by + 16.2 ± 14.8 points with posterior tunnel fixation and +20.7 ± 14.3 with centralization (adjusted difference + 4.5; 95% CI + 0.4 to +8.6; p = 0.032). KOOS QoL improved by + 12.1 ± 15.6 versus + 18.0 ± 15.0 (adjusted difference + 5.9; 95% CI + 1.3 to +10.5; p = 0.013). KOOS ADL improved by + 10.8 ± 13.7 versus + 13.9 ± 13.1 (adjusted difference +3.0; 95% CI −0.7 to +6.8; p = 0.11). Lysholm increased by + 14.2 ± 16.0 versus + 17.9 ± 15.5 (adjusted difference + 3.7; 95% CI 0.0 to +7.5; p = 0.050). VAS pain decreased by −2.0 ± 2.1 versus −2.6 ± 2.0 (adjusted difference −0.6; 95% CI −1.1 to −0.0; p = 0.042) (Table 4). After Holm–Bonferroni correction for multiple secondary endpoints, differences remained statistically significant for IKDC and KOOS QoL; other secondary comparisons did not retain significance.
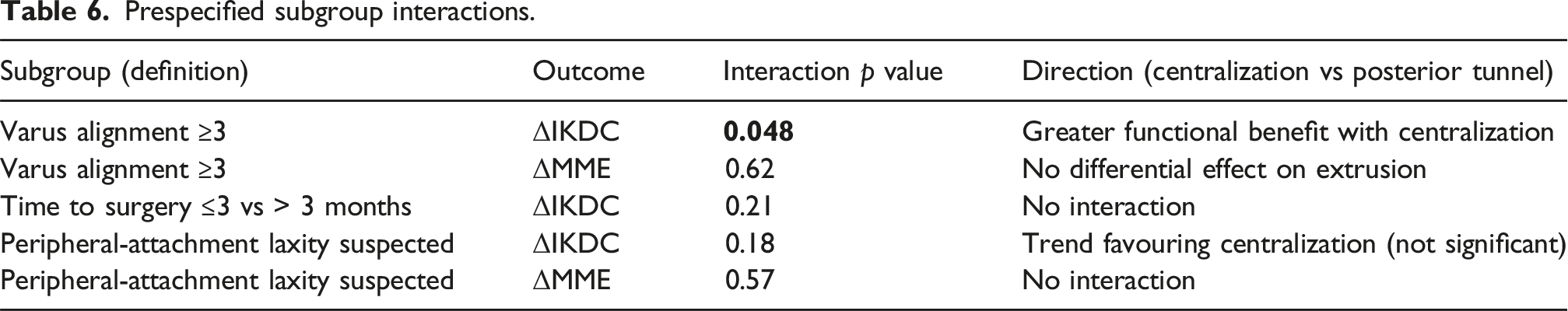
Prespecified subgroup interactions.
Adverse events and reoperations.

aManaged with partial meniscectomy. DVT, deep-vein thrombosis; PE, pulmonary embolism; MUA, manipulation under anesthesia.
Discussion
In this retrospective matched cohort, MRI‐based medial meniscus extrusion (MME) did not change meaningfully from preoperative to 12 ± 4 months in either group, whereas patient‐reported outcomes improved in both cohorts with a modest advantage for double-tunnel centralization. The structural finding aligns with recent syntheses indicating that while posterior medial meniscus root repair (PMMRR) reliably improves symptoms and joint preservation versus meniscectomy or nonoperative care,
While the mean between-group difference in IKDC scores (+5.7 points) was statistically significant, it remained below the established MCID threshold of 10–11 points. However, this mean value obscures the individual benefit; the ‘responder analysis’ revealed that adding centralization significantly increased the probability of a patient achieving a clinically meaningful improvement (77% vs 63%, p = 0.046). Thus, while the average effect size is modest, the intervention increases the likelihood of surgical success for the individual patient.
Several longitudinal MRI series also suggest that MME may
Against this backdrop, our finding of
The dissociation between our improved clinical scores and the unchanged static MME warrants careful interpretation. While static supine MRI failed to show structural restoration, recent biomechanical evidence by
Early clinical series and reviews published since 2021 also trend in the same direction. Case series of
Technique nuances may also matter. Recent biomechanical work indicates that
It is worth situating our findings within the broader treatment paradigm. Contemporary meta-analyses continue to show that
Several factors could explain why we did not detect a between-group difference in MME. First, our imaging window (12 ± 4 months) may be
Strengths of our study include a clearly defined imaging protocol with excellent inter/intra-rater reliability, propensity methods to mitigate selection bias, and a focus on a
A significant limitation is the lack of long-term joint preservation data, such as progression of Kellgren-Lawrence grade or conversion to total knee arthroplasty (TKA). As shown by Lamba et al. 6 clinical failure and TKA conversion often manifest over a decade. Therefore, our 2-years follow-up captures early functional recovery but cannot yet confirm whether centralization confers a durable chondroprotective benefit compared to isolated root repair. 3
We excluded patients with >5 varus alignment to isolate the effect of the soft-tissue procedure from bony malalignment. However, biomechanical data from Deichsel et al. 15 demonstrate that centralization is most effective in unloading the root repair specifically under varus loading conditions. By excluding severe varus, we may have paradoxically underestimated the potential benefit of centralization in the population that biomechanically needs it most. Our finding that even mild varus (≥3°) showed a significant interaction favoring centralization supports this hypothesis.
In adult PMMRTs suitable for repair, surgeons can reasonably expect
Conclusions
Anatomic posterior root repair reliably improved knee function despite largely unchanged static MRI extrusion. The addition of a second-tunnel centralization provided a statistically significant incremental benefit.
Footnotes
Ethical considerations
This retrospective cohort study was conducted across two centers—the Departments of Orthopaedics and Traumatology at Aksaray University Training and Research Hospital and Sanliurfa Mehmet Akif Inan Training and Research Hospital. The study protocol was approved by the Institutional Ethics Committee of Aksaray University (Protocol: SAGETİK 2025-197, Approval number: 2026/03, Date: January 08, 2026). All participants provided written informed consent for the use of their clinical and imaging data for research purposes, and all procedures were conducted in compliance with institutional ethical standards and the Declaration of Helsinki.
Consent for publication
All participants provided written informed consent for the use of their clinical and imaging data for research purposes, and all procedures were conducted in compliance with institutional ethical standards and the Declaration of Helsinki.
Authors’ contributions
Y.S.G. and M.F.D. contributed to the study conception and design. Material preparation, data collection and analysis were performed by Y.S.G. The first draft of the manuscript was written by Y.S.G. and M.F.D., and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
