Abstract
Background
Dorsal wrist ganglia (DWGs) are common, yet the contribution of carpal morphology to their formation is underexplored. We investigated whether scapholunate joint, lunate, and capitate morphology differ between DWG wrists and controls.
Methods
Retrospective MRI case–control study of adults aged 18–60 years, comprising 70 DWG wrists and 70 controls. Categorical variables were lunate type (medial hamatolunate facet present or absent), scapholunate joint morphology (parallel, inverted-Y, point-like parallel), and capitate head shape (flat, round, V-shaped). Quantitative measures included capitate–triquetral distance, mid-joint scapholunate gap, and medial hamatolunate facet length, recorded only when a facet was present.
Results
Interobserver agreement was generally good across both categorical and quantitative measures (categorical κ range: 0.50–0.88; quantitative ICC range: 0.72–0.95). Capitate morphology differed between groups: round heads were more frequent in DWG (71.4%) than in controls (44.3%) (p < 0.05). The scapholunate gap was larger in DWG (mean 1.8 mm) than in controls (1.5 mm) (p < 0.05). No between-group differences were observed for lunate type (Type II: 51.4% vs 38.6%; p = 0.107), scapholunate joint type (p = 0.787), capitate–triquetral distance (p = 0.223), or medial hamatolunate facet length (p = 0.395).
Conclusion
DWG wrists more often exhibit a round capitate head and a larger scapholunate gap than matched controls, whereas lunate type, scapholunate joint configuration, capitate–triquetral distance, and medial hamatolunate facet length are similar. Capitate head shape may be associated with DWG; however, given the lower agreement for this parameter, this finding should be considered exploratory and requires further validation before any recommendation for routine reporting can be made. The scapholunate gap can be considered supportive context alongside clinical findings. Prospective multicenter studies with multiplanar/loaded MRI or arthroscopic correlation are warranted.
Introduction
Dorsal wrist ganglia are the most common benign soft-tissue masses of the hand, and roughly 60–70% of wrist cysts arise on the dorsal aspect. Their prevalence is likely underestimated because they are frequently overlooked and predominantly asymptomatic.1–4 Although the exact cause remains contested, primary mechanisms include capsuloligamentous weakening and a one-way valve effect at the scapholunate joint that permits synovial fluid to leak into adjacent tissues.1,2,5 This synovial herniation is believed to arise from either trauma or myxoid degeneration of periarticular connective tissue. 6 Underlying anatomical factors that may predispose to dorsal wrist ganglion (DWG) formation, especially carpal morphology, are not yet well characterized.
Anatomical variations of the carpal bones and their articulations can alter load transfer and capsuloligamentous strain across the radiocarpal and midcarpal joints. In particular, morphology at the scapholunate joint (SLJ), lunate type, and capitate head geometry may directly or indirectly affect wrist kinematics.7–10 Prior wrist magnetic resonance imaging (MRI) studies have emphasized ligament integrity and cyst characteristics; comparatively fewer have examined whether specific bony and joint-shape features are over-represented in patients with DWG.3,11,12 As a result, the potential contribution of carpal morphology to ganglion formation remains insufficiently defined.
For lesions with a typical clinical presentation, imaging is often unnecessary; when evaluation is pursued, plain radiographs may be obtained to screen for concomitant osseous or articular abnormalities. These are usually unremarkable; ultrasound or MRI are appropriate next steps.13,14 Among imaging options, MRI additionally enables concurrent assessment of soft tissues and osseous morphology using standardized planes suitable for reproducible morphometrics. 15 With advances in MRI technology and image quality, carpal bone and joint morphology can now be delineated with greater clarity and examined for their relationship to dorsal wrist ganglia—a premise that motivated the present study.
Accordingly, we conducted an MRI-based study comparing patients with DWG to controls without ganglia. Our hypothesis was that the pathophysiology of DWG formation is influenced not only by capsuloligamentous weakness but also by carpal bone–joint morphology. We therefore evaluated whether morphologic parameters of the SLJ, lunate, and capitate are associated with the presence of DWG.
Material and methods
Study groups
Following institutional review board approval, we queried the university hospital’s digital archive to assemble two study groups.
The first group comprised patients with DWG. We queried the hospital’s digital archives to retrieve wrist MRI examinations labeled with a ganglion diagnosis between 2015 and 2024. Adults aged 18–60 years with an imaging-reported dorsal midline wrist ganglion were initially considered eligible. In total, 126 patients were identified for preliminary screening. Single-chamber dorsal midline lesions with a cross-sectional area >0.5 cm2 were included; radial and volar ganglia were excluded. In line with Anderson et al., 16 crescentic ganglia interpreted as synovitis and occult ganglia <0.5 cm2 were excluded. Multiloculated (multiseptated) cysts and cysts with a maximal diameter >20 mm were also excluded. Additional exclusions comprised joint effusion, osteophytes/arthrosis, and soft-tissue injuries (e.g., triangular fibrocartilage complex tears), as well as traumatic conditions (acute or prior fractures) and structural bone pathologies, including intraosseous cysts, ulnar-positive variance, and Kienböck disease. A total of 70 wrist MRI examinations from 70 patients were included for analysis in the DWG group.
The second group comprised controls. We queried the same hospital archive (2015–2024) to identify wrist MRI examinations from adults aged 18–60 years with radiology reports interpreted as normal. In total, 358 patients were identified for preliminary screening. Candidates with any trauma history (recent or prior), previous wrist surgery, or findings that could influence morphometric measurements were excluded (joint effusion, osteophytes/arthrosis, ligament or TFCC tears, occult or intraosseous cysts, scaphoid nonunion, scaphoid nonunion advanced collapse [SNAC], scapholunate advanced collapse [SLAC], Kienböck disease, ulnar impaction syndrome, or other structural bone pathology). Exams with motion or metal artifacts were excluded. All candidate MRIs were second-read by study observers to confirm normal status.
We performed 1:1 nearest-neighbor matching without replacement on sex (exact), wrist side (exact), and age (caliper ±2 years). When multiple candidates were eligible, ties were resolved by the closest.
Imaging technique and MRI protocol
All wrist MRI examinations were performed on a 1.5-T scanner (MAGNETOM Aera; Siemens Healthineers, Erlangen, Germany) using a multi-channel extremity coil. Imaging was obtained with the patient in the prone “superman” position, with the wrist maintained in a neutral position. Slice thickness was 3.0 mm with an inter-slice gap of 0.3 mm according to the routine institutional protocol. All morphometric measurements were performed on coronal T2-weighted gradient-echo (GRE) images using electronic calipers on a PACS workstation.
Morphological assessment and quantitative measurements
Measurements were performed by two orthopedic hand surgeons (10 and 4 years of experience) and one orthopedic surgeon (5 years of experience). Prior to formal measurements, the raters completed a calibration session on a subset of cases to harmonize anatomical landmarks and measurement rules. All subsequent assessments were performed independently and blinded to group allocation. For quantitative variables, the arithmetic mean of the three readings was used as the final value. For categorical variables, the final value was defined by simple majority (≥2 of 3). In the rare absence of a majority, disagreements were resolved by consensus meeting among the three observers.
For each dorsal wrist ganglion cyst, the presumed origin/attachment was recorded on MRI as the site of closest apparent continuity with the dorsal capsuloligamentous structures (e.g., dorsal scapholunate, capitolunate, radiolunate); when no clear attachment could be identified, this was recorded as ‘not identifiable’.
Lunate morphology was categorized according to Viegas et al.
7
as type I in the absence of the medial hamatolunate facet and type II in its presence (Figure 1). Scapholunate (SL) joint morphology was classified on coronal images according to Schimmerl-Metz et al.
17
into three patterns: type I (parallel)—parallel, straight articular surfaces across the midportion of the scapholunate interval; type II (inverted-Y)—an inverted-Y configuration with progressive widening of the joint space from distal to proximal; and type III (point-like parallel)—parallel, congruent mid-joint surfaces measuring <2 mm in length (Figure 2). Coronal wrist MR images. (a) Viegas type I lunate (no medial hamatolunate facet); (b) Viegas type II lunate; the yellow arrow indicates the medial hamatolunate facet forming the hamatolunate joint. Coronal MR images demonstrating scapholunate joint morphology. (a) Parallel type; (b) point-like parallel type; (c) inverted-Y type. Yellow arrows indicate the scapholunate joint space.
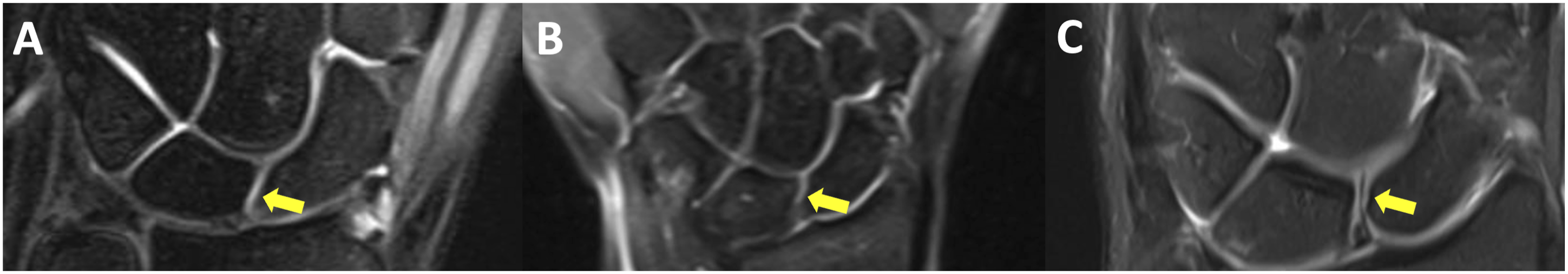
Capitate head morphology was classified using the previously described three-type framework (flat, round, V-shaped).
18
On coronal T2-weighted GRE images, the capitate head was assessed by reviewing 2–3 consecutive mid-coronal slices in which the proximal capitate articular surface was most sharply visualized. Classification was based on the contour of the proximal capitate head: round was defined as a smooth, continuously convex arc; flat as a predominantly straight or minimally curved proximal contour; and V-shaped as an angulated contour formed by two converging surfaces creating an apex (“V” configuration). When the appearance varied slightly across the evaluated slices, the predominant pattern across these consecutive images was recorded (Figure 3). Coronal MR images demonstrating capitate head morphologies. (a) Flat head; (b) round head; (c) V-shaped head. Yellow lines highlight the capitate head contour.
Capitate–triquetral (CT) distance was measured as the minimum cortex-to-cortex distance between the capitate and triquetrum on coronal MRI, following the anatomic principle of the classic radiograph-based definition.
19
Observers reviewed 2–3 consecutive mid-coronal slices with the sharpest opposing cortices, and the smallest value was recorded (mm) (Figure 4). Measurements on coronal MR image: yellow line = capitate–triquetrum (CT) shortest distance; blue line = scapholunate (SL) gap at the midportion as the shortest perpendicular cortex-to-cortex distance; red line = medial hamatolunate (HL) facet maximum linear dimension.
In type II lunates, medial hamatolunate (HL) facet length was measured on coronal MRI as the maximum linear dimension of the medial HL facet, recorded as the longest value across visible slices (mm) (Figure 4).
Statistical analysis
Given the fixed sample size of this retrospective cohort, a post hoc sensitivity power analysis was performed for the between-group comparison of the mid-scapholunate (SL) gap (primary outcome). Using the dispersion observed in the cohort (pooled SD ≈ 0.35 mm), with 70 wrists per group and a two-sided α = 0.05, the study has 80% power to detect a between-group difference of approximately 0.17 mm and >90% power to detect a difference of 0.20 mm. Power analysis was performed using G*Power (version 3.1; Heinrich Heine University Düsseldorf, Düsseldorf, Germany).
Interobserver agreement for categorical variables (lunate type, scapholunate joint type, capitate morphology) was quantified with Fleiss’ kappa; pairwise agreement between rater dyads was summarized with Cohen’s kappa and percent agreement. Ninety-five percent confidence intervals (CIs) for κ were obtained by percentile bootstrap (B = 2000). For continuous (mm) measurements, reliability was assessed using a two-way random-effects, absolute-agreement, single-measurement intraclass correlation coefficient, ICC(2,1), with 95% CIs. Measurement error was reported as the standard error of measurement (SEM), and the minimal detectable change at 95% confidence (MDC95) was calculated. Between-group comparisons of continuous variables were performed using the Mann–Whitney U test, and categorical variables were compared with Pearson’s chi-square (χ2) test. Homogeneity of variances was assessed with Levene’s test. Where applicable, 95% confidence intervals (CIs) were computed for effect estimates. Two-sided p values <0.05 were considered statistically significant.
Results
Interobserver agreement for categorical variables in the dorsal wrist ganglion and control groups.

CI = confidence interval, min = minimum, max = maximum.
Interobserver agreement for continuous variables in the dorsal wrist ganglion and control groups.

ICC = Intraclass Correlation Coefficient, CI = Confidence Interval (95%), SEM = Standard Error of Measurement, MDC95 = Minimal Detectable Change (95%).
Group comparisons of demographic balance, quantitative MRI measures, and carpal morphology distributions.

a= Mann Whitney U test (Monte Carlo).
b= Pearson Chi-Square Test (Monte Carlo). Bold values denote statistical significance.
In the ganglion group, most cysts were centered at the dorsal scapholunate region (59/70, 84.3%). The remaining cases showed a presumed capitolunate attachment (7/70, 10.0%) or radiolunate attachment (2/70, 2.9%); in 2 cases (2/70, 2.9%), no definite ligamentous attachment could be identified on MRI.
Lunate morphology did not differ significantly between groups; Type I predominated in controls (61.4%), whereas Type II was most frequent in the ganglion group (51.4%) (overall: Type I [n = 77; 55%], Type II [n = 63; 45%]). For SL joint morphology, the parallel configuration was the most common in both groups (54.3% each), with a similar overall distribution (overall: parallel [n = 76; 54.3%]). In contrast, capitate morphology differed: controls most often had a flat head (50.0%), whereas the ganglion group predominantly showed a round head (71.4%) (overall round [n = 81; 57.9%], flat [n = 51; 36.4%]) (Table 3).
CT distance did not differ between groups (overall mean [4.2] mm). The SL gap was larger in the ganglion group than in controls (1.8 vs 1.5 mm; p < 0.05; overall mean [1.6] mm). The HL facet length was similar between groups, with no significant difference (overall mean [3.1] mm) (Table 3).
Discussion
In this study, coronal wrist MRI scans of patients with DWG were examined for morphological features of the carpal bones and joints. The main results showed no between-group differences in lunate or scapholunate joint morphology, whereas round-type capitate heads were more frequent in the DWG group; among quantitative parameters, only the scapholunate gap was larger in DWG, while capitate–triquetral distance and hamatolunate facet length did not differ significantly.
Methodologically, this MRI-based case–control design leveraged standardized coronal images to enable reproducible morphometrics, with 1:1 nearest-neighbor matching on sex and wrist side (exact) and age within a ±2-years caliper to limit confounding. Measurements were obtained by three observers who routinely interpret wrist MRI in daily practice, and interobserver agreement was good for both categorical and continuous variables (Tables 1 and 2). Collectively, the standardized imaging, balanced matching, and strong interobserver metrics indicate that the observed group differences—and lack thereof—are unlikely to be artifacts of measurement inconsistency.
Despite the presence of several classification schemes for assessing lunate morphology, the system proposed by Viegas is the most widely accepted. This classification is predicated on whether the lunate articulates with the hamate via a medial hamatolunate facet (Type II) or lacks this articulation (Type I). 7 Lunate morphology can be assessed using plain radiographs or MRI. Park et al. 20 reported that MRI-based evaluation delineates lunate type more accurately than radiography. Accordingly, we planned the study using MRI-based assessment.
Numerous studies have examined lunate morphology and suggested that the presence of a medial hamatolunate facet alters load transmission across the radiocarpal joint and influences carpal kinematics.8,20,21 In line with these biomechanical considerations, several reports have linked distinct lunate types to diverse carpal pathologies, including scapholunate intraosseous ligament injury, Kienböck disease, and scaphoid fractures.22–24 However, to the best of our knowledge, no prior study has specifically evaluated the relationship between dorsal wrist ganglia (DWG) and lunate morphology. While carpal kinematics may differ across lunate types, in the present study the DWG group exhibited a higher proportion of Viegas Type II lunates than controls (51.4% vs 38.6%), yet this difference did not reach statistical significance (Table 3).
Beyond the Viegas classification, we assessed two secondary markers linked to lunate type: the CT distance and, when present, the length of the medial HL facet. Because CT distance is typically greater in Viegas Type II lunates—owing to the medial HL facet—any between-group difference would be expected to mirror lunate-type distribution. In our cohort, Type II lunates were more frequent in DWG than in controls but not significantly so (51.4% vs 38.6%; Table 3). Consistent with this, CT distance did not differ between groups (Table 3). HL facet length was measured only in Type II lunates; accordingly, no direct between-group association was anticipated. As expected, HL facet length did not differ between DWG and controls (mean [SD], 3.0 [0.8] vs 3.2 [0.9] mm; p = 0.395), indicating broadly similar facet morphology across groups and suggesting that facet length alone is unlikely to discriminate DWG.
Because most dorsal wrist ganglia (DWGs) arise from the scapholunate complex, the association between the scapholunate ligament (SLIL) and DWG has been widely examined. Edwards and Johansen 25 reported SLIL laxity in 45 of 55 patients undergoing arthroscopic ganglion excision. Similarly, McKeon et al. 26 found higher rates of generalized ligamentous laxity and a positive scaphoid shift test in patients with symptomatic ganglia than in comparison populations. Although we did not assess SLIL laxity directly, we measured the SL gap at the mid-joint—the literature-recommended site that yields the least-variable subchondral-to-subchondral distance. 17 In addition, we classified scapholunate joint morphology according to the scheme described by Schimmerl-Metz et al. 17 The scapholunate gap was greater in the DWG group than in controls (mean [SD], 1.8 [0.4] vs 1.5 [0.3] mm; p < 0.05), a finding consistent with subtle SLIL laxity reported previously. Notably, the distribution of SL-joint morphology types was similar between groups—the parallel configuration predominated in both (54.3% in DWG vs 54.3% in controls)—indicating that the gap difference is not attributable to a shift in gross joint shape.
Most studies evaluating carpal morphology have focused on the lunate and the scapholunate joint.8,10,17,19,21–24 In the present study, we additionally assessed capitate morphology. The literature on the capitate morphology is limited; available data suggest a potential association between lunate and capitate morphology. However, to our knowledge, no prior study has examined the relationship between capitate morphology and DWG. In cadaveric study, Yazaki et al. 18 reported a predominance of the flat-head capitate (69 of 107 wrists [65%]), whereas a radiographic series by Wollstein et al. 27 found the round (spherical) head to be most common (50 of 70 wrists [71%]). In our study, capitate-type distributions also differed between groups: the round head predominated in the DWG group (71.4%), whereas the flat head was most frequent among controls (50.0%); the overall distributions were significantly different (Table 3). However, interobserver agreement for capitate morphology was more variable compared with other categorical measures (Table 1); therefore, this finding should be interpreted with appropriate caution. In interpreting the observed difference in capitate-type distributions, a plausible explanation is altered midcarpal load transfer related to capitate head geometry. A more spherical capitate may reduce curvature congruity at the capitolunate and scaphocapitate interfaces, concentrating contact pressures dorsally and increasing shear on the dorsal capsule and the scapholunate complex, thereby lowering the threshold for the valve-like capsular attenuation implicated in DWG formation. While causality cannot be inferred from our cross-sectional design, these findings suggest that capitate geometry may modulate dorsal capsuloligamentous stress, providing a biomechanical substrate for DWG in susceptible wrists.
This study has several limitations. First, its retrospective, single-center design and reliance on archived reports may introduce selection and spectrum bias. Controls were drawn from patients who underwent wrist MRI for clinical indications and had reports interpreted as normal; therefore, their morphology may not fully represent the general asymptomatic population, which may limit generalizability. Although we mitigated this risk by sourcing cases and controls from the same archive and time window, applying strict exclusions for degenerative/traumatic and soft-tissue pathology, and confirming “normal” status by second-read, residual sampling bias cannot be excluded. The findings may also not generalize beyond adults aged 18–60 years imaged on a 1.5-T system; pediatric and older patients and 3-T or specialized protocols were not assessed. Second, measurements were limited to coronal images. Without axial or sagittal views, loaded MRI, or arthroscopic correlation, subtle SLIL laxity and the ganglion stalk could not be directly confirmed and were inferred. Third, matching was limited to sex, side, and age; unmeasured confounders such as hand dominance, occupational/sport exposure, generalized hyperlaxity, and prior microtrauma could influence carpal shape and the SL gap. Fourth, categorical classifications (particularly capitate head type) retain some subjectivity, and interobserver agreement for capitate morphology was more variable than for other measures; therefore, any capitate-shape association should be interpreted as exploratory and warrants further validation. In addition, all measurements were performed by orthopedic surgeons; confirmation by a musculoskeletal radiologist was not available, which may limit external validity despite the calibration session and standardized decision rules. Fifth, the inclusion threshold for DWG (>0.5 cm2 single-chamber dorsal midline cysts) and the exclusion of multiloculated and very large cysts (>20 mm) and concomitant degenerative or traumatic pathology improve internal validity but may limit applicability to the broader clinical spectrum of wrist ganglia. Although we documented presumed ganglion origin/attachment, subgroup analyses by origin were not performed because non–dorsal scapholunate origins were infrequent, limiting statistical power. Despite these limitations, to our knowledge this is the first MRI case–control study to assess capitate, lunate, and scapholunate morphology jointly in DWG, helping address a recognized paucity in the literature.
Conclusion
In this MRI-based case–control study of adults with dorsal wrist ganglion, DWG wrists more often showed a round capitate head and a larger scapholunate gap than matched controls, whereas lunate type, SL joint configuration, CT distance, and HL facet length did not differ meaningfully. Clinically, capitate head morphology may be associated with DWG; however, given the lower agreement for this parameter, this finding should be considered exploratory and requires further validation before any recommendation for routine reporting can be made. The SL gap may be interpreted as supportive context alongside symptoms and other imaging signs; confirmation in prospective multicenter cohorts with multiplanar/loaded MRI or arthroscopic correlation is warranted.
Footnotes
Ethical considerations
This study was approved by the Mersin University Clinical Research Ethics Committee (Decision No: 2022/832, Date: 21-12-2022).
Consent to participate
Waived due to the retrospective design.
Author contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
De-identified data and analysis materials are available from the corresponding author upon reasonable request.
