Abstract
Purpose
To assess whether the preoperative triglyceride–glucose (TyG) index is associated with 30-day surgical site infection (SSI) after instrumented posterior lumbar fusion (PLF) and to examine its predictive performance.
Methods
We retrospectively reviewed consecutive adults who underwent elective one- or two-level instrumented PLF between 2017 and 2024 at a tertiary center. Patients with active infection, revision surgery, tumor, trauma, or incomplete 30-day follow-up were excluded. Preoperative fasting triglycerides and glucose were used to calculate the TyG index as ln[(triglycerides × glucose)/2]. The primary outcome was 30-day SSI defined by Centers for Disease Control and Prevention criteria. Multivariable logistic regression evaluated the association between TyG and SSI, and receiver operating characteristic analysis assessed discrimination and identified an optimal cut-off. Incremental predictive value was examined by comparing a clinical model with and without TyG using AUC, likelihood ratio testing, reclassification metrics, calibration, Brier score, and decision curve analysis with bootstrap internal validation.
Results
Among 438 patients, 29 (6.6%) developed SSI (24 superficial, 2 deep, 3 organ/space). Patients with SSI had higher TyG values than non-SSI patients (9.1 ± 0.5 vs 8.6 ± 0.6; p < 0.001). Each 1-unit increase in TyG was independently associated with higher odds of SSI (adjusted odds ratio 3.65; 95% confidence interval 1.62–8.24; p = 0.002). TyG alone yielded an area under the curve of 0.73, and a cut-off of 8.80 identified a high-risk group with an SSI rate of 11.8% versus 2.5% in the low-TyG group. Beyond standard clinical factors, adding TyG improved model fit (LRT χ2 = 10.98; p = 0.001) and improved reclassification.
Conclusion
A higher preoperative TyG index is independently associated with 30-day SSI after PLF and provides moderate discriminative ability as a simple predictor. Incorporating TyG into preoperative risk assessment may help refine perioperative optimization and infection surveillance strategies in lumbar fusion surgery.
Keywords
Introduction
Posterior lumbar fusion (PLF) is widely performed to treat degenerative lumbar conditions such as spinal stenosis, spondylolisthesis, and instability. 1 Although clinical outcomes are generally favorable, surgical site infection (SSI) remains one of the most devastating complications after lumbar fusion, leading to prolonged hospitalization, reoperations, implant removal, and increased healthcare costs. 2 Reported SSI rates after instrumented lumbar fusion range from 2% to over 10%, depending on patient comorbidities, surgical complexity, and prophylactic measures.3,4
Metabolic dysfunction and insulin resistance have emerged as important contributors to postoperative infectious complications. 5 Obesity and diabetes mellitus are well-established risk factors for SSI across various surgical specialties, including spine surgery. However, conventional markers such as body mass index (BMI) and the presence of diabetes may not fully capture the spectrum of metabolic risk, particularly in patients with normal BMI or undiagnosed insulin resistance.6,7 Notably, insulin resistance can exist before clinically recognized diabetes and may contribute to impaired immune responses and delayed tissue repair, thereby increasing susceptibility to postoperative infection even among patients without overt diabetes or marked obesity. 8 Therefore, a continuous marker reflecting insulin resistance may help identify hidden metabolic risk that is not adequately represented by BMI or a binary diabetes diagnosis.
The triglyceride-glucose (TyG) index, calculated from fasting triglyceride and glucose levels, has been proposed as a simple surrogate marker of insulin resistance.9,10 Prior epidemiologic studies have shown that higher TyG is associated with type 2 diabetes, cardiovascular events, and all-cause mortality.11–13 More recently, elevated TyG has been linked to increased risk of infectious complications and poor outcomes in critically ill or surgical populations. 14 In an elderly cohort undergoing abdominal and pelvic surgery, higher preoperative TyG was independently associated with SSI and showed modest discrimination. 15 In addition, among patients with Rutherford grade 5 chronic limb-threatening ischemia undergoing endovascular therapy, TyG was associated with delayed wound healing and was retained in a predictive nomogram. 16 Nevertheless, data on the relationship between TyG and SSI specifically after lumbar fusion surgery remain scarce.
We hypothesized that a higher preoperative TyG index would be associated with an increased risk of SSI after PLF, and that TyG might provide incremental predictive value for identifying high-risk patients. The objectives of this study were: (1) to investigate the association between preoperative TyG and 30-day SSI after instrumented PLF; (2) to evaluate the discriminative performance of TyG as a single predictor of SSI and identify an optimal cut-off; and (3) to explore the dose–response relationship between TyG and SSI using restricted cubic spline analysis.
Materials and methods
Study design and patient population
We conducted a retrospective cohort study of consecutive adult patients who underwent instrumented posterior lumbar fusion at a tertiary academic center between January 2017 and December 2024. The study was approved by the institutional review board and provided informed consent. All protocols were conducted in accordance with the research principles set forth in the Declaration of Helsinki.
Inclusion criteria were: (1) age ≥ 18 years; (2) elective single- or two-level posterior lumbar fusion with pedicle screw instrumentation via a posterior approach; and (3) availability of complete preoperative laboratory data including fasting triglycerides and fasting plasma glucose within 7 days before surgery. Exclusion criteria included: (1) active systemic or local infection at the time of surgery; (2) revision lumbar fusion; (3) tumor, trauma, or spinal infection requiring emergent surgery; and (4) incomplete follow-up within 30 days postoperatively. After applying these criteria, 438 patients were included in the final analysis.
Surgical procedure and perioperative care
All procedures were performed by experienced spine surgeons using a standardized operative technique. Under general anesthesia, patients were placed in the prone position, and a midline posterior approach was used. Pedicle screws were inserted under fluoroscopic guidance, followed by decompression as indicated and posterolateral fusion with autograft and/or allograft. Interbody fusion was performed at the surgeon’s discretion. All patients received standard perioperative antibiotic prophylaxis according to institutional protocol, typically a first-generation cephalosporin administered within 30 min before incision and continued for 24 h postoperatively. Wound drains were used in most cases and removed when drainage output was <50 mL over 24 h. Postoperative glycemic control and wound care followed institutional guidelines, patients with known diabetes or perioperative hyperglycemia underwent capillary glucose monitoring and insulin therapy as needed, aiming for a target range of approximately 100–180 mg/dL in noncritically ill patients.
Data collection and TyG index calculation
Demographic and clinical data were extracted from electronic medical records, including age, sex, BMI, smoking status, comorbidities (with particular attention to diabetes mellitus), and American Society of Anesthesiologists (ASA) physical status classification. Perioperative variables included operative time, estimated blood loss (EBL), number of fused levels, intraoperative blood transfusion, and use of wound drains.
Preoperative laboratory values were obtained from the preoperative assessment, focusing on fasting plasma glucose, fasting triglycerides, and serum albumin. The TyG index was calculated as:
Serum albumin <3.5 g/dL was defined as hypoalbuminemia. BMI ≥ 30 kg/m2 was considered obesity.
SSI definition
The primary outcome was 30-day SSI, defined according to the Centers for Disease Control and Prevention (CDC) criteria. SSI was classified as superficial incisional, deep incisional, and organ/space infection. Consistent with SSI surveillance definitions, superficial incisional SSI is monitored within 30 days for all procedures, whereas deep or organ/space SSI may be monitored for longer periods depending on procedure category; therefore, this study focused on early postoperative infections occurring within 30 days. All SSIs were identified through review of inpatient and outpatient records, microbiology reports, and operative notes. Reoperation for irrigation and debridement, with or without implant retention, within 30 days was recorded.
Statistical analysis
Continuous variables are reported as mean ± standard deviation and categorical variables as counts and percentages. Baseline characteristics were compared between patients with and without 30-day SSI using the Student’s t test for continuous variables and the chi-square test or Fisher’s exact test for categorical variables, as appropriate.
The primary exposure, TyG index, was analyzed as a continuous variable. Associations with 30-day SSI were evaluated using multivariable logistic regression, with effect estimates reported as odds ratios (ORs) and 95% confidence intervals (CIs). Given the limited number of events, the primary adjusted model included a prespecified set of clinically relevant covariates to reduce overfitting (diabetes mellitus, current smoking, ASA class ≥ III, and hypoalbuminemia). A bias-reduced (Firth) logistic regression was additionally performed as a sensitivity analysis using an expanded model that further adjusted for BMI, operative time, estimated blood loss, fusion levels, intraoperative transfusion, and drain use.
Discriminative performance of TyG as a single predictor was assessed using receiver operating characteristic (ROC) analysis and the area under the ROC curve (AUC). The optimal TyG cut-off was identified by maximizing the Youden index and used to define low- and high-TyG groups. For this dichotomization, we compared SSI incidence between groups and reported sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and likelihood ratios (LR+ and LR−).
To assess incremental predictive value beyond standard clinical factors, we developed a clinical prediction model including diabetes mellitus, current smoking, ASA class ≥ III, and hypoalbuminemia, and then added TyG to create an augmented model. Model discrimination (AUC), calibration, and overall accuracy were evaluated using AUC, calibration plots (patients grouped into deciles of predicted risk), and the Brier score, respectively. Improvement in model fit after adding TyG was tested with a likelihood ratio test (LRT), and model parsimony was compared using Akaike and Bayesian information criteria (AIC and BIC). Internal validation was performed by bootstrap resampling to estimate optimism-corrected AUCs and Brier scores. Reclassification performance was quantified using the integrated discrimination improvement (IDI) and continuous net reclassification improvement (NRI) with bootstrap confidence intervals. Clinical utility was explored using decision curve analysis across clinically relevant threshold probabilities.
Prespecified subgroup analyses were conducted by diabetes status (yes vs no) and BMI category (<30 vs ≥30 kg/m2). Potential effect modification was assessed by including interaction terms between TyG and subgroup variables. All analyses were performed in R, and a two-sided p value <0.05 was considered statistically significant.
Results
Incidence and characteristics of SSI
Among the 438 patients included in the cohort, 29 (6.6%) developed SSI within 30 days of PLF. Of these, 24 (5.5%) were superficial incisional infections, 2 (0.5%) were deep incisional infections, and 3 (0.7%) were organ/space infections. Accordingly, 5 patients (1.1%) met criteria for deep SSI (deep incisional or organ/space infection). Seven patients (1.6%) underwent operative irrigation and debridement within 30 days. Five had deep SSI and two had persistent superficial infections prompting operative management, and instrumentation was generally retained.
Baseline demographic and perioperative characteristics according to 30-day SSI status.

*Hypoalbuminemia was defined as preoperative serum albumin < 3.5 g/dL.
Operative time and EBL were slightly higher in patients with SSI (189.2 ± 42.9 vs 188.1 ± 46.0 min; 309.8 ± 117.4 vs 276.5 ± 116.3 mL), but these differences did not reach statistical significance. The proportion of one-level PLF, intraoperative transfusion, and drain use was similar between groups.
Association between TyG and SSI
Multivariable logistic regression analysis of risk factors for 30-day SSI.

In a bias-reduced expanded model additionally adjusting for BMI, operative time, estimated blood loss, fusion levels, transfusion, and drain use, TyG remained independently associated with SSI (Supplemental Table S1).
In prespecified subgroup analyses, the association between TyG and SSI was directionally consistent across diabetes and BMI strata, with no evidence of effect modification (p for interaction > 0.05 for both; Supplemental Table S2).
Predictive performance and optimal cut-off of TyG
The TyG index alone demonstrated good discriminative ability for predicting SSI, with an AUC of 0.73(Figure 1(a)). ROC analysis identified a TyG value of 8.80 as the optimal cut-off based on the Youden index. At this threshold, TyG predicted SSI with a sensitivity of 0.79 and a specificity of 0.58. The corresponding positive predictive value (PPV) and negative predictive value NPV were 0.12 and 0.98, respectively, reflecting the low event rate in this cohort, with likelihood ratios (LR) of LR+ = 1.89 and LR− = 0.36. Predictive performance of the TyG index for 30-day surgical site infection (SSI) after posterior lumbar fusion. (a) Receiver operating characteristic (ROC) curve for the preoperative TyG index predicting 30-day SSI. (b) Bar chart showing the incidence of 30-day SSI according to TyG category.
When patients were stratified by this cut-off, 195 (44.5%) were classified as having high TyG (≥8.80) and 243 (55.5%) as low TyG (<8.80). The incidence of 30-day SSI was 11.8% (23/195) in the high-TyG group and 2.5% (6/243) in the low-TyG group (p < 0.001) (Figure 1(b)). The crude odds of SSI were 5.28 times higher in the high-TyG group than in the low-TyG group (95% CI 2.11–13.25; p < 0.001), indicating marked risk separation based on a simple laboratory index.
Incremental predictive value of TyG beyond standard clinical factors
Model performance, calibration, and incremental predictive value of adding the TyG index to the clinical prediction model.

Reclassification metrics (IDI and continuous NRI) for evaluating incremental discrimination after adding the TyG index to the clinical prediction model.

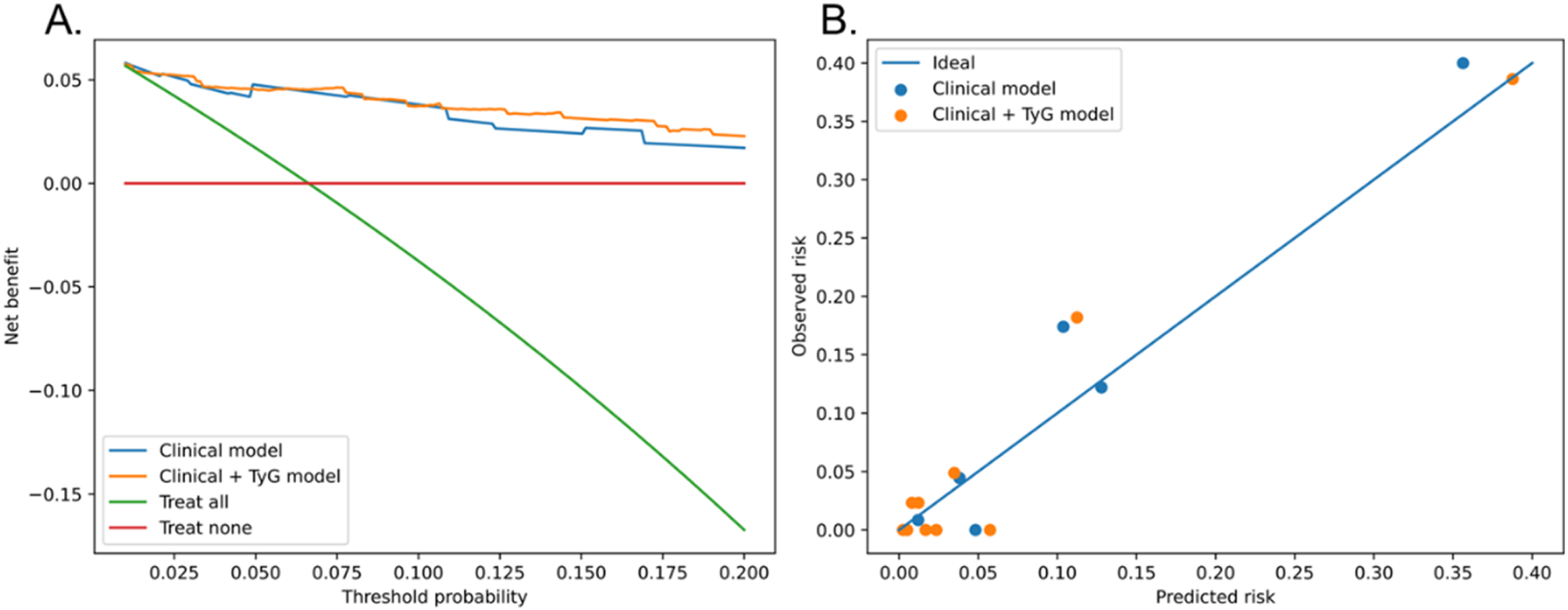
(a) Decision curve analysis for models predicting 30-day SSI after posterior lumbar fusion. (b) Calibration plots for the clinical-only and TyG-augmented models predicting 30-day SSI, patients were grouped into deciles of predicted risk, and for each group the mean predicted probability is plotted against the observed event rate. The diagonal line represents perfect calibration. Both models showed acceptable agreement between predicted and observed risks, with similar calibration performance after adding TyG.
Association of TyG index with SSI subtypes
We next explored whether higher TyG values were preferentially associated with deeper SSI phenotypes. When patients were stratified by SSI subtype, the mean TyG index increased progressively from those without infection to those with superficial, deep incisional, and organ/space SSI (8.64 ± 0.60, 8.96 ± 0.44, 9.64 ± 0.02, and 9.77 ± 0.11, respectively). Overall differences in TyG across these four categories were statistically significant (p < 0.001) (Figure 3(a)). Among the 29 patients who developed SSI, the TyG index was strongly correlated with an ordinal indicator of infection depth (superficial = 1, deep incisional = 2, organ/space = 3), with a Spearman correlation coefficient of 0.53 (p < 0.001). Relationship between TyG index and SSI subtype. (a) Distribution of the TyG index according to SSI type. (b) Dot-matrix plot illustrating the distribution of SSI subtypes within low- and high-TyG groups.
Consistent with these findings, deep incisional and organ/space infections occurred exclusively in the high-TyG group (TyG ≥ 8.80), whereas all 6 SSIs in the low-TyG group (TyG < 8.80) were superficial. In the high-TyG group, superficial, deep incisional, and organ/space SSI accounted for 9.2%, 1.0%, and 1.5% of patients, respectively, compared with 2.5%, 0%, and 0% in the low-TyG group (Figure 3(b)). These patterns suggest that higher TyG is associated not only with a greater overall risk of SSI but also with a shift towards more severe, deep and organ/space infections.
Discussion
The present study evaluated the association between the triglyceride–glucose (TyG) index and early postoperative SSI after posterior lumbar fusion in a large single-center cohort. Several key observations emerged. First, the overall 30-day SSI rate was 6.6%, with deep or organ/space infection occurring in 1.1% of patients and reoperation required in 1.6%. Second, patients who developed SSI had a distinctly more adverse metabolic and systemic profile, characterized by a markedly higher prevalence of diabetes, current smoking, higher ASA class, hypoalbuminemia, and an increased TyG index. Third, after adjustment for these conventional risk factors, TyG remained an independent predictor of SSI, with each 1-unit increase associated with more than a threefold increase in odds. Fourth, the TyG index showed reasonable discriminatory performance (AUC 0.73), and an empiric cut-off of 8.80 separated patients into low- and high-risk strata with almost a fivefold difference in SSI incidence. In addition, beyond standard clinical factors, adding TyG improved overall model performance and fit, with lower Brier score, significantly better model fit by likelihood ratio testing, lower information criteria, improved reclassification (IDI and continuous NRI), and small but consistent net-benefit gains on decision curve analysis, while calibration remained acceptable. Finally, TyG was not only related to the occurrence of SSI, but also to its infection depth: higher TyG values were associated with a greater likelihood of deep incisional and organ/space infections, which occurred exclusively in the high-TyG group. However, these depth-related findings should be interpreted cautiously given the small number of deep and organ/space events and should be viewed as exploratory.
Our data extended the growing body of evidence that the TyG index is a simple but informative marker of adverse outcomes in metabolic and cardiovascular disease and suggest that it also has relevance in the perioperative spine surgery setting. TyG, calculated from fasting triglyceride and glucose concentrations, correlates closely with insulin resistance and has been linked to endothelial dysfunction, low-grade inflammation, and a prothrombotic state.17–19 Prior observational studies in non-surgical populations have associated elevated TyG with incident diabetes, cardiovascular events, stroke, and mortality, while perioperative studies have suggested relationships between TyG and complications such as acute kidney injury or poor functional recovery.20–22 Our findings add postoperative SSI after PLF to the list of TyG-related outcomes and demonstrate that the association persists even after accounting for the presence of overt diabetes and other systemic risk factors. In spine surgery, several traditional metabolic and nutrition-related predictors have been repeatedly associated with SSI risk, most notably diabetes and perioperative dysglycemia, obesity/BMI, and malnutrition or hypoalbuminemia. Contemporary spine-focused evidence syntheses and guidelines consistently identify diabetes as an important risk factor for postoperative SSI, while also emphasizing that perioperative hyperglycemia may confer added risk even beyond a binary diabetes diagnosis. 23 Likewise, obesity has been linked to increased SSI risk in instrumented procedures, and low albumin/malnutrition has been associated with higher postoperative infectious complications, 24 supporting the clinical relevance of nutritional–metabolic reserve. 25 However, these conventional variables can be coarse or incompletely captured in routine practice (e.g., “diabetes: yes/no,” BMI as an imperfect proxy for metabolic health), and direct insulin-resistance assessments are rarely feasible at scale. 26 In this context, TyG offers a simple, continuous, and readily available surrogate that may better reflect the spectrum of metabolic vulnerability, particularly among patients with normal BMI, impaired glucose tolerance, or subclinical insulin resistance, thereby complementing established SSI risk factors rather than replacing them. These findings address a clinically relevant gap in current risk assessment: conventional markers such as BMI and a binary diabetes diagnosis may not fully reflect subclinical insulin resistance, whereas TyG offers a continuous and readily available metric that may better capture the spectrum of metabolic vulnerability in surgical candidates.
The mechanisms by which a higher TyG index predisposes to SSI are likely multifactorial. Insulin resistance and chronic hyperglycemia impair neutrophil chemotaxis, phagocytosis, and oxidative burst, thereby weakening first-line host defenses at the surgical site.27,28 Hypertriglyceridemia and associated lipotoxicity contribute to endothelial damage and microvascular dysfunction, which can compromise tissue oxygenation and antibiotic penetration within the wound bed.29,30 In addition, the TyG index captures aspects of metabolic stress that are not fully represented by fasting glucose or HbA1c alone, including post-prandial excursions and hepatic insulin resistance.10,31 In our cohort, TyG remained independently associated with SSI after adjustment for clinically diagnosed diabetes, suggesting that it may reflect “hidden” metabolic vulnerability even in patients without established diabetes or in those with apparently adequate glycemic control. The graded increase in TyG across no SSI, superficial, deep, and organ/space infection further supports a dose–response relationship between the severity of metabolic derangement and the depth and extent of postoperative infection. Notably, this association was observed despite institutionally standardized perioperative glycemic management (target approximately 100–180 mg/dL in noncritically ill patients), implying that the preoperative metabolic milieu captured by TyG may convey risk beyond perioperative glucose targets alone. Given the biological proximity between TyG and diabetes, some overlap is expected; therefore, we intentionally used a parsimonious primary model and confirmed robustness in a bias-reduced expanded model.
The predictive analyses suggest that TyG has practical value for risk stratification. As a continuous variable, TyG exhibited a roughly linear relationship with SSI risk across the observed range. For clinical decision-making, dichotomization at an empirically derived cut-off can be useful. In our data, a TyG value of 8.80 provided a reasonable trade-off between sensitivity (0.79) and specificity (0.58), with a clear separation in infection rates between low- and high-TyG groups (2.5% vs 11.8%). Because SSI events were uncommon, the positive predictive value at this threshold was modest (PPV 0.12) whereas the negative predictive value was high (NPV 0.98). Consistently, the likelihood ratios (LR+ 1.89; LR− 0.36) suggest that the cut-off is more useful for identifying patients at very low risk (rule-out) than for confidently confirming SSI risk (rule-in). Such a simple index, readily obtainable from routine fasting blood tests, could therefore be incorporated into preoperative risk discussions and used to flag patients who might benefit from intensified metabolic optimization, stricter perioperative glycemic control, more aggressive wound surveillance, or prolonged antibiotic prophylaxis, although these strategies require prospective evaluation. Importantly, the 8.80 threshold is data-driven and should be externally validated across different populations and care pathways before being adopted for routine clinical triage.
The association between TyG and SSI subtypes also has clinical and biological implications. While superficial incisional infections are often manageable with local care and short-term oral antibiotics, deep and organ/space infections can necessitate multiple debridements, prolonged intravenous therapy, and, in some cases, implant removal or nonunion. The finding that patients with deep or organ/space SSI had the highest TyG values and that the TyG–infection-depth correlation among infected patients was moderate to strong suggests that metabolic dysfunction may influence not only the likelihood of bacterial contamination progressing to infection, but also the ability of the host to contain infection to superficial tissues. 32 However, because deep and organ/space infections were uncommon, these subtype and depth analyses should be interpreted as hypothesis-generating rather than definitive evidence that TyG predicts infection severity. This observation supports a paradigm in which TyG is viewed not merely as a binary risk flag but as a continuous marker of impaired host defense and tissue repair capacity.
Nevertheless, the findings should be interpreted in light of certain limitations. First, this was a retrospective analysis from a single institution, which may limit generalizability to other practice settings, surgical approaches, or patient populations. External validation in independent cohorts and in different types of spine surgery will be important. Second, the outcome window was restricted to 30-day SSI. While this is consistent with surveillance for superficial incisional SSI and aligns with our focus on early postoperative infections, it may underestimate later-presenting deep or organ/space infections after instrumentation; thus, our results should be interpreted as pertaining primarily to early SSI risk. Third, this was a single-center retrospective study, which may introduce selection bias and temporal bias, thereby limiting the generalizability of our findings to other settings. Multicenter prospective validation studies may be further conducted in the future. Fourth, pre-existing dermatological or skin conditions could plausibly influence incision healing and susceptibility to postoperative infection. However, detailed dermatologic histories were not uniformly captured as structured variables in our retrospective dataset, precluding reliable adjustment and leaving the possibility of residual confounding. This factor should be incorporated into future prospective spine SSI registries and risk models. Additionally, TyG was calculated from a single preoperative measurement; intra-individual variability and perioperative changes in triglyceride and glucose levels were not captured. Serial assessment might provide additional insight into dynamic metabolic risk. Moreover, the number of deep and organ/space infections was relatively small, and estimates for these subgroups should therefore be interpreted with caution, despite the clear qualitative pattern observed. Finally, the study was not designed to test whether modifying TyG through lifestyle or pharmacologic interventions can reduce SSI risk, such causal relationships remain speculative.
Conclusions
In summary, this study demonstrates that an elevated preoperative TyG index is independently associated with the occurrence of 30-day SSI following posterior lumbar fusion. TyG provides moderate discriminative ability as a stand-alone predictor and identifies a subset of patients at substantially higher risk of deep and organ/space infections. Given its simplicity, low cost, and strong biological rationale as a surrogate of insulin resistance and metabolic dysfunction, TyG may represent a useful addition to existing SSI risk assessment tools in spine surgery, but the empiric cut-off and overall performance require external validation. Future prospective, multicenter studies should validate these results, refine TyG-integrated prediction tools, and test whether preoperative metabolic optimization can reduce SSI risk and improve postoperative outcomes.
Supplemental material
Supplemental material - Preoperative triglyceride–glucose index as a metabolic predictor of surgical site infection after posterior lumbar fusion
Supplemental material for Preoperative triglyceride–glucose index as a metabolic predictor of surgical site infection after posterior lumbar fusion by Yu Hua, Shaoxing Li, Yuan Jiang, Jinwang Liu in Journal of Orthopaedic Surgery
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated and analyzed during the current study are not publicly available because of institutional privacy regulations but are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

