Abstract
Total hip and knee arthroplasty are common surgical procedures aimed at improving mobility and quality of life. Despite surgical advances, postoperative complications remain a concern, particularly for patients with comorbidities and poor nutritional status. Immunonutrition, the targeted administration of nutrients that modulate inflammation, such as arginine, omega-3 fatty acids, and antioxidants, has been shown to reduce infections, complications in wound healing, and shorten hospital length of stay. This review examines current evidence on the role of immunonutrition in hip and knee arthroplasty, focusing on the mechanisms of key nutrients, clinical application of supplementation, and areas for future investigation. Despite promising outcomes, further research is needed within orthopedics to establish guidelines for patient screening, nutrient dosage, and timing of administration.
Keywords
Introduction
Nutrition is becoming increasingly relevant within the literature for orthopedic surgery patients. Surgical outcomes continue to gain importance, particularly in total hip arthroplasty (THA) and total knee arthroplasty (TKA), in part due to the increased amount performed annually. 1 There are ongoing concerns regarding postoperative complications and healthcare-related costs, highlighting the need to identify risk factors and optimize perioperative care. For instance, surgical wound complications following THA and TKA increase the risk of postoperative infections, which contribute to prolonged hospital stays, higher readmission rates, and elevated procedural costs.2,3 Although outpatient total joint arthroplasty (TJA) procedures have been introduced to reduce costs, they can pose a heightened risk of complications, particularly in high-risk patients with comorbidities, such as malnutrition, diabetes, or cardiovascular disease.4,5 These challenges, along with the increasing prevalence of osteoarthritis, highlight the need for perioperative and postoperative strategies that target modifiable risk factors to improve surgical outcomes associated with THA and TKA.
Orthopedic surgeries, including THA and TKA, are known to trigger catabolic and localized inflammatory stress responses, potentially progressing to systemic inflammation and activation of the innate immune system.6,7 The innate immune system plays a central role in regulating the inflammatory response following joint prosthesis implantation.8,9 These local and systemic inflammatory responses can impair immune function, delaying tissue repair. Many orthopedic patients, particularly geriatric patients with pre-existing comorbidities, are subclinically malnourished.10,11 This insufficiency of essential nutrients in the body compromises immune function and wound healing, which increases the risk of postoperative complications.12,13 TJA can trigger an intense immunological and physiological stress response due to surgical trauma, anesthesia, perioperative interventions, and increase the potential for complications. These physiological and immunological responses include immune activation, inflammation, and neuroendocrine changes, which are fundamental for healing. When prolonged and dysregulated, this may result in delayed recovery, poor wound healing, and extended hospital length of stays, potentially increasing the likelihood of developing nosocomial infections.14,15 Given these factors, supportive adjuvant strategies, such as immunonutrition, may be valuable in modulating inflammation, reinforcing immune function, and optimizing surgical outcomes. Therefore, an evaluation of the inflammatory and immune mechanisms, neuroendocrine activation patterns, and surgical considerations is key when considering the relationship between malnutrition and inflammation, and highlights the impact of nutrition on orthopedic surgeries, especially in THA and TKA.
To help address the role of nutrition regarding surgical outcomes, immunonutrition – the targeted administration of nutrients that modulate inflammation – can be incorporated into perioperative care, and has been shown to promote wound healing and reduce the risk of postoperative infections.16,17 Specific nutrients, such as amino acids, omega-3 fatty acids, and certain vitamins, can reduce pro-inflammatory cytokines and enhance immune function, which can improve orthopedic surgical outcomes. 18
Historically, immunonutrition has been investigated extensively in oncologic and gastrointestinal surgeries.19–21 Findings suggest that the utilization of immunonutrition interventions has been associated with reduced infection rates and enhanced postoperative recovery. Therefore, immunonutrition considerations present a promising therapy for patients undergoing orthopedic surgery with the potential to reduce both postoperative complications and healthcare costs. Despite positive results in other fields, its application in orthopedic surgery remains limited. As there are no current standardized regimens within orthopedic surgery, this review aims to address a critical gap in the existing literature, and identify a role for immunonutrition in orthopedic perioperative and postoperative care in TJA.
Methods
This study is a narrative review that aims to summarize and explore the role of perioperative nutrition in improving outcomes following total hip arthroplasty (THA) and total knee arthroplasty (TKA), with a focus on immunonutrition and amino acid supplementation.
A comprehensive literature search was conducted using PubMed, EMBASE, Web of Science, and Google Scholar databases for articles published between 1999 and 2025. The search strategy encompassed combinations of keywords and Boolean operators, such as “total joint arthroplasty,” “total knee arthroplasty,” “total hip arthroplasty,” “immunonutrition,” “surgical outcomes,” “perioperative nutrition,” and “orthopedic surgery.” Additional articles were identified through manual searches of reference lists from relevant literature. Titles and abstracts were initially screened to identify relevant studies, and full-text articles were then reviewed according to the inclusion and exclusion criteria described below.
Studies were eligible for inclusion if they were clinical trials, observational studies, systematic reviews, or meta-analyses that involved adult patients undergoing THA or TKA. These studies were evaluated further to include only those that investigated immunonutrition formulations or amino acid supplementation and reported clinical outcomes, such as infection rates, inflammatory markers, wound healing, muscle strength, or length of hospital stay.
Studies were excluded if they were non-peer-reviewed articles, case series, or case reports, or if they involved surgical procedures other than THA and TKA. Studies were also excluded if they were unrelated or lacked specific immunonutrition or amino acid supplementation, and mechanistic or laboratory-based studies without clinical outcomes.
Five studies met the inclusion criteria and were included in the narrative synthesis, focusing mainly on their reported effects on postoperative outcomes. Additional literature was examined to provide context and background on perioperative nutrition in THA and TKA. The workflow of the literature search and study selection process is summarized in Figure 1. Literature search and study selection workflow. This flowchart outlines the steps used to identify, review, and select studies for the narrative synthesis.
Total joint arthroplasty and immunonutrition
Inflammatory and immune response
Orthopedic surgeries elicit an acute inflammatory response characterized by the activation of various inflammatory markers and acute-phase reactants.
22
Key cytokines, such as Interleukin 1 (IL-1), Interleukin 6 (IL-6), and Tumor Necrosis Factor-alpha (TNF-α), play essential roles in coordinating pathogen clearance, promoting inflammation, and ultimately initiating repair and resolution.
22
Prolonged inflammation leads to an imbalance between pro-inflammatory and anti-inflammatory cytokines, such as interleukin 10 (IL-10) and Transforming Growth Factor-beta (TGF-β), which may result in insufficient regulation of immune responses and contribute to chronic tissue damage and disease.
23
This imbalance can result in delayed wound healing, periprosthetic joint infections, oxidative stress, tissue necrosis, and systemic infections, as outlined in Figure 2.
24
The persistence of this imbalance is critical to recognize, as it can lead to poor outcomes for patients. Inflammatory cascade following surgical trauma in arthroplasty. This flow diagram summarizes the key stages from surgical trauma triggering cytokine release and immune cell recruitment, to dysregulated inflammation causing oxidative stress and reduced collagen synthesis, leading to clinical consequences such as delayed wound healing and increased infection risk.
Persistent inflammation is clinically detrimental as it impairs normal tissue repair, mobility, and recovery. For example, patients undergoing revision surgery many years after failed TKAs exhibited elevated levels of active pro-inflammatory mediators. 25 These mediators limit recovery as they induce skeletal muscle proteolysis, delay wound healing, and increase the risk of periprosthetic joint infections. 24 This evidence suggests that the knee exists in a state of chronic inflammation in patients with poor outcomes following TKA. Further, there is an association between sustained inflammation and the development of fibrosis and stiffness, which manifests as a restricted range of motion. 25 Therefore, unresolved inflammation plays a critical role in recovery as it can delay muscular and wound healing, increase the risk of infection, and the development of pain and stiffness, all of which lead to poor outcomes. While acute inflammation is necessary for tissue repair, persistent inflammation can be harmful through the prolonged activation of leukocytes and other immune cells.
Inflammation begins with the recruitment and activation of leukocytes, particularly neutrophils and macrophages, and the release of proinflammatory cytokines. Neutrophils contribute to microbial clearance and the removal of damaged tissues via phagocytosis, proteolytic enzymes, cytokines, and reactive oxygen species (ROS). While critical in the short-term, excessive ROS and proteolytic enzyme activity can result in collateral tissue damage and prolonged inflammation.26,27 Macrophages similarly play a significant role in inflammation as well as resolution of infection and inflammation. The M1 immune response releases cytokines and acts as an antigen presenter to CD4 T-Helper cells to promote immune activation. Alternatively, the M2 immune response secretes IL-10 and TGF-β – anti-inflammatory and restorative cytokines – known to promote tissue remodeling, angiogenesis, and inflammation resolution. 28 Discordance between M1 and M2 immune responses is posited to be linked to poor wound healing and increased susceptibility to postoperative complications, especially in patients with comorbidities. 29 While the immune and inflammatory responses are necessary for tissue repair, infection control, and resolution, their imbalance can contribute to postoperative complications. Therefore, it is important to understand the dynamic between pro- and anti-inflammatory signals to optimize recovery, support immune resolution, and maximize favorable outcomes.
Neuroendocrine activation
Total joint arthroplasty (TJA) can elicit a significant neuroendocrine response by activating the hypothalamic-pituitary-adrenal (HPA) axis, resulting in increased cortisol and catecholamine levels. 30 This occurs through the activation of afferent nerves at the surgical site, which stimulate the hypothalamus to release corticotropin-releasing hormone (CRH) and arginine vasopressin (AVP). 30 In turn, both hormones work cooperatively to activate the secretion of adrenocorticotropic hormone, ultimately leading to the release of cortisol from the adrenal cortex. 30 Long-term activation suppresses the immune system, leading to increased infection risk, reduced tissue repair, and disrupted metabolism. 31 In addition, increased levels of cortisol activate gluconeogenesis and proteolysis, leading to pronounced protein turnover and a depletion of nitrogen, which is essential for protein synthesis and muscle preservation.32,33 Overall, this response serves an initial protective and anti-inflammatory function. However, prolonged cortisol elevation can impair immune function, hinder tissue regeneration, and disrupt metabolic balance (as shown in Figure 2).
Surgical and patient factors
Total joint arthroplasty (TJA), which includes procedures like total knee arthroplasty (TKA) and total hip arthroplasty (THA), often has substantial bone and soft tissue trauma, which can impact the local immune environment and healing dynamics. 17 These specific operations, in combination with individual patient factors, can impact postoperative recovery and outcomes. Specifically, patients with osteoporosis, diabetes, obesity, malnourishment, and other chronic conditions can have an increased risk of complications. These comorbidities are often related to the patient’s underlying immunonutritional status, highlighting the value of adjuvant immunonutrients in these settings to enhance host defenses and attenuate inflammation. A systematic review conducted by Gregori et al. emphasized the importance of tailored immunonutrition protocols to promote collagen synthesis and regulate systemic inflammation to prevent implant rejection. 17 For this reason, the administration of immune and inflammatory modulators serves to maintain an appropriate physiological stress response that may optimize postoperative outcomes, which should be considered in patient populations with greater risk factors.
Key nutrients and relevance in arthroplasty
Immune and inflammatory modulators, such as omega-3 fatty acids, arginine, glutamine, antioxidants, and nucleotides, are essential in limiting surgical stress in total joint arthroplasty (TJA). Each of these immunonutrients has specific mechanisms of action that contribute to reducing inflammation and promoting recovery. Findings from other fields, such as oncologic and gastrointestinal surgery, have shown that these mechanistic factors improve postoperative outcomes. However, their relevance to arthroplasty remains limited. Insights from these other surgical fields may help clarify the potential role of immunonutrition supplementation in TJA.
Omega-3 fatty acids are known for their anti-inflammatory properties, specifically, their active components, eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA). 34 They reduce pro-inflammatory eicosanoids, downregulate the production of Tumor Necrosis Factor-alpha (TNF-α), and stimulate glutathione production, which produces a shift from a pro-inflammatory to an anti-inflammatory state.34,35 Regarding arthroplasty, this shift translates into decreased inflammation and pain, which may shorten hospital stays and facilitate early mobilization.
Arginine is a non-essential amino acid that is crucial for collagen synthesis, enhancing immune function, and nitric oxide synthesis, leading to vasodilation. 36 Surgery can lead to a disturbance in arginine metabolism, which can cause its levels to drop ≥50% within a few hours after surgery. 37 Thus, it has the potential to be important for supplementation in the recovery period. 36 The combination of improved blood flow and collagen synthesis may play a supportive role in wound healing after arthroplasty, leading to decreased wound complications and faster recovery.
Glutamine is a non-essential amino acid that protects against inflammation, fuels rapidly dividing epithelial cells, and preserves muscle mass. 36 Its depletion has been associated with impaired muscle repair and regeneration due to the lack of substrates for protein synthesis. 36 Therefore, supplementation of glutamine has been shown to restore these substrates, which have been shown to promote wound healing, limit inflammation and stress, and prevent muscle atrophy. The enhancements seen postoperatively may decrease wound healing complications, shorten hospital length of stay, and accelerate recovery.
Antioxidants, such as vitamin C, vitamin E, zinc, and selenium play an important role in minimizing cellular damage from the production of reactive oxygen and nitrogen species. 35 TJA can produce excessive reactive oxygen species, which damage healthy cells, proteins, and lipids, resulting in oxidative stress. 35 Antioxidants can combat the damage by neutralizing free radicals, maintaining the integrity of cell membranes, and promoting tissue repair after surgical stress. 35 Altogether, antioxidants play a supportive role in limiting postoperative damage and aiding in recovery after arthroplasty.
Vitamin D and calcium are essential in supporting bone health and recovery after arthroplasty. 38 Specifically, vitamin D acts to maintain bone health through the absorption and regulation of calcium and phosphorus, while calcium is critical for bone mineralization.38,39 The combination of vitamin D and calcium acts cooperatively to maintain bone health, implant stability, and facilitate healing postoperatively.
Nucleotides serve as building blocks for deoxyribonucleic acid (DNA), ribonucleic acid (RNA), and adenosine triphosphate (ATP). They play an important role in cell proliferation and immune modulation, primarily through the enhancement of lymphocyte blastogenesis. 34 As a result of adequate nucleotide availability, which may involve supplementation, the immune system has the necessary building blocks to respond efficiently to the demands of surgical stress. Clinically, this can improve wound healing, shorten hospital length of stay, and facilitate recovery after arthroplasty.
Key nutrient summary and takeaways.

Abbreviations: EPA, eicosapentaenoic acid; DHA, docosahexaenoic acid; TNF-α, tumor necrosis factor-alpha; DNA, deoxyribonucleic acid; RNA, ribonucleic acid; ATP, adenosine triphosphate; GI, gastrointestinal;
Nutritional screening, patient selection, and timing for immunonutrition
Immunonutrition can be highly beneficial in orthopedic surgery, especially when administered to high-risk, malnourished patients. The incidence of malnutrition varies by patient population, with those most at risk including geriatric patients, trauma patients, and those with obesity. 40 For instance, geriatric patients have been found to have high rates of malnutrition at 24%–32%. 40 Although those with the highest risk are similar across surgical specialties, the prevalence of malnutrition among patients undergoing total joint arthroplasty (TJA) varies in the literature. Some studies report rates as high as 50%, while one study focusing on TJA in patients under 55 years old found a malnutrition rate of only 1.7%.41,42 Patients undergoing TJA who have diagnoses of malnutrition and obesity have three times the rate of periprosthetic joint infections and overall complications compared to those with normal nutritional status. 43 In addition, an estimated 48% of people worldwide are vitamin D deficient, when considering the clinical cutoff of <50 nmol/L. 44 These individual risk factors contribute to a higher incidence of wound healing issues, infection, increased length of stay, renal complications, and mortality. 40 Therefore, evaluating malnutrition status is an important consideration for patients undergoing TJA as it identifies those who may be susceptible to postoperative complications.
Given its importance in identifying at-risk patients, nutritional screening in TJA can be performed through various measures, including laboratory tests, anthropometric values, and screening tools. Serologic laboratory markers, such as albumin, prealbumin, transferrin, and total lymphocyte count, are commonly used to evaluate nutritional status preoperatively. 40 While each is linked to predicting postoperative complications and has its limitations, several studies report that albumin is the most widely studied and reliable marker.40,45,46 Albumin, prealbumin, and transferrin are negative acute-phase proteins, which means their plasma concentrations decrease typically by 25% during inflammatory states, such as after surgery. 47 This decrease can limit their reliability as nutritional markers during inflammation, potentially providing a false impression of malnutrition. Other measures can also be used to evaluate body composition and nutritional status, which include body weight, body mass index (BMI), mid-upper arm/calf circumference, and triceps skinfold thickness. 48 Further, various screening tools can be utilized that place additional focus on patient history and physical examination. Those most used in the literature and clinical settings include the Nutritional Risk Screening 2002 (NRS-2002), Subjective Global Assessment (SGA), Mini Nutritional Assessment (MNA), and Malnutrition Universal Screening Tool (MUST). 40 Each screening tool examines specific patient populations and core components, which can produce variability in their findings. Ultimately, there is no consensus regarding the best screening tool. Therefore, to obtain a greater understanding of a patient’s nutritional status, it is important to consider serologic laboratory markers, a patient’s history, their physical examination, and ultimately use clinical judgment for screening selection.
Determining which patients should undergo nutritional screening and receive immunonutrition has not been established in the literature. There is ongoing controversy as to whether screening should be performed routinely for all patients or selectively based on individual factors. Some studies have recommended selective nutritional screening for those with high-risk characteristics, such as diabetes mellitus, high BMI, and those undergoing revision arthroplasty.49,50 In contrast, several studies have advocated for routine nutritional screening, given the numerous risk factors present in patients undergoing TJA at baseline.42,51,52 Phillips et al. recommend that selection and screening be performed at the surgeon’s discretion using methods that best fit their practice. 43 Once a patient is screened and found to have a nutritional imbalance, it is beneficial to consider administering immunonutrition interventions, such as vitamin supplement education or prescriptions. Most studies recommend administration of immunointerventions within 5–10 days before the day of surgery.53,54 However, there is variation in postoperative use and no consensus in the orthopedic literature currently.53,54 Ultimately, appropriate screening and administration of immunonutrition can be an effective perioperative tool to reduce the risk of complications in TJA. Due to the lack of clinical guidelines and consensus, immunonutrition screening and treatment should be performed at the surgeon’s discretion.
Clinical impact of immunonutrition on surgical outcomes
Immunonutrition supplementation and surgical outcomes in THA/TKA patients.
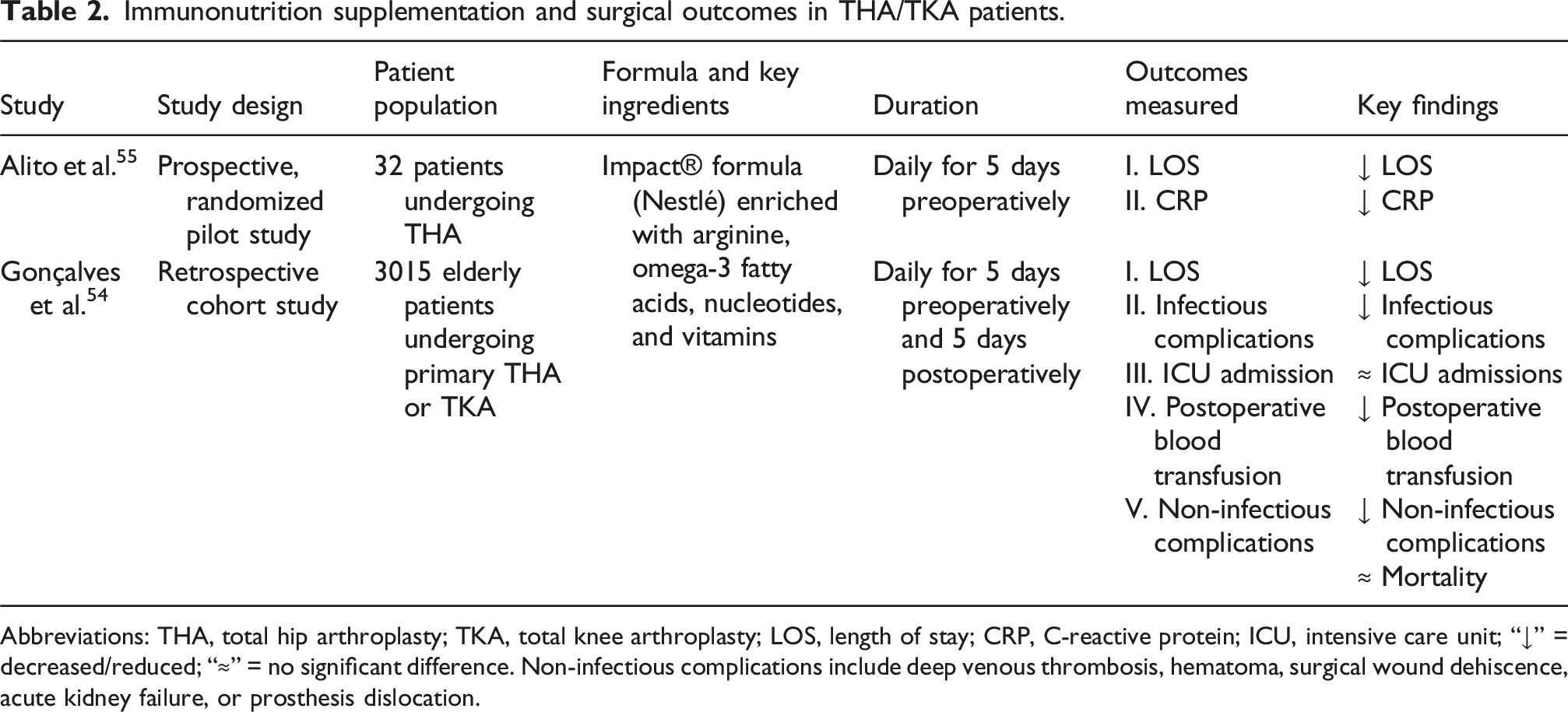
Abbreviations: THA, total hip arthroplasty; TKA, total knee arthroplasty; LOS, length of stay; CRP, C-reactive protein; ICU, intensive care unit; “↓” = decreased/reduced; “≈” = no significant difference. Non-infectious complications include deep venous thrombosis, hematoma, surgical wound dehiscence, acute kidney failure, or prosthesis dislocation.
As shown in Table 2, patients who received an immunonutrition intervention had better postoperative outcomes compared to their respective control group.54,55 According to Alito et al., patients had a significantly shortened length of stay (LOS) and maintained lower C-Reactive Protein (CRP) values. 55 In particular, the immunonutrition group had 17.5% lower CRP values (80.6 vs 66.5 mg/L, p < 0.01) and an average length of stay of three days (range two to five days), which was three days shorter than the control group (median six days, range three to eight days; p < 0.01). 55 While not included in their outcome variables, no patients in either group developed an infection, required a postoperative blood transfusion, subsequent surgical intervention, or re-admission. 55 Although generalizability is limited due to the study being a pilot with a small sample size, similar outcomes are reported in the study by Gonçalves et al., featuring a much larger patient population (see Table 2).
In the study by Gonçalves et al., patients had significantly decreased LOS, infection-related complications, and postoperative blood transfusions. More specifically, the immunonutrition intervention group had a 42% lower mean LOS in hours (32.0 ± 19.4 vs 56.0 ± 26.4; p < 0.001) and lower rates of infectious complications (2.2% vs 4.6%; p < 0.001). 54 Regarding secondary outcomes, the immunonutrition group had 55% lower CRP values, 55% reduction in the chance of infectious complications, 50% reduction in the chance of non-infectious complications, and a 75% reduction in the chance of postoperative blood transfusion. 54 However, there were no significant differences between immunonutrition and control groups for intensive care unit (ICU) admission and 90-days mortality. 54 These findings suggest that supplementation with immunonutrients can have a significant enhancement on postoperative recovery. While there are promising but limited studies on immunonutrition supplementation in elective total joint arthroplasty (TJA), other nutritional studies have demonstrated comparable findings in TJA.
Amino acid supplementation and surgical outcomes in TKA patients.

Abbreviations: TKA, total knee arthroplasty, AAs, amino acids, “↓” = decreased, “↑” = increased.
Improved postoperative clinical outcomes can be appreciated in all three studies seen in Table 3. Patients receiving amino acid supplementation consistently experienced less postoperative muscle atrophy and accelerated return to functional mobility.56–58 When compared to the control groups, supplemented groups showed either no significant muscle atrophy or significantly less muscle atrophy, greater increases in quadriceps muscle area, and faster return to functional mobility.56–58 Although the included studies are distinct from typical immunonutrition compositions, the findings reflect a similar postoperative trend. Supplementation with immunonutrients consistently demonstrates improved outcomes through the modulation and enhancement of immune and inflammatory pathways (as shown in Figure 3). For this reason, the role of nutritional supplementation during orthopedic surgeries, such as TJA, should be considered, as it can alter the trajectory of a patient’s recovery. Inflammatory cascade modulated by immunonutrition in arthroplasty. Immunonutrition attenuates excessive cytokine release and supports balanced immune cell recruitment following surgical trauma. These effects promote controlled inflammation with reduced oxidative stress and enhanced collagen synthesis, leading to accelerated wound healing and reduced infection risk.
Challenges and limitations
A thorough discussion of the role of immunonutrition in optimizing total hip arthroplasty (THA) and total knee arthroplasty (TKA) would be incomplete without a dive into current limitations and challenges to applicability. While immunonutrition in oncologic patients has been well documented, applications to orthopedic surgery patients remain limited, particularly in the total joint arthroplasty (TJA) literature. This creates a challenge for orthopedic professionals seeking guidance regarding perioperative nutrition optimization in these patients, as protocols are relatively new and varied. In contrast, several countries in Europe have long-standing guidelines on immunonutrition for patients undergoing surgery for gastrointestinal cancer.59–63 Still, there are promising protocols underway for perioperative nutrition in surgical patients. 64 Duke University developed the Perioperative Nutrition Score (PONS) to screen for malnutrition risk in preoperative patients. During preoperative phone screening, if patients answer “Yes” to the question “Have you lost more than 8–10 pounds in the last 6 months without trying?”, they undergo further screening at an in-person office visit. 64 During this visit, their PONS score is assessed based on the following criteria: Body Mass Index (BMI) <18.5 (BMI <20 if age is >65), weight loss >10% in the last 6 months without trying, oral intake <50% of the normal diet in the last week, and serum albumin <3.0. 64 Any positive (i.e., “Yes”) responses are escalated to “high-risk” status for surgical malnutrition and are subsequently referred to a registered dietitian. In the high-risk nutrition care pathway, patients receive high-protein oral nutrition supplements for two to four weeks prior to surgery, immunonutrition supplements for five days prior to surgery, and a complex carbohydrate loading drink two hours prior to anesthesia. 64 After surgery, these patients continue to be followed and treated for their nutritional status. Post-operatively, patients receive one week of immunonutrition and one month of high-protein oral nutritional supplements. 64 Screening tools, along with a multidisciplinary approach incorporating dietitians, have the potential to improve patient outcomes if implemented on a larger scale.
Although immunonutrition has demonstrated statistically significant reductions in the costs associated with postoperative complications and is generally considered cost-effective, accessibility concern persists (as shown in Figure 4).65,66 Most of the literature surrounding immunonutrition’s cost caters to research from larger, academic hospitals in high-income settings. This focus on higher income and resource-rich settings limits the generalizability of findings. Particularly, individuals in rural or resource-limited settings may face challenges, including unreliable transportation for multiple visits, inconsistent supply of immunonutrition supplementation, and challenges to a multidisciplinary approach to treatment in areas where nutritional specialists are not available. Without addressing these systemic and structural barriers, the benefits of immunonutrition can remain limited for patients who may need it most. Barriers to immunonutrition supplementation in total joint arthroplasty. Implementation of perioperative immunonutrition is hindered by multiple factors, including limited clinical evidence in orthopedics, health literacy gaps, systemic barriers, and accessibility concerns.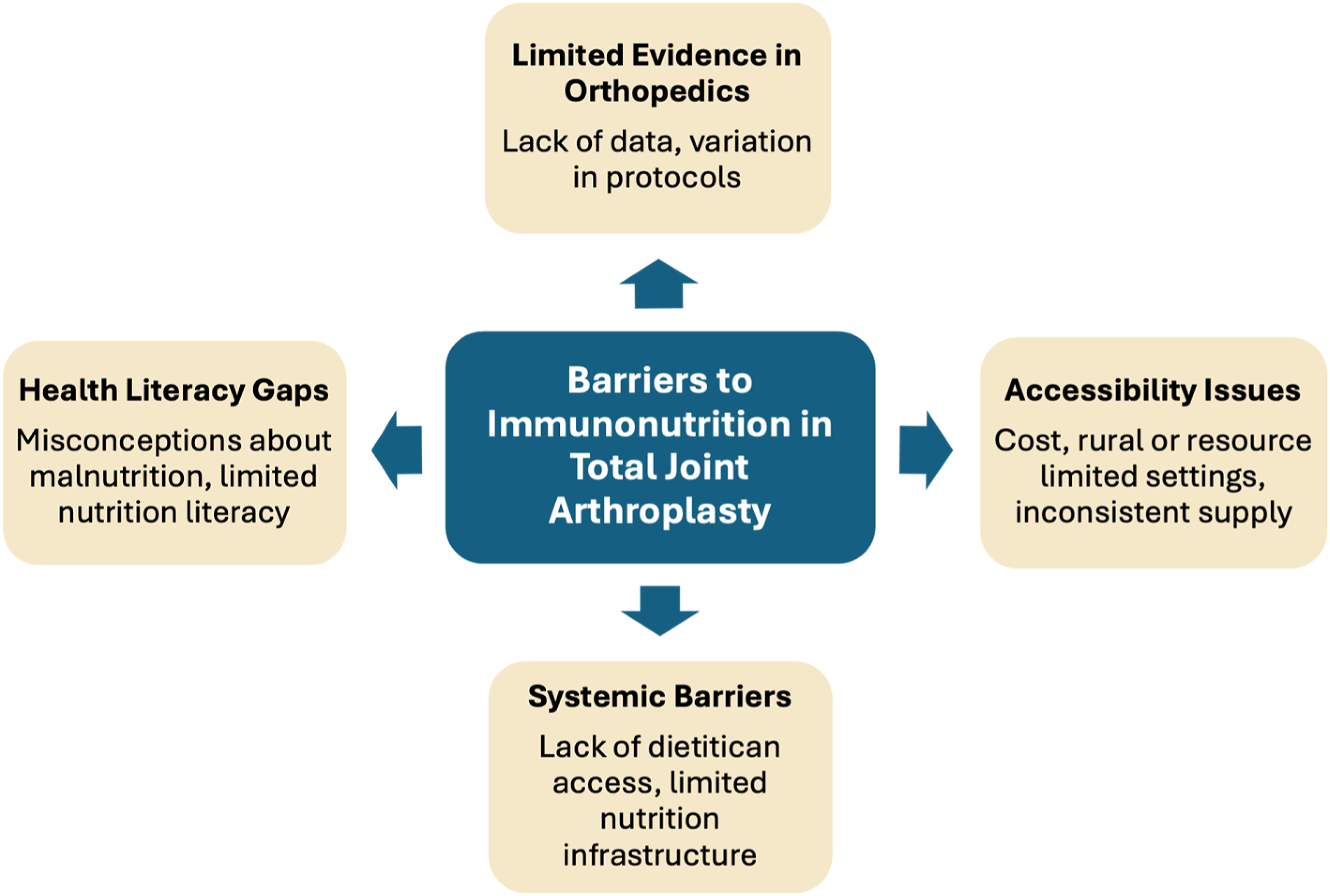
Health literacy is an additional challenge to addressing malnutrition for surgical patients. Though several definitions of health literacy have existed, it is currently defined as the ability to find, understand, and use information and services to inform health-related decisions and actions for themselves and others. 67 Poor health literacy is a key contributor to why malnutrition often goes unnoticed. 50% of patients undergoing joint or spinal surgery are at risk of malnutrition, are already malnourished, or will become malnourished during postoperative recovery. 68 Misconceptions regarding what malnutrition looks like (i.e., thinner body habitus, low weight) can lead to underdiagnosis in patients with higher caloric intake and lower nutritional value. While immunonutrition in the short-term perioperative period has potential for improving recovery outcomes, limited nutrition literacy can pose a barrier to long-term overall health. The Newest Vital Sign is an interviewer-administered health literacy assessment tool in which patients read an ice cream nutrition label and subsequently answer six questions about the content. Answering fewer than four of six questions correctly may indicate limited literacy. 69 This assessment tool, which has been used for almost two decades, has implications for guiding future patient-physician conversations regarding nutrition. Though plain language is always recommended when interacting with patients, for patients with lower health literacy, healthcare providers may also incorporate the “teach back” method and visual imagery to ensure thorough patient understanding. 67 Evaluating health literacy, considering accessibility to care, and identifying and addressing malnutrition are essential and may involve interdisciplinary support, including nutritionist referrals, especially in the perioperative setting.
Future directions
Future research is needed to further clarify the role of immunonutrition in the perioperative setting, the possible benefits of standardization, and cost-effective mechanisms to eliminate the gap between socioeconomic status and available medical care. While not conducted specifically in orthopedic populations, multiple studies have shown that incorporating preoperative immunonutrition into Enhanced Recovery after Surgery (ERAS) protocols can reduce complications, shorten hospital length of stays, and lower costs.70,71 Future research should examine its application within orthopedic ERAS protocols and consider complementary interventions that could enhance its effects.
Additionally, few orthopedic surgeons receive training or education on immunonutrition, reflecting the current state of professional guidelines, which often omit such recommendations. The broader educational landscape shows similar gaps: nutrition education during medical school, residency training, and practice often remains rudimentary, inconsistent, and insufficient.72,73 Incorporating immunonutrition into the professional guidelines of the American Academy of Orthopaedic Surgeons (AAOS) could provide an important educational foundation and facilitate its adoption in orthopedic practice, with the European Society for Clinical Nutrition and Metabolism (ESPEN) serving as a model, as its guidelines explicitly endorse perioperative immunonutrition for high-risk surgical patients. 74
Other nutritional strategies also warrant consideration. High-protein supplementation and anti-inflammatory diets have shown clinically meaningful benefits in malnourished patients undergoing total joint arthroplasty (TJA), including decreased muscle atrophy, improved muscle function, and reduced length of stay.75,76 These findings support that comprehensive nutritional approaches may improve perioperative outcomes and represent a promising direction for continued study.
Nutritional screening within orthopedics has demonstrated measurable clinical and economic benefits. Torchia et al. conducted the first cost-effectiveness analysis of malnutrition screening and treatment before total knee arthroplasty (TKA), reporting an incremental cost-effectiveness ratio of $6,454 per quality-adjusted life year (QALY) gained. 77 This equates to $6,454 for each extra year of life lived in good health, which is considered highly cost-effective in most health care systems. This highlights the potential value of integrating nutritional screening into orthopedic care, as it improves patient outcomes while simultaneously lowering costs. Although not commonly used in orthopedic settings, the Geriatric Nutritional Risk Index (GNRI) was used in a recent study by Fujita et al., which found that patients classified as at-risk experienced significantly poorer postoperative muscle strength following total hip arthroplasty (THA). 78 Incorporating GNRI could help identify elderly patients at risk for delayed recovery, suggesting it may be a worthy tool for future consideration alongside more widely used nutritional screening methods. Together, these studies demonstrate the ongoing need for improved recommendations, as a central feature of ERAS is the development of evidence-based protocols, including unified approaches to nutritional screening and assessment. Later strategies may be optimized beyond standardized formulations, such as the Impact® formula (Nestlé), with personalized immunonutrition approaches tailored to individual biologic profiles. Additionally, biomarkers, such as Nucleotide-binding Oligomerization Domain 1 (NOD-1), C-Reactive Protein (CRP), and Interleukin 6 (IL-6), may assist in assessing anti-inflammatory response to immunonutrition.79,80 Limited current use and research on immunonutrition in orthopedics underscores the urgency of further investigation to guide practice and optimize outcomes.
Socioeconomic status and geographical location are important factors that influence access to immunonutrition and warrant further investigation to address these disparities. Accessibility concerns align with a broader pattern of inequities in postoperative outcomes. For instance, Upfill-Brown et al. found that Black and Hispanic patients experience higher rates of venous thromboembolism (VTE) and major complications following TKA, THA, or both. 81 These findings were suggested to stem from reduced access to care among other possible factors. 81 Similarly, malnutrition itself has been shown to increase risks of wound complications and surgical site infections following total joint arthroplasty.82,83 Affordability is another barrier, as low-income patients may struggle to pay for over-the-counter vitamins or supplements, raising concern that nutritional recommendations could create added financial strain.84,85 Ignoring disparities may exacerbate existing inequalities in healthcare access and widen health outcome gaps, especially as immunonutrition may inadvertently worsen these inequities if issues of equitable access, delivery, and implementation are not addressed. To mitigate this, future strategies should prioritize equitable access, affordability, and context-sensitive delivery of immunonutrition, especially for patients in underserved or resource-limited settings. This includes integrating patient-centered approaches, strengthening multidisciplinary care models, and ensuring the availability of trained nutritional support across diverse healthcare environments. In addition, future studies should assess whether orthopedic surgeons could play a role in improving patient access to nutritional information, such as providing standardized education materials or facilitating referrals for nutritional counseling, as these strategies may enhance patient understanding, adherence, and outcomes. These efforts must be prioritized to ensure that all segments of the population can benefit from immunonutrition interventions. Successful integration of immunonutrition into orthopedic surgery protocols represents an area of significant potential, but it will depend on future collaborative research, standardization of care, and equitable implementation to ensure widespread and inclusive adoption.
Conclusion
Perioperative care should consider immunonutrition as patients undergoing total joint arthroplasty (TJA) are more susceptible to an altered immune environment, complicating postoperative recovery. Challenging comorbidities commonly found, such as osteoporosis, diabetes, and obesity, can increase the risk of postoperative complications, especially with poor nutrition and vitamin or mineral deficiencies. Understanding these complexities, the importance of immunonutrition in TJA is evident in regard to its role in immune dysfunction and inflammation, metabolic imbalance, and delayed healing, and that appropriate supplementation supports a more favorable recovery environment.
The current, but limited, evidence consistently demonstrates that perioperative immunonutrition improves surgical outcomes in patients undergoing TJA. This presents as shortened hospital stays, reduced postoperative complications, and enhanced physiological recovery, which reduces healthcare costs and improves resource allocation. In addition, supplementation has been found to reduce muscle atrophy and improve the rate of return to functional mobility. This is critical for accelerating both postoperative rehabilitation times and a return to independence for patients. Evidence also indicates that tailored immunonutrition protocols enhance wound healing and may lead to improved implant acceptance. These findings highlight the practical value of immunonutrition and promote its consideration in standard TJA perioperative care.
Although immunonutrition is shown to be advantageous, several challenges must be addressed before it can be widely adopted. These challenges include the lack of standardized formulations, disparities in patient access, and the absence of a single consensus tool for identifying at-risk individuals. Tackling these obstacles would help ensure effective, efficient, and equitable administration of this intervention. Future research should be focused on large-scale clinical trials aimed at establishing evidence-based guidelines for patient screening, nutrient dosage, and timing of administration. This would not only solve the “at-risk identification” debate between common screening tools but would also serve as the crucial next step for integrating immunonutrition into ERAS protocols. Ultimately, immunonutrition must become a standardized way of providing perioperative care for patients undergoing TJA procedures.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
