Abstract
Purpose
Heart failure (HF), classified as preserved (HFpEF) or reduced ejection fraction (HFrEF), is associated with heightened perioperative risk. This study assessed postoperative outcomes after total knee arthroplasty (TKA) in HF patients, by ejection fraction (EF), comorbidities, laboratory values, and pharmacotherapy on complications and mortality.
Methods
Using the TriNetX database (2005–2025), adults undergoing elective primary TKA with HFpEF or HFrEF were propensity score–matched to healthy controls. Matching included demographics, comorbidities, medication use, and labs: BMI, INR, hemoglobin A1c, troponin, and B-type natriuretic peptide (BNP). Outcomes included 90-days and 1-year systemic complications and mortality. Cox regression evaluated independent risk predictors.
Results
Final cohorts included 2611 HFpEF and 554 HFrEF patients with matched controls. Both subtypes demonstrated elevated 90-days mortality (OR 7.48 HFpEF; OR 7.43 HFrEF). HFpEF patients had significantly increased risks of MI (OR 13.96), atrial fibrillation (OR 19.31), stroke (OR 9.93), sepsis (OR 10.36), pulmonary embolism (OR 6.55), and AKI (OR 5.25). HFrEF patients showed similar patterns, including atrial fibrillation (OR 17.01), AKI (OR 9.17), and MI (OR 6.91). Cox regression identified age, male sex, elevated BNP, dialysis, chronic kidney disease, and prior MI or stroke as predictors of mortality. Medication analysis revealed ACE inhibitors, ARBs, SGLT2 inhibitors, GLP-1 agonists, and beta blockers as protective, whereas calcium channel blockers and loop diuretics increased risk.
Conclusion
HF patients undergoing TKA face increased complications and mortality. Stratifying risk by EF, comorbidities, and laboratory markers, particularly BNP and INR, along with tailored pharmacotherapy, may improve perioperative management and outcomes.
Keywords
Background
Total knee arthroplasty (TKA) is among the most commonly performed orthopaedic procedures in the United States, with demand rising alongside an aging population. Among Medicare beneficiaries annual primary TKA volume more than doubled between 2000 and 2019, and projections anticipate over one million procedures annually by 2030. 1 Concurrently, heart failure (HF) affects approximately 6–7 million Americans and is expected to reach 8.7 million by 2030, with prevalence exceeding 10% among older adults. 2 Advances in medical management have significantly extended survival, with 5-years survival after HF diagnosis improving from ∼29% in the 1970s to ∼60% in recent decades. 3 As a result, more patients with HF are becoming candidates for TKA, highlighting the need to understand perioperative risks in this growing population.
HF is a well-established risk factor for adverse outcomes after TKA.4–6 Studies have linked HF to longer hospitalizations, increased postoperative complications, and higher odds of reoperation and readmission following TKA.4–6 However, most analyses treat HF as a single entity, overlooking the syndrome’s clinical heterogeneity. HF is broadly categorized into HF with preserved ejection fraction (HFpEF; left ventricular ejection fraction [LVEF] ≥50%) and HF with reduced ejection fraction (HFrEF; LVEF≤40%), which differ in pathophysiology, demographics, and response to therapy.7,8 HFpEF now represents nearly half of all HF cases, particularly among older adults with metabolic comorbidities, while HFrEF has been more extensively studied.9,10
Despite the growing recognition of HF subtypes, large-scale investigations evaluating their differential impact on surgical outcomes remain limited. 11 Prior orthopaedic studies rarely stratify by ejection fraction due to limitations in datasets, leaving a gap in understanding how HFpEF and HFrEF influence TKA outcomes.5,12,13
In this context, this study aims to evaluate postoperative complications following TKA in patients with HFpEF and HFrEF, and to identify patient factors independently associated with mortality. We hypothesize that both HFpEF and HFrEF patients undergoing TKA experience higher rates of postoperative complications compared to matched controls, and that select comorbidities and perioperative medications are independently associated with increased hazard of mortality. To our knowledge, this represents the first large-scale, EF-stratified analysis of TKA outcomes in HF patients.
Methods
Data collection
Data were obtained from the US Collaborative Network of the TriNetX Research Platform, which aggregates de-identified health records from over 122 million patients across 70+ U.S. healthcare organizations. We identified patients who underwent elective primary TKA between January 2005 and March 2025, extracting demographic, clinical, and outcome variables for analysis. Because TriNetX provides only de-identified patient data, this study did not require institutional review board approval, and informed consent was waived.
Inclusion and exclusion criteria
Adults aged ≥18 who underwent elective primary TKA were eligible for inclusion if they had a documented diagnosis of HFpEF (LVEF≥50%) or HFrEF (LVEF≤40%), forming two separate cohorts. Patients were excluded if they had missing data, underwent revision or bilateral TKA, or had prior documentation of any study outcome. Diagnostic and procedural definitions were based on the International Classification of Diseases, 10th Revision, Clinical Modification (ICD-10-CM) and Current Procedural Terminology (CPT) codes. The index procedure, elective primary TKA, was identified using CPT 27447. HF was defined using ICD-10-CM I50 and LVEF values derived from Logical Observation Identifiers Names and Codes (LOINC) 10230-1. Full code listings, including those used to define exclusion criteria such as acute kidney injury (AKI), myocardial infarction (MI), sepsis, pneumonia, stroke, pulmonary embolism (PE), deep vein thrombosis (DVT), cardiac arrest, atrial fibrillation, and revision TKA, are available in Appendix A. Adults without HF who underwent elective primary TKA during the same period were selected as control patients.
Cohort selection
Patients undergoing TKA were categorized into three groups: HFrEF (LVEF ≤40%), HFpEF (LVEF ≥50%), and controls without HF. Initially, 844 HFrEF, 5637 HFpEF, and 238,195 control patients were identified. After excluding those with prior systemic complications or revision TKA, 526 HFrEF and 2659 HFpEF patients remained. These were each propensity score–matched 1:1 to eligible control patients, yielding final matched cohorts of 554 HFrEF patients with 554 controls, and 2611 HFpEF patients with 2611 controls. The full selection process is shown in Figure 1. Flowchart outlining selection process of HFrEF and HFpEF cohorts.
To minimize selection bias, we conducted 1:1 nearest-neighbor propensity score matching without replacement. Matching variables included: age, sex, race, hypertension, hyperlipidemia, ischemic heart disease, diabetes, hypothyroidism, body mass index (BMI), international normalized ratio (INR), hemoglobin A1c, B-type natriuretic peptide (BNP), troponin, and use of beta blockers, calcium channel blockers, angiotensin-converting enzyme (ACE) inhibitors, angiotensin II receptor blockers (ARBs), and antiarrhythmics. These variables were selected based on established risk factors for both HF severity and perioperative complications in orthopaedic surgery, as outlined in prior guidelines and studies of preoperative risk stratification.14,15 The full list of codes used to define these comorbidities and medications is provided in Appendix A.
Outcomes assessed
Twelve complications were assessed, including AKI, MI, blood loss anemia, pneumonia, PE, DVT, emergency department (ED) visits, mortality, cardiac arrest, stroke, sepsis, and atrial fibrillation. Corresponding diagnostic codes used to define these outcomes are detailed in Appendix A.
Statistical analysis
Statistical analysis was conducted using TriNetX Analytics and GraphPad Prism (v10.1.2). Patients were grouped as HFpEF, HFrEF, or control. Outcomes were summarized using descriptive statistics; continuous variables were compared with T-tests, and categorical variables with chi-square or Fisher exact tests. Multivariable logistic regression estimated odds ratios (ORs) with 95% confidence intervals (CIs); significance was set at p < 0.05. Cox regression evaluated time-to-event outcomes (cardiac arrest or mortality) and risk modifiers in HF patients. Hazard ratios (HRs) with corresponding 95% confidence intervals were calculated to assess associations between HF status and time-to-event outcomes.
Results
Baseline patient characteristics
Baseline patient characteristics after matching.

*Statistically significant.
HFrEF = Heart Failure with Reduced Ejection Fraction; HFpEF = Heart Failure with Preserved Ejection Fraction; BMI = Body Mass Index; INR = International Normalized Ratio; BNP = Brain Natriuretic Peptide; ACE = Angiotensin-Converting Enzyme; ARB = Angiotensin II Receptor Blocker.
After matching, cohorts were well-balanced. In the HFpEF group, mean age was 73.0 years ± 9.3 versus 72.8 years ± 8.6 in controls (p = 0.59); for HFrEF, 72.0 years ± 8.9 versus 72.1 years ± 8.4 (p = 0.72), with no significant differences in sex, race, or comorbidities (p > 0.05 for all).
Preoperative medication use remained higher in HF patients. HFpEF patients had greater use of beta blockers (83.7% vs 52.5%), ACE inhibitors (51.5% vs 42.8%), and calcium channel blockers (59.9% vs 41.3%) (p < 0.0001 for all). Similar trends were seen in HFrEF. Lab differences persisted post-matching, with slightly elevated BMI and INR in HF groups, while hemoglobin A1c and troponin levels were comparable.
Outcomes in HFpEF patients
Figure 2 summarizes the association between HFpEF and postoperative complications following TKA. At 90 days, patients with HFpEF experienced significantly worse outcomes compared to healthy matched controls (P < 0.0001 for all outcomes unless otherwise noted). Effect of heart failure with preserved ejection fraction (HFpEF) on postoperative outcomes following TKA (p-values are listed in the format 90 days/1 year).
Mortality was markedly increased (OR 7.48, 95% CI 3.85–14.52), along with AKI (OR 5.25, 95% CI 3.88–7.09) and sepsis (OR 10.36, 95% CI 5.39–19.89). Cardiovascular complications were also elevated, including MI (OR 13.96, 95% CI 7.32–26.60), atrial fibrillation (OR 19.31, 95% CI 12.85–29.03), stroke (OR 9.93, 95% CI 5.16–19.08), and cardiac arrest (OR 2.51, 95% CI 1.21–5.25, P = 0.011). PE (OR 6.55, 95% CI 3.72–11.53) and pneumonia (OR 13.41, 95% CI 7.03–25.57) were also more frequent in HFpEF patients. Blood loss anemia showed a modest but significant increase (OR 1.35, 95% CI 1.08–1.69, P = 0.0087), as did ED utilization (OR 2.00, 95% CI 1.72–2.34). DVT showed less increase (OR 1.76, 95% CI 1.21–2.54, P = 0.0026), though still statistically significant. Similar patterns persisted at 1-year follow-up (Figure 2).
Patients with baseline HFpEF (LVEF ≥50%) who developed LVEF <50% at 1 Year.

Figure 3 illustrates the impact of HFrEF on postoperative outcomes at 90 days and 1 year following TKA. At 90 days, patients with HFrEF demonstrated significantly higher rates of complications compared to matched controls (p < 0.0001 for all outcomes unless otherwise noted). Effect of heart failure with reduced ejection fraction (HFrEF) on postoperative outcomes following total knee arthroplasty (TKA) (p-values are listed in the format 90 days/1 year).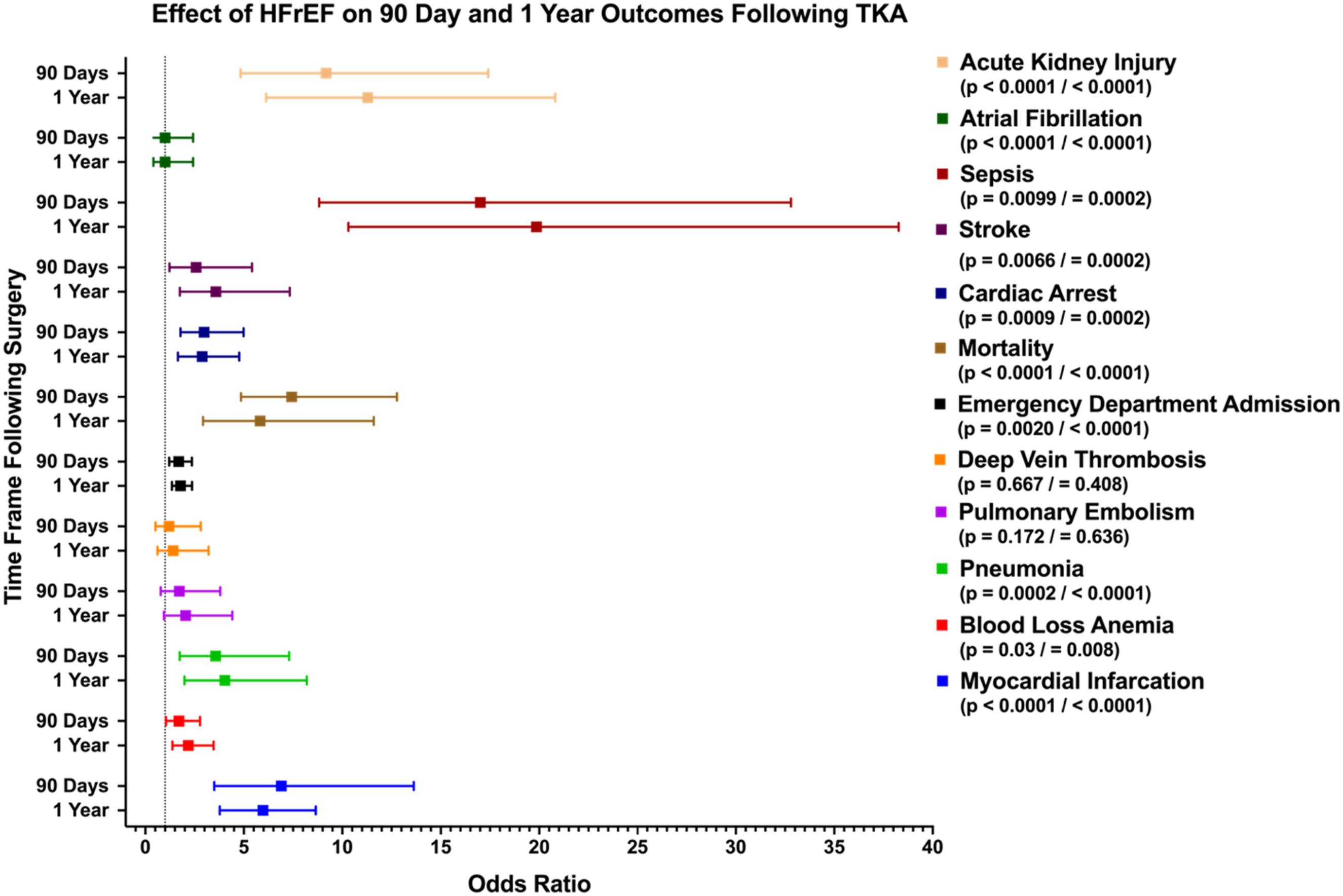
Mortality was elevated (OR 7.43, 95% CI 4.86–12.78), as were AKI (OR 9.17, 95% CI 4.83–17.41) and sepsis (OR 2.58, 95% CI 1.22–5.42, p = 0.0099). Cardiovascular events were notably increased, including atrial fibrillation (OR 17.01, 95% CI 8.82–32.80), MI (OR 6.91, 95% CI 3.50–13.63), stroke (OR 2.68, 95% CI 1.28–5.63, p = 0.0066), and cardiac arrest (OR 2.98, 95% CI 1.79–4.99, p = 0.0009). Increased odds were also observed for pneumonia (OR 3.57, 95% CI 1.74–7.30, p = 0.0002), blood loss anemia (OR 1.71, 95% CI 1.05–2.77, p = 0.03), and ED utilization (OR 1.69, 95% CI 1.21–2.36, p = 0.002). DVT showed a non-significant increase (OR 1.21, 95% CI 0.52–2.81, p = 0.667). At 1-year follow-up, elevated risks of mortality and most complications persisted, except for DVT and PE, which were no longer significantly different (Figure 3).
Subset analysis
To determine predictive factors influencing the overall incidence of cardiac arrest or mortality, we conducted a multivariate Cox regression analysis in patients undergoing TKA. Figure 4 illustrates the impact of demographic characteristics, comorbidities, medications, BNP levels, and substance use on 90-days and 1-year systemic outcomes in HF patients (p < 0.0001 for all variables unless otherwise specified). Impact of clinical factors and biomarkers outcomes following total knee arthroplasty (TKA) in heart failure (HF) Patients (p-values are listed in the format 90 days/1 year).
Male sex was associated with increased hazard (HR 1.16, 95% CI 1.15–1.17 at 90 days; HR 1.18, 95% CI 1.16–1.19 at 1 year), and advancing age similarly predicted elevated risk (HR 1.02, 95% CI 1.02–1.02; HR 1.03, 95% CI 1.03–1.03).
BNP levels demonstrated a graded relationship with postoperative risk. Patients with BNP 0–100 pg/mL had reduced hazard (HR 0.76, 95% CI 0.75–0.78; HR 0.71, 95% CI 0.70–0.73), while those with BNP 100–400 pg/mL showed a modest increase (HR 1.03, 95% CI 1.01–1.05, p = 0.0015; HR 1.02, 95% CI 1.00–1.04, p = 0.015). Risk rose further in the 400–800 pg/mL group (HR 1.14, 95% CI 1.11–1.17; HR 1.18, 95% CI 1.15–1.20) and was highest among those with BNP >800 pg/mL (HR 1.41, 95% CI 1.38–1.44; HR 1.44, 95% CI 1.41–1.47).
Several comorbidities were independently predictive of worse outcomes, including acute MI (HR 1.28, 95% CI 1.26–1.30; HR 1.19, 95% CI 1.17–1.21), chronic kidney disease (HR 1.15, 95% CI 1.14–1.17; HR 1.24, 95% CI 1.23–1.25), cerebrovascular disease (HR 1.22, 95% CI 1.20–1.23; HR 1.21, 95% CI 1.20–1.23), and dialysis dependence (HR 1.56, 95% CI 1.51–1.61; HR 1.67, 95% CI 1.64–1.72). Nonrheumatic aortic valve disorders (HR 1.04, 95% CI 1.03–1.06; HR 1.07, 95% CI 1.05–1.08), essential hypertension (HR 1.25, 95% CI 1.24–1.26; HR 1.29, 95% CI 1.28–1.29), atherosclerosis (HR 1.31, 95% CI 1.29–1.33; HR 1.31, 95% CI 1.29–1.32), and atrial fibrillation (HR 1.08, 95% CI 1.07–1.09; HR 1.12, 95% CI 1.11–1.17) were also associated with elevated risk.
Type 2 diabetes mellitus was not associated with elevated hazard at 90 days (HR 1.00, 95% CI 0.99–1.02, p = 0.62), but reached significance at 1 year (HR 1.01, 95% CI 1.00–1.02, p = 0.024).
Substance use also contributed to postoperative risk. Alcohol abuse was associated with increased hazard (HR 1.10, 95% CI 1.07–1.13 at 90 days; HR 1.08, 95% CI 1.05–1.10 at 1 year), while nicotine dependence was linked to increased hazard at 90 days (HR 1.09, 95% CI 1.01–1.10, P = 0.039), but a slight reduction at 1 year (HR 0.99, 95% CI 0.98–1.00, p = 0.010).
Figure 5 summarizes the effects of common HF medications on 90-days and 1-year postoperative outcomes following TKA. P < 0.0001 for all variables unless otherwise specified. Among the agents evaluated, calcium channel blockers (HR 1.07, 95% CI 1.05–1.08 at 90 days; HR 1.07, 95% CI 1.06–1.08 at 1 year) and loop diuretics (HR 1.21, 95% CI 1.20–1.23; HR 1.32, 95% CI 1.31–1.33) were associated with increased postoperative risk at both timepoints. Effect of medications on 90-day and 1-year outcomes following total knee arthroplasty (TKA) in heart failure (HF) patients (p-values are listed in the format 90-days/1-year).
In contrast, all other medication classes demonstrated protective associations. These included angiotensin-converting enzyme (ACE) inhibitors (HR 0.80, 95% CI 0.79–0.81 at 90 days; HR 0.82, 95% CI 0.81–0.82), angiotensin II receptor blockers (ARBs) (HR 0.76, 95% CI 0.75–0.77; HR 0.78, 95% CI 0.77–0.79), thiazide diuretics (HR 0.92, 95% CI 0.90–0.93; HR 0.90, 95% CI 0.89–0.91), potassium-sparing diuretics (HR 0.88, 95% CI 0.86–0.90; HR 0.91, 95% CI 0.89–0.92), sodium-glucose cotransporter 2 (SGLT2) inhibitors (HR 0.68, 95% CI 0.66–0.71; HR 0.72, 95% CI 0.70–0.74), and glucagon-like peptide-1 (GLP-1) receptor agonists (HR 0.65, 95% CI 0.62–0.67; HR 0.67, 95% CI 0.65–0.69).
Beta blockers showed a modest protective effect at 90 days (HR 0.97, 95% CI 0.96–0.98), but the association did not reach statistical significance at 1 year (HR 0.99, 95% CI 0.98–1.00; p = 0.0589). (Figure 5).
These drug-related findings could potentially be explained by the empirically guided cardioprotective effect of certain HF medications. Whereas the majority of HF drugs evaluated have a proven survival benefit, loop diuretics and CCBs (dihydropyridine/non-dihydropyridine) show no survival or hospitalization benefit in HF patients. Furthermore, patients prescribed either of these two drugs are more likely to be in a more severe state of HF upon the time of prescription than patients that were not prescribed these same medications. The proven survival benefit of other medications likely masked this selection bias.
Discussion
Our findings indicate that HF patients face significant risks after undergoing TKA. Across HFpEF and HFrEF cohorts, there was a consistent postoperative risk of mortality, readmission at 90 days, and systemic complications, with cardiovascular and pulmonary incidents predominating up to 1 year after TKA. While a number of studies have assessed HF patients after joint replacement, there is a lack of literature which delineates outcomes based on ejection fraction.4,16 Given the expected rise in prevalence of HF in the United States and increased risk of HF-related mortality in the elderly population, proactive medical optimization of patients stratified by HF subtype is required to prevent adverse postoperative outcomes.2,17
Primary outcomes
Mortality and 90-day readmission
Both HFpEF and HFrEF patients exhibited higher mortality rates at 90-days, with odds ratios of 7.64 (95% CI: 4.58 to 12.72) and 7.43 (95% CI: 4.86 to 12.78), respectively. Bozic et al. reported that HF was an independent risk factor for both 90-days postoperative mortality (HR: 2.15, CI: 1.13 to 1.46) and periprosthetic joint infection, alongside diagnoses of diabetes, metastatic cancer, and dementia. 18 Our findings also showed that HF was associated with a greater 90-days readmission after TKA. In a recent retrospective review of Medicare claims data, Young et al. found HF patients undergoing TKA to have a longer in-hospital length of stay and a greater risk of developing complications within 90 days (49.2% vs 7.4%). 19 Accordingly, Rozell and colleagues advocate for extended postoperative monitoring in this patient cohort and advise against early discharge. 20 In patients with a history of congestive HF, the risk of requiring postoperative intervention far exceeded any other comorbidity (OR: 24.26, 95% CI: 9.51–61.91), demonstrating the need for nuanced clinical management after surgery. 21
Cardiovascular complications: Cardiac risk factors before TKA
Despite improvements in cardiac monitoring and perioperative management of HF, patients undergoing noncardiac surgery remain at heightened risk for adverse cardiac events. 22 Multiple studies identify prior heart disease as a significant risk factor for cardiac-related complications after both hip and knee arthroplasty.23–25 Our findings are consistent with the literature, showing that HF patients were at increased odds of developing atrial fibrillation, MI, and cardiac arrest within 90 days. Among the complications studied, the odds of developing atrial fibrillation after TKA were at least 15 times higher in both HF subtypes. In a review of more than two million hip fracture outcomes, patients with HFpEF experienced a greater risk of major adverse cardiac events (OR: 1.69, 95% CI: 1.51–1.89) when compared to controls. 12 In patients noted to have any cardiac complication after total joint arthroplasty (TJA), Belmont Jr. et al. identified nearly 80% of adverse cardiac events occurred within 7 days, further necessitating extended hospital observance for high-risk patients. 26
Venous thromboembolism (VTE) and pulmonary complications
Patients with HFpEF demonstrated markedly elevated odds of PE (OR 6.55 at 90 days; OR 6.07 at 1 year), DVT (OR 1.76; OR 2.17), and pneumonia (OR 13.41; OR 7.26). The discrepancy observed between complication risks in HFrEF likely reflect early mortality. Prior studies confirm PE and pneumonia as major complications after TKA in elderly and high-risk populations.27–29 In a large database review, Bohl et al. established that postoperative pneumonia increases the risk of mortality and readmissions.30,31 Although VTE prophylaxis is routine in orthopaedic surgery, its benefit in HF patients is uncertain. Some studies suggest aspirin alone is less effective, with higher symptomatic VTE rates than anticoagulants, 32 while others report low-dose aspirin as protective in select groups.33,34 Notably, anticoagulants themselves may not significantly reduce PE rates after TKA, emphasizing the complexity of prophylaxis in this vulnerable population. 35
Kidney disease and sepsis
Although relatively uncommon after TKA, patients with a history of cardiac disease face a significantly higher risk of postoperative complications related to AKI.36–38 Multiple studies have shown HF to be an independent risk factor for AKI following noncardiac surgery.39–41 In our analysis, both HFpEF (OR 5.25 at 90 days; OR 5.31 at 1 year) and HFrEF (OR 9.17; OR 11.3) cohorts had markedly increased risk. Thorsdottir et al. in their study concluded that AKI after orthopaedic procedures correlated with increased long-term mortality, 42 underscoring its clinical significance. However, early detection of HF and careful perioperative management may help mitigate this risk. 43
Biomarker and comorbidity-associated risks
Our analysis revealed a graded relationship between BNP levels, postoperative cardiac arrest, and mortality rates in HF patients undergoing TKA; as BNP levels increased, mortality increased while patients with normal BNP levels below 100 pg/mL were were found to be protective. This graded response highlights the potential role of BNP as a valuable biomarker for risk stratification in this patient population. Previous cardiovascular research emphasizes this role, with a notable study in the Journal of the American College of Cardiology reporting that a BNP level ≈ 400 pg/mL was associated with a 21% 3-years mortality risk in HF. 44 Additionally, perioperative research revealed that an NT-proBNP cutoff of 450 pg/mL independently predicted morbidity at 30 days more effectively than the Revised Cardiac Risk Index or ASA classification. 45 Together, these findings highlight BNP’s value as a prognostic tool to guide perioperative management and improve outcomes.
Our analysis identified acute MI and cerebrovascular disease as major predictors of cardiac arrest and mortality, consistent with a Korean cohort of 560,954 TKA patients that found cerebrovascular disease (HR 1.401) and MI (HR 2.111) significantly increased in-hospital mortality. 46
Similarly, CKD also predicted worse outcomes (HR 1.153 at 90 days; HR 1.242 at 1 year), aligning with a systematic review linking CKD to postoperative death and cardiovascular events. Furthermore, patients undergoing dialysis had even greater risk (HR 1.56; HR 1.67), corroborating findings of higher perioperative complications and costs in dialysis-dependent TKA patients. 47
In addition, several comorbidities—including nonrheumatic aortic valve disorders, hypertension, atherosclerosis, alcohol abuse, and atrial fibrillation—further elevated mortality risk. While these comorbidities independently elevate postoperative risks, their specific impact on HF patients undergoing TKA is not fully documented. However, it is logical to infer that their effects are particularly pronounced in this population due to their vulnerable preoperative risk profile. For instance, a retrospective cohort study demonstrated that patients with valvular dysfunction experienced worse hospital metrics, higher perioperative complications, and increased mortality following TKA. 5 Similarly, a study by Li et al. utilizing found that hypertensive patients undergoing TKA had a higher risk of developing AKI and electrolyte disorders postoperatively. 48 Moreover, patients diagnosed with alcohol misuse were independently associated with higher odds of in-hospital complications (OR 1.334), surgery-related complications (OR 1.293), and general medical complications (OR: 1.300) following joint arthroplasty. 49 Regarding atrial fibrillation, a retrospective study involving 453 patients with preexisting atrial fibrillation who underwent TKA revealed significantly higher odds of intraoperative bleeding, periprosthetic joint infection, postoperative stroke, and postoperative cardiovascular events compared to matched controls. 50 Collectively, these findings emphasize the compounded vulnerability of HF patients and the need for careful preoperative optimization.
Medication-associated risks and benefits
Medication use was significantly associated with postoperative outcomes in patients with HF undergoing TKA. We selected several prescription drugs as part of guideline-directed medical therapy (GDMT) as well as diuretics and antihypertensives commonly used in treating HF patients to analyze their role in overall mortality in TKA patients.
While beta blockers showed modest protective effects in our cohort, prior studies have raised concern over increased risks of intraoperative hypotension (RR 1.37; 95% CI: 1.20–1.56) and bradycardia (RR 2.41; 95% CI: 1.75–3.32), particularly with poor perioperative management. 51 Moreover, abrupt discontinuation has been associated with a twofold increase in MI and mortality risk (OR 2.0 for both). 52 This may explain the diminished 1 year benefit of beta blockers observed in our cohort.
Conversely, calcium channel blockers (HR 1.06) and loop diuretics (HR 1.32) were associated with worse outcomes, consistent with prior reports linking them to stroke, hemodynamic instability, and AKI in patients with hypertension or HF.53–56
Unsurprisingly, ACE inhibitors (HR 0.82 at 1 year) and ARBs (HR 0.78 at 1 year) were protective in both short and long-term outcomes in our analysis, given their known cardioprotective effects and impact on preventing worsening ventricular function. 57 Novel therapies such as SGLT2 inhibitors (HR 0.72 at 1 year) and GLP-1 receptor agonists (HR 0.67 at 1 year) offer significant benefits in the perioperative setting. SGLT2 inhibitors have demonstrated a 31% reduction in HF hospitalizations and a 38% relative risk reduction in progression of kidney disease through not only glycemic control, but also by natriuresis, reduced intraglomerular pressure, and improved myocardial energetics, which together alleviate volume overload and enhance renal perfusion. 14 GLP-1 receptor agonists have shown a 12% reduction in major adverse cardiovascular events and appear to improve perioperative stability through pharmacologic weight loss, glycemic regulation, and anti-inflammatory effects.58–61 Together, these findings underscore the value of tailoring pharmacotherapy to the physiologic vulnerabilities of HF patients, particularly in those at high risk for volume overload, metabolic stress, or renal dysfunction.
Limitations
This study has several limitations. Its retrospective, observational design precludes causal inference and introduces potential biases, including selection bias and residual confounding. Although extensive propensity score matching was performed, unmeasured factors such as socioeconomic status, medication adherence, perioperative optimization, and institutional care differences may have influenced outcomes. Reliance on ICD and CPT coding carries risk of misclassification due to interinstitutional variability and incomplete capture of disease severity or complications. The smaller matched HFrEF cohort limited direct comparisons between HF subtypes, reflecting surgical selection bias, as patients with reduced ejection fraction are less often selected for elective TKA. Additionally, since TKA patients tend to be older, HFpEF was overrepresented relative to HFrEF despite similar prevalence in the general population. While these limitations restrict the ability to make specific, practice-changing recommendations regarding perioperative management, the findings underscore clinically meaningful trends in how heart failure status influences postoperative outcomes. Future studies with granular access to perioperative hemodynamics, medication adjustments, anesthesia type, and fluid management are needed to refine guidelines and develop targeted interventions that better inform surgical decision-making in this high-risk population.
Conclusion
In this propensity-matched study, HFpEF and HFrEF patients undergoing TKA faced significantly higher risks of cardiovascular, pulmonary, renal, and mortality complications compared with controls. Cox regression identified advanced age, male sex, elevated BNP, and comorbidities as independent predictors of poor outcomes. Medication analysis showed ACE inhibitors, ARBs, and beta-blockers offered protection, whereas calcium channel blockers and loop diuretics were associated with harm. Together, these findings highlight the need for comprehensive risk stratification and individualized perioperative management to improve outcomes in this high-risk population.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Appendix
Codes used for cohort, matching, and outcome definitions.
Category
Code
Description
Index procedure code
CPT 27447
Arthroplasty, knee, condyle and plateau; medial AND lateral compartments with or without patella resurfacing (Total knee arthroplasty)
Heart failure inclusion (HFpEF cohort)
ICD-10-CM I50
Heart failure
Heart failure inclusion (HFpEF cohort)
LOINC 10230-1
Left ventricular ejection fraction (set LVEF ≥ 50%)
Heart failure inclusion (HFrEF cohort)
ICD-10-CM I50
Heart failure
Heart failure inclusion (HFrEF cohort)
LOINC 10230-1
Left ventricular ejection fraction (set LVEF ≤ 40%)
Exclusion criteria
ICD-10-CM I21
Acute myocardial infarction
Exclusion criteria
ICD-10-CM N17
Acute kidney failure
Exclusion criteria
ICD-10-CM A41.9
Sepsis, unspecified organism
Exclusion criteria
ICD-10-CM J18
Pneumonia, unspecified organism
Exclusion criteria
ICD-10-CM I63
Cerebral infarction (stroke)
Exclusion criteria
ICD-10-CM I26
Pulmonary embolism
Exclusion criteria
ICD-10-CM I82.4
Acute embolism and thrombosis of deep veins of lower extremity (DVT)
Exclusion criteria
ICD-10-CM I46
Cardiac arrest
Exclusion criteria
ICD-10-CM I48.91
Unspecified atrial fibrillation
Exclusion criteria
CPT 27486
Revision of total knee arthroplasty; one component
Exclusion criteria
CPT 27487
Revision of total knee arthroplasty; femoral and entire tibial component
Propensity score matching
ICD-10-CM I10–I11 A
Hypertensive diseases
Propensity score matching
ICD-10-CM I10–I15
Hypertensive diseases (deprecated 2018)
Propensity score matching
ICD-10-CM E78.5
Hyperlipidemia, unspecified
Propensity score matching
ICD-10-CM I20–I25
Ischemic heart diseases
Propensity score matching
ICD-10-CM E08–E13
Diabetes mellitus
Propensity score matching
ICD-10-CM E03.9
Hypothyroidism, unspecified
Propensity score matching
ICD-10-CM N18
Chronic kidney disease (CKD)
Propensity score matching
LOINC 9083
Body Mass index
Propensity score matching
LOINC 9032
INR in plasma or blood
Propensity score matching
LOINC 9037
Hemoglobin A1c
Propensity score matching
LOINC 9003
Natriuretic peptide B [Mass/volume] in serum, plasma or blood
Propensity score matching
LOINC 9005
Troponin I.cardiac [Mass/volume] in serum, plasma or blood
Propensity score matching
RxNorm CV300
Antiarrhythmics
Propensity score matching
RxNorm CV100
Beta blockers/related
Propensity score matching
RxNorm CV200
Calcium channel blockers
Propensity score matching
RxNorm CV800
ACE inhibitors
Propensity score matching
RxNorm CV805
Angiotensin II inhibitors
Outcomes
ICD-10-CM N17
Acute kidney failure (acute kidney injury)
Outcomes
ICD-10-CM I21
Acute myocardial infarction
Outcomes
ICD-10-CM D62
Acute posthemorrhagic anemia (blood loss anemia)
Outcomes
ICD-10-CM J18.9
Pneumonia, unspecified organism
Outcomes
ICD-10-CM J15.9
Unspecified bacterial pneumonia
Outcomes
ICD-10-CM J22
Unspecified acute lower respiratory
Outcomes
ICD-10-CM I26
Pulmonary embolism
Outcomes
ICD-10-CM I82.4
Acute embolism and thrombosis of deep veins of lower extremity
Outcomes
CPT 1013711
Emergency department services
Outcomes
ICD-10-CM I46
Cardiac arrest
Outcomes
ICD-10-CM I63
Cerebral infarction (stroke)
Outcomes
ICD-10-CM A41.9
Sepsis, unspecified organism
Outcomes
ICD-10-CM I48.9
Unspecified atrial fibrillation and atrial flutter
