Abstract
Background
In many countries, disposable high-value consumables are sterilized and reused for economic reasons. We aimed to study the feasibility and ways to reduce potential risks of reusing these consumables in shoulder arthroscopy.
Methods
We reported cases with complications potentially related to reusing high-value consumables. We evaluated the outflow water temperature from new and reused radiofrequency (RF) devices of different brands and under varying surgical practices. We also conducted fatigue tests on suture-passing needles from different brands subjected to various disinfection methods.
Results
Reused RF devices from MECHAN demonstrated significantly higher peak temperature (PT) (68.3 ± 2.3°C vs 63.9 ± 3.0°C, P < 0.001) and peak sustaining temperature (PST) (62.6 ± 2.3°C vs 58.5 ± 3.1°C, P < 0.001) compared to new devices. The mean flow rate of MECHAN devices was higher than that of DePuy (63.2 ± 8.2 ml/min vs 42.0 ± 11.1 ml/min, P < 0.001). Ethylene oxide disinfection did not significantly affect the excitation times before fatigue fracture of Arthrex (291.6 ± 76.9 vs 305.1 ± 86.6, P = 0.717) and DePuy (173.3 ± 26.8 vs 174.6 ± 31.7, P = 0.922) suture-passing needles. High-pressure steam disinfection also did not markedly affect the fatigue time of Arthrex (291.6 ± 76.9 vs 292.0 ± 65.7, P = 0.990) and DePuy (173.3 ± 26.8 vs 168.8 ± 36.1, P = 0.755) suture-passing needles.
Conclusions
For both RF devices and suture-passing needles, being reused after ethylene oxide disinfection for appropriate times can be feasible and safe if surgeons and disinfection suppliers exercise caution.
Keywords
Introduction
In both developed and developing countries, reuse of high-value consumables is very common in clinical practice. Reusing high-value consumables is environmentally friendly and significantly reduce medical costs, potentially expanding access to treatment for more patients. Though reuse of consumables is widely implemented, few documents reported its feasibility and potential risks. Our inter- and intra-institutional communication with high-volume arthroscopic surgeons indicated that certain complications may be associated with device reuse. For example, dermal burns have occasionally occurred due to hot water discharged from the drainage tubes of arthroscopy radiofrequency (RF) devices of certain brand (Figure 1). Additionally, the tips of suture-passing needles have been known to break during shoulder arthroscopy, sometimes remaining undetected until postoperative radiographs revealed their presence (Figure 2).
1
These complications are rarely reported and the prevalence is underestimated, particularly when they cause no symptoms or mild symptoms that tended to resolve over time. However, these complications may result in both physiological and psychological effects on patients and even litigation issues. The goal of this study is to provide surgeons, public-health policymakers, and medical consumable manufacturers with a clearer understanding of the feasibility and potential risks associated with consumable reuse. (a) Intraoperative image showing the mechanism of burn injuries. Case 1: A 62-year-old woman underwent right-sided arthroscopic rotator cuff repair with subacromial decompression. The patient’s skin was intact before the surgery, and the operated arm was placed in traction and sterilized with povidoneiodine. At the end of the procedure, no redness or swelling was observed at the surgical site. (b) During a follow-up visit 3 days later, four areas of ulceration were identified, with the largest measuring 5 × 3 cm. A diagnosis of superficial second-degree burn was made, and treatment with silver sulfadiazine cream and human epidermal growth factor gel was initiated to prevent infection and promote wound healing. (c) After 2 weeks of treatment, only mild erythema and itching remained. (d) At the 2-years follow-up, the wound had completely healed. Case 2: A 48-year-old woman underwent a surgical procedure similar to Case 1. (e) At the end of the procedure, an area of redness was observed at the surgical site. (f) By the third postoperative day, an ulceration measuring 8 × 4 cm was noted, and the patient was diagnosed with deep second-degree burns. A treatment strategy similar to Case 1 was employed. However, the patient opted out of the keloid treatment due to financial constraints. (g, h) At the 2-years follow-up, a keloid measuring 4 × 2 cm was still present. RF devices were reused for 6th and 9th cycles for case1 and case2 respectively. Both patients were satisfied due to significant pain relief and functional improvement. Case 1: A 58-year-old woman with Parkinson’s disease and a massive rotator cuff tear underwent shoulder arthroscopy. The tip of a DePuy suture needle broke during subscapular tendon repair. (a, arrow). A postoperative X-ray revealed the needle tip lodged in the subscapular tendon area. (b arrow). At the 1-month follow-up, imaging showed that the broken needle tip had migrated along the anterior space of the subscapular muscle. Case 2: A 66-year-old woman with a massive rotator cuff tear underwent shoulder arthroscopy. The tip of a DePuy suture needle broke during supraspinatus tendon repair (c, d, arrow). Postoperative X-ray and MRI revealed the needle tip along the supraspinatus tendon area. Case 3: A 47-year-old man underwent supraspinatus tendon repair, during which the Arthrex suture-passing needle tip broke but was not visible. An intraoperative radiograph of the shoulder joint identified the needle tip positioned superior to the glenoid and is relatively long (e, arrow). An extended incision at the anterior portal between the deltoid muscle and pectoralis major was made, and the broken piece was retrieved from the belly of the supraspinatus. A photograph of the broken nitinol needle from the Scorpion suturing device is shown (f). Case 4: A 68-year-old man with a massive rotator cuff tear underwent shoulder arthroscopy. The tip of a Arthrex suture needle broke during supraspinatus tendon repair (g, h, arrow). Postoperative MRI revealed the needle tip and associated metal artifact along the supraspinatus tendon area.

Methods
Institutional ethics committee approved this study. According to the committee, informed consent was not required as the experiments did not interfere with the diagnosis or treatment process. No financial support was received from any third party for this study.
Temperature measurement of RF outflow water
The outflow water temperature from the device’s drainage tube was measured during subacromial decompression using a thermometer (Testo 905T1). The irrigation water tube was positioned at a height of 2m, and the energy levels for electrocision and electrocution were set to the highest grades. The measurements were performed at a room temperature of 24°C by two assistants, ensuring that the surgical area is sterile and not interfering with the surgical procedures (Figure 3a). The metal portion of the thermometer were disinfected using iodophor. Only the end of the outflow tube contacted the thermometer and the outflow water collecting basin was a sterilely packaged disposable consumable item. Once the measurement finished, the outflow tube on the RF device was cut by a sterile scissor and discarded. The brand, number of reuse cycles (new or reused for 5 times), and the corresponding outflow temperatures were recorded for each patient. The peak temperature (PT) was defined as the highest temperature recorded on the thermometer, while the peak sustaining temperature (PST) was defined as the highest temperature maintained for at least 5 s. We chose to measure PT and PST based on the work by Andrews et al.
2
Additionally, drainage quantity was measured using a measuring glass. Picture showing how the outflow water temperature of RF devices was measured (a). DePuy (b, e) and MECHAN (c, f) RF devices. The reused tip of the DePuy device (d).
Four surgeons each performed 20 surgeries using RF devices from DePuy (VAPR S90; DePuy Mitek, Raynham, MA, USA) (Figure 3b, d, and e). Additionally, Surgeon 2 conducted 45 surgeries using devices from MECHAN (MC407; MECHAN, Chengdu, China) (Figure 3c and f). All the RF devices were collected beforehand and randomly assigned to each surgeon. These surgeries were performed with irrigation water at a temperature of 24°C. Surgeon 2 also performed an additional 20 surgeries using RF devices from DePuy, with irrigation water heated to 40°C. Surgeons 1 and 2 habitually used the RF devices continuously, keeping in contact with the target tissue. Surgeons 3 and 4 did the opposite, using the devices intermittently and frequently moving away from the target tissue during the procedure.
Suture-passing needle fatigue tests
Sixty suture-passing needles, designated for disposal as medical waste, were collected for this experiment. Thirty needles were from Arthrex (FastPass Scorpion; Arthrex, Naples, FL, USA) (Figure 4a), and 30 were from DePuy (ExpresSew; DePuy Mitek, Raynham, MA, USA) (Figure 4b). For each brand, 30 needles were randomized into 3 groups. Ten needles were left unsterilized, 10 underwent ethylene oxide disinfection for 5 times, and 10 underwent high-pressure steam disinfection for 5 times. The disinfection procedures followed standard clinical practice guidelines. During the surgery, the number of excitations was less than 10, and the needle tip did not contact any hard objects, such as the acromion or coracoid process. Each suture-passing needle was cut to a length of 5 cm and placed on an electronic universal mechanical testing machine (UTM2102, Shenzhen SUNS Technology Stock Co, Ltd) (Figure 4c). The needles were stimulated at a frequency of 0.125 Hz, an amplitude of 3 mm, and a uniform bending direction (Figure 4d and e). A stress diagram of the procedure was recorded for each needle, and the number of recycling the suture-passing needles underwent before breaking was documented (Figure 4f). The tip of the Arthrex (a) and DePuy (b) suture-passing needles. Electronic universal mechanical testing machine and stress diagram monitor (c). Bending mode of Arthrex (d) and Depuy (e) suturepassing needles. Blue arrow showed needle breaking on the stress diagram (f). Ethylene oxide sterilized Arthrex (black arrow) and Depuy (blue arrow) suture-passing needles. High-pressure steam sterilized Arthrex (white arrow) and Depuy (red arrow) suture-passing needles (g).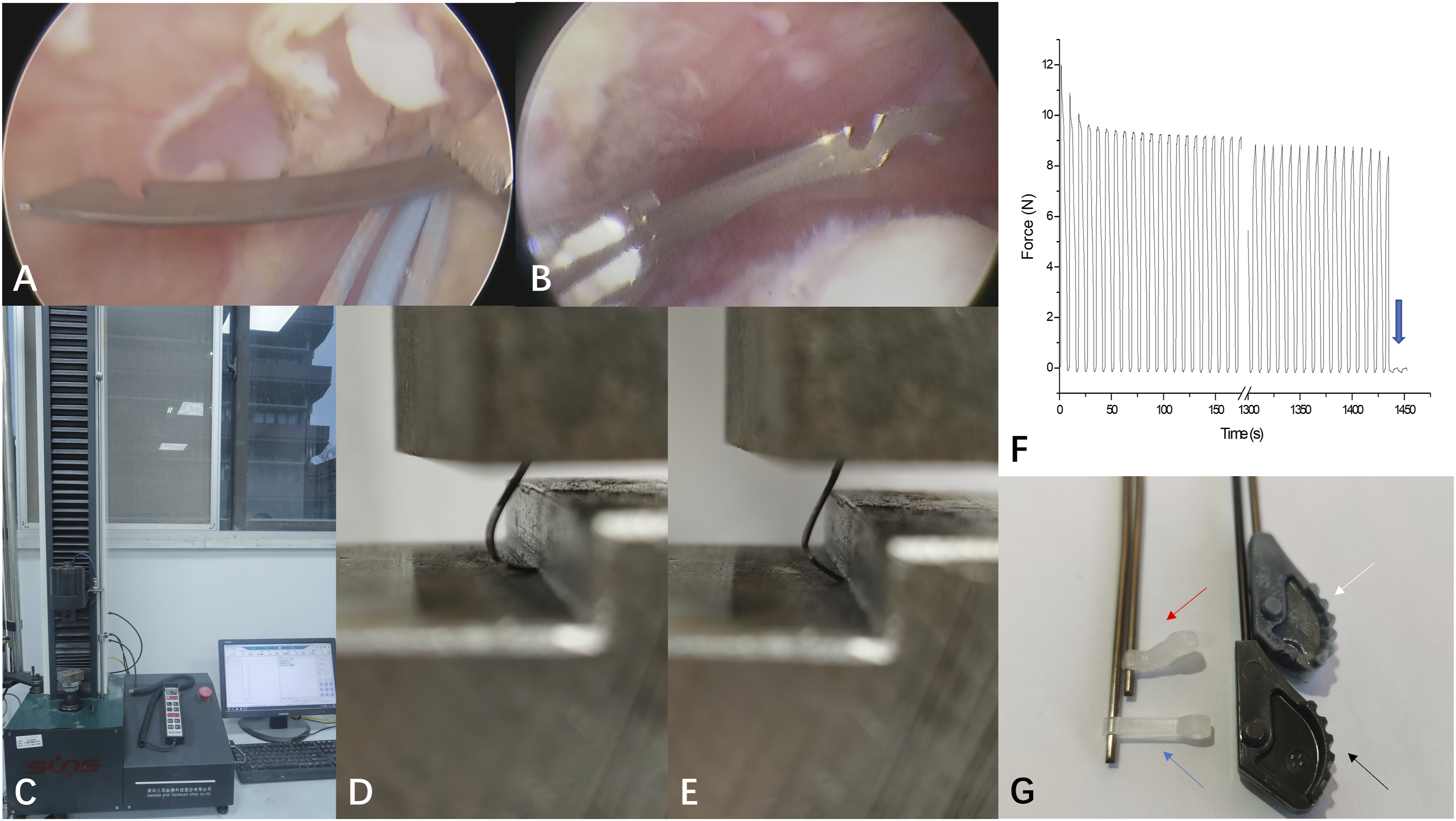
Statistical analysis
An independent sample t-test was employed to compare quantitative data between two groups, while the one-way ANOVA was used to compare quantitative data for more than two groups. A P-value <0.05 was considered statistically significant. All analyses and calculations were performed using SPSS (version 25; IBM, Chicago, IL, USA).
Results
Water temperature measurement
Of the 145 patients included in the study, 96 were female and 49 were male, with an average age of 59.4 years.
Comparison between new and reused (5 times) RF devices.

Comparison between different surgeons using RF devices from DePuy.

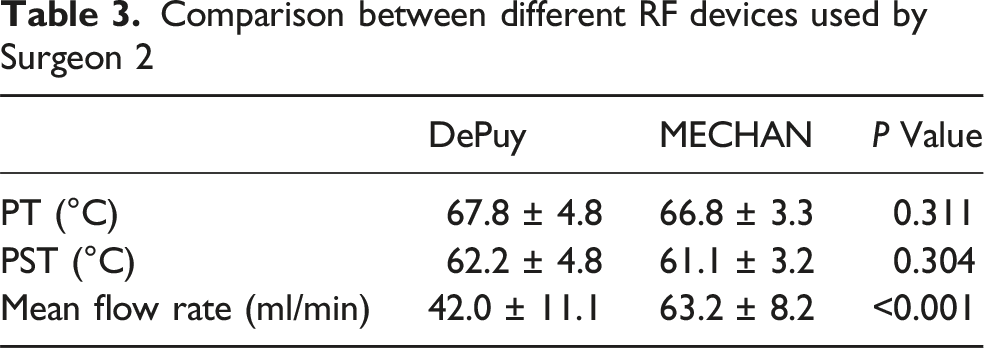
Comparison between different RF devices used by Surgeon 2
Comparison between irrigation water at 24°C and 40°C by Surgeon 2 using RF devices from DePuy.

Suture-passing needle fatigue tests
For Arthrex suture-passing needles, the number of excitation cycles before fatigue fracture was similar for unsterilized and ethylene oxide-sterilized needles (291.6 ± 76.9 vs 305.1 ± 86.6, P = 0.717). It was also similar after high-pressure steam disinfection (291.6 ± 76.9 vs 292.0 ± 65.7, P = 0.990). For DePuy suture-passing needles, the number of excitation cycles before fatigue fracture was comparable between unsterilized and ethylene oxide-sterilized needles (173.3 ± 26.8 vs 174.6 ± 31.7, P = 0.922). It was also comparable after high-pressure steam disinfection (173.3 ± 26.8 vs 168.8 ± 36.1, P = 0.755).
Arthrex needles demonstrated a higher number of excitation times before fatigue fracture than DePuy needles for the unsterilized (291.6 ± 76.9 vs 173.3 ± 26.8, P = 0.001), ethylene oxide-sterilized (305.1 ± 86.6 vs 174.6 ± 31.7, P = 0.001) and high-pressure steam sterilized (292.0 ± 65.7 vs 168.8 ± 36.1, P < 0.001) needles.
Discussion
Reuse of RF devices and dermal thermal injuries
Interestingly, all five reported cases of dermal burns as far as we can find in the literature involved RF devices from a single manufacturer (TurboVac; ArthroCare, Sunnyvale, CA).3–6 Alejandro et al. attributed these burns to the outflow tube being too short and recommended lengthening the tube with a 30-cm aspiration probe to direct hot water into the collection bag. 4 All iatrogenic dermal burns after arthroscope in our institution happened in patients treated with reused RF devices from MECHAN. However, the tube lengths of RF devices from different brands were comparable (DePuy: wand 31.3 cm, tube 38.0 cm; MECHAN: wand 30.0 cm, tube 40.7 cm). This led us to explore other possibilities causing dermal complications.
Previous studies have explored the relationship between elevated intra-articular temperature and articular or neural injuries, particularly because of the widespread use of thermal capsulorrhaphy for treating shoulder instability. Axillary nerve injuries have been reported following capsular shrinkage, likely caused by penetration of thermal energy through soft tissues.7,8 Additionally, partial or full-thickness chondrolysis after capsular shrinkage has been implicated to be caused by overheated irrigation water.9,10 An in vitro study demonstrated morphological changes in arthritic and non-arthritic articular cartilage at temperatures of 56.5°C and 60.9°C, respectively. 11 A cadaveric study reported that mean intra-articular temperatures during the 9 min of thermal capsulorrhaphy with a monopolar device did not reach levels considered physiologically deleterious. 12 Similarly, Scott et al. measured intra-articular temperatures in 15 patients during subacromial decompression using a bipolar device and observed a mean PT of 32°C (23.5°C - 43.1°C) in the subacromial bursa, which was insufficient to cause tissue damage. 13 However, when thermometer probes were placed closer to the RF device and water flow was obstructed, intra-articular temperatures exceeded 50°C within approximately 40 s of intermittent activation.14,15 Continuous water flow and intermittent RF application are reliable methods for maintaining safe intra-articular temperatures.14–16
Our study demonstrated that the outflow water temperature of RF devices could exceed 60°C, which is high enough to cause dermal thermal injuries. 2 Notably, the outflow temperature of reused MECHAN devices was higher than that of the new devices, likely due to repeated use affecting the temperature control mechanism. The plastic insulative temperature control unit may be partly damaged after repeated use, resulting in a local short circuit. Additionally, the water outflow rate of MECHAN devices was higher than that of DePuy. The elevated temperature and increased outflow rate both contributed to the risk of cutaneous complications. The bending of the outflow tube after disinfection may further increase the likelihood of hot outflow water contacting with patients’ skin (Figure 1a). In contrast, reused RF devices from DePuy demonstrated comparable outflow temperatures and lower water outflow rates than new devices. This may be due to the gasification of the metal head in DePuy devices after repeated use (Figure 3d) and the accumulation of tissue debris which can obstruct the outflow cannula, separately. Our study also revealed that surgeons’ operating practices significantly influenced the outflow water temperature. Intermittent use of RF devices and frequently moving away from the target tissue were associated with significantly lower outflow water temperatures. Hypothermia occasionally occurs due to continuous heat removal by the irrigation water, particularly during prolonged surgeries. Using irrigation water at higher temperatures can mitigate hypothermia, which is often suggested by anesthetists. However, as shown in this study, it also significantly increases the outflow water temperature, elevating the dermal thermal injury risk. We recommend using heated irrigation water cautiously and only when RF devices are used intermittently to minimize the risk of thermal injury.
We employed a simple and cost-effective method by cutting the drainage tube at the root. This allowed the hot water to flow to the surgeon’s hand first, prompting immediate adjustments if it was too hot. Connecting the RF tube to suction is another effective strategy to prevent hot water from contacting the patient’s skin directly. However, frequent switching of the suction connection between the RF device and the shaver can be inconvenient. Moreover, this practice accelerates water flow, and increases bleeding by Bernoulli Effect. 17 In several instances, we encountered total blockage of the outflow tract in reused RF devices. This is particularly concerning when arthroscopic cannulas are used, as natural water outflow through the incision is restricted or blocked, leaving the RF device’s outflow tract as the sole drainage pathway. To avoid overheating the intracavitary water, it is essential to ensure the patency of the RF device’s drainage tube. Applying unobstructed suction is essential to prevent both cutaneous and intracavitary thermal injuries on this occasion. 15 One surgeon at our institution used an antimicrobial incise drape to protect the skin from heat injuries. However, the effectiveness of this method remains uncertain.
In our study, the RF device was set at the highest energy level as our daily practice. Ahrens, P. et al. Pointed out that the energy level of the RF device (Turbo Vac, ArthoCare, Austin, Texas, USA) did not affect the intra-articular fluid temperature. 18 However, this conclusion was not validated in RF of different brands. It is difficult to duplicate the exact same condition as the dermal burn happened. As a result, we could not determine whether these burns were inherent to other technical errors. Besides, inter-surgeon variability may lead to bias. Our results only showed that reusing RF of certain brand will raise the outflow water temperature which will raise the chance of dermal burns. The chances can be minimized with awareness and precautionary measures mentioned above. In Zhejiang province, the reusing cycles of RF devices were determined at 5 times to ensure device efficacy and safety. With a permanent population of 66.7 million and more than 50 thousand arthroscopes per year, this policy has saved more than 150 million USD of medical expense during the past 20 years. No severe complications were reported according to the reginal medical products administration.
Reuse of suture-passing needles and needle breakage
Chung et al. reported remnant needle tip rate of 5.7% in a series of 283 patients. 19 Risk factors identified included thicker tendons, higher tendinosis degrees, and delaminated tears. They concluded that remnant needle tips did not result in prolonged negative effects. Similarly, Yang et al. observed a 4.3% rate of suture-passing needle breakage in a consecutive series of 437 shoulders, although the broken tip was retained in the joint in only one case. 20 Their study investigated the mechanical features of the needles and suggested that the notch on the needle might be unnecessary. Interestingly, we also encountered several cases of suture needle tip breakage but no apparent difficulty in tendon penetration or suture passage. However, whether the intact suture needle protected tendon fibers by smaller entrance should be further studied.
Our result demonstrated that the two disinfection methods did not affect the lifespan of suture-passing needles. However, high-pressure steam disinfection may cause plastic part of the suture-passing needle to deform or fall off (Figure 4g). Despite the disinfection method, reuse inevitably increases the number of excitation times, thereby elevating the risk of needle breakage. The increased fatigue cycles of Arthrex suture-passing needles did not mean its superiority of mechanical property. It may also result from the different bending mode due to different shape of tip (Figure 4d and e). In our study, the needles struck hard metal during each excitation, which we considered harsh working conditions. All the needles experimented exhibited more than 100 fatigue cycles. Considering the intraoperative usage conditions are much milder than the experimental conditions, it is conservative estimated that the suture passing needles can be safely reused for 5 times. The disinfection suppliers should also be careful to operate gently to avoid bending of the needles.
Several strategies can help prevent the breakage of suture-passing needles. When dealing with thick tendon tissue, the surgeon should clamp the tendon first, activating the device quickly to ensure a successful puncture. In the meantime, the suture-passing device should always be operated under direct arthroscopic supervision. Slight rotation of the device can help guide the needle puncture within the visual field, avoiding unintentional contact with the acromion and observing the integrity of the needle tip. The scrub nurse should also carefully inspect the needle’s integrity before and after every excitation. The subscapular tendon is thicker than the supraspinatus tendon, and it presents a greater challenge to avoid coracoid process at the exit position. Thus, we recommend using a suture hook to repair the subscapular tendon rather than a suture-passing needle. If the reused needle failed to pass smoothly through the tendon repeatedly, a new needle should be considered. Although a broken needle tip may initially cause no symptoms, its potential morbidities, such as migration to major vascular tissue, cannot be ignored. This risk is particularly concerning for patients with underlying conditions, such as Parkinson’s disease (Figure 2a and b).21,22
Beside striking the bony structures, which is mostly encountered, the failures also could be caused by other patterns like that the device was disengaged from the rotator cuff before the needle had completely retracted into the device or repeated bending within or beneath the tendon tissue. As a result, we could not provide the same condition as the suture needle broke during the surgery. As far as we know, there is no severe complication reported in the literature and recorded in the Rigional Medical Products Administration related to needle failures. In the past, many institutions around the world are reusing suture passing needles but the appropriate reusing cycles are not determined. After reusing limits were determined at 5 times and exercising practitioner education, needle failure rate had decreased to 1/10,000 within the regional hospitals.
Ethical and regulatory implications
It is ethically justifiable to adopt a less than perfect choice as an alternative to the best possible treatment due to limited resources. Reusing these consumables is part of the regional policy of public medical insurance. It aimed to cover and benefit more patients who are candidate for advanced treatment methods and reduce unfair health inequalities. To ensure the right to know, patients should be informed of the rare complications of reusing these consumables and given the right to choose whether to reuse them. The feasibility of reusing medical devices depends on safety and effectiveness assessment. Devices should undergo comprehensive evaluation in laboratory and clinical settings for appropriate recycling limits and potential risks before being adopted for widespread reuse. Low-temperature sterilization methods like hydrogen peroxide plasma sterilization and ethylene oxide sterilization have been proved effective and friendly to medical consumables. However, for reused medical devices, the quality of cleaning directly affects the effectiveness of sterilization. Insufficient cleaning may result in a high residual biological load, which could affect the sterilization outcome. Reutilization should be subject to regulation, with strict requirements for reprocessing and reprocessing verification, risk assessment and management, as well as employee training. All hospital personnel involved in arthroscopic surgeries should be educated and trained to avoid the associated complications before encountering them.
Conclusion
Reusing RF devices and suture-passing needles is feasible but carries the potential risk of morbidity for patients. These risks can be effectively mitigated by implementing preventive measures and determining appropriate recycling limits. Low-temperature sterilization methods are recommended for reused consumables. Furthermore, establishing robust educating, reporting and feedback mechanisms is crucial.
Footnotes
Ethical considerations
This study was approved by our institutional committee for clinical research.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. No benefits in any form have been received or will be received from a commercial party related directly or indirectly to the subject of this article.
