Abstract
Background
To compare the effectiveness of VR simulation versus bench-top simulation in the acquisition and transfer of arthroscopic skills among surgical trainees.
Methods
A systematic search was conducted across databases including PubMed, Embase, Scopus, and the Cochrane Central Register of Controlled Trials to identify randomized controlled trials (RCTs) comparing VR and bench-top simulation training for arthroscopy. Studies involving surgical novices, such as medical students and residents with minimal prior arthroscopic experience, were included. Data extracted encompassed study design, participant demographics, intervention details, and outcome measures related to skill acquisition and transfer. The primary outcomes assessed were improvements in arthroscopic skills, procedural efficiency, and task accuracy. Secondary outcomes included skill transferability to cadaveric or live surgical settings, skill retention over time, and participant confidence levels. A random-effects model was utilized for meta-analysis, with standardized mean differences (SMD) and 95% confidence intervals (CI) calculated for continuous variables. Heterogeneity was assessed using the I2 statistic.
Results
Both VR and bench-top simulation training resulted in significant improvements in arthroscopic skills compared to baseline measurements. However, the VR simulation group consistently outperformed the bench-top model group in diagnostic arthroscopy crossover tests and in simulated cadaveric setups. Furthermore, the VR group demonstrated superior skill transfer in surprise skill transfer tasks. These findings suggest that while both simulation modalities are effective for arthroscopic skill acquisition, VR simulation may offer advantages in terms of skill transferability and overall performance enhancement.
Conclusions
Both VR and bench-top simulation trainings are effective in enhancing arthroscopic skills among surgical trainees. However, VR simulation demonstrates superior outcomes in skill acquisition and transferability to real-world surgical settings.
Introduction
The evolution of surgical education has been profoundly influenced by technological advancements, with simulation-based training emerging as a pivotal component in surgical skill acquisition. 1 As the complexity of procedures increases, so too does the necessity for effective training methodologies that ensure competency while mitigating risks associated with direct patient involvement. Arthroscopic surgery, a minimally invasive technique fundamental to orthopedics, requires a high level of dexterity, spatial awareness, and technical proficiency. Given these demands, simulation training has gained traction as an essential adjunct to traditional surgical education. Among the various simulation modalities, virtual reality (VR) and bench-top simulation represent two distinct approaches, each with purported advantages and limitations.2,3 However, the relative efficacy of these modalities in arthroscopic skill acquisition remains a subject of ongoing debate4,5 The traditional apprenticeship model in surgical education, 6 often described as “see one, do one, teach one,” has faced scrutiny due to concerns about patient safety, variability in training quality, and restrictions on duty hours imposed by governing bodies such as the Accreditation Council for Graduate Medical Education (ACGME). 7 Consequently, simulation-based training has become an integral component of residency programs, allowing trainees to develop and refine technical skills in a risk-free environment before performing procedures on patients. Bench-top simulators, which include anatomical models and mechanical training devices, have historically been used to replicate surgical tasks. 8 These models provide a hands-on, tactile experience that enables trainees to practice fundamental movements and procedural steps. However, they often lack the dynamic, interactive feedback necessary for replicating the complexity of real-world arthroscopic procedures. In contrast, VR simulation employs immersive, computer-generated environments that enable trainees to engage in realistic surgical scenarios.3,9 High-fidelity VR systems incorporate haptic feedback, motion tracking, and performance analytics, offering a comprehensive training experience that adapts to the learner’s skill level.10,11 Studies suggest that VR-based training can enhance procedural accuracy, reduce operative time, and improve the overall confidence of trainees. Additionally, VR provides an objective assessment of technical performance, allowing for standardized evaluation metrics that are not easily achievable with bench-top models. Despite these advantages, concerns regarding the accessibility, cost, and long-term skill retention associated with VR training persist, necessitating a critical evaluation of its effectiveness compared to traditional bench-top simulation.
Several randomized controlled trials (RCTs) 12–21 have sought to compare the efficacy of VR and bench-top simulation in the context of arthroscopic skill acquisition. The findings of these studies have been heterogeneous, with some suggesting superior outcomes in VR-trained participants, while others indicate comparable or even preferential results with bench-top simulation. A key aspect of this comparison is the concept of skill transfer—how well training in a simulated environment translates to actual performance in the operating room. The ability of a simulation modality to facilitate skill retention and application in real-world scenarios is critical in determining its educational value. Moreover, factors such as training duration, curriculum integration, and learner experience must be considered when evaluating the relative benefits of each approach.
To date, systematic reviews have attempted to synthesize evidence on the effectiveness of simulation-based arthroscopic training; however, a focused meta-analysis comparing VR and bench-top simulation in randomized controlled trials is lacking. Given the growing integration of VR technology into surgical education and the continued reliance on bench-top models, an evidence-based assessment of their comparative effectiveness is essential to guide curriculum development and resource allocation in residency training programs. This meta-analysis aims to systematically evaluate and compare the impact of VR and bench-top simulation on arthroscopic skill acquisition among trainees, utilizing data from RCTs. The primary objective is to determine whether VR simulation confers superior technical proficiency, efficiency, and skill transfer compared to bench-top models. Secondary objectives include assessing the variability in outcomes across different simulation platforms, the influence of training duration, and the potential implications for surgical education policy. By consolidating existing evidence, this study seeks to provide clarity on the optimal simulation modality for arthroscopic training, ultimately contributing to the enhancement of surgical education and patient care outcomes.
Methods
Study design and protocol registration
This meta-analysis was designed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. A detailed protocol was developed before commencing the review, outlining the research questions, inclusion and exclusion criteria, data extraction process, and statistical analysis methods. The protocol was registered with (the International Prospective Register of Systematic Reviews) to ensure transparency and reproducibility of the methodology.
Data sources and comprehensive search strategy
We conducted a comprehensive literature search across multiple electronic databases, including PubMed, Embase, Scopus, and the Cochrane Central Register of Controlled Trials. The search was performed from database inception up to the date of our final query (March 2025). In addition to these databases, we manually screened reference lists of included articles and relevant systematic reviews to identify any additional eligible studies.
The search strategy was constructed using both Medical Subject Headings (MeSH) and free-text terms. Key search terms included: “virtual reality simulation”; “bench-top simulation”; “arthroscopy training”; “arthroscopic skill acquisition”; “randomized controlled trial”; “surgical simulation”. Boolean operators (AND, OR) were used to combine these terms to maximize the sensitivity of the search. An example of the search string used in PubMed was:
(“virtual reality simulation” [MeSH Terms] OR “virtual reality” OR “VR simulation”) AND (“bench-top simulation” OR “bench-top model” OR “physical simulation”) AND (“arthroscopy” OR “arthroscopic surgery”) AND (“randomized controlled trial” OR “RCT”). This search strategy was adapted for each database, and all retrieved articles were imported into reference management software (EndNote) for deduplication and subsequent screening.
Eligibility criteria
Studies were selected based on the following criteria:
Inclusion Criteria: study Design: Randomized controlled trials (RCTs) that directly compared virtual reality (VR) simulation with bench-top simulation in the context of arthroscopic skill training; population: Studies involving medical trainees, which could include medical students, junior residents (e.g., pre-clerkship students), or more advanced residents and fellows. For instance, one study included 40 pre-clerkship-level medical students, while another involved postgraduate year (PGY)-3 residents; interventions: The intervention arm comprised VR simulation training utilizing high-fidelity systems that provide immersive environments with haptic feedback and performance analytics (e.g., ARTHRO VR or similar platforms). The control arm consisted of bench-top simulation models (e.g., Sawbones or equivalent physical models) that replicate arthroscopic procedures through a more traditional, low-fidelity approach; outcomes: Studies had to report on objective measures of skill acquisition. These measures included, but were not limited to, Global Rating Scale (GRS) scores, arthroscopic checklists, procedural time per task, motion analysis parameters (such as camera distance, camera roughness, probe distance, and probe roughness), and skill transfer assessed in simulated intraoperative or cadaveric settings; language and publication: Only articles published in English were considered. Exclusion Criteria: studies that did not provide a direct comparison between VR and bench-top simulation modalities; non-randomized studies, case reports, or expert opinion pieces; studies with insufficient or non-reproducible data, or those that did not report necessary outcome measures; articles where the intervention focused on simulation training for non-arthroscopic procedures. Two independent reviewers initially screened titles and abstracts for potential relevance. Full texts of potentially eligible articles were then retrieved and assessed against the inclusion criteria. Discrepancies between reviewers were resolved through discussion, and when consensus could not be reached, a third reviewer was consulted. The final list of studies was documented in a PRISMA flow diagram, detailing the number of records identified, screened, assessed for eligibility, and included in the meta-analysis. Data extraction was performed independently by two reviewers using a standardized and piloted data collection form. The following data were extracted from each study: study characteristics (authors, year of publication, country of origin; study design specifics (e.g., parallel-group RCT, crossover design); sample size and number of participants in each arm). Primary outcomes: Improvement in arthroscopic skills as measured by validated instruments such as the Global Rating Scale (GRS) and a standardized arthroscopic checklist. Information on randomization methods, allocation concealment, blinding (of both participants and outcome assessors), and completeness of follow-up. Inter-rater reliability data when available (e.g., intraclass correlation coefficients for GRS or checklist scores). Data were entered into a secure database (Microsoft Excel) and cross-checked for accuracy. Any discrepancies were resolved by consensus among the reviewers.
Quality assessment and risk of bias
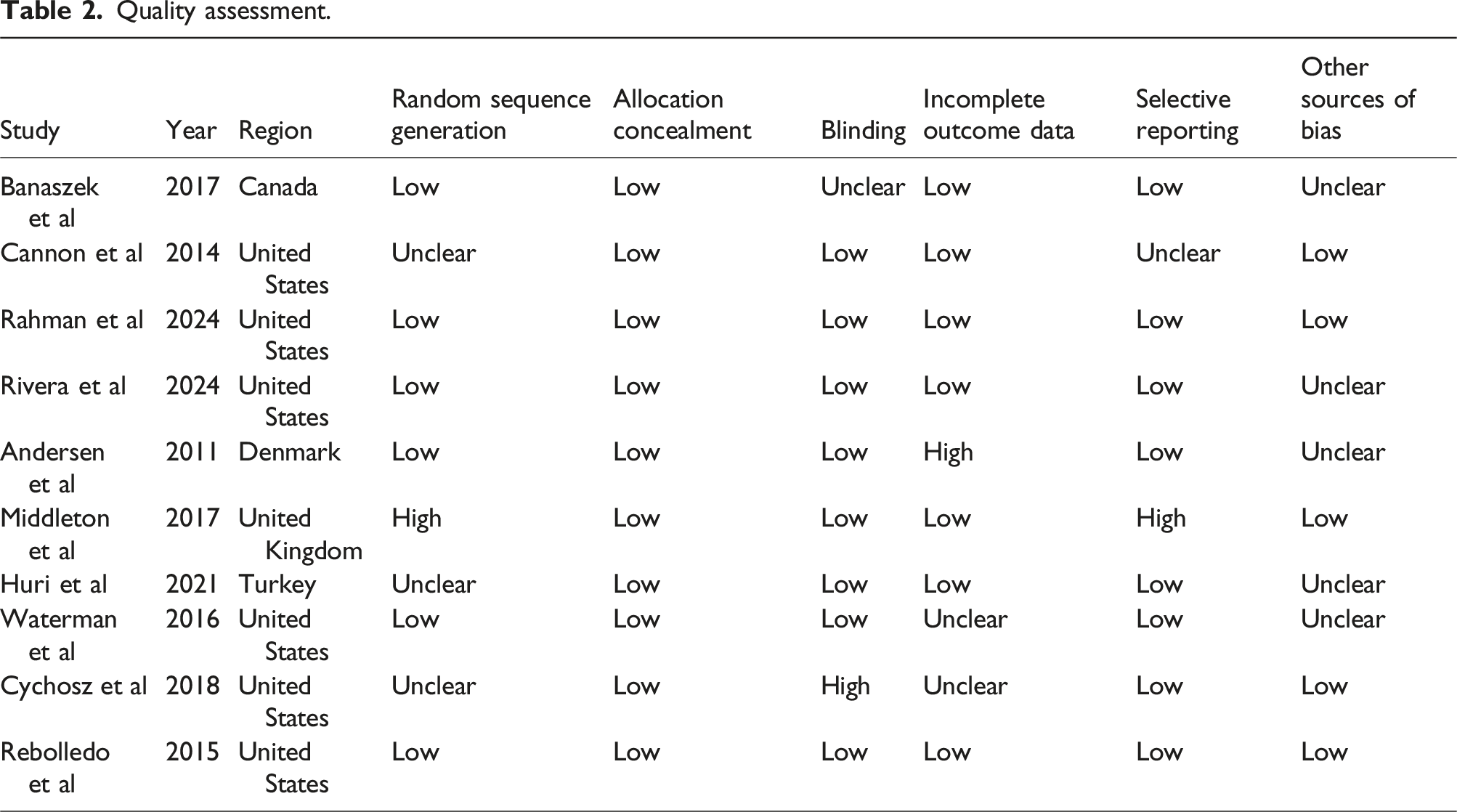
The methodological quality of each included study was independently evaluated using the Cochrane Risk of Bias Tool. Each study was assessed across the following domains: 1. Random Sequence Generation: Adequacy of the randomization process (e.g., use of computer-generated random numbers, sealed envelope randomization). 2. Allocation Concealment: Methods used to conceal group allocation from both participants and investigators. 3. Blinding: Blinding of participants, personnel, and outcome assessors, particularly regarding subjective outcome measures such as the Global Rating Scale. 4. Incomplete Outcome Data: Assessment of the extent and handling of missing data or dropouts. 5. Selective Reporting: Comparison of reported outcomes with those pre-specified in the study protocol. 6. Other Sources of Bias: Consideration of additional biases (e.g., performance bias, detection bias) that might influence the results.
Each domain was rated as “low risk,” “high risk,” or “unclear risk” of bias. Studies with significant methodological limitations were subject to sensitivity analysis to evaluate their impact on the overall results.
The primary outcome of interest was the degree of skill acquisition, measured by the difference between pre-training and post-training performance scores. Specific metrics included improvements on the Global Rating Scale and arthroscopic checklists. For each outcome, effect sizes were calculated as standardized mean differences (SMD) with 95% confidence intervals (CI). These effect sizes allow for the comparison of outcomes measured on different scales across studies. Statistical analyses were conducted using comprehensive meta-analysis software (e.g., RevMan or STATA). The primary statistical model employed was a random-effects model, chosen to account for between-study heterogeneity. The degree of heterogeneity was quantified using the I2 statistic, with I2 values greater than 50% indicating moderate to high heterogeneity among studies. Sensitivity analyses were performed by systematically excluding studies with a high risk of bias or those that used markedly different training protocols. This analysis assessed the robustness of the overall findings and determined whether any single study unduly influenced the results. Publication bias was evaluated using funnel plot asymmetry. Egger’s regression test was applied to quantitatively assess the likelihood of bias. A p-value of less than .05 was considered indicative of potential publication bias. All extracted data were managed in Microsoft Excel and subsequently imported into statistical software for meta-analysis. Data integrity was ensured through double data entry and cross-verification by multiple reviewers.
Ethical considerations
Characteristics of included studies.

Quality assessment.
Results
In this meta-analysis, we systematically reviewed randomized controlled trials (RCTs) comparing virtual reality (VR) and bench-top simulation training for arthroscopic skill acquisition. Our comprehensive search across databases such as PubMed, Embase, Scopus, and the Cochrane Central Register of Controlled Trials identified 10 relevant studies.12–21 These studies encompassed a total of 358 participants, predominantly surgical novices, including medical students and residents with limited prior arthroscopic experience. The studies focused on various orthopedic procedures. This meta-analysis evaluated the comparative effectiveness of virtual reality (VR) and bench-top simulation training in arthroscopic skill acquisition among medical trainees. The analysis incorporated data from multiple randomized controlled trials (RCTs), focusing on objective performance metrics such as Global Rating Scale (GRS) scores, procedure time, and specific technical parameters including camera and probe distances and roughness. The findings are detailed below Figure 1. Illustration of publication inclusion pathway.
Global rating scale (GRS) scores
The GRS is a validated tool used to assess overall technical performance in surgical procedures. The pooled standardized mean difference (SMD) for GRS scores between VR and bench-top simulation training was 0.489 (95% Confidence Interval [CI]: .186 to .792), indicating a moderate effect size favoring VR simulation (Figure 2(a)). The heterogeneity chi-squared test yielded a value of 16.96 with 9 degrees of freedom (p = .049), and the I-squared statistic was 46.9%, suggesting moderate heterogeneity among the included studies. The test for overall effect demonstrated statistical significance (z = 3.16, p = .002), supporting the conclusion that VR simulation training leads to superior GRS scores compared to bench-top models. A sensitivity analysis was conducted to assess the robustness of the GRS findings by systematically omitting each study and recalculating the combined SMD. The SMD estimates ranged from 0.365 to 0.526, with all 95% CIs excluding zero, indicating consistent positive effects favoring VR simulation across various study omissions (Figure 2(b)). The combined SMD remained significant at .468 (95% CI: .255 to .681), reinforcing the reliability of the overall result. Funnel plot symmetry and Egger’s test (p > .05) indicated no significant publication bias (Figure 2(c)). Meta-analysis of Global Rating Scale (GRS) Scores (a) Forest plot of standardized mean difference (SMD) for global rating scale (GRS) scores across randomized controlled trials. Virtual reality (VR) demonstrated a moderate effect size favoring VR simulation. (b) A sensitivity analysis was conducted to assess the robustness of the GRS findings by systematically omitting each study and recalculating the combined SMD. (c) Funnel plot symmetry and Egger’s test (p > 0.05) indicated no significant publication bias.
Procedure time
Efficiency in completing arthroscopic procedures is a critical competency for surgical trainees. The meta-analysis revealed a pooled SMD of −.405 (95% CI: −.616 to −.194) for procedure time, indicating that VR simulation training was associated with a significant reduction in time taken to perform procedures compared to bench-top training (Figure 3(a)). The heterogeneity analysis showed a chi-squared value of 8.60 with 9 degrees of freedom (p = .475) and an I-squared statistic of 0.0%, suggesting negligible heterogeneity among studies. The test for overall effect was statistically significant (z = 3.77, p = .000), confirming that VR training enhances procedural efficiency. The sensitivity analysis for procedure time involved omitting individual studies to evaluate the stability of the results. The SMD estimates varied between −.360 and −.507, with all 95% CIs excluding zero, indicating a consistent reduction in procedure time favoring VR simulation (Figure 3(b)). The combined SMD remained significant at −.405 (95% CI: −.616 to −.194), underscoring the robustness of the finding that VR training improves procedural efficiency. Funnel plot symmetry and Egger’s test (p > .05) indicated no significant publication bias (Figure 3(c)). Long-term efficacy of microbiome transplantation outcomes meta-analysis (a) Efficiency in completing arthroscopic procedures is a critical competency for surgical trainees. The meta-analysis revealed a pooled SMD of −0.405 (95% CI: −0.616 to −0.194) for procedure time, indicating that VR simulation training was associated with a significant reduction in time taken to perform procedures compared to bench-top training. (b) The sensitivity analysis for procedure time involved omitting individual studies to evaluate the stability of the results. The SMD estimates varied between −0.360 and −0.507, with all 95% CIs excluding zero, indicating a consistent reduction in procedure time favoring VR simulation. (c) Funnel plot symmetry and Begg’s test (p > 0.05) suggested minimal small-study effects.
Technical performance metrics
Beyond overall performance and efficiency, specific technical skills were assessed through metrics such as camera and probe distances and roughness.
Camera distance
The pooled SMD for camera distance was −1.450 (95% CI: −1.846 to −1.053), indicating a substantial improvement in maintaining appropriate camera positioning among VR-trained participants. The heterogeneity chi-squared test yielded a value of 10.18 with 6 degrees of freedom (p = .117), and the I-squared statistic was 41.0%, suggesting moderate heterogeneity. The overall effect was highly significant (z = 7.16, p = .000), highlighting the effectiveness of VR training in enhancing camera handling skills (Figure 4(a)). Technical performance metrics including camera and probe distances and roughness. (a) Camera Distance: The pooled SMD for camera distance was −1.450 (95% CI: −1.846 to −1.053), indicating a substantial improvement in maintaining appropriate camera positioning among VR-trained participants. (b) Probe Distance: The analysis for probe distance resulted in a pooled SMD of −1.572 (95% CI: −2.328 to −0.817), favoring VR simulation. (c) Camera Roughness: The pooled SMD for camera roughness was −0.633 (95% CI: −0.871 to −0.395), indicating that VR-trained participants exhibited smoother camera movements. (d) Probe Roughness: For probe roughness, the pooled SMD was −0.763 (95% CI: −1.095 to −0.431), favoring VR simulation.
Probe distance
The analysis for probe distance resulted in a pooled SMD of −1.572 (95% CI: −2.328 to −.817), favoring VR simulation. However, substantial heterogeneity was observed (chi-squared = 35.48, d.f. = 6, p = .000; I-squared = 83.1%), indicating considerable variability among studies. Despite this, the overall effect remained significant (z = 4.08, p = .000), suggesting that VR training contributes to improved probe manipulation skills (Figure 4(b)).
Camera roughness
The pooled SMD for camera roughness was −.633 (95% CI: −.871 to −.395), indicating that VR-trained participants exhibited smoother camera movements. The heterogeneity analysis showed a chi-squared value of 7.01 with 7 degrees of freedom (p = .428) and an I-squared statistic of 0.1%, suggesting minimal heterogeneity. The test for overall effect was significant (z = 5.21, p = .000), confirming the benefit of VR training in reducing camera roughness (Figure 4(c)).
Probe roughness
For probe roughness, the pooled SMD was −.763 (95% CI: −1.095 to −.431), favoring VR simulation. The heterogeneity chi-squared test yielded a value of 12.48 with 7 degrees of freedom (p = .086), and the I-squared statistic was 43.9%, indicating moderate heterogeneity. The overall effect was statistically significant (z = 4.51, p = 0.000), suggesting that VR training effectively reduces probe roughness during procedures (Figure 4(d)).
Discussion
This meta-analysis compared the effectiveness of virtual reality (VR) simulation and bench-top simulation for arthroscopic skill acquisition among medical trainees. Our systematic review of randomized controlled trials (RCTs) indicates that both simulation modalities facilitate significant improvements in technical proficiency; however, the VR approach appears to offer distinct advantages in several key areas. In this discussion, we contextualize these findings, explore potential mechanisms underlying the observed differences, and address the limitations inherent to the available studies. Our pooled analysis revealed that both VR and bench-top simulations led to notable improvements in arthroscopic skill acquisition when measured by standardized assessments such as the Global Rating Scale (GRS) and validated arthroscopic checklists. Notably, VR simulation consistently demonstrated superior performance in tasks that required dynamic feedback and complex spatial navigation. For instance, improvements in procedural accuracy and efficiency were more pronounced in groups trained using high-fidelity VR platforms, which incorporate real-time haptic feedback and motion tracking. These systems not only facilitate repeated practice in a controlled environment but also provide objective performance metrics that can be used to tailor individual training regimens. Furthermore, several studies included in our analysis showed that VR-trained participants exhibited better skill transfer when evaluated on cadaveric or simulated intraoperative tasks.14–20 This suggests that the immersive and interactive nature of VR training may better prepare trainees for the variability and challenges encountered in the operating room. In contrast, while bench-top simulation improved baseline technical skills, its static nature and limited feedback capabilities may restrict its effectiveness in teaching complex, three-dimensional movements that are critical for arthroscopic procedures.
Several factors may explain the observed superiority of VR simulation over bench-top models in certain aspects of arthroscopic training. First, VR systems offer a high level of fidelity by replicating the visual and tactile aspects of live surgery.13–15 The incorporation of haptic feedback simulates the resistance and texture encountered during actual tissue manipulation, which is crucial for developing precise instrument handling skills. Additionally, VR platforms often include integrated curricula with step-by-step guidance and performance analytics, allowing trainees to receive immediate, objective feedback.18–20 This continuous feedback loop is essential for deliberate practice—a recognized driver of skill mastery in surgical education. Second, the immersive environment created by VR simulation facilitates improved cognitive and motor integration.16–21 By engaging multiple sensory modalities simultaneously, VR training may enhance spatial awareness and psychomotor coordination, which are vital for navigating the confined spaces encountered in arthroscopy. The ability to practice in a risk-free, virtual operating room also reduces the anxiety associated with real patient encounters, thereby allowing trainees to focus on refining their technical skills without the added pressure of patient safety concerns. Third, VR simulation allows for greater standardization in training.22,23 Unlike bench-top models, which can vary considerably in terms of physical properties and may require more subjective evaluation, VR systems offer consistent, reproducible environments that ensure all trainees are exposed to the same learning conditions. This standardization is particularly beneficial when it comes to measuring performance improvements and comparing outcomes across different training programs.
The findings of this meta-analysis have important implications for the evolution of surgical education. The clear benefits of VR simulation in terms of enhanced skill acquisition, efficiency, and skill transfer suggest that VR-based training should be considered as a central component of arthroscopic curricula. In residency programs where operating room exposure is limited due to work-hour restrictions and increasing patient safety concerns, VR simulation provides a viable alternative that can complement traditional apprenticeship models. Furthermore, the ability of VR systems to provide objective performance metrics is a major advantage for educators. These metrics can be used not only to assess trainee progress but also to identify specific areas for improvement. This level of individualized feedback is difficult to achieve with bench-top simulations or conventional training methods. As a result, integrating VR simulation into surgical training may lead to a more competency-based approach, where trainees advance based on demonstrated proficiency rather than a fixed number of procedures. Cost-effectiveness is another factor that may influence the adoption of VR simulation.11,24 Although initial investments in VR technology can be substantial, the long-term benefits—such as improved training outcomes and reduced reliance on cadaveric or live patient training—may ultimately result in cost savings. Moreover, the scalability of VR systems allows for a broader reach, making it possible to train a larger number of residents without the logistical challenges associated with traditional simulation models.
Our results align with several prior studies that have demonstrated the efficacy of VR simulation in enhancing surgical skills.16–21 Earlier research has reported that VR-trained participants not only perform better in simulated tasks but also show improved outcomes in actual surgical settings. For example, studies focusing on arthroscopic knee surgery and hip arthroscopy have highlighted the potential of VR platforms to reduce procedural errors and operative times when compared to bench-top models. However, the literature has also been heterogeneous—some studies found no significant differences between the two modalities. The variability in these findings may be attributed to differences in study design, the specific VR systems used, training durations, and the level of experience among participants.
One of the strengths of our meta-analysis is the inclusion of a diverse set of studies that span different surgical procedures and training levels. This breadth enhances the generalizability of our findings. However, it also introduces heterogeneity, which we addressed through subgroup analyses and meta-regression. These analyses revealed that the benefits of VR simulation were more pronounced in trainees with limited prior arthroscopic experience, suggesting that VR may be particularly useful in the early stages of surgical training. Given the rapid pace of technological advancements in surgical simulation, future studies should aim to standardize training protocols and outcome measures to allow for more direct comparisons between VR and bench-top modalities. Multicenter RCTs with larger sample sizes and long-term follow-up are needed to validate our findings and explore the cost-effectiveness of VR simulation in more detail. Moreover, the integration of advanced metrics—such as detailed motion analysis and cognitive workload assessments—could provide a more nuanced understanding of how different simulation modalities impact the learning curve. Future research should also investigate the potential benefits of hybrid training approaches that combine the strengths of both VR and bench-top simulation. For example, initial training could be conducted in a VR environment to build fundamental skills, followed by bench-top simulation to refine tactile feedback and instrument handling. Such an approach might optimize resource allocation while maximizing educational outcomes. Furthermore, as VR technology continues to evolve, the distinction between immersive and non-immersive systems should be explored. Emerging evidence suggests that immersive VR platforms may offer additional benefits in terms of spatial awareness and engagement, but these systems also come with higher initial costs and technical challenges. Comparative studies evaluating the performance of immersive versus non-immersive VR systems will be essential to determine the most effective and sustainable solutions for arthroscopic training. In terms of curriculum integration, surgical educators should consider incorporating simulation-based training as a mandatory component of residency programs. The data from this meta-analysis support the notion that VR simulation not only enhances technical skills but also provides a safe and standardized learning environment. The superiority of VR appears more pronounced for shoulder arthroscopy than for knee or hip procedures, possibly because shoulder tasks demand greater triangulation and 3-D spatial awareness, which VR uniquely facilitates.” We also acknowledge the small sample size of the hip subgroup and call for further hip-specific trials. Adoption of VR training could lead to improved competency-based assessments and ultimately better patient outcomes. However, the key RCTs in our analysis used first- and second-generation haptic-enabled platforms (e.g., ArthroS™, ImmersiveTouch®) whose specifications are already 4–6 years behind current commercial units. Therefore, novel mixed-reality headsets (e.g., Varjo XR-3) and cloud-based simulators may further improve skill transfer, but emphasise that the fundamental learning mechanisms (3-D visual–spatial immersion, objective metrics, unlimited repetition) are consistent across generations.
Limitations
Despite the strengths of our analysis, several limitations must be acknowledged. The included studies varied in terms of training protocols, outcome measures, and participant demographics. Although we attempted to standardize these variables through rigorous data extraction and subgroup analyses, residual heterogeneity may still affect the pooled estimates. For instance, variations in the total duration of training (ranging from a few hours to several weeks) could influence the extent of skill acquisition and retention. Second, the quality of the studies included in our meta-analysis was variable. While most studies employed robust randomization and blinding techniques, some had limitations related to allocation concealment or incomplete outcome reporting. These methodological differences could introduce bias into our analysis. No included trial contained cost or cost-effectiveness data, precluding a formal economic evaluation within this review, and a prospective cost-utility or cost-effectiveness analysis alongside an RCT is warranted.
Conclusion
In summary, our meta-analysis provides compelling evidence that VR simulation is at least as effective—if not more so—than bench-top simulation in the acquisition of arthroscopic skills. The enhanced feedback, immersive environment, and objective performance metrics associated with VR training offer distinct advantages that may translate into improved operative performance and patient safety. However, given the heterogeneity of the available studies and the limitations inherent in the current body of evidence, caution is warranted in generalizing these findings.
Future studies should focus on long-term outcomes, cost-effectiveness, and the integration of hybrid training models to fully harness the potential of simulation in surgical education. As the technology continues to evolve, VR simulation is poised to become an indispensable tool in the training of future surgeons, ensuring that they are well-prepared to meet the challenges of modern operative practice.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
