Abstract
Background
Non-traumatic femoral head necrosis results from insufficient blood supply to the femoral head, causing hip pain, restricted movement, and eventual collapse. This study compares the efficacy of endoscopy-assisted lesion debridement with free vascularized fibular grafting versus core decompression (CD) to identify a more effective surgical approach for treating osteonecrosis of the femoral head (ONFH).
Methods
This retrospective study included 83 patients with femoral head necrosis who were treated between January 2010 and December 2020. The patients were divided into Group A (intraosseous endoscopic-assisted lesion debridement combined with free fibular grafting, n = 45) and Group B (CD, n = 38) based on their treatment approach. Preoperative general data, intraoperative variables such as operation time and blood loss, and postoperative assessments of Harris Hip Score, Visual Analog Scale (VAS), as well as imaging evaluations through X-ray and MRI for femoral head shape restoration and healing, were conducted at 1 week, 4 weeks, 3 months, 6 months, and 12 months post-surgery.
Results
A follow-up of more than 2 years was conducted for both groups of patients. Preoperative general data, such as age, showed no significant difference between the two groups (p > 0.05). Group A patients had significantly better Harris Hip Scores at 3, 6, and 12 months postoperatively, as well as significantly lower VAS scores at 4 weeks, 3 months, 6 months, and 12 months post-surgery compared to Group B (p < 0.05). In Group A, 3 patients experienced elevated body temperature (maximum 38.6°C), which was considered to be postoperative absorption heat; their temperatures returned to normal within 2–3 days following symptomatic treatment. All surgical incisions in both groups healed well, with no vascular or nerve injuries or other related complications. Additionally, imaging results showed that 4 hips in Group A developed femoral head collapse, compared to 10 hips in Group B. In Group A, 4 patients (4/45, 8.9%) with collapse were in ARCO stage II, while in Group B, 6 patients (6/38, 15.7%) were in ARCO stage I and 4 patients (4/38, 10.5%) were in ARCO stage II. Overall, Group A had a non-collapse rate of 91.1% (41/45) in early-stage femoral head necrosis, which was significantly higher than Group B’s 73.7% (28/38) (p < 0.05).
Conclusion
Intraosseous Endoscopic-assisted lesion debridement combined with free fibular grafting shows superior clinical efficacy compared to CD in the treatment of femoral head necrosis. It effectively improves femoral head morphology, alleviates pain, restores joint function, and has a lower complication rate, making it a promising approach for further clinical application and promotion.
Keywords
Introduction
Osteonecrosis of the femoral head (ONFH) is an orthopedic condition caused by various factors, such as steroid use, and alcohol abuse, leading to ischemic necrosis of the femoral head and, if untreated, resulting in femoral head collapse and loss of hip joint function, significantly affecting the quality of life.1,2 The early symptoms of ONFH typically include intermittent hip pain and restricted movement. As the disease progresses, the mechanical strength of the femoral head weakens, and structural changes occur, ultimately leading to the potential need for total hip arthroplasty.3–6 However, total hip arthroplasty is associated with complications such as infection, prosthesis loosening, and a limited lifespan of the implant.7–9 Therefore, preserving the natural joint and delaying arthroplasty are key goals in ONFH treatment. Non-surgical options, such as medication and physical therapy, help relieve pain and improve function in the early stages but have limited effect on disease progression.10,11 For patients in whom conservative treatment is ineffective, surgical treatment is crucial, with common methods including femoral head core decompression (CD) and bone grafting. 12 Among these, femoral head drilling CD is currently one of the most commonly used methods for treating ONFH. 13 For pre-collapse ONFH, CD is an effective surgical procedure that has been proven over more than 50 years to be more effective than non-surgical treatments for pre-collapse lesions.14,15 It works by relieving intramedullary pressure, improving local blood circulation, and promoting the formation of new bone tissue.16,17 However, this method has limitations, including inaccurate localization and incomplete removal of the necrotic area. 18 A study highlighted the benefits of flexible intraosseous endoscopy during CD, as it enabled the identification of avascular bone not visible on preoperative MRI, facilitated a more precise and thorough femoral head debridement, and contributed to improved clinical outcomes in 85% of patients at a follow-up of 27–45 months. 19 Fibular grafting can accelerate femoral head repair by providing structural support and promoting vascularization, especially in cases of extensive necrosis, offering significant advantages.20–22 This study aims to compare the efficacy of endoscopy-assisted lesion debridement combined with free fibular vascularized transplantation with traditional femoral head drilling CD, providing scientific reference for the clinical treatment of ONFH.
Methods
Study subjects and grouping
A retrospective analysis was conducted on 90 early ONFH patients treated at hospital from January 2010 to December 2020. After excluding patients lost to follow-up, a total of 83 patients who met the inclusion criteria were included in the study. The inclusion criteria were as follows: (1) patients met the surgical indications for early ONFH, including pain or functional limitation 23 ; (2) Classified as Association Research Circulation Osseous (ARCO) stage I or II 24 ; (3) Aged 18–65; (4) Signed informed consent for surgery; (5) Follow-up≥2 years. The exclusion criteria were as follows: (1) Femoral head necrosis due to trauma or fracture; (2) Hip dysplasia, femoroacetabular impingement, or labral tears; (3) Tumor-related diseases; (4) Patients with a history of hip joint surgery. The patients were divided into two groups based on the surgical method: 45 patients underwent endoscopy-assisted lesion debridement combined with free fibular vascularized transplantation (Group A), and 38 patients underwent traditional CD (Group B). Preoperative assessment included anterior-posterior and frog-leg X-rays of the pelvis, as well as hip joint magnetic resonance imaging (MRI) for all patients. All patients included in the study were evaluated for the necrotic area and location based on the Japanese Investigation Committee (JIC) classification system. 25 The ARCO staging and JIC classification of ONFH were determined by two experts based on the patient’s symptoms, X-rays, and MRI images. If their opinions differed, a third expert’s opinion was used to finalize the preoperative staging. Kaplan-Meier survival analysis was then performed to compare the proportion of patients who did not experience femoral head collapse (ARCO stage III or above) within 3 years postoperatively. This study was approved by the Institutional Review Board of hospital, and informed consent was obtained from all patients (Ethics Approval No. 202412180637000547623).
Data collection and outcome assessment
Data collection included the clinical characteristics of both groups of patients, such as age, sex, body mass index (BMI), cause of osteonecrosis, ARCO staging, JIC classification, preoperative Visual Analog Scale (VAS) scores, and Harris Hip Scores during the perioperative period. During the surgery, the operation time and blood loss were recorded. Postoperatively, VAS scores and Harris Hip Function Scores were assessed at 1 week, 4 weeks, 3 months, 6 months, and 12 months.
Surgical procedure
Group A (endoscopy-assisted lesion debridement combined with free fibular vascularized transplantation)
Under general anesthesia, the patient is positioned in the supine position. For the fibular graft, a 10 cm incision is made on the lateral side of the calf, 6 cm below the fibular head. The deep fascia is incised to expose the lateral surface of the fibula, which is then cut 5 cm and 12 cm below the fibular head, with careful preservation of the peroneal artery and veins. The fibula is freed and trimmed to a length of 6 cm, wrapped in heparinized saline-soaked gauze for later use. For the hip procedure, a 3–5 cm longitudinal incision was made over the greater trochanter to establish an endoscopic portal for debridement. The femoral-neck tunnel was reamed to a final diameter of 10–13 mm, as confirmed intra-operatively with a calibrated reamer. Under endoscopic visualization, necrotic bone was excised, after which the vascularized fibular graft was introduced through the tunnel and secured with an absorbable screw. Finally, the graft’s peroneal artery and veins were anastomosed to branches of the lateral circumflex femoral vessels.
Postoperatively, patients receive anticoagulation therapy, vasospasm prevention, and vasodilation treatment. The drainage tube is removed within 48 h, and antibiotics are administered for 3 days. Rehabilitation begins on day 2 with lower limb muscle strengthening, followed by sitting and standing exercises on day 10, and weight-bearing exercises starting at 3 months. The incision is closed in layers using sterile techniques to ensure proper sealing and prevent infection. Patients then enter a short-term non-weight-bearing phase (4–6 weeks), during which physical therapy helps maintain joint mobility and prevent muscle atrophy. Regular imaging (e.g., X-rays, MRI) is performed to assess the integration of the fibular graft and femoral head repair progress. Weight-bearing is gradually reintroduced starting at 3 months, beginning with light weight-bearing and progressively increasing. Full weight-bearing is permitted after 6 months, with activity levels adjusted according to recovery. Follow-up visits occur every 3–6 months to monitor the restoration of blood supply to the femoral head and the fusion of the fibular graft, as shown in Figure 1. Schematic of the surgical procedure (a) establishment of the lateral approach prior to endoscopic examination; (b) clear visualization and debridement of the necrotic lesion under endoscopy; (c–e) demonstration of fibular grafting and successful vascular anastomosis during the procedure; (f–h) intraoperative fluoroscopy showing complete lesion removal and full implantation of the fibula into the lesion area.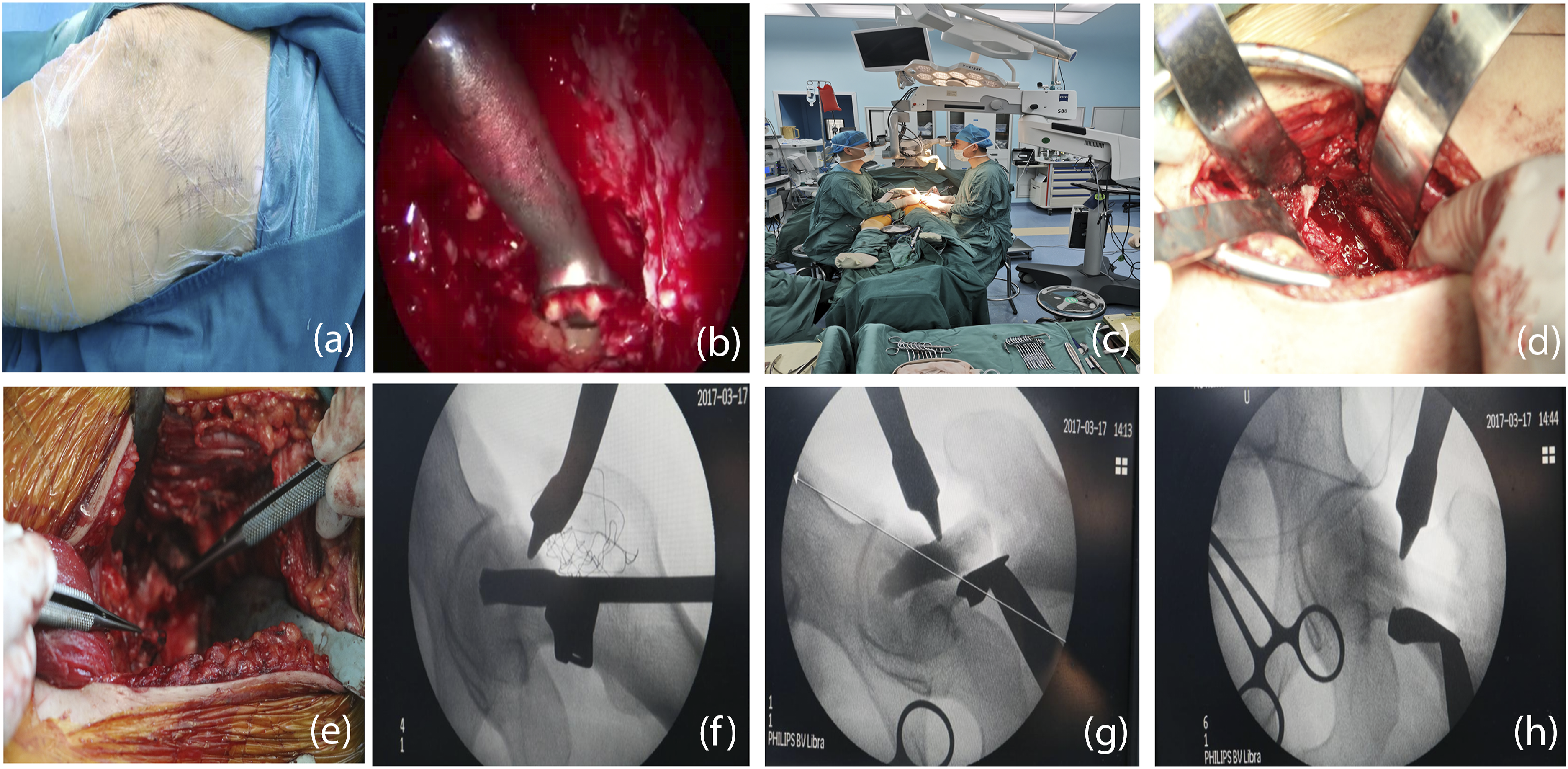
Group B (CD of traditional femoral head)
Under general anesthesia, the patient was positioned in the supine position and a percutaneous approach was used. Under fluoroscopic guidance, a guidewire (1.5 mm in diameter) was inserted through the proximal lateral cortex of the femur, penetrating the necrotic lesion. A cannula (3.0 mm in diameter) was then used to debride the necrotic tissue, followed by the creation of multiple drill holes (1.5 mm steel wire) to enhance decompression. No complications were observed during the procedure. Postoperatively, the patient entered a short-term non-weight-bearing phase, typically lasting 4 to 6 weeks, during which weight-bearing activities were strictly avoided. Physical therapy was initiated to maintain joint mobility. As recovery progressed, weight-bearing was gradually resumed at 3 months, with increasing load until full recovery was achieved.
Radiological evaluation
X-rays were obtained at 1 week, 4 weeks, and 12 months to monitor graft positioning. During each session, non-target areas—including gonadal regions—were shielded with a lead apron to minimize unnecessary radiation exposure. MRI was performed at 3, 6, and 12 months to assess graft revascularization and femoral head morphology.
Complication assessment
Postoperative complications were recorded, including infection, deep vein thrombosis of the lower limbs, and others.
Statistical analysis
Data analysis was performed using Statistical Package for the Social Sciences (SPSS) 26.0 statistical software (IBM SPSS Inc., Chicago, USA). Continuous data were expressed as mean ± standard deviation, and intergroup comparisons were performed using independent samples t-test. Categorical data were analyzed using the chi-square test. Kaplan-Meier survival curves were used to compare femoral head collapse-free rates between the two groups. A p-value of <0.05 was considered statistically significant.
Results
General data comparison
Characteristics of the participants between the two groups.

Comparison of surgery-related indicators
Comparison of surgical clinical outcomes between the two groups.

Comparison of postoperative hip VAS and Harris scores in both groups
During the postoperative follow-up, we compared the recovery of VAS and Harris scores between Group A and Group B. For VAS scores, no significant difference was observed between the two groups at 1 week postoperatively (Group A: 3.38 ± 1.35, Group B: 3.24 ± 1.57, p > 0.05). However, at 4 weeks postoperatively, Group A showed a significantly lower VAS score than Group B (p < 0.05). At 3 months, Group A’s VAS score was 1.72 ± 0.58, significantly lower than Group B’s 2.61 ± 0.65 (p < 0.05). At 6 months, Group A’s VAS score was 1.55 ± 0.71, again significantly lower than Group B’s 2.02 ± 0.85 (p < 0.05). At 12 months, Group A’s VAS score further decreased to 1.20 ± 0.43, significantly lower than Group B’s 1.58 ± 0.49 (p < 0.05).
Comparision of the mean post-operative data between the two groups.

Comparison of postoperative complications and collapse conditions
Both groups of patients were followed up for more than 2 years. In Group A, three patients experienced a mild fever (up to 38.6°C), which was considered to be post-surgical heat absorption. After symptomatic treatment, the fever subsided, and their body temperature returned to normal within 2–3 days. All incisions in both groups healed well, with no vascular or nerve injuries reported. Radiological results revealed that four hips in Group A experienced femoral head collapse, while ten hips in Group B showed collapse. In Group A, all four patients (4/45, 8.9%) with femoral head collapse were in ARCO stage II. In Group B, six patients (6/38, 15.7%) with collapse were in ARCO stage I, and four patients (4/38, 10.5%) were in ARCO stage II. Overall, the non-collapse rate of femoral heads in Group A treated for ONFH was 91.1% (41/45), significantly higher than the 73.7% (28/38) observed in Group B (p < 0.05) (Figure 2). Survival probability. The non-collapse rate of femoral heads in Group A treated for ONFH was 91.1% (41/45), significantly higher than the 73.7% (28/38) observed in Group B (p < 0.05).
Discussion
The treatment methods for ONFH are diverse, and the primary clinical focus has always been on effectively restoring femoral head morphology, alleviating pain, and improving joint function. 26 Many studies have reported the effectiveness of CD in reducing intraosseous pressure, alleviating pain, restoring blood flow, and promoting healing of necrotic tissue.27–30 However, for patients with extensive necrosis, CD alone is often insufficient and its effectiveness is limited.31,32 In contrast, free fibular vascular grafting provides additional bone support and enhances blood supply to the femoral head, significantly improving treatment outcomes.33–36
Our study results indicate that intraosseous endoscopy-assisted lesion debridement combined with free vascularized fibular grafting significantly improves hip joint function, restores femoral head morphology, alleviates pain, and accelerates functional recovery compared to core decompression alone. Additionally, our research demonstrates that this combined approach has a high safety profile in hip joint preservation surgeries. In the group A, three patients experienced a mild increase in body temperature, likely due to postoperative absorptive heat, which returned to normal within 2–3 days following symptomatic treatment.
Previous studies have also shown that free vascularized fibular grafting is an effective approach for treating ONFH by supporting the joint surface, reducing intraosseous pressure, removing necrotic tissue, and improving the local biological microenvironment.36–38 Interestingly, other study have shown that fibular vascular grafting, compared to CD, improves the vascular distribution and blood supply to the femoral head, with SPECT/CT analysis indicating significantly higher vascular distribution in the fibular vascular graft group at 6 and 36 months postoperatively. 39
The theoretical advantage of free vascularized fibular grafting lies in its provision of a new blood supply during surgery, allowing osteoinductive progenitor cells to infiltrate the previously necrotic area, thus revitalizing it and restoring healthy subchondral bone. 37 The fibula itself also has significant bone healing potential, and as an autograft, it is less likely to cause immune rejection. 40 Then,A study compared 98 hips treated with CD and 614 hips treated with free vascularized fibular grafting in patients with Ficat stages I-III osteonecrosis, showing that at 50 months of follow-up, the survival rate for stage II hips treated with free vascularized fibular grafting was 89%, compared to 65% for those treated with CD, and for stage III hips, the survival rate was 50% with free vascularized fibular grafting versus 21% with CD. 41 Additionally, A research suggests that scopically navigated CD of the femoral head reduces radiation exposure, enhances precision, and allows simultaneous treatment of additional pathologies, offering advantages over conventional techniques despite increased preparation time. 42 Our findings are consistent with previous studies. Although the surgical time and blood loss in Group A (vascularized fibular graft) were greater than in Group B (core decompression, CD) (p < 0.05), the VAS and Harris scores at 4 weeks postoperatively were significantly better in Group A (p < 0.05). Additionally, the non-collapse rate of the femoral head in Group A was 91.1% (41/45), significantly higher than the 73.7% (28/38) observed in Group B (p < 0.05). On the other hand, our innovative approach of using intraosseous endoscopy to treat ONFH, which enhances the removal of necrotic lesions, may have a positive impact on postoperative outcomes. This concept was initially suggested by Govaers et al., who intuitively recognized the role of endoscopic examination, using a 0° laparoscope to assess lesion penetration and the integrity of the subchondral bone. 43
And endoscopy can assist surgeons in precisely targeting lesions, performing thorough debridement, and providing accurate visual confirmation. 43
However, this study has some limitations. First, it is a single-center study with a small sample size. Future multi-center, large-scale randomized controlled trials are needed to further validate the efficacy of endoscopy-assisted lesion debridement combined with free vascularized fibula grafting. In addition, the optimization of postoperative rehabilitation protocols and the assessment of long-term follow-up outcomes require further investigation.
Conclusion
Intraosseous endoscopic-assisted lesion debridement combined with free vascularized fibula grafting demonstrates significantly better clinical efficacy than CD in the treatment of femoral head osteonecrosis. This approach effectively alleviates pain, restores joint function, and has a lower incidence of complications. It is a promising method that deserves further clinical application and promotion.
Supplemental Material
Supplemental Material - Intraosseous endoscopic-assisted lesion debridement combined with free fibular vascularized transplantation for non-traumatic femoral head necrosis: A comparative study with core decompression
Supplemental Material for Intraosseous endoscopic-assisted lesion debridement combined with free fibular vascularized transplantation for non-traumatic femoral head necrosis: A comparative study with core decompression by Yalong Yang, Zhao Zhang, Yuanyuan Wei, Yang Li, Junliang Liu, Bo Wang, Shouhu Mi, Jintong Liu, Yongqiang Zhang, Yongming Guo and Meng Wu in Journal of Orthopaedic Surgery
Footnotes
Author contributions
Conceptualization, Z.Z. and W.Y.Y.; methodology, L.Y.; software, L.J.L.; validation, W.B., M. S.H. and L. J.T.; formal analysis, Z. Y.Q.; investigation, G.Y.M.; resources, W.M.; data curation, Y. Y.L.; writing—original draft preparation, W.M.; writing—review and editing, Z. Y.Q.; visualization, W.M.; supervision, W.M.; project administration, Z.Z.; funding acquisition, Z.Z.. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Technology Development Fund of the Institute of Weapon Industry Health Research, project number K7202216.
Ethics statement
Data Availability Statement
The datasets generated and analyzed in this study are not publicly available as they are subject to the hospital’s privacy policies, but they can be obtained from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
