Abstract
Objective
This study investigates the effectiveness of tibia periosteum distraction (TPD) applied to the tibial periosteum, an innovative approach grounded in Ilizarov’s tension-stress theory, for the treatment of ischemic diabetic foot and vasculitic foot conditions.
Methods
A retrospective analysis was conducted on 33 patients (36 limbs) who underwent TPD between June 2019 and May 2022. The study comprised 21 males (23 limbs) and 12 females (13 limbs), aged 41 to 80 years (mean age: 63.4 years). Diabetic foot accounted for 27 cases, thromboangiitis obliterans for 2 cases, and arterial occlusive disease for 4 cases. The distribution of affected limbs included 15 left feet and 21 right feet. Periosteum traction commenced on the third postoperative day at a rate of approximately 0.75 mm/day, adjusted biweekly. The traction device was removed after two weeks. Evaluation included capillary refill and wound healing assessment, along with pre- and postoperative analysis of foot skin temperature, ankle-brachial index (ABI), visual analogue scale (VAS) pain scores, and peripheral blood oxygen saturation. CT angiography (CTA) was utilized to assess vascular conditions in both lower limbs.
Results
All 33 patients were successfully followed up for a duration ranging from 4 to 24 weeks (mean: 11.03 weeks). VAS pain scores significantly decreased from preoperative (5.09 ± 1.70, range: 2-8) to postoperative two weeks (2.24 ± 0.90, range: 1-4) (t = 9.44, p < .001). Oxygen saturation levels increased from 83.88% ± 11.82% (range: 58%-97%) preoperatively to 91.36% ± 5.69% (range: 76%-98%) at two weeks postoperatively (t = −4.21, p < .001). Foot skin temperature also showed a significant increase (t = −3.98, p < .001). Capillary refill test at two weeks postoperatively demonstrated notable improvement. CTA revealed evident neovascularization in the operated limbs compared to preoperative conditions. Wound improvement was significant in all 33 patients within two months postoperatively.
Conclusion
TPD emerges as a promising technique for chronic ischemic lower limb diseases, demonstrating favorable preliminary outcomes in wound healing promotion and amputation rate reduction. Nevertheless, large-scale randomized controlled trials are essential to further validate its efficacy.
Introduction
Chronic lower limb ischemic diseases, including diabetic foot, arterial occlusive disease, and thromboangiitis obliterans, are characterized by impaired microcirculation and varying degrees of distal lower limb ischemic symptoms.1,2 The pathophysiology of foot ulcers is influenced by neuropathy, vascular dysfunction, and immune system disorders. The underlying pathology involves stenosis and occlusion of the lower limb arteries, resulting in reduced local blood supply, ischemic rest pain, intermittent claudication, and ultimately ischemic necrosis of the toes. 3 These conditions often lead to foot infections, ulcers, and/or deep tissue damage associated with peripheral neuropathy and peripheral vascular disease. 4 Due to the high prevalence of comorbidities and compromised peripheral microcirculation, ulcers in these patients often exhibit delayed healing, which increases the risk of systemic infections. 5 Severe limb pain due to ischemia may eventually necessitate amputation.6–8
Current treatment approaches include medical management, local wound care, interventional procedures, and surgical interventions.9,10 However, medical and local wound care alone often yield unsatisfactory outcomes, prompting a shift towards interventional procedures and surgical interventions. Interventional methods currently employed include intraluminal stent intervention and balloon angioplasty. Interventional treatments offer the advantage of minimal invasiveness and are preferred for elderly and frail patients who cannot tolerate bypass grafting surgery. 2 However, it should be noted that interventional treatments carry the risk of intravascular intimal hyperplasia and thrombosis, which may lead to recurrent stenosis and occlusion. The long-term clinical efficacy of these interventions requires further investigation.7,11 Surgical vascular reconstruction is another viable treatment option, but it requires patients to meet certain physical requirements, including the ability to tolerate anesthesia and the presence of at least one patent outflow vessel. 12 Clinical observations have shown that in patients with occluded vessels in diabetic foot, most of the occlusions occur in the peripheral vessels, which are often unsuitable for interventional recanalization or bypass reconstruction, leading to limited clinical efficacy.13,14
Current therapeutic options include optimized conservative medical management, endovascular techniques like balloon angioplasty and stenting, surgical arterial bypass grafting, and some emerging regenerative medicine modalities. 15 However, complete revascularization is often precluded by extensive multilevel atherosclerotic occlusive disease and lack of suitable autologous vein conduits. 16 Endovascular recanalization alone may have suboptimal long-term patency for complex below-the-knee tibioperoneal lesions. 17 Nearly a third of diabetic patients still undergo major amputation within 5 years of diagnosis of critical limb ischemia.18–20 This underscores the need for disruptive innovations to achieve effective limb salvage and wound healing.
In recent years, although the Ilizarov technique-based transverse tibial bone transport has demonstrated satisfactory clinical results in the treatment of mid-to-late-stage ankle and foot diseases caused by chronic lower limb ischemic diseases, the method requires osteotomy in the middle of the tibia.15,21,22 This not only results in significant surgical trauma but also compromises the integrity of the bone structure, increasing the risk of postoperative fractures. 23 In clinical practice, we have observed that periosteal reaction plays a vital role in diagnosing bone tumors, indicating the essential role of periosteum in the musculoskeletal system. Therefore, we have designed a novel surgical technique called TPD, in which a traction device is inserted beneath the periosteum to apply controlled distraction forces and stimulate periosteal detachment from bone. Our preliminary canine model demonstrated significantly increased neovascularization after periosteal distraction. Since June 2019, we have been utilizing TPD for the treatment of chronic lower limb ischemic diseases.
Ilizarov’s principles of distraction osteogenesis provides a promising alternative approach to stimulate neovascularization. 24 Gradual controlled cyclic distraction forces applied to bone tissues elicit cellular proliferation responses not just in osseous cells but also in the surrounding soft tissues including muscle, fascia, vasculature, and nerves.25,26 This process is mediated through mechanotransduction pathways that convert biomechanical forces into cellular signaling.27,28 Animal studies reveal exuberant angiogenesis around the distraction site in response to progressive tensile strain.29,30 While tibial corticotomy with gradual bone transport has demonstrated potential in promoting angiogenesis, wound healing, and limb salvage in ischemic diabetic limbs,15,31 the risk of complications such as fracture, malunion, and infection remains a concern, accompanied by high costs and technical complexity associated with traditional techniques.
This study conducts a retrospective analysis of the TPD technique to: (1) Investigate the mechanism of periosteal distraction in improving limb ischemia; (2) Analyze its efficacy in patients with chronic lower limb ischemic diseases; (3) Summarize its potential advantages over conventional tibial bone transport techniques.
Materials and methods
Study design and participants
This was a retrospective case series study conducted at our hospital. We reviewed the medical records of patients who underwent TPD surgery for chronic limb ischemia between June 2019 and May 2022. The study was approved by the institutional ethics review board.
The inclusion criteria were: (1) Age 18-80 years; (2) Chronic lower limb ischemic diseases including diabetic foot, arterial occlusive disease, and thromboangiitis obliterans; (3) Treatment with TPD technique; (4) Availability of outcome data on foot skin temperature, peripheral oxygen saturation, pain scores, capillary refill, and wound healing.
The exclusion criteria were: (1) Severe arterial stenosis or occlusion on CT angiography with no distal runoff vessels; (2) Inability to comply with treatment due to mental disorders; (3) Active infection in periosteal traction area; (4) Incomplete follow-up data; (5) Advanced heart failure or renal failure.
General information
From June 2019 to May 2022, 47 patients underwent TPD treatment. Based on the inclusion and exclusion criteria, 33 patients (36 feet) were included in the final analysis. Among these patients, there were 21 males and 12 females, aged 41 to 80 years (mean age: 63.4 years). Diabetic foot was diagnosed in 27 patients, thromboangiitis obliterans in 2 patients, and arteriosclerosis obliterans in 4 patients. Exclusions were due to loss to follow-up (5 patients), death (6 patients), and refusal to continue participation (3 patients). Among the 33 patients, who experienced symptoms of rest pain before surgery, 11 patients had symptoms of decreased foot skin temperature, and 21 patients had wound infection and/or necrosis. Among the 33 patients in this group, 25 had hypertension, 16 had coronary heart disease, 29 patients with infection and/or necrosis in a single foot. 3 patients had infection and ulcers in both feet, and 1 patient with thromboangiitis obliterans had no wound but primarily experienced pain. The duration of foot ulcer necrosis ranged from 1 week to over 3 years. All patients underwent bilateral lower limb CTA before surgery but 29 patients 2 weeks after surgery underwent CTA.
This study was approved by the ethics committee, and all patients provided informed consent.
Surgical technique
Figures 1, 2 and 3. Illustration of the Surgical Procedure (I) (a) Longitudinal incision of approximately 1 cm is made below the tibial tuberosity on the medial side of the tibia. (b) After making a transverse incision on the tibial periosteum, a periosteal elevator is inserted beneath the periosteum towards the caudal direction. (c) The periosteum is carefully and slowly separated from the tibia to create a tunnel. Illustration of the Surgical Procedure(II) (a) A thin steel plate measuring 0.8 cm × 8.0 cm is inserted beneath the periosteum. (b) The steel plate is inserted distally beneath the periosteum, with its proximal end parallel to the periosteal incision. (c) Drill holes are made in the incision area using a 2.0 mm diameter drill bit for medullary decompression. Illustration of the Surgical Procedure(III) (a) The plate then moved in the reverse direction. (b) The plate is positioned centrally across the periosteal incision. (c) A countersunk hollow screw is screwed into the central hole of the plate, and a 1.5 mm diameter Kirschner wire is drilled into the hollow screw to secure the plate and the screw.

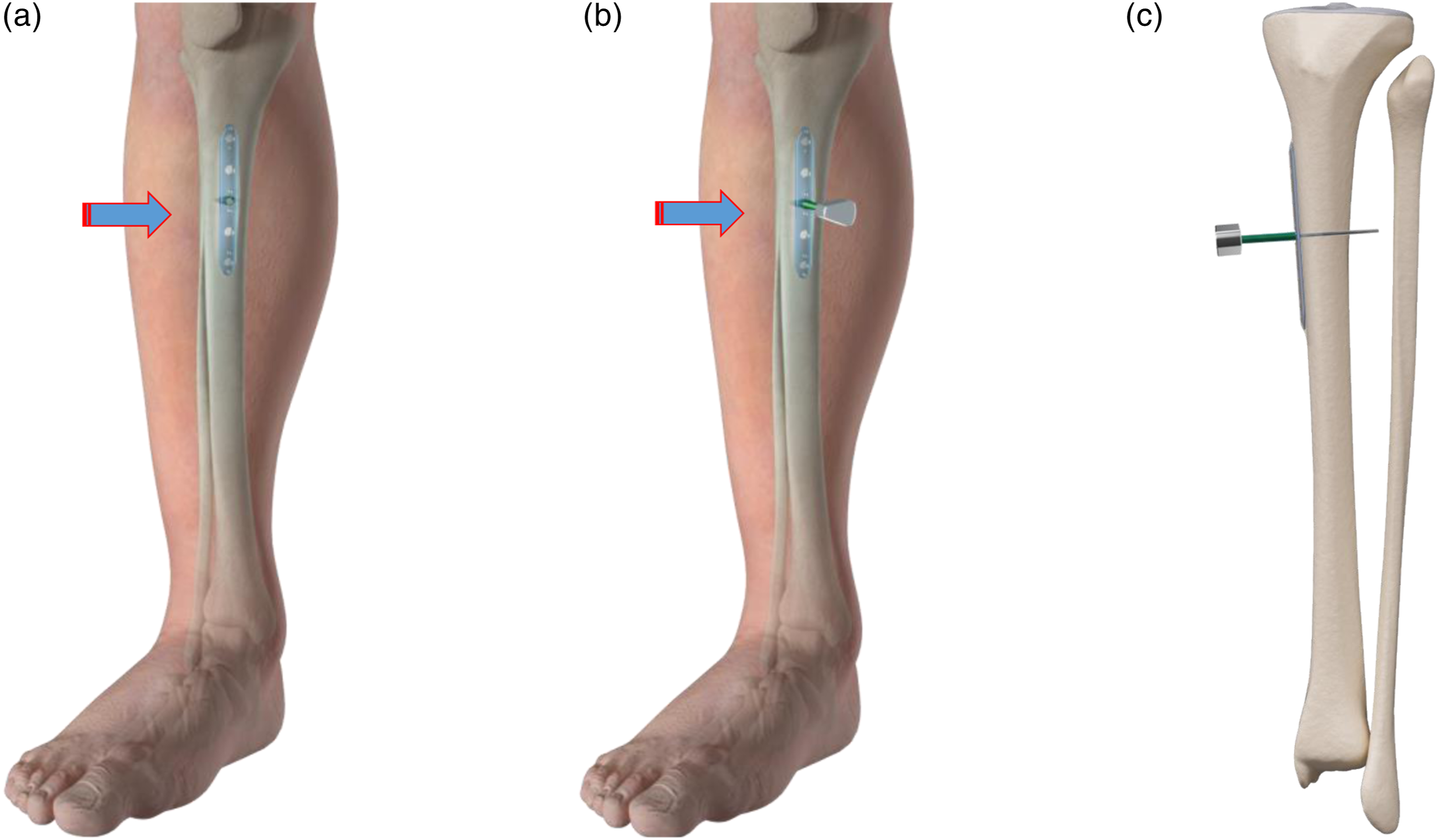
The detailed steps of the surgical technique are as follows
Following Subarachnoid anesthesia, spinal anesthesia or epidural anesthesia, the patient was placed in the supine position (Figure 4). Illustration of the Surgical Procedure(IV) (a) The exposed tail of the screw is rotated clockwise outside the skin to observe if the periosteal plate can be lifted along with the rotation of the screw. (b) Successful installation is indicated if the plate is lifted. (c) The cross-section shows that the steel plate has been placed between the tibia and the periosteum. The exposed tail of the screw outside the skin is rotated clockwise to lift the plate. (d) The steel plate has been elevated, and the periosteum is consequently lifted relative to the tibia.
Make a longitudinal incision approximately 1 cm in length along the medial side of the tibia, just below the tibial tuberosity. After exposing the periosteum, make a transverse incision on it, and insert a nerve stripper beneath it. Gently elevate and peel the periosteum to create a subperiosteal tunnel. Insert a specially designed periosteal detacher. along the direction in which the steel plate will be inserted, and strip the periosteum subperiosteally. The length of the periosteal detacher. should be the same as the length of the steel plate. Remove the periosteal detacher.
Use a 2.0 mm drill bit to drill four holes into the tibia for decompression of the medullary cavity. These four holes should only penetrate the cortex of one side of the bone. Two holes should be drilled above the transverse periosteal incision, and the other two should be drilled below it (these four holes should be located on either side of the longitudinal skin incision).
Insert a 0.8 cm × 8.0 cm thin steel plate into the deep layer of the periosteum at the incision site, towards the distal end of the body. Wait for the steel plate to be fully inserted beneath the deep layer of the periosteum before performing the reverse operation. Continue inserting this steel plate from the distal end of the body towards the head side along the deep surface of the periosteum until the plate is fully inserted. Once the periosteal incision is aligned with the center of the steel plate, insert a hollow screw with a flat head into the central hole of the steel plate. Insert a Kirschner wire with a diameter of 1.5 mm into the center of the hollow screw to fix the steel plate to the hollow screw. After completing these steps, the periosteal distractor is installed.
Turn the exposed screw clockwise, and the subperiosteal steel plate will be elevated along with the screw, lifting the periosteum. This demonstrates that the periosteal distractor has been successfully installed. Then, turn the screw counterclockwise, and the steel plate will gradually adhere to the cortical bone, further demonstrating that the periosteal distractor has been successfully installed. No traction treatment is performed during the operation.
After suturing the periosteum, suture the skin incision to close it. On the third day after surgery, begin periosteal distraction therapy. Turn the screw clockwise, and the steel plate on the deep side of the periosteum gradually moves away from the cortical bone at a rate of approximately 0.75 mm/day. Stop adjusting when the patient feels tension and/or pain. Take X-rays to confirm that the distraction device has elevated the periosteum. After approximately two weeks of adjustment, remove the Kirschner wire and the distractor.
Postoperative distraction protocol
Initiation of periosteal distraction
Distraction treatment of the periosteum commenced on the third day post-surgery (Figure 2). Rotating the screw clockwise facilitated the gradual displacement of the steel plate beneath the periosteum from the cortical bone, achieving a controlled rate of approximately less than 0.75 mm per day. Adjustment ceased upon the patient’s report of tension and/or pain. X-ray imaging was employed to verify the elevation of the periosteum by the distraction device. Following approximately two weeks of adjustment, the removal of both the Kirschner wire and the distraction device was undertaken. Figure 5. • Distraction treatment commenced on the third postoperative day. • The steel plate was gradually moved from the cortical bone at a controlled rate (∼0.75 mm/day). • Adjustments stopped upon patient reporting tension or pain. • X-ray imaging confirmed periosteum elevation. • The Kirschner wire and distraction device were removed after two weeks. The surgical procedure of TPD. (A): Make a longitudinal surgical incision about 1 cm long below the tibial tubercle on the medial aspect of the tibia. (B): Expose the tibial periosteum. (C): Make a transverse incision on the periosteum about 1 cm long. (D): At the periosteal incision site, place the periosteal elevator between the periosteum and the tibia. (E): Insert a specially designed periosteal elevator along the pre-determined direction for the insertion of the plate and strip the periosteum. (F): Drill holes with a 2.0 mm drill bit in the incision area for decompression of the medullary cavity. (G): Insert a 0.8 cm × 8.0 cm thin plate under the periosteum. (H): Screw an empty hollow lag screw into the central hole of the plate, and drill a 1.5 mm Kirschner wire through the center of the hollow lag screw to fix the plate and hollow lag screw, then suture the periosteum. (I): Intraoperative fluoroscopic examination to determine fixation status of the plate and hollow lag screw. (J): The plate is raised 2 weeks later.

Outcome assessments
(a)
Baseline: The average skin temperature on the body surface is (33.5 ± 0.5)°C.
Significance: Changes in skin temperature indirectly reflect the degree of microcirculation perfusion.
(b)
Baseline: Healthy arterial blood oxygen saturation is 98%, while venous blood oxygen saturation is 75%.
Significance: Measurement of oxygen saturation indirectly reflects the degree of peripheral microcirculation perfusion.
(c) Visual Analog Scale (VAS): Assessed pain intensity on a 10 cm scale from 0 (no pain) to 10 (unbearable pain).Significance: VAS is utilized to gauge the intensity of pain reported by the patient.
(d) Capillary Refill Test: Applied pressure to the skin, observing time for color return to normal (normal ≤ 2 seconds).
Significance: Reflects the microcirculation status in the lower extremities, with prolonged refill times indicating potential issues such as low blood pressure or shock.
These comprehensive assessments collectively contribute to evaluating the efficacy of
TPD in promoting microcirculation and facilitating postoperative recovery in patients with chronic lower limb ischemic diseases.
Research methods
To ensure measurement consistency, the first author exclusively conducted the measurement of skin temperature and oxygen saturation of the foot. The measurement method involved multiple measurements at 2-hour intervals (from 8:00 a.m. to 8:00 p.m.) over a 3-day period, resulting in 21 data points recorded and averaged for each patient.
Statistical analysis
Statistical analysis utilized SPSS version 20.0 software (SPSS Inc., USA). Continuous data were presented as mean ± standard deviation and compared using the paired t-test. Categorical data were presented as counts and percentages. The significance level (α) was set at 0.05 for two-tailed tests.
Results
Study participants
33 patients were included in the study, with a follow-up period ranging from 4 to 24 weeks and an average of 11.03 weeks.
Case presentation
Case 1, 2, 3. She XX: Female, 71 yrs, diabetes mellitus combined with atherosclerotic occlusion of both lower limbs, dry gangrene of the right foot for 4 months. (A). Preoperative necrosis of the second toe. (B). Preoperative CTA of both lower limb arteries. (C). Postoperative X-ray of the affected limb showed the position of periosteal retractor placement. (D). Postoperative 3 months, the skin blood flow of the foot has improved. (E). 3 months after surgery, CTA of both lower limb arteries, significant improvement compared with the preoperative period. (F). 9 months after surgery, the 2nd toe has fallen off and the wound has healed. (G). 4 years after surgery, good blood circulation in the foot, no recurrence of ulceration. Xiong XX, Male, 55 yrs old, lower limb arterial occlusive vasculitis, cold and blue feet, obvious pain. (A). right lower extremity aubergine. (B). Patient’s preoperative CTA. (C). CTA of both lower extremity arteries 4 weeks after periosteal distraction surgery. (D). 3 weeks after periosteal distraction surgery, the right foot became red and the pain symptoms were significantly relieved. (E). 3 weeks after tibial periosteal distraction surgery. Hou XX, Male, 69 yrs, diabetes mellitus combined with atherosclerotic occlusion, left foot infection and necrosis. (A). Intraoperative debridement of left foot. (B). CTA. (C). TPD surgery after infection control after 1 month. X-ray images confirmed successful periosteum elevation by the distraction device. (D). 1 month after periosteal distraction surgery, CTA of both lower limbs. (E). 5 months later, the left foot wound has healed. (F). 1 year later, the patient walked normally.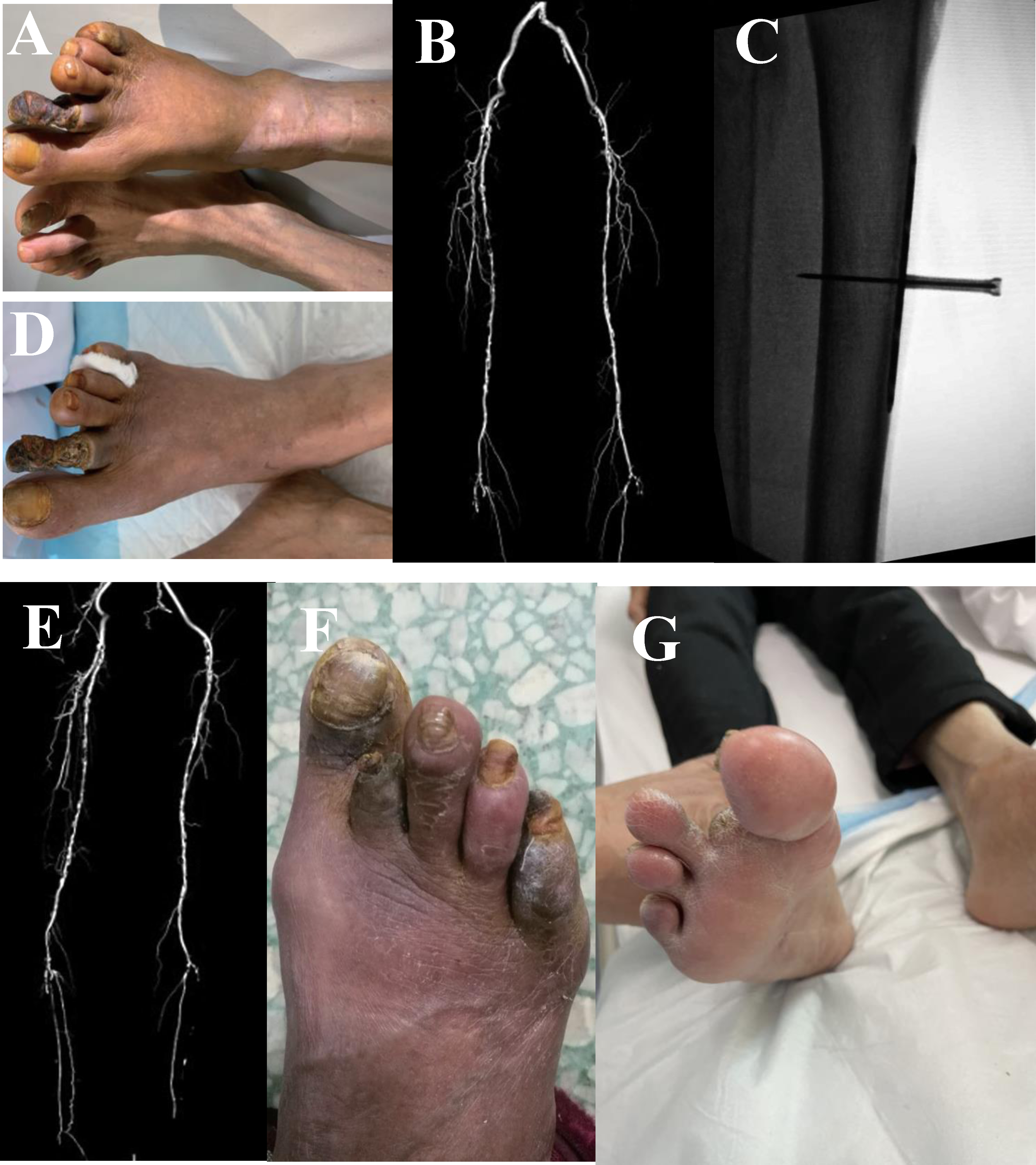


Computed Tomography angiography (CTA)
Preoperative CTA was performed on all patients; 25 underwent follow-up CTA two weeks post-distraction device removal, showing significant vascular network formation in the affected limb.
Foot Skin temperature
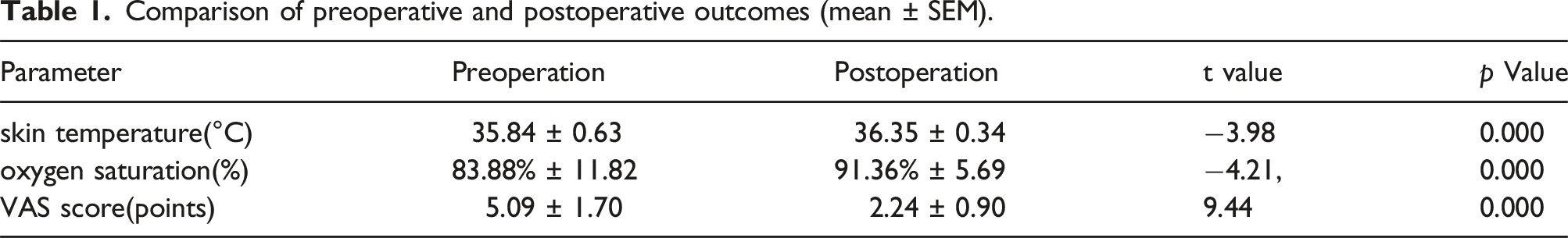
Among the 33 patients, 30 exhibited increased skin temperature on the dorsal foot after surgery. • Preoperative: (35.84 ± 0.63)°C (range: 34.70 °C–36.60 °C) • Postoperative (2 weeks): 36.35 ± 0.34°C (range: 35.50 °C–36.90 °C)
Peripheral oxygen saturation
• Preoperative: 83.88% ± 11.82% (range: 58%-97%) • Postoperative (2 weeks): 91.36% ± 5.69% (range: 76%-98%)
Pain scores (Visual Analog Scale - VAS)
• Preoperative: (5.09 ± 1.70, range: 2-8) • Postoperative (2 weeks): (2.24 ± 0.90, range: 1-4) (t = 9.44, P < .001)
Capillary perfusion and wound status
Capillary refill time was ≤2 seconds in all 33 patients at the 2-week postoperative follow-up, indicating significant improvement compared to preoperative measurements.
Complications
Comparison of preoperative and postoperative outcomes (mean ± SEM).
Discussion
Our findings demonstrate the clinical efficacy of TPD in improving limb ischemia and promoting wound healing in patients with chronic lower extremity peripheral vascular disease. The improvements in pain relief, tissue oxygenation, skin perfusion, capillary refill, and ulcer healing indicate enhanced microcirculatory perfusion in the affected limb. These results are consistent with previous studies on tibial bone transport techniques based on Ilizarov’s principles, which highlight the role of gradual cyclic distraction forces in stimulating cellular proliferation and neovascularization. 32
Biomechanical, molecular mechanisms and advantages
These results corroborate previous studies on tibial bone transport techniques grounded in Ilizarov’s principle of tension-induced tissue regeneration.33,34 Ilizarov first elucidated that gradual cyclic distraction forces applied to bone tissues provoke proliferative responses not just in osseous cells but also in the surrounding musculoskeletal and vascular tissues. 35 Animal experiments and clinical results have consistently evidenced exuberant neovascularization around the transport segment, attributable to upregulation of proangiogenic growth factors such as VEGF, angiopoietin, MMPs, PDGF, TGF-β, and cytokines like MCP-1.30,36,37 Our findings demonstrate that periosteal distraction, without disrupting bone integrity, can replicate these proangiogenic effects.
We postulate that the biomechanical loading on the highly vascular periosteum triggers reactive hyperplasia and activation of dormant vessels, thereby establishing collateral circulation in the ischemic limb. The periosteal distraction likely induces fluid shifts and hemorrhage within the medullary cavity, which communicates externally through the cortical drill holes. This resultant hematoma and inflammatory milieu provide chemotactic stimuli for inflammatory cell infiltration, extracellular matrix deposition, and endothelial sprouting.38,39 Macrophages transitioning from inflammatory M1 to pro-healing M2 phenotypes may release angiogenic factors like VEGF, PDGF, and FGF-2. 40 Mesenchymal stem cells recruited to the distraction site can also secrete angiogenic signals.41,42 The simultaneous periosteal and endosteal bleeding induced by this technique provides dual impetus for vascular proliferation.
The precise biomechanical mechanisms underlying this angiogenic response warrant further investigation but likely involve both biochemical and biophysical factors. The controlled cyclic tensile strain alters cell shape and cytoskeletal tension, triggering signaling cascades that activate proliferation, migration, and secretion of bioactive molecules.43,44 The mechanical forces may also directly impact the extracellular matrix, enhancing its permeability and release of sequestered growth factors [40]. Molecular pathways elucidated in distraction osteogenesis models like Wnt, BMP, Notch, HIF-1α, and PI3K/Akt signaling likely also play key roles. 45 Detailed understanding of these mechanobiology processes can help refine protocols to maximize angiogenesis.
Compared to conventional transverse tibial bone transport, our innovative approach offers several advantages. It is minimally invasive, preserving bone integrity and averting complications like nonunion, fracture, and osteomyelitis. The simplified distractor device curtails costs and permits outpatient surgery. Unhindered mobility and rapid rehabilitation improve patient comfort and compliance. The ability to target multiple limb segments addresses multilevel vascular disease.
Limitations and future research
However, our study has some limitations. The small sample size and retrospective nature constrain conclusions about efficacy and safety. The lack of a control group precludes comparison to standard wound care, endovascular treatment, or bone transport. The short follow-up provides no data on long-term outcomes like ulcer recurrence. While promising, these preliminary findings warrant verification in large multicenter randomized controlled trials with extended follow-up. Future research should also evaluate quantitative metrics of wound healing, impacts on neuropathic pain and sensory recovery, patient-reported outcomes, and cost-effectiveness. Elucidating the biological mechanisms and optimal protocols will facilitate continued innovation and clinical translation.
Nonetheless, our results indicate that distraction osteogenesis applied through periosteal traction offers a minimally invasive alternative to stimulate neovascularization and potentially revascularize critically ischemic limbs. This technique shows immense promise in the armamentarium for chronic limb ischemia and merits ongoing investigation. Larger data sets, mechanistic studies, and protocol refinements will help unlock its full potential to reduce amputation rates and improve patient quality of life.
The clinical success of this technique likely depends on several key factors. Careful patient selection is crucial, and severe multilevel occlusive disease precludes sufficient runoff vessels. Meticulous maintenance of sterile technique prevents local infection which can disrupt the regenerative process. Judicious titration of distraction forces avoids ischemia or periosteal detachment. Early ambulation and offloading help durability of wound healing. Ongoing medical management including glycemic control, smoking cessation, and medical therapy is still warranted.
In addition to the future research directions outlined earlier, other areas warranting further investigation include: • Determining the effects of patient factors like age, sex, comorbidities, inflammation, microbiome on treatment response. • Defining optimal eligibility criteria based on location, extent of arterial occlusion and runoff vessel status. • Evaluating adjunctive therapies like hyperbaric oxygen, growth factor or stem cell injections to enhance efficacy. • Assessing the synergistic potential of combining periosteal distraction with other novel revascularization techniques. • Exploring alternate distraction modalities like ultrasound, electromagnetic or piezoelectric actuation. • Developing fully implantable distraction devices to enable prolonged therapy. • Identifying systemic biomarkers of treatment response and limb salvage. • Correlating clinical, angiographic and molecular outcomes to optimize personalized treatment. • Elucidating impacts on wound strength, scarring, sensory and motor nerve regeneration after healing. • Analyzing long-term ulcer recurrence, limb salvage rates and functional outcomes. • Evaluating patient-reported quality of life, pain, activities of daily living after treatment. • Performing pharmacoeconomic analysis to compare cost-effectiveness against current standards. • Establishing multidisciplinary collaborations to facilitate clinical translation and adoption.
Conclusion
TPD is a promising minimally invasive technique for stimulating neovascularization and revascularizing critically ischemic limbs. This approach offers significant potential for limb salvage in patients unsuitable for conventional revascularization methods. Continued research and protocol refinement are essential to fully unlock its potential and integrate it into the clinical management of chronic limb ischemia.
Footnotes
Author contributions
Conceptualization: Meng Gan, Yun Wu, Naxin Zeng.
Data curation: Meng Gan, Yi You, Wei Xu, Xinyu Peng, Jinjun Xu, Wengao Wu, Yinkui Tang, Qiong Chen, Yun Wu, Naxin Zeng.
Formal analysis: Yi You, Yinkui Tang, Yinkui Tang,.
Investigation: Wei Xu, Jinjun Xu, Xinyu Peng, Qiong Chen,.
Software: Xinyu Peng, Wengao Wu.
Supervision: Meng Gan, Yun Wu, Naxin Zeng.
Writing – original draft: Xiqin Xia, Meng Gan.
Writing – review & editing: Meng Gan, Xiqin Xia.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The paper is supported by the Planned Science and Technology Project of Hunan Province of China (Grant nos. 2020SK53801).
