Abstract
Background
Chordoma is a bone tumor that tends to occur in middle-aged and elderly people. It grows relatively slowly but is aggressive. The prognosis of middle-aged and elderly patients with chordoma is quite different from that of young patients with chordoma.
Objectives
The purpose of the research was to construct a nomogram to predict the Individualized prognosis of middle-aged and elderly (age greater than or equal to 40 years) patients with chordoma.
Methods
In this study, we screened 658 patients diagnosed with chordoma from 1983 to 2015 in the Surveillance, Epidemiology, and End Results (SEER) database. We determined the independently prognostic factors that affect the survival of patients by univariate and multivariate Cox proportional hazards model. Based on the independent prognostic factors, we constructed a nomogram to predict the overall survival (OS) rates of middle-aged and elderly patients with chordoma at 3 and 5 years. The validation of this nomogram was completed by evaluating the calibration curve and the C-index.
Results
We screened a total of 658 patients and divided them into two cohort. Training cohort had 462 samples and validation cohort had 196 samples. The multivariate Cox proportional hazards model of the training group showed an association of age, tumor size, histology, primary site, surgery, and extent of disease with OS rates. Based on these results, we constructed the corresponding nomogram. The calibration curve and C-index showed the satisfactory ability of the nomogram in terms of predictive ability.
Conclusion
Nomogram can be an effective prognostic tool to assess the prognosis of middle-aged and elderly patients with chordoma and can help clinicians in medical decision-making and enable patients to receive more accurate and reasonable treatment.
Introduction
Chordoma is a rare malignant bone tumor with a prevalence rate of less than one in a million. Its proportion in malignant bone tumors is about 1%–4%. 1 Chordoma originates from the remaining embryonic notochord. 2 Under normal circumstances, notochord remnants will disappear completely in the first few years of life, 3 but there will be residual notochord tissue in some people. Most of the remaining notochord tissue is harmless, and only a small part will be transformed into chordoma. 4 The mechanism of transformation is still unclear, but some genetic changes have been reported to be related to the occurrence of chordoma. 5 Chordoma is a moderate sarcoma that grows relatively slowly but is aggressive. 6 It mostly occurs in middle-aged and elderly patients around 60 years old, and the incidence is very low in patients under 40 years old. 7 The sacrum is the most common site of chordoma (50%), followed by the base of the skull (30%) and the spine (20%). 8 Chordomas in other locations have also been reported, but they are very rare. Generally, chordoma is classified into three histologic types: conventional, chondroid, and dedifferentiated. Most chordomas are of the conventional type, and the undifferentiated type is the least. Patients with undifferentiated chordoma account for about 2%–8% of patients with chordoma, with the worst prognosis. 9 Patients usually have no obvious symptoms in the early stage, until the late-stage tumor invades adjacent tissues and causes bone destruction or neurological dysfunction. 10 Chordoma is usually close to blood vessels and nerve tissues, so it has a strong tendency to recur locally. Epidemiological data found that the local recurrence rate was as high as 40%. 11
At present, the treatment of chordoma is mainly surgical treatment with or without radiotherapy.11–13 However, chemotherapy has limited efficacy for most chordomas. 14 Complete surgical resection based on negative surgical margins is recommended by chordoma treatment guidelines. 15 However, due to the aggressive growth pattern of chordoma and its primary site is often adjacent to blood vessels and nerves, it is difficult even for experienced surgeons to completely remove the tumor. 16 Determining the prognostic factors of patients with chordoma and evaluating the prognosis of patients is an important step in the treatment process. Studies have confirmed that factors such as age, surgical margin, tumor size, metastasis and other factors affect the survival of patients with chordoma.7,16–18 However, the role of a single prognostic factor in individualized prognostic evaluation is limited. Nomogram is an effective statistical tool that can integrate multiple prognostic factors and enhance the accuracy of single prognostic factor prediction, and finally visually evaluate the prognosis of patients through graphs. This method has been applied to a variety of cancers and achieved good results.19–21 Because most of the patients with chordoma are middle-aged and elderly, and the prognosis of middle-aged and young patients is quite different.7,22 Therefore, it is of great significance to accurately assess the prognosis of middle-aged and elderly patients with chordoma. So far, we have not found any research that provides nomogram to predict the 3-years and 5-years OS of middle-aged and elderly patients with chordoma.
The SEER database is a cancer database based on the U.S. population, including nearly 30% of the U.S. population. Therefore, this study aims to develop an effective nomogram to accurately assess the 3-years, 5-years overall survival rate of middle-aged and elderly patients with chordoma. We collected the clinical information of middle-aged and elderly patients with chordoma from 1983 to 2015 from the SEER database and performed statistical analysis.
Methods
Data selection
After the approval of our registered account, we could use the patient data in the SEER database for research. This study complied with all guidelines of the “Declaration of Helsinki” on ethical considerations in human trials. The data published in the SEER database does not require any patient informed consent. We collected data on patient age, gender, race, primary site, histology, diagnosis year, tumor size, surgery, the extent of disease (EOD), and other clinical characteristics.
The inclusion criteria of this study were: (1) Diagnosis age was greater than or equal to 40 years old. (2) Diagnosed between 1983 to 2015. (3) Chordoma as primary malignancy. (4) Complete follow-up. (5) The primary site limited to skull, vertebral column and pelvis.
The exclusion criteria were: (1) Unknown tumor size. (2) Use of unknown surgery. (3) Unknown tumor EOD.
We used X-tile software to determine the best cut-off value for diagnostic age and tumor size, and its accuracy had been proven in previous studies.
23
In this study, the best cut-off values for diagnosis age were 63 years and 80 years, and the best cut-off values for tumor size were 35 mm and 119 mm (Figure 1). Therefore, the age was divided into three groups: 40–62, 63–80, >80 years old. Tumor sizes were categorized into three groups: <34 mm, 34–119 mm, and >119 mm. The diagnosis durations of the screened patients were divided into three groups: 1983–1993, 1994–2004 and 2005–2015. Surgery is classified as yes or no. AJCC bone tumours staging system has divided the extent of disease (EOD) into three types:
24
localized (tumor confined to the periosteum), regional (tumor extended beyond the periosteum without distant metastasis), and distant (having the metastatic disease at presentation). In this study, there were fewer patients with distant metastases. So these patients were included in the regional group. Identification of optimal cutoff values of age of diagnosis (A and B) and tumor size (C and D) via X-tile analysis.
Statistical analysis
According to the above inclusion and exclusion criteria (Figure 2), we collected 658 samples and randomly divided them into two cohort in the ratio of 7:3 to construct and verify the nomogram. Training cohort had 462 samples and validation cohort had 196 samples. The chi-square test was employed to compare the clinicopathological characteristics of patients between the training group and the verification group. In the training cohort, univariate and multivariate Cox proportional hazard models were used to evaluate the prognostic factors included in the study. We also examined the hazard ratio of each variable and used the independent prognostic factors selected in the multivariate Cox analysis to construct a nomogram showing 3- and 5-years OS. OS indicates the time from the date of diagnosis to death due to any cause. We evaluated the predictive ability of the nomogram through the calibration curve and the consistency index (C-index). The C-index ranges from 0.5 to 1.0 (0.5 means complete disagreement; 1.0 means complete agreement, c-index >0.7 means that the model has good accuracy). We established a calibration curve to determine the consistency of the predicted survival period with the actual survival period. The above statistical analyses were performed by using statistical software SPSS, version 25.0 (IBM Inc, USA) and R software (version 4.0.2). Flow chart.
Results
Clinical data of the studied cases
Clinical and pathological features of the study population.

Prognostic factors affecting OS
Univariate and multivariate analyses of overall survival in the Training cohort.

Construction and validation of OS nomogram
Based on the results of the Cox analysis, we included six important independent factors: age, tumor size, primary site, surgery, histology, tumor size, and EOD into the prognostic nomogram in order to estimate 3- and 5-years OS in patients with chordoma (Figure 3). The nomogram gives score of every prognostic variable on the point scale (Table 3).The following steps are involved in development of the nomogram: 1. Based on individual chordoma patient’s prognostic factors, scores related to each prognostic factor can be obtained. 2. Add all the points to get the “total point”. 3. Draw a vertical line from the “total point” column to the survival probability column to get the corresponding survival rate. Graphs showing nomograms predicting 3-and 5-years OS of middle-aged and elderly patients with chordoma. Detailed points of each predictor in the nomograms.
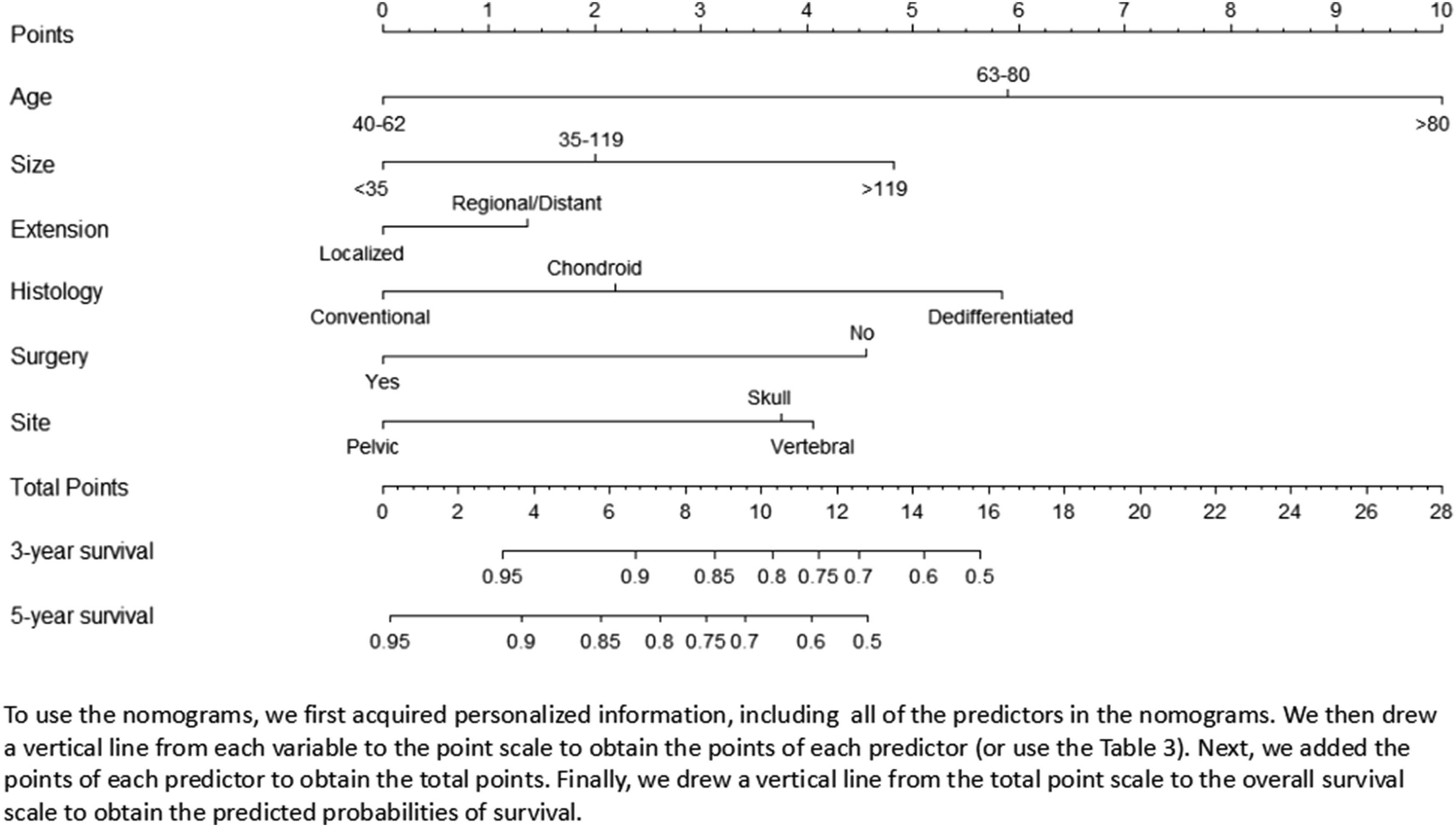

As an example, a patient diagnosed with Conventional chordoma was aged 58 and the site of the disease was in the spine. Imaging revealed that the size of the tumor was 82 mm and the EOD did not break through the periosteum, and he underwent surgery. According to our nomogram, the patient’s OS scores were 6.1 points. The 3-years OS and the 5-years OS rates were 0.91 and 0.83, respectively.
The nomogram was verified both internally and externally. For OS, the C-indexes of the training group is 0.759 (95% CI: 0.723–0.785) and the C-indexes of the validation group is 0.746 (95%CI: 0.694-0.798). The internal and external calibration curves of 3- and 5-years OS showed strong consistency of the predicted results of the nomogram with the actual results (Figure 4). Internal calibration plots of 3-year (a) and 5-year (b) overall survival nomogram calibration curves; External calibration plots of 3-year (c) and 5-year (d) overall survival nomogram calibration curves.
Discussion
The survival of patients with tumours is affected by many factors. Researchers have done a lot of research on the prognostic factors of chordoma, including tumor size, surgical margin status, recurrence and metastasis, etc.7,16–18,25,26 However, it is obviously inaccurate to evaluate the prognosis of middle-aged and elderly patients with chordoma using a single variable. The current treatment of chordoma is still a huge problem for clinicians. Most patients with chordoma are middle-aged and elderly, 7 and the prognosis of middle-aged and elderly patients is quite different from that of younger patients. 22 Therefore, it is meaningful for clinicians to construct a simple and accurate nomogram for middle-aged and elderly patients with chordoma. A nomogram can intuitively and effectively evaluate prognosis based on a multivariate regression model. It can provide a graphical calculation scale method for estimating the probability of overall patient survival. Nomogram has been widely used in the survival prediction of individual patients in recent years.19–21 At present, prognostic model for middle-aged and elderly patients has not been constructed in the literature. We obtained the clinical data of 658 middle-aged and elderly patients with chordoma from the SEER database and constructed a nomogram to predict the 3-years and 5-years overall survival rates of middle-aged and elderly patients with chordoma.
In this study, based on the data after screening, through univariate and multivariate Cox analysis, we determined the independent prognostic factors for the overall survival of middle-aged and elderly patients with chordoma. These factors included age, primary site, histology, surgery, tumor size, and EOD. Patients older than 80 years had the worst prognosis. This may be related to the poor general condition of older patients, and another possibility is that the neurological symptoms caused by chordoma are similar to those caused by some benign spinal diseases, such as degenerative disc disease. This can prevent chordoma patients from getting a clear diagnosis and prompt treatment. We determined the best cut-off values of tumor size through X-tile software to be 35 mm and 119 mm respectively. In our study, tumor size was also an independent factor affecting the prognosis of middle-aged and elderly patients with chordoma. Patients with larger tumors had a poor prognosis. This may be because the smaller the tumor, the easier it is to completely remove it by surgery. But researchers had different views on the correlation between tumor size and OS in patients with chordoma. Bergh et al. pointed out that patients with larger tumor size in the sacrum and active spinal chordoma had the lowest survival rate. 25 The study by Meng et al. showed that tumor size was not an independent prognostic factor for spinal chordoma OS. 16 The extent of disease as an independent prognostic factor for middle-aged and elderly patients with chordoma had also been confirmed by us. In the results of COX multivariate analysis, the tumor breaking through the periosteum indicated that the patient had a poor prognosis. We speculated that this may be because the tumor that broke through the periosteum invaded important nerve and blood vessel tissues. This increased the risk of tumor recurrence and metastasis. Research by Sun et al. showed that patients with dedifferentiated chordoma often had a poor prognosis. 27 The reason was that dedifferentiated chordoma was the most aggressive among all histological types of chordoma. This was also confirmed in our study.
Whether radiotherapy is effective in the treatment of chordoma is controversial. A study showed that postoperative radiotherapy was effective in prolonging the overall survival of patients with chordoma. 28 Research by Boriani et al. proved that radiotherapy had no effect on the treatment of chordoma. 29 The radiotherapy information was not included in this study because the SEER database had a “No/Unknown” field in the radiotherapy information column, and the radiotherapy dose was not specified. Chemotherapy had a limited role in the treatment of chordoma. Commonly used drugs for chemotherapy are cisplatin, anthracyclines, alkylating drugs, etc., but none of these drugs have been proven effective. 30 Surgical treatment is still the most effective treatment at present. Multiple studies have shown that patients with chordoma who undergo surgery have a higher survival rate than patients with chordoma who have not undergone surgery.2,31,32 In our study, surgical treatment was also an independent prognostic factor affecting OS in middle-aged and elderly patients with chordoma. Surgical treatment can improve the 3-years and 5-years overall survival rate of middle-aged and elderly patients with chordoma. The rise of tumor targeted therapy technology provides another way for the treatment of chordoma. Some targeted molecules such as tyrosine kinase inhibitors (TKIs) are used in the treatment of patients with advanced chordoma. 33
In this study, we found out the independent prognostic factors of middle-aged and elderly patients with chordoma through statistical analysis of a large number of cases in the SEER database, and incorporated these factors into the construction of the nomogram. The C index and the calibration curve indicated that the model had a good predictive ability. Clinicians can use this nomogram to judge the prognosis of each patient and adjust the treatment plan in time to achieve individualized treatment.
It is also important to consider the potential limitations of our study. There may be some important independent prognostic factors such as the status of tumor margins, spinal segment, and whether there is nerve/vascular invasion that might not have been identified by researchers or recorded in the SEER database. 34 These factors may be related with the prognosis of middle-aged and elderly patients with chordoma, but were not included in this study. All our data came from the SEER database. Although we randomly divided patients into training and validation groups to verify the internal and external nomograms, we should do some prospective studies or at least verify the nomograms in another database.
Conclusion
Taken together, based on the large database, we analyzed the factors affecting middle-aged and elderly patients with chordoma. Also, we determined that tumor size, age, histology, primary site, surgery and EOD were the independent prognostic factors of OS in middle-aged and elderly patients with chordoma. We also developed and validated the nomogram to evaluate the 3- and 5-years OS of middle-aged and elderly patients with chordoma. Nomogram has high accuracy and applicability. Hopefully, our nomogram could be used as a convenient and effective tool. It can help doctors conduct personalized survival assessments and screen out patients with high mortality rates, to provide a reference for subsequent treatment options and follow-up strategies.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Changzhou International Science and Technology Cooperation Project (CZ20200037).
