Abstract
Purpose
This study explores the use of ultrasound-guided Hyaluronic Acid (HA) injections for Insertional Achilles Tendinopathy (IAT).
Methods
A cohort of 15 ankles diagnosed with IAT received three weekly ultrasound-guided HA injections. The Victorian Institute of Sport Assessment – Achilles (VISA-A) questionnaire scored the severity of symptoms and functional impairment before treatment, and at one and six months post-treatment.
Results
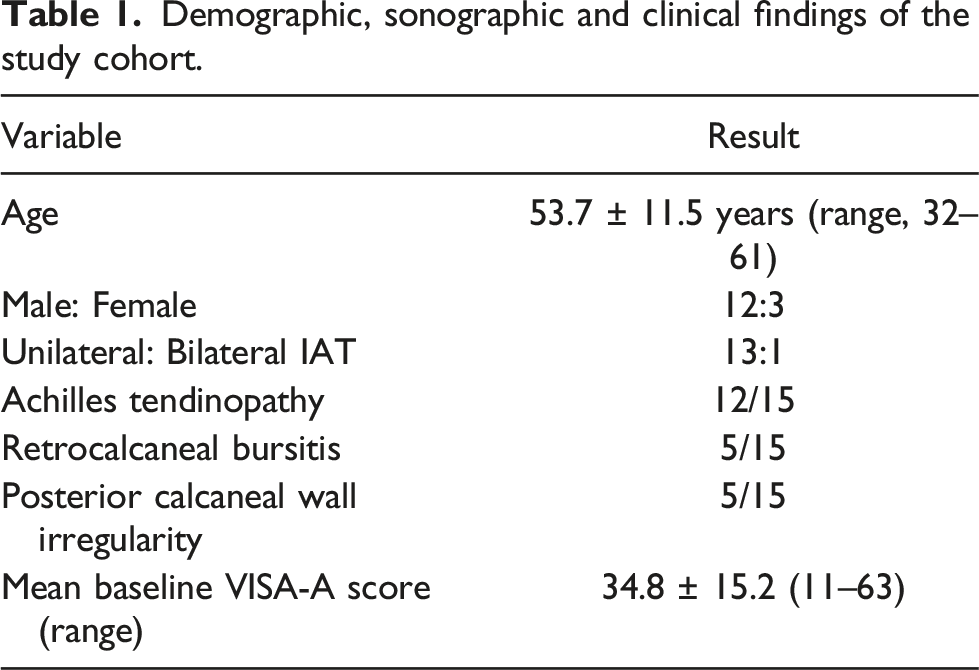
Significant improvement was observed in VISA-A scores post-treatment, rising from an average baseline of 34.8 ± 15.2 (11-63) to 53.6 ± 20.9 (15-77) after one month, and then to 50.7 ± 18.6 (20-75) after six months. No adverse reactions were noted, underscoring the safety of the intervention.
Conclusion
The pilot study presents HA injections as a potentially effective treatment for IAT, while interpretation of these findings must take into account the variability in results, indicating a range of patient responses. It encourages further research to confirm these findings and to explore HA’s full potential in managing IAT, despite the limitations of a small sample size and lack of control group.
Keywords
Introduction
Insertional Achilles Tendinopathy (IAT) manifests as a condition characterized by significant pain, affects a wide range of individuals, from the physically active to the sedentary. Our intervention focuses on pain relief as its primary outcome, recognizing that such pathology, characterized by chronic degenerative changes, 1 may not always be symptomatic. The diagnosis is mainly clinical and is based on posterior heel pain, localized to the insertion of the Achilles tendon, accompanied by local swelling and impaired function. 2 About 24% of the patients suffering from heel pain have clinical and histopathological signs of IAT. 3 The pathological process in IAT may also involve a retrocalcaneal bursitis. 4 Significant histopathological degenerative changes in IAT were found not only in the tendon, but also in the calcaneal fibrocartilage layers surrounding the distal part of the retrocalcaneal bursa. 5 These degenerative changes were found to resemble changes to articular cartilage that occur in osteoarthritis.5,6 Furthermore, the inflammatory changes, such as retrocalcaneal bursitis, were suggested to be secondary to the fibrocartilage degeneration. 6 Those findings imply that IAT may not be merely an activity-related pathology, but rather a more complex process involving all parts of the enthesis.7–9
The degenerative component in IAT has recently led researchers to suspect that arthritis-modifying agents, such as hyaluronic acid (HA), may be applicable in the treatment of IAT. 5 Several histological studies that investigated the effects of exogenous HA on enthesopathies have shown that HA can reduce inflammatory mediators, 10 inhibit cartilage degeneration, 11 and promote neovascularization and growth of new sensory nerves. 12 Due to the osteoarthritic-like changes in the fibrocartilage, mainly in the calcaneal posterior wall, we hypothesized that retrocalcaneal intra-bursal HA injections would have a beneficial effect on IAT patients. The primary goal of this study was to evaluate the effect of this treatment modality for IAT. The secondary goal was to assess whether IAT can be categorized according to four distinct sonographic findings and to correlate these findings with the treatment outcomes.
Material and methods
With approval from our institutional review board (MMC-0082-14), we conducted this prospective, uncontrolled single-center pilot study.
Patients
Fourteen patients that were diagnosed with IAT (15 ankles) were asked if they are willing to participate in the study. Following their preliminary approval, a detailed explanation about the study purposes, benefits and potential risks was given and an informed consent was sign by each participant. Each individual was treated by three consecutive weekly ultrasound-guided retrocalcaneal intra-bursal injections of high-molecular hyalin G-F 20 (Synvisc®; Genzyme Biosurgery, Genzyme Corporation, NJ, USA). Patients were followed in a specialized foot and ankle clinic for at least 6 months.
Inclusion criteria were a diagnosis of chronic IAT that was established by a foot and ankle orthopedic surgeon, and a rheumatology clinic assessment in which a systemic arthropathy was ruled out. The diagnosis was based on a history of pain at the calcaneal insertion of the Achilles tendon and a physical examination that revealed tenderness in the calcaneal insertion of the Achilles tendon. A mandatory radiographic work-up included a Magnetic resonance imaging (MRI) study of the relevant foot and ankle, by which the soft tissue was evaluated for both insertional and peritendinous edema. Exclusion criteria included previous surgical treatment such as retrocalcaneal osteotomy, retrocalcaneal injection of platelet-rich plasma (PRP) or steroids during the 6 months prior to the enrollment, previous two or more steroids injection, a history of Achilles tendon rupture or a previous calcaneal fracture.
During the first visit to the clinic, detailed demographic data, medical history and previous treatment modalities were documented. A physical examination was performed by one of the two abovementioned authors and the patients were asked to fill out the “VISA-A” questionnaire (see below). All the injections were performed by one experienced radiologist by the following technique.
Sonography
The patients were placed in a prone position with the knee slightly flexed. The ultrasound probe was placed transverse to the Achilles tendon and the retrocalcaneal bursa. In the transverse sonographic view (Figure 1), the hypoechoic area (retrocalcaneal bursa) was situated between the Achilles tendon and the calcaneus. Sonography images of the retrocalcaneal intrabursal injection of HA. Abbreviations: AT: Achilles tendon, BUR: Retrocalcaneal bursa, CAL: Calcaneus.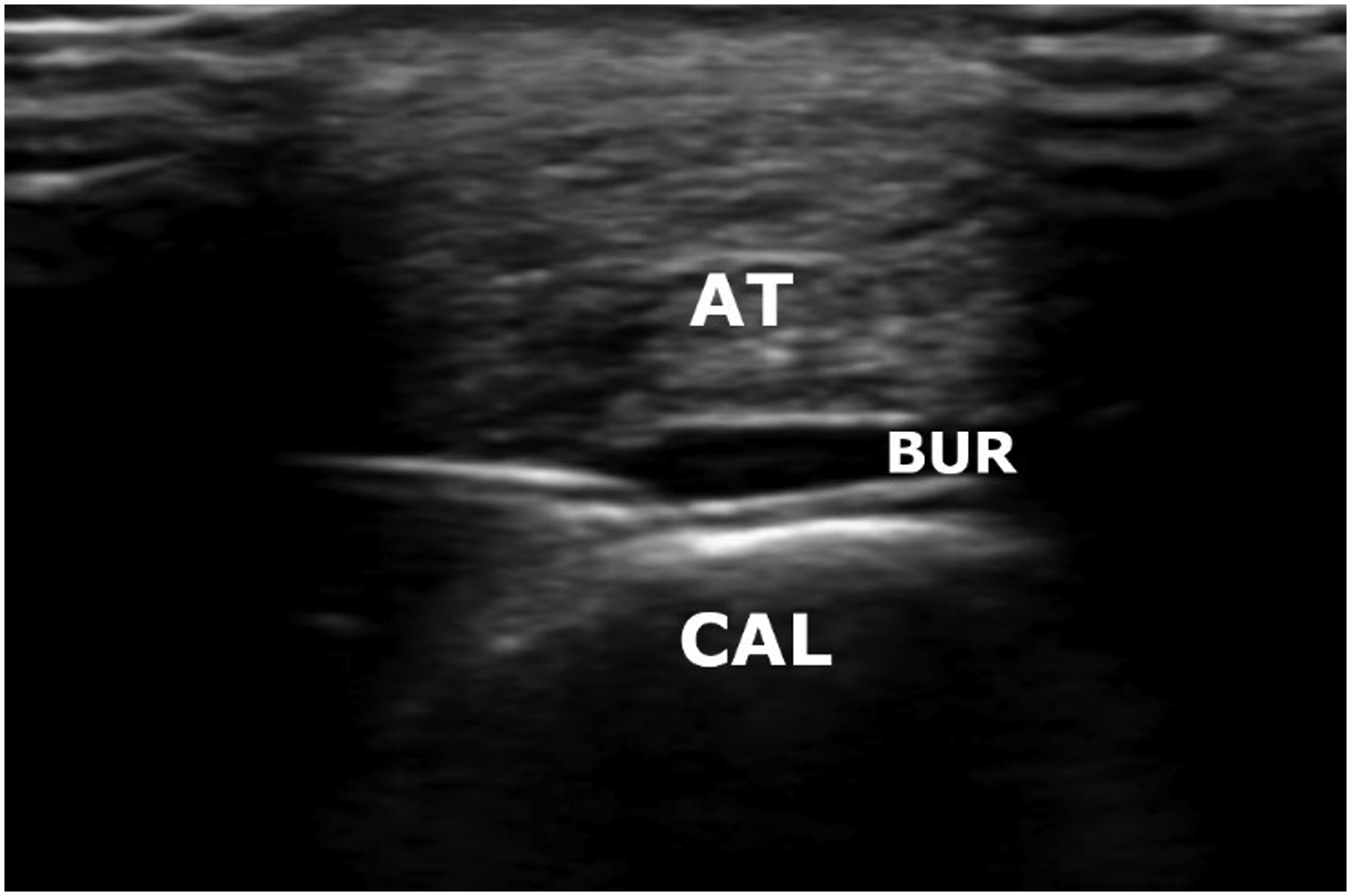
During the first test, four different areas were examined to characterize the pathology process: A swelling and irregularity in the retrocalcaneal bursa, representing an inflammatory process (Figure 2(a)), an irregularity in the posterior calcaneal wall that represents reactive degenerative changes (Figure 2(b)), an irregularity in the Achilles tendon that represents tendinitis (Figure 2(c)), and an enthesitis reflected as an irregularity in the Achilles tendon insertion (Figure 2(d)). The sonographic pathological patterns in insertional achilles tendinopathy (a) retrocalcaneal bursitis. (b) Posterior calcaneal wall irregularity. (c) Enthesitis. (d) achilles tendon irregularity. Abbreviations: At, achilles tendon; BUR, retrocalcaneal bursa; CAL, calcaneus; INSR, achilles tendon insertion.
Hyaluronic acid
A 21-gauge needle was inserted from the lateral side into a point just anterior to the Achilles tendon and a volume of 1-2 ml of Synvisc® was injected into the retrocalcaneal bursa.
The study product is a sterile, nonpyrogenic, elastoviscous fluid containing hylans, a derivative of hyaluronan, and the sodium salt of hyaluronic acid. Each 2 mL glass syringe of Synvisc® contains 16 mg of hylan G-F 20 (cross-linked hylan polymers; hylan A and B), 17 mg sodium chloride, 0.32 mg disodium hydrogen phosphate, and 0.08 mg sodium dihydrogen phosphate monohydrate.
The hyaluronan is extracted from chicken combs and the purified material has an average molecular weight of 6,000 kDa. The dose was adapted according to intra-bursal pressure and the procedure was terminated when the retrocalcaneal bursa appeared brimful on the sonographic view. The patients were asked to avoid vigorous exercise during the first two days following the injection. Thereafter, the patients were instructed to try to return to full activity.
Follow-up
For outcome evaluation, we used the Victorian Institute of Sport Assessment – Achilles (VISA-A) questionnaire. 13 This questionnaire was designed specifically to assess the severity of Achilles tendinopathy. The questionnaire is a self-administered form that evaluates pain, function and activity level 13 and has been commonly used to monitor outcomes after different treatment modalities for Achilles tendinopathy. It was previously found to be valid, reliable and clinically relevant.13,14 The scores range from 0 to 100 points, with higher scores indicative of better function. The minimum clinically important difference (MCID), which is defined as the smallest change on a scale that would be considered important to a patient, has been estimated to be between 6 and 12 points.15,16
A detailed history of daily and sport activities, physical examination results and physical adverse events were assessed 1 month and 6 months following the primary injection. The patients were asked to answer the VISA-A questionnaire before every injection and on each follow-up visit.
Statistical analysis
In this study, we employed the student’s t-test and ANOVA to rigorously evaluate changes in VISA-A scores at three time points: baseline, one month, and six months following a regimen of hyaluronic acid injections. The student’s t-test was utilized to assess the significance of changes in scores pre- and post-treatment for individual patients, allowing for an assessment of treatment impact at a personal level. Concurrently, ANOVA was used to analyze overall patterns and consistency in score changes across the study duration. We set our significance threshold at a p-value of less than 0.05, adhering to conventional standards for statistical significance. Reflecting on the multiple comparisons undertaken in our analysis, we considered the necessity of adjusting for the number of tests to mitigate the risk of type I errors, employing a Bonferroni correction to maintain the integrity of our statistical findings.
Results
Demographic, sonographic and clinical findings of the study cohort.
At follow-up, two patients did not experience any functional improvement. Six patients reported a significant pain relief, mainly during daily life activity with various levels of pain and functional improvement in low-intensity physical activity. One patient reported a significant pain relief and functional improvement during the first 5 months, followed by a significant functional decline. Analysis of the VISA-A questionnaires showed that the improvement in the mean functional score following three consecutive HA injections was statistically significant (p < .05). The mean VISA-A score, before entering the study, was 34.8 ± 15.2 (range, 11-63) points and these was considered as baseline. Following the first injection, it raised to 53.6 ± 20.9 (range, 15-77) points after 4 weeks, and 50.7 ± 18.6 (range, 20-75) points after 6 months (Figure 3). The change in function following HA injections. The improvement is noticed from 1 month on. The differences are significant (p < .05).
Only two patients from the study group had to undergo further surgical treatment due to insufficient improvement. No adverse drug reaction or infection following injection were noted in any of the patients. We did not find a correlation between specific sonographic features (Figure 2(a)-(d)) and the functional level at presentation or following treatment, as indicated by the VISA-A scores (p > .05).
Discussion
The key finding of the present study is the demonstration of safety associated with retrocalcaneal intra-bursal injections of HA, which were well-tolerated and led to clinical improvement in IAT-related symptoms without any meaningful complications.
The observed functional improvements post-HA injection could be attributed to the intricate histopathology associated with IAT, as indicated by the latest findings showing that the degenerative changes are not only in the tendon, but also in the calcaneal fibrocartilage layers surrounding the distal part of the retrocalcaneal bursa. 5
Until recently, the main etiology of IAT was considered overuse in patients with poor physical training habits, which include repetitive traction force. 2 However, it was lately suggested that the fibrocartilage degenerative lesions might be the result of a “stress-shielding” reaction to long periods of less tensile load in an anterior part of the enthesis.2,7,8 Moreover, the degenerative changes, particularly fissuring and chondrocyte clustering, were found to resemble osteoarthritic articular cartilage changes5,6 and the severity of these histological changes, as indicated by an osteoarthritic grading system, correlated with the degree of functional impairment. 5 Those findings imply that IAT may be a complex pathological process7,8 involving the periosteal and sesamoid fibrocartilages, the retrocalcaneal bursa and the fat pad, 9 which are all parts of the enthesis.
Overall, the majority of patients with IAT are successfully treated non-operatively, with activity modification, non-steroidal anti-inflammatory medications and physical therapy with eccentric training of gastrocnemius and soleus muscles. 2
Despite transient increases in pain during stretching, no serious adverse effects have been reported. Future research should focus on long-term effects and the potential benefits of incorporating additional muscle stretches. 17
A variety of local injection-based therapies that deliver a drug directly to the damaged tendon or to the retrocalcaneal bursa were described, including PRP and autologous blood and corticosteroid.
While PRP therapy has been explored for its potential in tendon healing for Achilles tendinopathy, its clinical effectiveness, especially regarding pain relief and functional improvement, has yielded inconsistent results. Some studies have found no significant advantage over placebo treatments. 18 This suggests a need to prioritize pain and function rather than structural tendon changes in the management of IAT.
Corticosteroid injections for tendinopathy may reduce symptoms and improve imaging findings, but their mechanism remains uncertain, and they pose significant risks, including tendon atrophy and rupture. Evidence suggests that the potential harms likely outweigh the benefits, with recent analyses indicating minimal advantage. 18 Combining these injections with rehabilitation strategies like eccentric exercises could offer improved outcomes, yet cautious application, preferably under imaging guidance, is advised due to the substantial risk of adverse effects. NSAIDs’ role in chronic tendinopathy treatment is controversial, with evidence suggesting they might impede tendon healing and offer minimal clinical improvement. Their short-term pain relief can mask symptoms, risking further tendon damage, alongside potential side effects like ulcers and renal issues in older adults. 18
A recently published review by the Cochrane library found that the currently available evidence is insufficient to support the routine use of these modalities. 19
When the non-operative management fails to improve symptoms, a surgical approach may be indicated. The surgical treatment includes a debridement of the ill portion of the tendon, an excision of the retrocalcaneal bursa and a resection of the calcaneal posterior wall or superior prominence.2,5
Hyaluronic acid is a polysaccharide with relatively high molecular weight (between 100 kDa and 10 MDa) that is found mostly in the extracellular matrix of numerous tissue types in the human body. 20 The primary role of HA in connective tissues is to increase the viscoelasticity by binding water and to lubricate the synovial joints.20,21 Currently, intra-articular injection of HA is mainly used as a visco-supplementation as part of the non-operative treatment of knee osteoarthritis. 21 Recently, it has been proposed to utilize the viscoelastic and anti-inflammatory properties of HA for the treatment of different tendinopathies and enthesopathies.20,21 Although in vitro and animal studies produced encouraging results, including better tendon architectural organization, reduced adhesions and enhanced tendon gliding, only a limited number of clinical studies were published on the use of HA for tendinopathies, mainly regarding rotator cuff tendinopathy and elbow epicondylitis Kumai et al. 21 published their experience with 61 patients having a verity of enthesopathies (14 with IAT) that were treated with a single HA injection, applied locally. The authors reported a mean reduction of 2.20 points in the patient’s VAS up to 1 week following one injection of HA. Although we have not used the VAS scale as a tool to evaluate pain reduction and treatment success, our results show the same pattern of pain relief following 3 HA injections. Injecting HA under sonar guidance to the heel has not been published routinely until short while ago. Ferreira and his colleagues 23 have recently reported their experience injecting only one dose of HA for heel pain in 25 patients. They have shown a significant clinical improvement in the majority of patients and concluded that a single dose HA injection is a safe procedure that improve function and reduces pain for six months. Our results are similar.
Comparing Kumai 21 and Ferreira 22 results to ours, one may ask if three consecutive injections of HA are superior to only one. Unfortunately, we cannot give a strict answer for that due to uneven research methods. In the Kumai study, no functional evaluation has been performed and pain scoring was measured only once, one week following injection. The Ferreira study although evaluate function in a similar time interval, has used a different scale.
To date, much has been published on different non-operative treatments for non-insertional Achilles tendinopathy, including PRP injection, 16 high-energy extracorporeal shock wave therapy, 24 topical glyceryl trinitrate patches, 25 cryotherapy, 25 laser therapy and eccentric exercise. 26 However, less evidence is available regarding the non-operative treatment of IAT. 28 Reports on treatment outcomes of injection therapies for IAT are even scarcer. Ohberg et al. prospectively analyzed 11 patients, who were treated with a sclerosing agent (polidocanol), which was injected against the local neovascularization found in the distal tendon portion. 29 They found that the mean Visual Analogue Scale (VAS) pain score decreased by 5.9 at 8 months follow-up, and eight out of 11 (73 %) patients found the treatment satisfactory. Ryan et al. retrospectively analyzed 22 patients treated with ultrasound-guided hyperosmolar dextrose (20 mg/mL) injections. 30 The mean VAS on daily activity decreased by 4.1 points (p = .001) at a follow-up of 28.6 months. Rompe et al. compared extracorporeal shockwave therapy (ESWT) with an eccentric training regime in a randomized controlled trial and found a 10-point increase in the VISA score in patients treated with eccentric training and a 27-point increase in patients treated with ESWT. 31 In light of these findings, the mean improvement produced by the modality offered in the current study, namely an increase of 15.9 points in the VISA-A score at 6 months follow-up with no adverse effects, seems to be a legitimate treatment option in IAT.
The wide range of improvement on the VISA-A scale (6 – 44 points) on the last follow-up, along with the diversity in imaging findings, raise the suspicion that IAT may in fact be a combination of several pathologies. Distinguishing between those entities, especially in terms of therapeutic implications, has barely been discussed in the literature. Dijk et al. reviewed the terminology for Achilles tendon injuries and proposed new definitions, based on the anatomic location, symptoms, clinical findings and histopathology. 32 The authors have made a clear distinction between mid-portion Achilles tendinopathy, Achilles para-tendinopathy, insertional Achilles tendinopathy, retrocalcaneal bursitis and superficial calcaneal bursitis. In their systematic review on the treatment for IAT, Wiegerinck et al. hypothesized that patients with IAT and bony disorder located at the calcaneal insertion of the Achilles tendon may have different treatment outcomes when compared with IAT patients without bony pathology. 27 Five of the patients in our series had radiographic findings compatible with retrocalcaneal bursitis. The inflammatory characteristics of retrocalcaneal bursitis, combined with the degenerative process found in fibrocartilage and tendon tissues, implies that IAT has a complex pathogenesis. Since the major advantage of HA injection is improving the viscoelasticity and affecting degenerative cartilage tissue, the impact of HA injection for IAT was expected to be greater in patients with an irregularity in the posterior calcaneal wall, but without retrocalcaneal bursitis. This assumption could not be proved in this cohort of patients.
Our study bears several limitations that are shown in the Ferreira study 23 as well. Clearly, as this is a pilot study, we did not include a control group, and thus, the interpretation of this research must take into account the variability in results, indicating a range of patient responses to the treatment. Moreover, the number of participants is insufficient for drawing a clear therapeutic recommendation or for high statistical power. Nevertheless, the study was based on a biological rationale, included an advanced imaging modalities-based diagnosis, and yield promising results. Aligned with findings from similar studies, our results lay the groundwork for further investigation into HA injections as a treatment for IAT across various severities. However before recommending widespread clinical application, a prospective controlled clinical trial is needed.
Conclusion
This pilot study, despite variability in outcomes, underscores the safety of ultrasound-guided intra-bursal retrocalcaneal injections of HA. While the results are promising, further research through prospective randomized controlled trials is essential to conclusively determine its efficacy.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
