Abstract
Background
Total knee arthroplasty (TKA) is a routine orthopedic procedure often associated with significant postoperative pain. Efficient pain management is paramount for patient recovery, with nonsteroidal anti-inflammatory drugs (NSAIDs) being a common choice. Nevertheless, the specific NSAID and its dosing regimen can have varying impacts on outcomes.
Methods
In this retrospective cohort study spanning from January 2016 to December 2020, we analyzed patients who underwent TKA. These patients were divided into two groups: one receiving preemptive low-dose ketorolac (15 mg) followed by 15 mg every 6 h for 48 h, and the other receiving parecoxib (40 mg) every 12 h for the same duration. We assessed pain scores, opioid consumption, and monitored adverse events.
Results
Our findings reveal that ketorolac yielded superior results compared to parecoxib. Specifically, patients receiving ketorolac reported significantly lower Visual Numeric Rating Scale (VNRS) scores at 8- and 20-h post-surgery. This trend was further confirmed by linear mixed models (p = .0084). Additionally, ketorolac was associated with reduced opioid consumption during the initial 24 h. Importantly, the rates of adverse events were comparable between the two groups.
Conclusion
The utilization of preemptive low-dose ketorolac demonstrates promising potential in bolstering pain control within the initial 24 h post-TKA, potentially reducing the need for opioids. However, further exploration is required to thoroughly assess its prolonged analgesic effects and safety across various surgical contexts. These investigations could provide invaluable insights for optimizing pain management protocols.
Keywords
Introduction
Total knee arthroplasty (TKA) is a well-established procedure that improves patients’ quality of life and functional status. However, this procedure is commonly associated with moderate to severe postoperative pain.1,2 Nonsteroidal anti-inflammatory drugs (NSAIDs) are frequently prescribed after TKA, as they have proven effective in reducing postoperative pain and opioid consumption during hospitalization.1–3 Despite their benefits, caution should be exercised, particularly in geriatric patients, due to potential side effects.4–6
Among the NSAIDs used during the perioperative period after TKA, ketorolac is commonly administered. This drug has shown efficacy in providing analgesia; however, evidence suggests a ceiling effect for its potency.7,8 In addition, ketorolac is associated with adverse gastrointestinal, renal, and cardiovascular events, some of which may be dose-dependent or duration-dependent. 5 Studies on low-dose ketorolac have demonstrated effective analgesia after spinal fusion surgery compared with a placebo.9,10 However, limited evidence exists regarding its use in TKA, with a study by Zhou et al. reporting no significant difference in pain relief after a single dose of ketorolac (15 mg or 30 mg) compared with placebo following TKA. 11 Importantly, no study compares low-dose ketorolac to other NSAIDs for patients undergoing TKA.
Given the lack of evidence regarding the efficacy of low-dose ketorolac for analgesia after TKA, we conducted a retrospective study on patients who underwent TKA and received a preemptive dose of 15 mg of ketorolac followed by 15 mg intravenously every 6 h for 48 h, comparing the outcomes with those of patients administered parecoxib. Our study aimed to explore whether low-dose ketorolac exhibits a significant difference in analgesic efficacy compared with parecoxib following TKA.
Methods
This was a retrospective cohort study. Data were extracted from the hospital’s electronic medical records of patients who underwent TKA (International Classification of Diseases, Ninth Revision, code: 81.54) between January 2016 and December 2020. The Ethics Committee and Institutional Review Board approved the study protocol. Informed consent was waived because of the study’s retrospective nature and the use of anonymous clinical data. The hospital provided permission to retrieve information from the database.
Patients who met the inclusion criteria for this study underwent primriorary TKA for end-stage primary knee osteoarthritis. Patients receiving general anesthesia who did not receive a peripheral nerve block, parecoxib, or ketorolac until 48 h postoperatively were excluded from the study.
This study aimed to compare the effectiveness of two specific dosing and medication regimens, parecoxib and ketorolac. In the parecoxib group, patients intravenously received 40 mg of parecoxib preoperatively, and every 12 h until 48 h postoperatively. In the ketorolac group, patients received 15 mg of ketorolac preoperatively, and every 6 h until 48 h postoperatively. Patients who received different dosing of parecoxib or ketorolac were excluded from the study.
Patients were administered either parecoxib or ketorolac as a preemptive medication 30 min preoperatively. All patients received spinal anesthesia and a preoperative peripheral nerve block during surgery. The decision to use a femoral nerve block or an adductor canal block was made based on the anesthesiologist’s judgment. A tourniquet was applied and maintained at an inflation pressure ranging from 250 to 280 mmHg throughout the operation. The total knee arthroplasty procedures utilized a cemented posterior-stabilized fixed-bearing design. Periarticular injections were not used at the time of this study. A single senior surgeon performed all surgical procedures.
Following the operation, patients were given either parecoxib or ketorolac for postoperative pain management until 48 h after the surgery. In both groups, patients were given intravenous morphine or fentanyl as needed every 2–3 h for severe pain. Both groups also had access to additional pain medications such as acetaminophen and gabapentin. As a preventive measure against deep vein thrombosis, patients were prescribed 162 mg of aspirin daily for 14 days. Furthermore, patients from both groups participated in a standardized rehabilitation program for total knee arthroplasty, which included quadriceps isometric exercise, ankle pumping, range-of-motion exercises, and early ambulation with the support of a device.
Age, sex, body mass index (BMI), American Society of Anesthesiologists (ASA) classification, Charlson comorbidity index, and type of peripheral nerve block were collected as baseline characteristics. Pain scores were collected with each vital sign assessment every 4 h until 48 h postoperatively and evaluated using a 10-point verbal numerical rating Score (VNRS). Cumulative opioid consumption at 24 and 48 h postoperatively was measured by conversion to morphine equivalent units. Postoperative adverse events within 1 year were evaluated, including cardiovascular events, pulmonary embolism (PE), deep vein thrombosis (DVT), and gastrointestinal bleeding.
Statistical analysis
We initiated our analysis with descriptive examinations of patient demographics. To ascertain the normality of continuous data, the Shapiro-Wilk test was applied. For normally distributed variables, categorical variables underwent Pearson’s chi-square tests, while continuous variables were subjected to Student’s t-tests. In cases of non-normal data distribution, we opted for Wilcoxon rank-sum tests.
To mitigate the marked intergroup variances in patient characteristics, we conducted propensity matching analysis, adhering to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) checklist. This entailed a 1:1 matching approach employing a nearest neighbor matching algorithm without replacement sampling. The chosen matching variable was restricted to peripheral nerve block, as it held potential influence over post-surgery pain scores. Variable selection was determined through logistic regression modeling.
To compare VNRS scores for specific time points, we utilized independent Student’s t-tests, and for an overarching evaluation of VNRS scores, linear mixed models were employed. Statistical significance was designated with a threshold of p < .05.
All statistical analyses were executed using R software version 4.0.5, developed by the R Foundation for Statistical Computing in Vienna, Austria.
Results
Propensity score matching
We retrieved and analyzed data from the electronic hospital medical records of 710 patients who underwent TKA. Among them, 200 patients met the inclusion criteria and were included in this study, whereas 510 were excluded (Figure 1). The exclusion criteria were: 198 patients who did not receive either ketorolac or parecoxib, 195 who received ketorolac or parecoxib but whose dosage and duration did not match the study protocol, 92 patients who did not receive the peripheral nerve block, and 25 who did not undergo spinal anesthesia. Study workflow.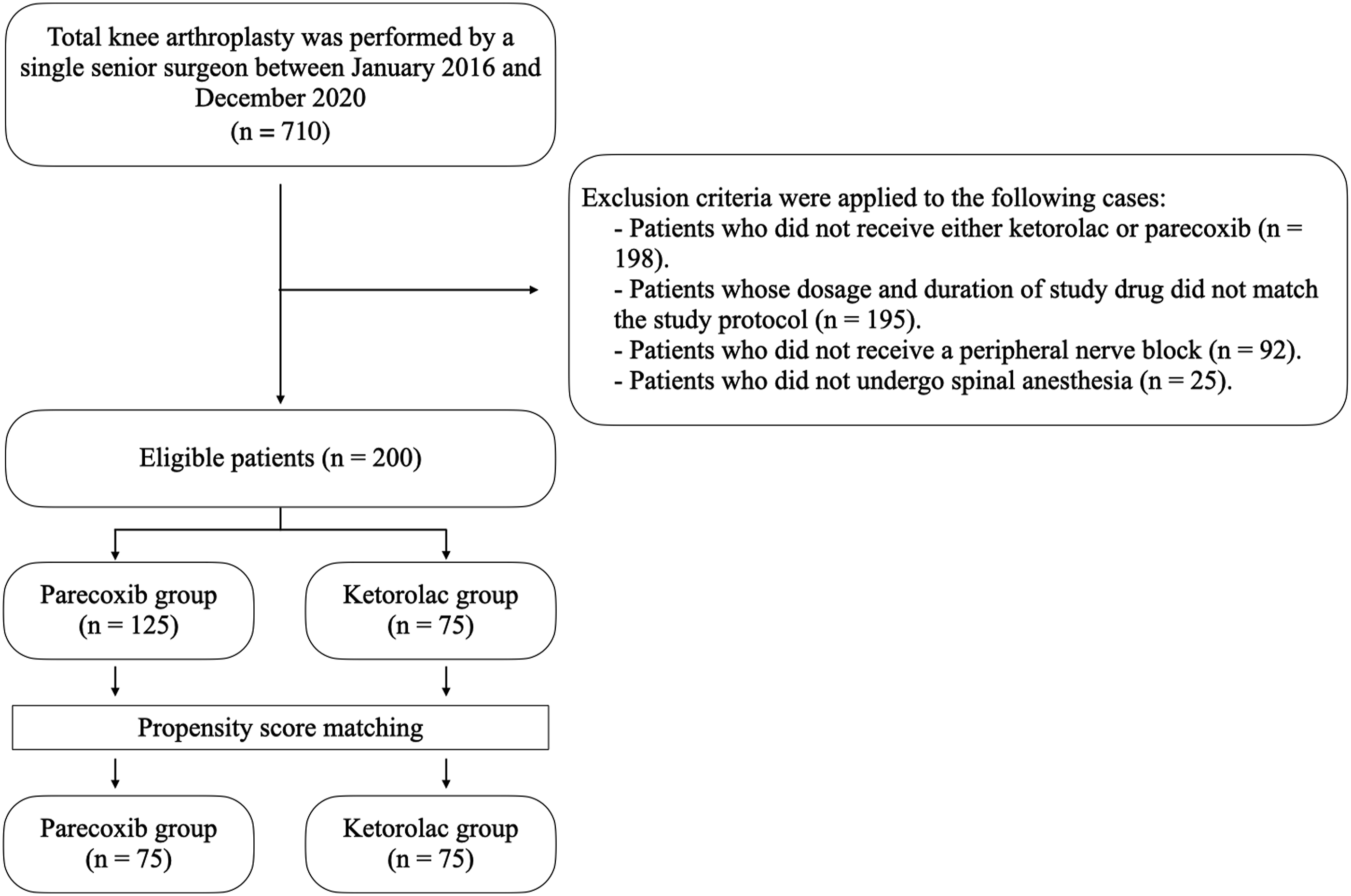
Comparison of patient demographics before and after propensity score matching for peripheral nerve block.

BMI: body mass index kg/m2; ASA: American society of anesthesiologists.
Significant p-values are written in bold letters.
Comparison between both NSAIDs
Our study demonstrates that the ketorolac group consistently reported lower Visual Numeric Rating Scale (VNRS) scores for postoperative pain in the 48-h period following surgery. Remarkably, significant reductions in postoperative pain were pinpointed at specific time intervals within the ketorolac group: 8 h (p-value = .026) and 20 h (p-value = .016) post-surgery (Figure 2). Comparison of postoperative visual numeric rating scale (VNRS) scores between the groups treated with parecoxib and ketorolac.
Moreover, our comprehensive analysis using linear mixed models reaffirmed the ketorolac group’s advantage, with significantly lower pain scores (p-value = .0084). These scores exhibited a noteworthy decline over time, particularly pronounced within the ketorolac group.
Comparison of opioid consumption (median [IQR]) in morphine equivalent units for each group.
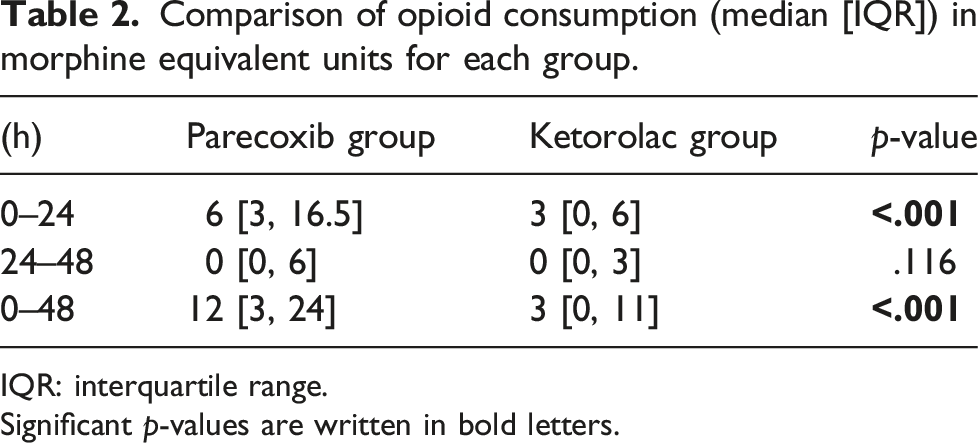
IQR: interquartile range.
Significant p-values are written in bold letters.
Furthermore, our study observed one case of pulmonary embolism (PE) in the ketorolac group and one deep vein thrombosis (DVT) in each group. Encouragingly, no other adverse events, such as cardiovascular events or gastrointestinal bleeding, were reported in either group.
Discussion
Our study’s main finding revealed that patients who underwent TKA and received a preemptive dose of 15 mg of ketorolac, followed by 15 mg intravenously every 6 h for 48 h, experienced significantly lower VNRS scores than those receiving parecoxib during the first 24 h postoperatively. Opioid consumption was also significantly lower in the ketorolac group during the initial 24 h. However, the differences did not reach a clinically significant level for VNRS and opioid consumption.12,13 Moreover, both groups showed no significant distinctions in adverse gastrointestinal and cardiovascular events.
Our results align with previous studies in patients undergoing spinal fusion surgery, where low-dose ketorolac demonstrated improved analgesia compared with placebo.9,10 In our research, low-dose ketorolac exhibited better analgesic effects than parecoxib during the first 24 h postoperatively. These findings are in accordance with the study conducted by Laoruengthana et al., 14 where periarticular injections (PAIs) with 30 mg of ketorolac, followed by intravenous injections of 30 mg of ketorolac every 12 h until 48 h postoperatively, resulted in significantly lower visual analog scales of pain compared with the parecoxib group at 6 h after TKA. In our study, the ketorolac group also had significantly lower VNRS pain scores at 24 h postoperatively. This raises the question of whether preemptive analgesia with ketorolac can provide longer-lasting pain relief compared with the PAIs technique.
Low-dose ketorolac has demonstrated effective pain control following TKA, hinting at a potential ceiling effect for its analgesic efficacy. This observation aligns with the findings of Forestell et al., who conducted a systematic review and meta-analysis comparing various ketorolac doses for acute pain management. Their analysis suggested that ketorolac doses between 10 and 20 mg are likely as effective in alleviating pain as higher doses exceeding 30 mg, although they may necessitate additional rescue analgesia. 15 However, our study yielded different results, showing lower opioid consumption in the ketorolac group. This divergence could be attributed to our administration of continuous intravenous ketorolac every 6 h, which may have contributed to more sustained pain control compared to the intermittent dosing regimens evaluated in prior research.
In our study, ketorolac was administered every 6 h, resulting in significantly lower morphine equivalent units used for rescue analgesia during the first 24 h after surgery compared with the parecoxib group. This finding suggests that the frequent dosing of ketorolac effectively controlled postoperative pain and reduced the need for additional analgesia.
Regarding adverse events, our study did not detect any significant differences between the ketorolac and parecoxib groups, which may be attributed to our study’s limited number of patients. However, evidence from large observational trials indicates that higher doses of ketorolac are associated with an increased risk of gastrointestinal bleeding.16,17 Considering the lack of additional benefits with higher doses of ketorolac, our findings support the use of low-dose ketorolac for postoperative pain management after TKA. This approach may help mitigate the risk of adverse events while maintaining effective pain control.
However, our study does have some limitations. Firstly, as a retrospective study lacking randomization, potential confounding factors could have influenced the results. However, we took measures to address this by utilizing propensity score matching to ensure comparability of patient characteristics between the two groups. Moreover, all TKA procedures were performed by a single experienced surgeon, and patients uniformly adhered to a standardized postoperative rehabilitation protocol and received identical medication regimens. This approach was adopted to minimize variability within the study population.
Secondly, there were slight disparities in mean age and ASA classification between the two groups. These differences arose due to our clinic’s practice of adjusting ketorolac dosages for patients aged over 65 years, driven by safety concerns. Additionally, we intentionally avoided using parecoxib in elderly patients and those with a history of cerebrovascular or cardiovascular events, which may account for the observed variations in these characteristics.18,19
Lastly, even with matching, our parecoxib group had more females and younger patients. Research indicates these factors may contribute to stronger post-op pain, possibly due to central sensitization.20,21 This adds complexity to our results, and it’s crucial to keep in mind during result interpretation.
In conclusion, preemptive analgesia with low-dose ketorolac demonstrated superior analgesic efficacy compared with parecoxib during the first 24 h after TKA. Our results contribute to the growing evidence suggesting a potential analgesic ceiling effect with ketorolac. This finding may also affect postoperative pain management in other orthopedic procedures. Further studies are warranted to explore the extended duration of analgesic effects and the safety profile of low-dose ketorolac in various surgical settings.
Footnotes
Acknowledgements
I extend my sincere appreciation to Ms Jirawan Jayuphan, the esteemed statistician whose expertise significantly contributed to the meticulous analysis and interpretation of the data in this study. Her invaluable efforts have been instrumental in the success of this research, and her dedication to precision and excellence is deeply acknowledged.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
