Abstract
Aim
To investigate the efficacy of fat grafting in primary tendon healing through immunohistochemical and biomechanical examinations.
Material and Method
The study material comprised a total of 10 male Sprague-Dawley rats, each approximately 10 weeks old. All 10 rats were operated on bilaterally. The right Achilles tendon in all the animals was defined as the study group. The tendon was cut and then repaired, and then fat graft was applied to the repair area. The left Achilles tendon of all the rats constituted the control group. The tendon was cut and repaired with no further application. After 4 weeks, the rats were euthanised and samples were taken from the tendons for immunohistochemical and biomechanical examinations.
Results
In the biomechanical evaluations, no statistically significant difference was determined between the groups in respect of peak load and stiffness values (p: .068, p: .089, respectively). In the histopathological evaluation, the tenocyte value of the study group was superior to that of the control group (p: .04). No statistically significant differences were determined between the groups in respect of the other histopathological parameters. In the immunohistochemical evaluations, the type I collagen and TGF values of the study group were found to be higher than those of the control group (p: .011, p: .012, respectively).
Conclusion
Compared to stem cell applications, the use of fat grafting is clinically easy to apply, has low costs, and has been shown to contribute to tendon healing at an immunohistochemical level with increased collagen and TGF beta values.
Introduction
Tendon healing is one of the most important problems in trauma and sports injury surgery. Tendons show resistance to high tension loading and this may be a reason for healing failure. Moreover, a healed tendon never regains full biomechanical and histopathological properties.1,2 As the Achilles tendon is the strongest tendon in the body and has to resist body weight, the healing of this tendon is more important. 3 Various types of augmentation have been developed and examined to obtain optimum function after tendon repair. Stem cell and growth factor treatments are among these, but the data obtained remain a matter of debate.4–6 In recent years, stem cell applications have become extremely common, and tendon healing is just one of the many areas where stem cell applications are used. 7 However, stem cell treatments have not yet become established in clinical practice because of accessibility, costs, and applicability. However, as stem cells have been shown to be found in adipose tissue, this has generated the idea that fat grafting could be used as treatment in clinical practice.8,9
Since Neuber et al. first used fat grafting in the treatment of large tissue defects after oncology operations, the use of fat grafting has become more widespread among surgeons. 10 Fat grafts are used in various conditions to accelerate healing. In a randomised controlled study by Del Papa et al., the application of autologous fat grafts in patients with systemic sclerosis was seen to accelerate the healing of digital ulcers. 11 In another study by Covarubias et al., the use of fat grafts in patients undergoing face-lift procedures was histopathologically shown to increase healing. 12 Patients with myocardial scar were examined radiologically by Gastelurrutia et al., and better healing results were reported to have been obtained with the application of fat tissue. 13 Bank et al. reported that in patients with Reynaud phenomenon, an increase in vascularity was seen with fat grafting and successful results were obtained. 14
To the best of our knowledge, there is no study in literature showing the immunohistochemical and biomechanical efficacy of fat grafting in primary tendon healing. Therefore, the aim of this study was to investigate the efficacy of fat grafting in primary tendon healing immunohistochemically and biomechanically, which is a cost-effective, readily available, and easy-to-apply method.
Material and method
The study was conducted in the Experimental Animals Research Laboratory with the approval of the Local Ethics Committee. The study material consisted of 10 male Sprague-Dawley rats, each approximately 10 weeks old and weighing approximately 300 gr. The rats were kept in separate cages at a controlled temperature under a 12-h light-dark cycle with ad libitum access to food and water.
All 10 rats were operated on bilaterally. The right Achilles tendon was identified in all the animals as the study group. The tendon was cut and then repaired, 6 with fat grafts placed as a strip surrounding the repair area, so that the repair area had not contact with the soft tissue. The left Achilles tendons of all the rats constituted the control group. The tendon was cut and repaired with no further application.
Preparation of the rats and the surgical procedure
The rats were anaesthetised with 40 mg/kg ketamine (Ketalar®; Pfizer, NYC, NY, USA) and 20 mg/kg xylazine (Rompun®; Bayer, Germany). Both left and right anterior groin and posterior ankle regions were shaved in each rat and antisepsis was provided with 10% povidon-iodine solution (Batticon, Adeka Ilaç, Türkiye). Antibiotic prophylaxis of 10 mg/kg cefazoline sodium (Sefazol®; Mustafa Nevzat İlaç, Türkiye) was then administered intraperitoneally.
Following this preparation, an incision of approximately 1 cm was made to the left and right groin region of each. A strip shaped fat graft weighing approximately 1 g was obtained, and it was of a width that would cover about 0.5 cm proximal and 0.5 cm distal to the repair site. The strip of fat graft was approximately 2 cm in length, designed to encircle the tendon 360°. The bleeding was controlled then the incisions were closed with non-absorbable sutures. The bilateral Achilles tendons were exposed by creating an incision of approximately 2 cm in length in the posterior region of both ankles. The tendons were then completely cut perpendicular to their long axis using a no. 15 scalpel, approximately 1 cm proximal to their insertion point. Then the tendons were repaired with prolene sutures using the modified Kessler method. In the study group involving the right-side tendons, fat grafts were meticulously applied as a strip encircling the repair area. These grafts were strategically positioned to cover approximately 0.5 cm proximal and 0.5 cm distal to the repair site, ensuring a comprehensive 360-degree coverage around the tendon. No further application was made to the control group of the left-side tendons. After bleeding was controlled, the incisions were closed with non-absorbable sutures (Figure 1). Surgical procedure, (a) harvesting of the fat graft from the inguinal region, (b) exposure then sharp cutting of the achilles tendon, (c) repair of the achilles tendon with the modified Kessler method, (d) placement of the fat graft in strips in the repair region in the study group.
Both legs of the rats were immobilised for 2 weeks with plaster of Paris cast as far as above the knee joint. The casts were removed at the end of second week and at the end of the 4th week all the rats were euthanised. For the histopathological and biomechanical examinations, the tendons were removed proximally from the gastrocnemius attachment point of the distal femur and distally to include the calcaneus.
Biomechanical evaluation
The Achilles tendons obtained from the rats were tested biomechanically with the MTS Acumen Electrodynamic Test System in the Biomechanical Laboratory of the manufacturer (Response Ortho Technology Production Inc.). The tendons were prepared for the test with one end in the calcaneus and one end in the femur. A system was created using sandpaper to provide stabilisation (Figure 3). The tensile test was performed with the device set at 0.1 mm/sec speed and 0.2 N initial load to be applied. The maximum load (N) and stiffness (N/mm) values at the breaking point of the tendons were recorded. Stiffness values of the tendons were calculated by dividing the maximum load values by the amount of elongation at the breaking point of the tendons. (Figure 2).
15
Biomechanical examination with a system formed using sandpaper.
Histopathological evaluation
All grafts were used for biomechanical testing followed by histological examination.
The harvested specimens were immediately fixed in 10% neutral buffered formalin, dehydrated through an alcohol gradient, cleaned, and then embedded in paraffin blocks. 3 μm Histological consecutive sections were cut longitudinally with the tendon ends facing each other. 16 They were prepared using a microtome and subsequently subjected to hematoxylin-eosin (HE) and Masson-trichrome (MT) staining using kit protocol. Samples were then examined under light microscopy and photographed. Evaluations were made separately by two histologists who were blind to samples code. Histological scoring was applied separately for tenocytes, ground substance, collagen and vascularity maturation and density. 15 Each parameter was graded from 0 to 5; 0 = the most pathological findings, 1 = a lot, 2 = moderate, 3 = few, 4 = very few, and 5 = no pathological finding. Preparates scored with different points were re-evaluated by the two histologists to reach consensus. 17
Immunohistochemical evaluation
The sections taken longitudinally from the paraffin blocks were stained with TGF-beta-1 and type I collagen antibody with primary antibodies TGF-beta-1 (Sigma, St. Louis, Mo, USA) and anti-Type-I collagen (Santa-Cruz Biotechnology, Calif, USA) were used. After dewaxing in xylene, the sections were dehydrated with ethanol. They were then incubated with 0.5% trypsin at 37°C for 15 min and endogenous peroxidase activity was inhibited using hydrogen peroxide (Merck). Blocking serum was applied for 1 h, followed by incubation with primary antibodies at 4°C overnight. The sections were then treated with the anti-mouse biotin-streptavidin hydrogen peroxidase secondary antibody (85–9043 Zymed Histostain kit). Immunoreactivity was made visible using diaminobenzidine (DAB, 00–2014, Invitrogen), and counterstaining was performed using Mayer’s hematoxylin (800-729-8350, ScyTek). Quantitative results of immunohistochemical staining were performed using free Fiji (Imageversion 2.1.0, National Institutes of Health, Bethesda, MD, USA). It was done as described. After the color deconvulsion is applied to the microphotographs belonging to the groups, DAB the average gray value was calculated from the image. Optical Density; Opticaldensity was calculated using the formula log (maximum gray density/average gray density). Data are presented as optical density ± standard deviation. 18
Statistical analysis
Data obtained in the study were analysed statistically using JAMOVI 2.3.17 open-source software. Conformity of the data to normal distribution was assessed with the Shapiro-Wilk test. Quantitative data were evaluated with the student’s t-test if distribution was normal and with the Mann Whitney U-test if distribution was not normal. Results are presented as mean +/− standard deviations. A value of p < .05 was accepted as statistically significant.
Results
All surgical procedures were successfully conducted. No rats experienced any loss of life throughout the course of the study. There were no rats that exhibited significant weight loss or alterations in behaviour. No instances of notable complications were encountered.
Biomechanical evaluation results
In the comparisons of peak load values between the groups, the mean peak load was determined to be 52.5 ± 32.9 N in the control group and 83.6 ± 38.4 N in the study group, with no statistically significant difference between the groups (p: .068). Stiffness values were 17.9 ± 12.6 N/mm in the control group and 23.9 ± 10.3 N/mm in the study group, with no statistically significant difference determined between the groups (p: .089) (Figure 3). The box plots graph presenting comparison of biomechanical parameters (peak load and stiffness) between the control and the study group.
Histopathological evaluation results
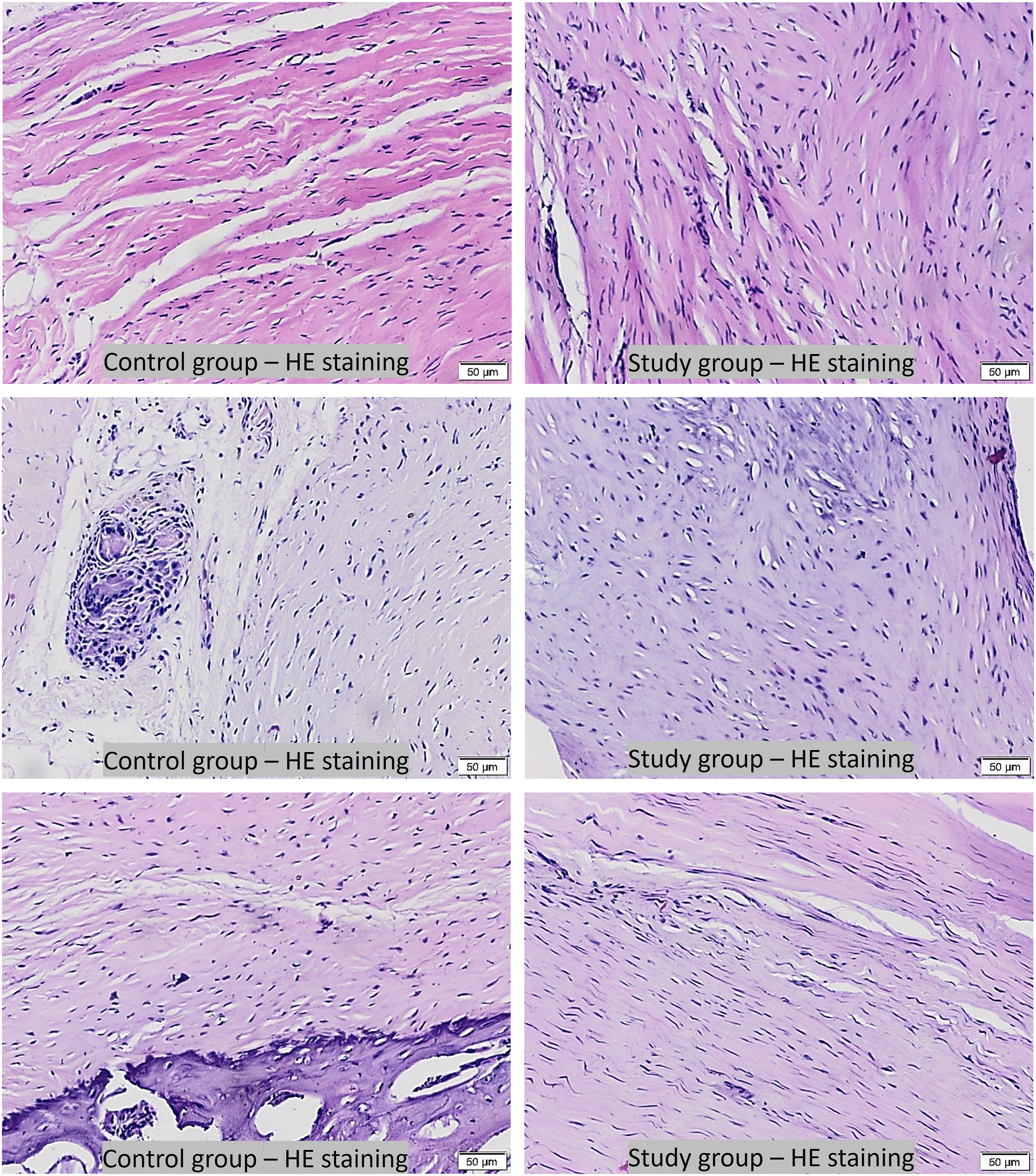
Histological examination of tendon sections stained with HE and MT revealed notable enhancements in the study group samples comparing to control group. These improvements were evident in terms of blood vessel density, fibroblast presence, and the organization of connective tissue. The sections exhibited a distinctive pattern of wavy, parallel mature tendon-like cells. Microscopic analysis further indicated heightened densities of tenocytes and collagen fibers, reflecting a more advanced level of maturity in the study group samples.
Tenocyte values were determined to be mean 1.82 ± 0.3 in the control group and statistically significantly higher at 2.2 ± 0.4 in the study group (p: .04). The ground substance values were determined to be 2.05 ± 0.4 in the control group and 2.46 ± 0.5 in the study group, with no statistically significant difference between the groups (p: .057). The collagen values were determined to be 1.81 ± 0.4 in the control group and 2.22 ± 0.5 in the study group, with no statistically significant difference between the groups (p: .076). The vascularity values were determined to be 2.32 ± 0.5 in the control group and 2.57 ± 0.4 in the study group, with no statistically significant difference between the groups (p: .054) (Figures 4–6). The box plots graph presenting comparison of histopathological parameters (tenocytes, ground substance, collagen and vascularity) between the control and the study group. Light microscope images: with hematoxylin-eosin staining. More mature tenocyte and organized ground substance with better vascularity were observed in different region from representative study group samples compared to those of in control group samples. Scale bar: 50 µm. Light microscope images: with masson-trichrome staining in the control group and the study group. More mature, organized and better collagen distribution were observed in different region from representative study group samples compared to those of in control samples. Scale bar: 50 µm.

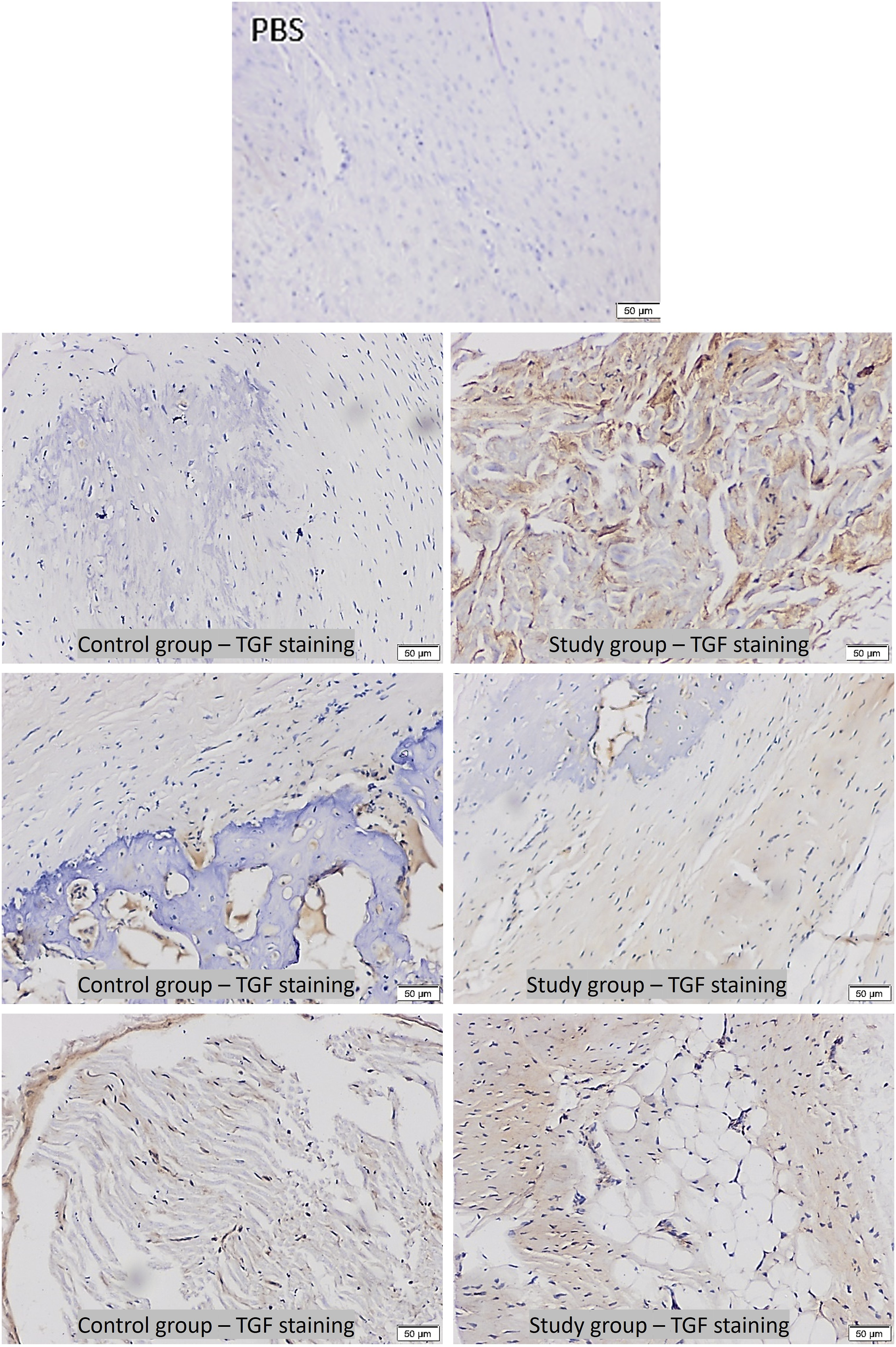
Immunohistochemical evaluation results
Increase in expression of TGF-β1 and collagen I were found in the study group samples comparing to control group. The heightened expression of TGF-β1 and collagen I, played a pivotal role in reshaping the inflammatory process, bolstering the extracellular matrix, and expediting the healing process. This orchestrated interplay culminated in a more rapid and efficient healing response.
The type I collagen values were determined to be mean 197 ± 32.8 in the control group, and statistically significantly higher at 238 ± 27.5 in the study group (p: .011). The TGF values were determined to be mean 84.8 ± 11.4 in the control group, and statistically significantly higher at 101 ± 15.1 in the study group (p: .012) (Figures 7–9) (Table 1). The box plots graph presenting comparison of immunohistochemical parameters (collagen one and TGF) between the control and the study group. Light microscope images with immunohistochemical for type I collagen staining. More number and intense labelling with primary antibody were observed in the study group compared to those of control group from different representative region. Primary antibody replaced by phosphate buffered saline (PBS) for isotype-staining to show specific labelling in control and study samples. Scale bar: 50 µm. Light microscope images with immunohistochemical for TGFbeta-1 staining. More number and intense labeling with primary antibody were observed in the study group compared to those of control group from different representative region. Primary antibody replaced by phosphate buffered saline (PBS) for isotype-staining to show specific labelling in control and study samples. Scale bar: 50 µm. Comparisons between the groups of all the parameters evaluated.


Discussion
There are many studies in literature that have been conducted to increase tendon healing and improve the quality of healed tendons. Although many augmentation methods, including stem cell applications, have been tried, the search is still ongoing for the ideal augmentation which will both strengthen clinical results and be cost-effective. As fat grafts have been shown to contain stem cells, the use of fat grafting rather than direct stem cell applications has been trialled in various areas and has been found to be successful.12–14 However, there is no study in the literature that has investigated the effect of fat grafts on tendon healing. The results of this experimental study showed that fat grafting increased tendon healing with specific immunohistochemical markers.
The application of fat grafting is a method widely used in many surgical areas. The revelation that adipose tissue contains stem cells engendered the idea that fat grafting could be used as an alternative to stem cell treatments which are expensive and difficult to access.8,9 Covarubias et al. applied fat grafting to the periauricular region during facelift surgery. When the results were examined histologically, the application of fat graft was seen to have a regenerative effect on the skin. 12 Similarly in another study of patients with systemic sclerosis and ischaemic finger ulcers, treatment with fat grafting was attempted and was found to be successful. 11 In another multicentre study, patients who had experienced myocardial infarcts were applied with grafting with cardiac adipose tissue to the scarred region of the cardiac muscle during coronary artery bypass operation, and the results were examined. In addition, some clinical studies have shown that fat grafting used in tendon repairs reduced adhesions and it has been recommended as a method with a positive effect on outcomes especially in flexor tendon repairs.19,20 Thus, local adipose tissue grafting has been shown to be an easy-to-apply, low-cost method that provides successful results. 13 In the current study, for the first time in literature, the application of fat graft, which is a low-cost, easy-to-apply, and easily accessible method, was used in experimental tendon healing.
In several studies of tendon healing in which serial biomechanical evaluations were made, it was shown that the mechanical tensile strength of the tendon in rats is due to fibrosis in the early period. 21 Decreases in tensile strength can be seen in the fourth week with decreasing fibrosis and remodelling. In the current study, histopathological and immunohistochemical evaluations were made in addition to biomechanical outcomes, and as in the study taken as reference, the biomechanical tests were performed in the late period. 7
The current study results demonstrated that the maximum load and stiffness values were higher in the study group than in the control group, but the difference was not statistically significant (p: .068, p: .089, respectively). Previous studies investigating the tendon healing biomechanically found better results in early period only.22,23 When these results were taken into consideration, the fact that no statistically significant difference was determined in the current study was thought to be due to the test having been performed in the 4th week. However, there are also studies in literature that have shown biomechanical healing with a test performed in the 4th week. 7
In the histopathological evaluation of the current study, the tenocyte values of the study group were found to be higher than those of the control group. However, no difference was determined between the groups in respect of the ground substance, collagen, and vascularity values. In a previous study conducted with stem cells of adipose tissue origin, the histological data were examined in general as a single data group, and better results of the study group were reported. 7 The histopathological examination of the current study was performed in more detail under specific separate headings. Although an increase was found in the collagen values in the histopathological examination, the difference was not statistically significant (p: .076). However, with a more specific staining method in the immunohistochemical examination, the application of fat grafting was seen to provide positive results in respect of the collagen values in tendon healing.
Type 1 collagen is the most important and most abundant protein in the extracellular matrix of tendon tissue. Good quality tendon repair requires a high amount of collagen synthesis during healing. 24 An increase in Type 1 collagen is known to be one of the leading factors in increasing the tensile strength of the tendon. 25 In an experimental study by Uysal et al., it was shown immunohistochemically that the amount of type 1 collagen increased in the tendon with the application of stem cells of adipose tissue origin. 7 With the fat grafting in the current study as an easily applicable and low-cost method, the amount of collagen in the study group was shown immunohistochemically to be statistically significantly higher in the study group than in the control group, similar to the findings of Uysal et al.
TGF-beta is one of the cytokines that play an important role in wound healing and inflammation. Numerous studies in the literature have explored the activity of TGF in relation to tendon healing, yielding conflicting results. An increase in the level of TGF-beta has been shown to be the reason for an increase in collagen synthesis in tendon healing. 26 In another study, the application of TGF-beta was shown to increase tendon and ligament healing. 27 However, in the research into tendon healing by Uysal et al. using stem cells of adipose tissue origin, the TGF-beta values were found to be lower in the study group. 7 We used TGF beta one staining in the study. The association between increased TGF-beta one activity and fibrosis has been previously demonstrated in the literature. However, in a study that comprehensively investigated Achilles tendon healing both immunohistochemically and biomechanically, the relationship between TGF-beta one activity and healing was unequivocally established. Across all parameters, the study clearly demonstrated that enhanced TGF-beta one activity corresponds to improved tendon healing. 28 In another study investigating the outcomes of fetal tendon healing compared to adult tendon healing, it was observed that in adult tendon healing, TGF-beta one activity was found to be higher. However, despite this difference, no biomechanical distinctions were observed between the two groups. 29 In outcome, we believe that the increase in expression of TGF-β1 in study samples alters the inflammatory process by increasing the extracellular matrix, ultimately contributing to the acceleration of the healing process. In the immunohistochemical evaluation results of the current study of fat grafting, the TGF-beta values of the study group were determined to be higher than those of the control group. These data were supported by the increase in collagen staining seen in this study.
There were some limitations to this study, primarily the relatively low number of subjects as it was an animal model experimental study. Another notable limitation can be attributed to economic constraints, which prevented us from conducting immunohistochemical examinations for all histopathological parameters. Furthermore, due to the same constraints, an investigation of all TGF subgroups within our study was not feasible. Nevertheless, the study did encompass immunohistochemical staining of TGF-beta and collagen Type 1, both of which hold substantial histological validity. Finally, the biomechanical test was only performed in the 4th week, and therefore, there is a need for further studies to conduct serial biomechanical tests to be able to obtain more definitive results.
Conclusions
It was seen from the increased collagen and TGF-beta values in the results of this study that fat grafting made a positive contribution to tendon healing at a histopathological and immunohistochemical level. Compared with direct stem cell applications, this method, which is low-cost and easy-to-apply, can be considered to provide an effective contribution to surgical practice. Based on the outcomes of our study, we believe that following the repair of Achilles tendon ruptures, utilizing a readily obtainable fat graft that can be harvested easily from the surrounding area, once the paratenon has been repaired as extensively as possible, could offer benefits for tendon healing. Nevertheless, there is a need for further prospective, randomised, clinical studies to be able to obtain more definitive results.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
