Abstract
Bone defects caused by osteomyelitis can lead to severe disability. Surgeons still face significant challenges in treating bone defects. Nano-hydroxyapatite (n-HA) plays an important role in bone tissue engineering due to its excellent biocompatibility and osteoconductivity. Levofloxacin (Levo) was encapsulated in mesoporous silica nanoparticles (MSNs) via electrostatic attraction to serve as a drug delivery system. MSNs were incorporated with n-HA and polyurethane (PU). The degradation and osteoconductivity properties of these novel composite scaffolds and their effectiveness in treating chronic osteomyelitis in a rabbit model were assessed. Gross pathology, radiographic imaging, micro-computed tomography, Van Gieson staining, and hematoxylin and eosin staining were conducted at 6 and 12 weeks. The group of composite scaffolds combining n-HA/PU with MSNs containing 5 mg Levo (n-HA/PU + Nano +5 mg Levo) composite scaffolds showed superior antibacterial properties compared to the other groups. At 12 weeks, the n-HA/PU + Nano +5 mg Levo composite scaffolds group exhibited significantly greater volume of new trabecular bone formation compared to the other three groups. The surface of the novel composite scaffolds exhibited degradation after 6 weeks implantation. The internal structure of the scaffolds collapsed noticeably after 12 weeks of implantation. The rate of material degradation corresponded to the rate of new bone ingrowth. This novel composite scaffold, which is biodegradable and osteoconductive, has potential as a drug delivery system for treating chronic osteomyelitis accompanied by bone defects.
Introduction
Chronic osteomyelitis is a bone infection that results from the delayed diagnosis and management of acute osteomyelitis leading to catastrophic consequence. 1 Osteomyelitis can be caused by open fractures and orthopedic surgery. 2 Sequestrum and sinus tract formation are common clinical manifestation of chronic osteomyelitis. Chronic osteomyelitis places a significant economic burden on patients and creates complex therapeutic challenges for surgeons. Chronic osteomyelitis is more prevalent in developing countries than in developed countries.3–5 The predominant microorganism is Gram-positive bacteria Staphylococcus aureus. 6 The incidence of chronic osteomyelitis caused by multi-resistant microorganisms, such as methicillin-resistant Staphylococcus aureus, 7 is increasing.
The primary treatment approaches for the treatment of chronic osteomyelitis is thorough sequestrum debridement and systematic use of antibiotics for 5 weeks. 8 Prolonged infection reaction and sequestrum formation lead to bone destruction, the final result is bone tissue defect lead to severe disability. Autologous bone, allogeneic bone and bioactive composite scaffold material are commonly used in clinical treatments for bone defect reconstruction. Autologous bone grafting is considered the gold standard for bone regeneration. 9 However, autologous bone has limitations, as allogeneic bone can result in adverse reactions or may carry a risk of disease transmission to patients.10,11 Therefore, there is a need for novel composite scaffolds for bone reconstruction are.
Nano-hydroxyapatite (n-HA) is the primary inorganic component found in human bones. It is commonly employed in tissue engineering as a bone replacement material due to its biocompatibility, and non-toxic and osteoconductive properties.12,13 More over, n-HA is utilized as a carrier for antibiotics, growth factors and anticancer drugs because of its effective adsorption ability.14–16 Consequently, n-HA is regarded as one of the most promising materials in the field of bone tissue engineering. Polyurethane (PU) is a commonly utilized biodegradable and biocompatible scaffold in the medical field, and it has been demonstrated to serve an effective drug delivery system.17,18
Mesoporous silica nanoparticles (MSNs), initially described by Kresge et al, 19 have been extensively studied in the fields of nanomedicine and bionanotechnology as a drug delivery system. This is due to their large volume, controllable pore size and adaptable structure. MSNs can be loaded with various antibiotics and small bioactive molecules for the treatment of cancer.20,21 Levofloxacin (Levo) is the S(-) enantiomer of ofloxacin racemate, and it demonstrates potent antibacterial ability against both gram-positive and -negative bacteria. Furthermore, due to its low molecular weight, it can readily be combined with MSNs through electrostatic attraction. 22
In the present study, we first synthesized MSNs and then loaded Levo into MSNs through electrostatic attraction. The MSNs were then adsorbed onto the surface of the n-HA/PU scaffold, resulting in the synthesis of a novel bioactive, biodegradable, antibacterial scaffold. The objectives of the present study were to investigate the degradation properties and osteoconductivity of this novel composite scaffold using micro-computed tomography (micro-CT), as well as to evaluate its effectiveness in treating chronic osteomyelitis.
Material and methods
Preparation of new biodegradable scaffolds
The detailed method for synthesizing composite scaffolds was described in our previous study. 23 Briefly, MSNs were synthesized in accordance with Hyeon’s method. 24 Cetyltrimethylammonium bromide (CTAB; 0.2 g) was mixed with 0.75 mL 2 mol/l NaOH, and the resulting solution was added to 100 mL H2O under stirring. Then 20 mL n-hexane was added to the above solution while stirring at 400 r/min. Fe3O4 was stabilized using oleic acid. The suspension was supplemented with 2 mL ethyl acetate and 0.5 mL tetraethyl orthosilicate at 70°C for 3 hours. The nanoparticles of CTAB and Fe3O4 were formed when the pH value of the suspension decreased. The solid product was obtained by filtering and vacuum drying at room temperature, resulting in the formation of MSNs. Finally, MSNs was dispersed in suspension and Levo was successfully encapsulated through electrostatic attraction.20,24
n-HA nanoparticles were synthesized using the wet chemical method. 25 The n-HA/PU bioactive scaffolds were propitiously fabricated using the in situ foaming method.26,27 Initially, 30 g castor oil was mixed with 40 g n-HA particles in a nitrogen atmosphere and under thorough stirring. Then, 30 g isophorone diisocyanate was added to the suspension. The solution was kept at 70°C for 3 hours to obtain the polymer. Subsequently, 1 mL 1,4-Butanediol was added to extend the prepolymer. The n-HA/PU bioactive scaffolds were thus obtained. The novel bioactive scaffolds were shaped into small cuboids with dimensions of 10x6x6 mm. The bioactive scaffolds were immersed in a solution of 2% (w/w) acetic acid and 0.5 wt% chitosan followed by the MSN +Levo solution for 30 min. Finally, they were dried in a vacuum oven at 40°C. 28 Subsequently, scaffolds combining n-HA/PU with nanoparticles containing 1 mg Levo (n-HA/PU + MSN +1 mg Levo) were successfully synthesized. Similarly, scaffolds combining n-HA/PU with nanoparticles containing 5 mg Levo (n-HA/PU + Nano +5 mg Levo) were synthesized using the same method. 23
Scanning electron microscopy (SEM)
SEM was employed to observe the morphological characteristics of novel composite scaffolds. Prior to imaging, the surface of the composite scaffolds was coated with gold powder, and SEM (JEOL JSM-7500F, USA) was used to evaluate the morphology of the nanosphere and the coated composite scaffolds.
Establishment of the animal model. All procedures were performed in accordance with recognized guidelines. All experimental schemes were approved by the Institutional Animal Care and Use Committee of Heze Medical College (Heze, China). The Staphylococcus aureus (cat. no. 25923; American Type Culture Collection) strain was used in the present study. The bacterial concentration was 3x107 colony-forming unit (CFU)/ml and was assessed using a UV spectrophotometer. A total of 36 New Zealand White rabbits were used. The rabbits were procured from the experimental animal center of Daping Hospital, Research Institute of Surgery of the Third Military University. The sex of the rabbits was random and they weighed between 2-4 kg. The rabbits were allowed to acclimatize for 2 weeks before surgery. The methods for establishing osteomyelitis models have been refined based on Norden’s method. 29 Briefly, the rabbits were anesthetized using 30 mg/kg sodium pentobarbital through the marginal auricular vein. They were then fixed, and the right tibia of the rabbit was shaved and disinfected with iodophor. A 2-cm longitudinal incision was operated on the medial side of the tibia. A round hole (diameter, 3 mm) was drilled into the proximal part of the tibia. Next, 0.1 mL solution containing 3x107 CFU/ml bacteria was injected into the medullary cavity. Bone wax was used to block the hole. Following surgery, all rabbits were fed individually. Postoperatively, the rabbits had free access to food and water and were monitored daily for complications or abnormal behaviors during the healing period.
Debridement and implant material
Six weeks after the surgery, radiological imaging, pathology and observations of local soft tissue appearance were employed as indicators to evaluating the effectiveness of the rabbit model of chronic osteomyelitis. The animal model of chronic osteomyelitis was successfully established in all rabbits. Complete debridement was performed on the tibia affected by chronic osteomyelitis, creating a fenestration of 10 × 6 mm. Subsequently, the rabbits were randomly divided into four groups. An overview of experimental grouping and indicator detection time points was showed in Figure 1. Group A (n = 9) consisted of rabbits that solely underwent debridement and sevred as the blank control. Group B (n = 9) rabbits were treated with n-HA/PU. Rabbits in group C (n = 9) were treated with n-HA/PU + Nano +1 mg Levo. Rabbits in group D (n = 9) were treated with n-HA/PU + Nano +5 mg Levo. Radiological imaging performance, pathology, observations of local soft tissue appearance, micro-CT scans and histological evaluation were analyzed at 1, 6 and 12 weeks post-surgery. The surgical procedure was conducted following previously documented procedures (Figure 2).
23
An overview of experimental grouping and indicator detection time points. The structural and morphological characterization of new synthetic composite scaffolds and implantation process. (a). nano-hydroxyapatite/polyurethane (n-HA/PU). (b). The SEM image of n-HA/PU. (c). 1 mg Levofloxacin @ mesoporous silica nanoparticle /nano-hydroxyapatite/polyurethane (1mg Lev@ MSNs/n-HA/PU). (d). The SEM image of 1mg Lev@ MSNs/n-HA/PU. (e). The molecular structural formula of Levofloxacin. (f). The threedimensional structural diagram of newly synthetic mesoporous silica microspheres, Levofloxacin was attached to the surface of the microspheres through electrostatic attraction. (g). The size of opened debridement is 10 mm×6 mm. H. The new composite scaffolds was implanted in the bone defects.

Radiological evaluation
Radiological imaging techniques was used to access the severity of chronic osteomyelitis. The animals were anesthetized and immobilized, and radiological images were obtained at 6 and 12 weeks post-implantation. The radiological machine (Carestream DRX) with an exposure time of 2.8 mAs and instrument parameters at 250 mAs and 44 kVp was utilized in this study.
Gross pathology
The rabbits in each group were euthanized at 1, 6 and 12 weeks after surgery. There were 3 rabbits per group at each time point. The surgical procedure was carried out as follows: Firstly, the rabbits were injected with sodium pentobarbital (60 mg/kg) through the marginal auricular vein to induce deep anesthesia. Then, 40 mL of air was rapidly injected into the marginal auricular rabbit vein using the air embolism method. Rabbit death was confirmed by the loss of righting reflex and cessation of breathing. The tibia was obtained after removing the surrounding soft tissue. The severity of bone defects, tibia infection, and the presence of the sequestrum were evaluated.
Micro-CT measurement
After gross pathology examination, the tibia was divided into small sections and then immersed in 4% paraformaldehyde. The samples were scanned using a micro-CT scanner (Scanco Medical Viva CT40; SCANCO Medical AG) with the following instrument parameters: 1 mm thickness, 300 mAs, 60 KV and 1,024x1,024 voxel matrix. Scanco medical systems (cat. no. SC5073; version μ CT v6.1) were used to generate two-dimensional and 3D reconstruction pictures of the composite scaffolds. A total of three rabbits from each group were calculated.
A cuboid region of the same size near the surface of the composite scaffolds was considered appropriate for calculating new bone formation. This region was selected. A threshold ranging from 450 to 1000 was utilized to identify the bone. The bone volume to total volume ratio (BV/TV), trabecular thickness (Tb. Th) and trabecular number (Tb. N) were assessed. A cube region of the same size at the center of the composite scaffold material was deemed appropriate for determining the degradation degree of the composite scaffolds. This region was chosen. A threshold ranging from 220 and 450 was utilized to recognize the hydroxyapatite. Scaffold volume (SV), SV to total volume (SV/TV), scaffold density (SD), scaffold number (SN), scaffold thickness (ST) and scaffold space (SS) were assessed. Micro-CT measurement were evaluated by two radiologist independently.
Histological evaluation
After obtaining the micro-CT measurements, the samples were immersed in 4% paraformaldehyde solution. The sample was subjected to dehydration using different concentrations of alcohol for several days. Subsequently, the samples were embedded in methyl methacrylate for 1 week. The non-decalcified tibia was secioned into 200-μm slices using EXAKT 300 CP cutters. The slices were polished to a thickness of 30 μm and then prepared for Van Gieson (VG) staining. Additionally, another sample was decalcified using 4% HNO3 and dehydrated with several different concentration gradients of alcohol. Subsequently, The slices were further cut into 5 μm slices using a microtome (Leica Microsystems, GmbH) and prepared for hematoxylin and eosin (H&E) staining. The formation of new bone and the severity of infection were observed. Pathological morphological changes were evaluated by two pathologists independently. They were stained with HE and VG, then examined under an optical microscope.
Statistical analysis
All quantitative data are presented as the mean ± standard deviation. To assess the normality of the data set, the Shapiro-Wilk test was employed. Significant differences between groups were determined using one-way ANOVA with SPSS version 19.0 (IBM, Corp.). A p-value of less than 0.05 was considered to indicate a statistically significant difference between two groups.
Results
Structural and morphological characterization
The novel bioactive materials were successfully synthesized. The n-HA/PU scaffolds are shown in Figure 2(a). A large number of pores could be seen on the surface of the n-HA/PU scaffolds. The average porosity of the bioactive materials was (54.46±5.68)%, with a size of 10x6x6 mm. The SEM image of the n-HA/PU scaffolds is shown in Figure 2(b). The morphology of n-HA/PU could be clearly observed. A total of 1 mg Levo was loaded on MSNs and combined with n-HA/PU scaffolds. The n-HA/PU + Nano +1 mg Levo scaffold was shown in Figure 2(c). The SEM image of the n-HA/PU + Nano +1 mg Levo scaffold was shown in Figure 2(d). A large number of MSNs were uniformly absorbed in the surface of n-HA/PU. The molecular structural formula of Levo is shown in Figure 2(e). The 3D structural diagram of newly synthesized mesoporous silica microspheres is shown in Figure 2(f). Several perforated channels were observed above the mesoporous silica microspheres. A high volume of Levo was attached to the surface of the microspheres through electrostatic attraction.
Animal models of chronic osteomyelitis
Following the injection of Staphylococcus aureus into the tibia for 6 weeks, all rabbits survived. Among them, 3 rabbits experienced complications of reduced diet and weight loss compared to before surgery. Animal models of chronic osteomyelitis were successfully created. There was sinus tract formation and pyogenic fluids outflow from the soft tissue of tibia. Bone destruction and sequestrum formation could been seen in the gross pathology pictures. A high volume of pyogenic fluids was found around the tibia in soft tissue. Radiological imaging showed that inflammation destroyed bone tissue and lead to bone defect. A large number of inflammatory cells could been seen from H&E staining pathological section. All rabbits were successfully established animal models of chronic osteomyelitis.
Radiographic evaluation
On week 12 from implantation, significant bone destruction, soft tissue swelling, sequestrum formation, bone destruction, obscure boundary and osteolytic lesions caused by chronic osteomyelitis were clearly observed in the blank control group (Figure 3). Inflammatory reaction of bone and sequestrum formation were discovered in group B. No obvious bone defects or tissue swelling were observed in the 1 mg Lev@MSNs/n-HA/PU group. No obvious osteomyelitis symptoms were discovered in the n-HA/PU + Nano +5 mg Levo group. Performance of X-ray imaging, GP, VG, HE in each group after implantation process for 12 weeks. BC, blank control group (Group A). HA+ 0 mg, n-HA/PU (Group B). HA+ 1 mg, 1mg Lev@ MSNs/n-HA/PU (Group C). HA+ 5 mg, 5 mg Lev@ MSNs/n-HA/PU (Group D).
Gross pathology
Gross pathology was assessed after sacrificing the rabbits at 12 weeks, and the gross pathology of the tibia was clearly observed. Group A exhibited sinus tract formation and pyogenic fluid outflow on the surface of the tibia. Bone destruction and defects could be seen in group A. Group B showed limb swelling, skin irritation and severe inflammation were observed in group B. Group C did not exhibit any obvious inflammatory reaction and limb swelling. Group D showed no signs of inflammatory reaction, and the wound had healed well in group D.
VG staining
At 12 weeks, group A exhibited reduced bone density and sparse collagen fibers. Hydroxyapatite composite scaffold particles (yellow arrow) were observed in the n-HA/PU, n-HA/PU + Nano +1 mg Levo and n-HA/PU + Nano +5 mg Levo groups. In group B there was a small amount of novel collagen fiber; red collagen fibers represent new bone trabeculae (blue arrow). The number of collagen fibers that grew into the n-HA/PU + Nano +5 mg Levo composite scaffolds was higher than that in the n-HA/PU and n-HA/PU + Nano +1 mg Levo groups. The n-HA/PU + Nano +5 mg Levo group showed abundant newly formed collagen fibers growing into the composite scaffolds and tightly adhering to the implant surface. The VG staining images of the bone are shown in Figure 3.
H&E staining
The H&E staining images at 12 weeks are shown in Figure 3. Group A exhibited a significant accumulation of inflammatory cells in the bone marrow cavity in. Minimal inflammatory reaction was observed in the n-HA/PU + Nano +1 mg Levo group. No inflammatory cells were observed in the n-HA/PU + Nano +5 mg Levo group. These results indicated that the sustained release of antibiotics from the composite scaffolds had a therapeutic effect on bone infection.
Micro-CT measurement
3D reconstruction of the composite scaffolds
Figure 4 displays the 3D reconstructed images of the bone and composite scaffolds. In group A, a significant inflammation response resulted in bone destruction, leading to the identification of a large bone defect. The volume of the new bone formation around the n-HA/PU bioactive material was relatively small. Conversely, the volume of the new trabecular formation around the n-HA/PU + Nano +1 mg Levo scaffold was big, as compared with that around the n-HA/PU scaffold. The newly generated trabeculae were closely attached to the composite scaffolds and almost filled the entire bone marrow cavity in the n-HA/PU + Nano +5 mg Levo group. Three-dimensional reconstruction images of the composite scaffolds. (a). Blank control group. (b). n-HA/PU, nano-hydroxyapatite/polyurethane group. (c). 1 mg Lev@ MSNs/n-HA/PU: 1 mg levofloxacin @ mesoporous silica nanoparticle /nano-ydroxyapatite/polyurethane. (d). 5 mg Lev@ MSNs/n-HA/PU: 5 mg levofloxacin @ mesoporous silica nanoparticle /nanohydroxyapatite/polyurethane.
New bone formation
The volume of newly formed trabecular bone (BV/TV) on week 1 showed no significant difference among the four groups (p > .05). However, on week 6, group D exhibited a significantly higher volume of novel trabecular bone formation compared to groups A (p = .003), B (p = .010) and C (p = .034). Similarly, on week 12, group D showed a significantly higher volume of novel trabecular bone formation compared to groups A (p = .001), B (p = .019) and C (p = .035). The statistical analysis results are shown in Figure 5(a). The volume of novel trabecular bone formation (a), the number of novel bone trabeculars (b) and the thickness of new bone trabecular (c) of each group. BC, blank control group (Group A). HA+ 0 mg, n-HA/PU (Group B). HA+ 1 mg, 1mg Lev@ MSNs/n-HA/PU (Group C). HA+ 5 mg, 5 mg Lev@ MSNs/n-HA/PU (Group D). Bar and error bars represent mean and sd values. *p < 0.05.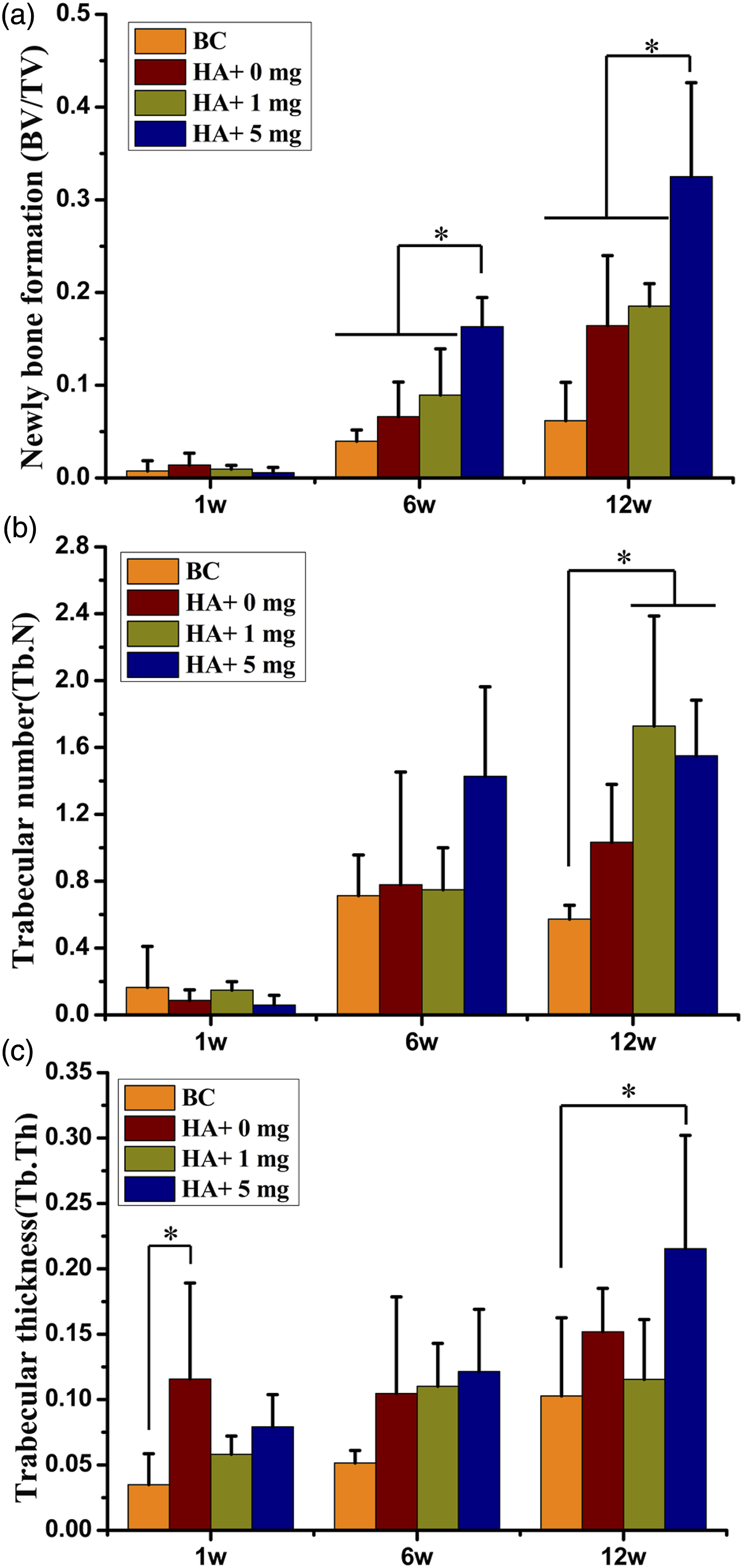
Tb. N
Regarding the number of Tb. N, no significant difference was observed among the four groups on both week 1 and week 6 (p>.05). However, on week 12, a significant difference was observed between groups D and A (p = .019), as well as between groups C and A (p = .009). The statistical analysis results are presented in Figure 5(b).
Tb. Th
On week 1, Tb. Th in group B was found to be significantly higher than that in group A (p = .043). However, no significant difference in Tb. Th was observed among the other groups (p > .05). On week 6, there was no significant difference in Tb. Th among the four groups (p > .05). Finally, on week 12, group D exhibited a significantly higher about Tb. Th compared to group A (p = .049). The statistical analysis results are depicted in Figure 5(c).
Degradation of novel composite scaffolds
The novel composite scaffolds were reconstructed using micro-CT, and Figure 6 presents the 3D images of composite scaffolds captured at different time points. On week 1, the n-HA/PU composite scaffolds exhibited no signs of degradation and remained intact. However, on week 6, degradation was observed at the edge of the composite scaffolds, accompanied by a decrease in the density of the internal structure compared to week 1. On week 12, the porosity of the composite scaffolds had increased, the internal structure of the material had started to collapse, and the density of the material had further decreased. Three-dimension reconstruction pictures of the composite scaffolds. BC, blank control group (Group A). HA+ 0 mg, n-HA/PU (Group B). HA+ 1 mg, 1mg Lev@ MSNs/n-HA/PU (Group C). HA+ 5 mg, 5 mg Lev@ MSNs/n-HA/PU (Group D).
Remaining SV
Regarding the the remaining SV, statistical analysis revealed significant differences between week1 and week 12 in various groups. In group C, the SV on week 1 was significantly higher than that on week 12 (p = .009). Similarly, the SV of the 1 mg Lev@MSNs/n-HA/PU group on week 6 was also significantly higher than that on week 12 (p = .033). Group D exhibited a significantly higher on week 1 compared to week 12 (p = .049). The SV of group B on week 1 was significantly higher than that on week 12 (p = .036). Detailed statistical analysis results of the SV for each group are shown in Figure 7(a). The quantification results of degradation of novel composite scaffolds. (a). The statistical result of the remaining scaffold volume (SV). (b). The statistical result of the percent scaffold volume (SV/ TV). (c). The statistical result of the scaffold density (SD). (d). The statistical result of scaffold number (SN). (e). The statistical result of scaffold thickness (ST). (f). The statistical result of scaffold space (SS). HA+ 0 mg, n-HA/PU (Group B). HA+ 1 mg, 1 mg Lev@ MSNs/n-HA/PU (Group C). HA+ 5 mg, 5 mg Lev@ MSNs/n-HA/PU (Group D). Bar and error bars represent mean and sd values. *p < 0.05.
SV/TV
Furthermore, the SV/TV also showed significant differences between week 1 and week 12 in certain groups. In group C, the SV/TV on week 1 was significantly higher than that on week 12 (p = .009). The SV/TV in group C on week 6 was significantly higher than that on week 12 (p = .034). The SV/TV in group D on week 1 was significantly higher than that on week 12 (p = .006). The SV/TV in group D on week 6 was significantly higher than that on week 12 (p = .002). No significant difference in the SV/TV was among the three different time points in group B (p > .05). The statistical analysis results of the SV/TV in each group are shown in Figure 7(b).
SD
No significant difference was observed in the SD among the three different time points in group C (p > .05). The SD in group D on week 6 was significantly higher than that on week 1 (p = .006) and 12 (p = .03). There was no significant difference in the SD at three different time points in group B (p > .05). The statistical analysis results of the SD in each group is shown in Figure 7(c).
SN
The SN in group C on week 1 was significantly higher than that on week 12 (p = .020). The SN in group D on week 1 was significantly higher than that on week 12 (p = .014). The SN in group B on week 1 was significantly higher than that on week 12 (p = .025). The statistical analysis results of the SN in each group is shown in Figure 7(d).
ST
There was no significant difference in ST among the three different time points in group C (p > .05). The ST in group D on week 6 was significantly higher than that on weeks 1 (p = .004) and 12 (p = .014). There was no significant difference in ST among the three different time points in group B (p > .05). The statistical analysis results of the ST in each group are shown in Figure 7(e).
SS
The SS in group C on week 12 was significantly higher than on week 1 (p = .025). The SS in group D on week 12 was significantly higher than that on week 1 (p = .005) and 6 (p = .038). The SS in group B on week 12 was significantly higher than that on week 1 (p = .021). The statistical analysis results of the SS in each group was shown in Figure 7(f).
Discussion
Chronic osteomyelitis is a progressive infection that cause an inflammatory reaction, sequestrum formation and bone neoformation. 30 It can invade various structures, including the bone marrow, the cortex and surrounding soft tissue. Chronic osteomyelitis cannot usually be eradicated, except by surgical debridement. Radical debridement involves removing sequestra and resecting scarred and infected bone and soft tissue. 2 Prolonged inflammatory response and radical debridement can results in bone defects, leading to disability. 31 Appropriate reconstruction of the bone defects and administration of antibiotics for infection control are necessary.
Autologous bone,32,33 allogeneic bone34,35 and bioactive composite scaffold material 36 are the most commonly used materials in clinical settings for reconstructing of bone defects. Autologous bone graft is considered the gold standard for bone regeneration. 9 Rodolfo et al 37 employed the osteoprogenitor cell-based tissue engineering approach to treat three patients with extensive bone defects. These cells were placed on macroporous hydroxyapatite scaffolds. However, the clinical application of autologous bones has the following drawbacks: A limited supply of autograft material, secondary infection and donor site morbidity.32,38 Allogeneic bone may lead to infectious diseases and immune reactions, and there are no effective treatments for it.
Bone tissue engineering has attracted significant attention for its outstanding performance in repairing bone defects. In the field of bone tissue engineering, researchers have investigated bioactive composite scaffolds that mimic the structure and mechanism of natural bone.39,40 An ideal scaffold should possess several essential properties: (i) Biocompatibility with human tissues; (ii) structural and mechanical resemblance to natural bone; (iii) degradation rate of the scaffolds consistent with natural bone osteogenesis; and (iv) provision of a suitable biological environment for cell adhesion, proliferation and differentiation.41,42
n-HA is the primary inorganic component of human bones, accounting for 60% of the bone composition. Previous studies have demonstrated that n-HA exhibits osteoinductivity and biocompatibility, enhances mechanical strength, and promotes the deposition of calcium phosphate. 43 Due to its satisfactory adsorption ability, n-HA has been utilized as a carrier for antibiotics, growth factors and anticancer drugs. 14 PU is a known biocompatible and biodegradable material that has demonstrated effective performance as a drug delivery system. Previous studies have shown the successful combination of n-HA and PU as an osteogenic platform. 44 Therefore, n-HA and PU were combined and used as a carrier in the present study.
Kresge 19 first described MSNs. MSNs have a unique structure, controllable pore size, chemical modification capability and large pore volume, making them promising for use as antibiotic carrier systems in the treatment of cancer or infectious diseases.45,46 MSNs can effectively load drugs and biologically active small molecules be loaded into for the treatment of cancer. Levo is the optical S(-) isomer of the racemic substance ofloxacin, exhibiting a broad spectrum of antibacterial properties against both G+ and G- bacteria. Due to its low molecular weight, Levo can easily bind with nanoparticles through electrostatic attraction. 47
The treatment of chronic osteomyelitis remains a challenge. The commonly used clinical method involves a 3-week course of oral antibiotics followed by 3 weeks of intravenous administration. 48 Due to the difficulty of penetrating the hard bone tissue, high doses of antibiotics must are required to achieve effective an effective antibacterial concentration at the infected site. However, this high dosage of antibiotics can lead to systemic toxic effects, including hepatotoxicity, nephrotoxicity and gastrointestinal syndromes. 49 In the present study, no significant differences were observed in the new BV/TV among the four groups at week 1. This lack of difference may be attributed to the persistent infection, which inhibited new bone formation. However, at the 6- and 12-week time points, the controlled release of Levo from the MSNs effectively managed the local inflammatory reaction, thereby creating a favorable environment for new bone formation. This may explain why the new BV/TV in the 5 mg Lev@MSNs/n-HA/PU group was significantly higher than that in the other groups at the 6- and 12-week timepoints. Additionally, at week 12, the Tb. N in groups D and C was significantly higher than that in group A. The remaining SV in groups B-D at week 12 was significantly lower than that on week 1. The SV in group C at week 12 was also significantly lower than that at week 6. These results demonstrated that these novel composite scaffolds had undergone degradation at week 6 from implantation. The internal porous pores of the scaffolds had clearly collapsed at week 12 from implantation. The rate of material degradation matches the speed of new bone ingrowth. Furthermore, H&E staining at week 12 revealed no obvious inflammatory cells in group D.
For the past three decades, micro-CT has been employed to analyze the characteristics of natural bone.50–52 Micro-CT can provide precise 3D datasets, and has been widely utilized to assess the architecture, in vitro degradation properties and new bone formation in composite scaffolds.53,54 Zhou et al 55 conducted an in vivo evaluation on the degradation of gelatin/β-TCP (G-TCP) composite porous scaffolds loaded with vancomycin. The results revealed that the G-TCP composite porous scaffolds exhibited a vancomycin release duration of 3 weeks, following which rapid degradation occurred. The vancomycin-loaded G-TCP5 scaffold could not fulfil the requirement of clinical treatment for chronic osteomyelitis. However, the assessment of degradation degree in relied solely on X-ray imaging without accurate quantification. The present study demonstrated that the novel composite scaffolds underwent degradation 6 weeks after implantation, with the internal structure collapsing at 12 weeks after implantation. In the present study, the remaining SV of the composite scaffolds at week 1 was significantly higher than that at week 12 in all three groups. The statistical analysis results of the SN were consistent with those of the remaining SV. The degradation performance of the scaffolds was also confirmed by the SN results. Hruschka et al 56 evaluated the performance of an n-HA aqueous paste in a novel scapula defect model. The results indicated that the degradation characteristics of n-HA pastes depends on their concentration and particle size. Ostim®, an aqueous paste containing n-HA, has been reported to be biocompatible and osteoconductive. Brandt et al 57 investigated the resorbability and osteointegration of the nanocrystalline hydroxyapatite Ostim® in a rabbit model. However, complete resorption of the implant material was not achieved until 12 weeks after surgery. In this experiment, the size of the scaffold was 10mm×5mm. The bone defect in rabbit tibia was also 10mm×5mm. The scaffold material perfectly fills the bone defect. When the scaffold material is used for the human body, the size of the scaffold was still 10mm×5mm, two or several scaffold could be used in the bone defect site.
Levo demonstrates suitability as a drug delivery system for the treatment of chronic osteomyelitis. In this study, electrostatic attraction was employed to successfully encapsulate 1 or 5 mg of Levo within MSNs. MSNs containing of 1 or 5 mg antibiotics were incorporated with n-HA/PU, and a novel osteoconductive and biodegradable composite scaffold was successfully synthesized. The group treated with n-HA/PU + Nano +5 mg Levo exhibited superior antibacterial properties compared to the other groups. This antibacterial effect contributed to the elimination of the inflammatory reaction, creating a favorable environment fo the promotion of osteogenesis by the composite scaffolds. After 6 weeks of implantation, degradation of the surface of the novel composite scaffolds was observed, with the internal structure of the material collapsing clearly collapsed after 12 weeks of implantation. This novel, biodegradable, osteoconductive composite scaffold holds promise for its application in bone tissue engineering for the treatment of chronic osteomyelitis accompanied by bone defects.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Development Program of the Affiliated Hospital of Heze Medical College (HYFY202108) and grant from the Medical and Health Science and Technology Development Plan Project of Shandong Province and Scientific Research (No: 2019WS349).
