Abstract
Purpose
3D-printed models rapidly evolving in orthopaedic. Studies show that 3D-printed models used for preoperative planning improve a better understanding of fracture morphology and reduce operative time, blood loss and frequency of fluoroscopy, but there are no studies that investigated possible advantages in the outcomes and complications for the treatment of distal radius fracture (DRF). Our study aims to evaluate short-term functional results and complications between two groups treated DRF using 3D-printed models for preoperative planning and without. We hypothesize that the addition of 3D-printed models would improve functional outcomes and reduce complication rates.
Methods
66 randomized cases of DRF AO/OTA C type were enrolled and divided into “Control group” (n = 33) and “3D-printed model group” (n = 33). Personalized 3D-printed models were created. The primary outcomes were: Patient-Rated Wrist Evaluation questionnaire, Quick Disabilities of the Arm, Shoulder and Hand Score questionnaire, and complications. The secondary outcomes were: measurement of the range of motions, grip strength, radiological evaluation, and the visual analogue scale. Assessments were measured at 6 weeks, 3 months, and 6 months intervals.
Results
We found that the integration of the 3D-printed model in preoperative planning decreased complication incidence significantly – from 30.3% in the “Control group” to 6.1% in the “3D-printed model group”, p = .022. But we did not find a difference in functional and radiological outcomes.
Conclusion
The 3D-printed models for preoperative planning surgically treating DRF AO/OTA C type can help minimize the complication rate, however, they can’t improve functional outcomes in the short-term results.
Level of evidence
Level I randomized controlled study.
Introduction
Distal radius fracture (DRF) became a more common injury in the population because young and older individuals promote an active lifestyle. 1 DRF in young individuals is mostly related to high-energy trauma, while for older patients prevails low-energy trauma. 2 Because trauma incidence increases, medical costs of DRF treatment are rising each year. 3
In the past decade, the overall rate of open reduction and internal fixation (ORIF) has increased.4,5 According to the AO Foundation (AO/OTA) classification, 25%–35% of distal radius fractures are complex intra-articular fractures and surgical management remains a challenge. 6
The overall complication rate following DRF internal plate fixation in the literature varies from 4% to 36%. 7 Management of the complex distal radius fracture AO/OTA C type has a higher incidence of complications such as loss of reduction, implant problems, infection, tendon irritation, carpal tunnel syndrome, malunion and traumatic osteoarthritis.8,9
3D-printed models rapidly evolving in orthopaedic for preoperative planning. 10 Recently published studies show that 3D-printed models used for surgical planning reduce operative time, blood loss and frequency of fluoroscopy.11,12 In addition, 3D-printed models improve a better understanding of fracture morphology and communication between doctors and patients.13,14 Chen et al. 14 randomised-controlled study show that 3D-printed models did not improve functional outcomes after 1-year follow-up. To our knowledge, there are no randomized-controlled clinical studies that investigated possible advantages in the short-term outcomes and complications of using 3D-printed models for the treatment of complex DRF. The main question for surgical planning remains determining - which fragments can be fixed and how they should be technically fixed with implants because there is a correlation between joint reduction and functional outcomes, especially in higher demand cases.15,16
Our study aims to evaluate short-term functional results and complication rates between two groups surgically treated intra-articular distal radius fracture AO/OTA C type using 3D-printed models for preoperative planning and without. We hypothesize that the addition of 3D-printed models for preoperative planning would improve functional outcomes and reduce complication rates in the short-term.
Materials and methods
Study design and preparations
The prospective randomized controlled study was approved by ******* Regional Biomedical Research Ethics Committee. From January 2021 to February 2022 70 randomized cases of unstable distal radius fracture AO/OTA C type surgically treated in the level I Trauma Centre were enrolled in this study according to the inclusion and exclusion criteria. Flowchart of the study (Figure 1). Flowchart of the study.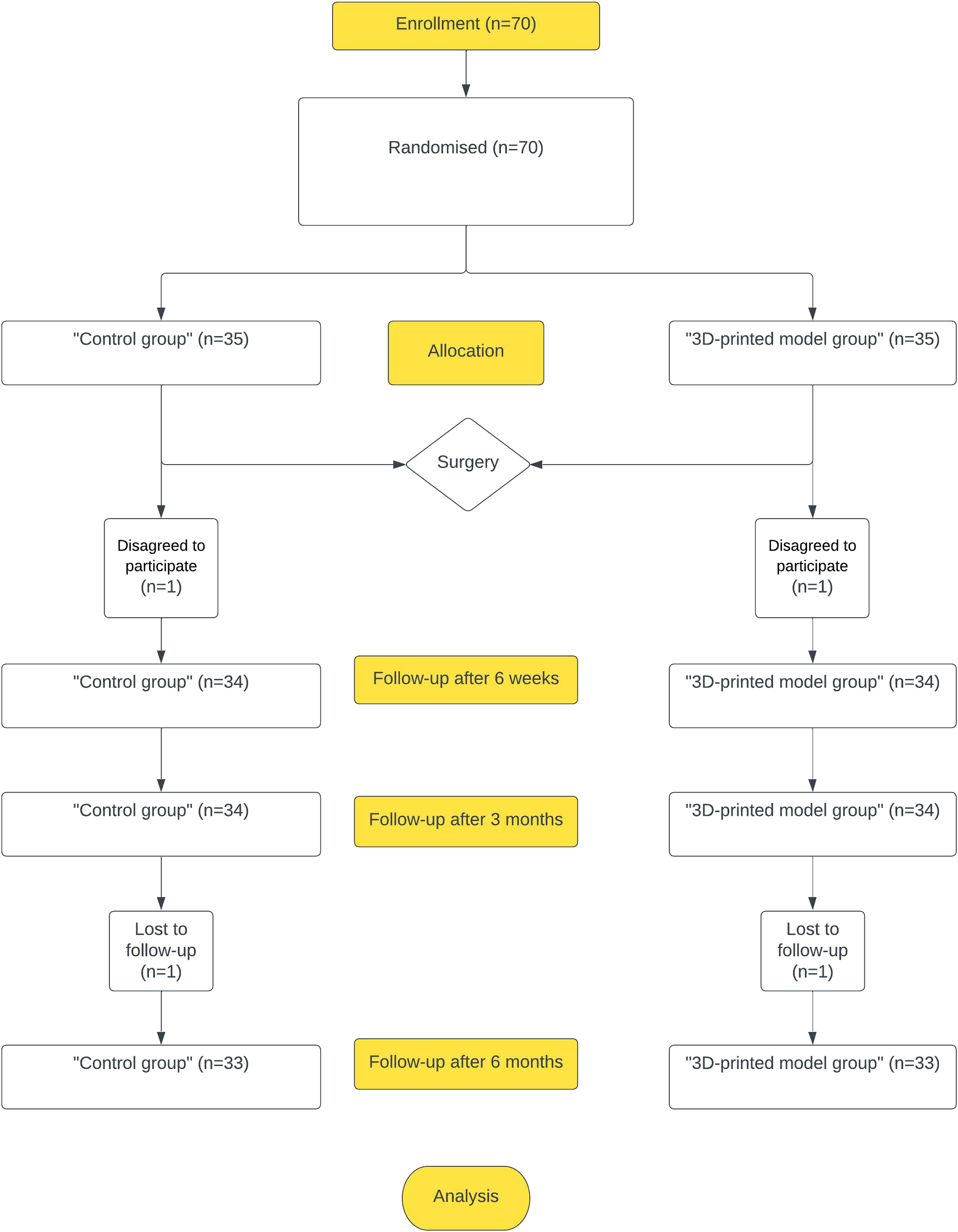
Study population
Patients selected were from 18 to 75 years of age who met inclusion criteria: isolated acute displaced distal radius intra-articular fracture AO/OTA C type with/or without ulnar styloid fracture diagnosed clinically and radiologically with no previous history of significant wrist pathology, agreed to participate in the study and gave written consent. The main exclusion criteria were: patients who disagree to participate in a clinical study, fractures beyond 3 weeks, open fractures, polytrauma, bilateral fractures, neurovascular injury, prior fractures of the wrists, hands or arms, other diseases: oncology, rheumatological diseases, neurologic deficiency, or mental disorders.
Randomisation
Participants (n = 70) were randomly divided with a 50/50 ratio into two groups: the “Control group” (n = 35) and the “3D-printed models group” (n = 35) by sealed envelope assistance. The envelope was taken randomly before each surgery by a third person who is not an author of this study. “Control group” –participants were surgically treated using preoperative planning pre-reduction and post-reduction X-rays and CT scans with 3D CT reconstructions. “3D-printed model group”- participants were surgically treated using preoperative planning same as the “Control group”, but with the addition of the 3D-printed model. All patients underwent ORIF with the same operative technique with volar or dorsal approach or combined approach9,17 and the same postoperative care algorithm was applied with wrist immobilization for 2 weeks and starting early motions exercises. 18
3D-printed models were created by a single surgeon, who neither evaluated nor had any influence on this study. Three orthopaedic surgeons performed the ORIF of the patients included in this study. All surgeons have more than 10 years (>35 cases in a year) of experience in the surgical management of DRF.
3D-printed models for preoperative planning
3D-printed models of the distal radius fracture were created using CT scan files in Digital Imaging and Communication in Medicine (DICOM) format obtained from the hospital’s MedDream software, version 7.6.0. Using 3D Slicer, version 4.10.2 software. DICOM files were processed and a 3D digital model of the distal radius fractures was created in Standard Triangle Language (STL) format. With Z-suite, version 2.16.2 software for the Fused Deposition Modeling (FDM) 3D printer Zortrax M200Plus (Olsztyn, Poland) 3D-printed model of distal radius fracture without carpus was printed from Polylactic Acid (PLA) material. 3D-printed models were personalized, real-size models, printed on a scale of 1:1, and support materials were removed. The 3D-printed models were not sterilized. Prior surgery surgeons could handle and rotate 3-D printed models in all directions for up-close inspection of articular surface dislocation, rotational deformity and test the position of the plate and screws (Figure 2). Preoperative planning using a 3D-printed model.
Setting outcomes
The primary outcomes of this study were a subjective 15-item Patient-Rated Wrist Evaluation (PRWE) self-reporting questionnaire focusing on wrist pain and disability in daily activities 19 and an 11-item Quick Disabilities of the Arm, Shoulder and Hand Score (QuickDash) self-reporting questionnaire for evaluating symptoms and function of the upper extremity. 20 Complications were registered. Major complications (requiring reoperation): loss of reduction, intra-articular screw penetration, implant failure, tendon rupture, and deep wound infection. Minor complications: too-long screw with symptoms, tendon irritation, superficial wound infection, carpal tunnel syndrome (CTS), nerve irritation, traumatic osteoarthritis. The secondary outcomes were: measurement of the difference in range of motions (ROM) of the injured wrist compare to the healthy wrist (flexion, extension, radial and ulnar deviation, supination, and pronation), radiological evaluation (ulnar variance, volar tilt, radial inclination, radial length), the visual analogue scale (VAS), 21 a measurement of the difference in grip strength compared with a healthy wrist. 22
Assessments for both groups were measured at 6 weeks, 3 months, and 6 months intervals during hospital outpatient clinic visits with the researchers.
Data collection and management
General data.

aVLP: volar locking plating.
bDLP: dorsal locking plating.
cFor two participants were applied only dorsal approach with dorsal locking plating.
Sample size
The sample size was calculated using the primary outcome PRWE questionnaire. Based on a previous study, a clinically important difference was set at 11.5 points 24 to detect the difference between the two groups. Effect size was calculated d = 0.869. The total sample size was calculated at n = 56 (n = 28 in each group) will have 90% power to detect a significant difference using a Two-sided independent means t test with a 0.05 significance level (G*Power software, Version 3.1.9.4, Germany). The dropout rate was set to 15% and the total sample size expanded to n = 70 (n = 35 in each group).
Statistical analyses
Statistical analyses were calculated using R software, version 4.0.4. Quantitative variables were tested for normal distribution using the Shapiro-Wilk test and variances were tested using the Two Variances F test. Normally distributed data were calculated using the Independent Samples t test. For data with non-normal distributions, nonparametric statistics Two-Sample Wilcoxon Test was used. Quantitative data were summarized as a mean with standard deviation (SD). Qualitative variables were tested using the Chi-squared test (or Fischer exact test when appropriate) and were summarized as numbers. For complications, data incidence rates (using Fisher exact test) were calculated and summarized as numbers and percentages. Statistical significance was considered p < .05.
Results
Two participants disagreed to participate in the study after the surgery (one in each group) and two patients were lost to follow-up (one in each group). The final sample size consisted of 66 patients (“control group” (n = 33) and “3D model group” (n = 33)) with follow-up points at 6 weeks, 3 and 6 months.
Primary outcomes
PRWE score, QuickDash score, VAS, grip strength.

Registered complications during 6 months follow-up.

aFPL: flexor pollicis longus irritation.
bEDT: extensor digitorum tendons irritation associated with dorsal plating.
CTS: carpal tunnel syndrome.
Secondary outcomes
Radiographic measurements.

Measurement of the difference in ROM.

Discussion
This study used 3D-printed models to facilitate preoperative planning for surgically treating DRF AO/OTA C type. In both groups, radiological and functional outcomes showed acceptable results. In the “Control group” were observed six major complications compared with none of such in the “3D-printed model group”. Retrospectively, there were no specific fracture types associated with major complications.
The study showed that DRF AO/OTA C type has a significantly higher incidence of complication rate up to 16.4% using for preoperative planning conventional radiological evaluation X-rays and CT scans. 7 In our study, in the “Control group” participants’ complications incidence was 30.3% because the majority of cases of DRF were AO/OTA C3 type, but this wasn’t higher compared to the other studies where complication rate was 44% 8 and 52.8%. 9
3D-printed models showed an improved understanding of fracture morphology and articular surface fragments displacement.26,27 We found that the integration of the 3D-printed model in preoperative planning decreased complication incidence significantly – from 30.3% in the “Control group” to 6.1% in the “3D model group”. Furthermore, the evaluation of CT scans alone can lead to confusion by misunderstanding irrelevant “small” or clinically insignificant fragment displacement leading surgeons to plan unrealistic fixation.28,29
We assume that our study presented X-rays and CT scans with carpal bones could provide less information about fracture personality, especially attention to the rotational dislocation of the fragments and implant selection and positioning. With 3D-printed models without carpal bones, surgeons could precisely evaluate the actual view of the articular joint from the inside and choose an appropriate surgical approach. In addition, 2D and 3D CT scans are still presented on a monitor screen and don’t show a true three-dimensional view
Chen et al. 14 in their study didn’t find improvements in the range of motion of the wrist with 3D-printed models. Our study confirmed a better wrist flexion and ulnar deviation at 6 weeks and at 3 months better wrist flexion, extension, ulnar deviation and supination in the “3D-printed model group”, p < .05. At 6 months there was no statistically significant difference between groups. We assume these changes hypothetically may be accrued because major complications had an impact on the early results in the “Control group” participants.
During the 6 months of follow-up, our study shows a tendency for better functional outcomes in the “3D-printed model group” comparing PRWE score, QuickDash score, VAS and grip strength with the “Control group”, but statistically non-significant. The lack of randomized clinical studies prevents comparing our results with other researchers.
3D-printed models may not provide the whole information for the fracture reduction simulation - because there is no soft tissue attachment to the fragments. In addition, dislocation of the fragments which are less than 0.4 mm. Can’t be presented. These disadvantages can affect the evaluation of the fracture and realistic reduction scenarios. In our opinion, the benefit of handling and rotating a 3D-printed model of DRF in all directions without carpus reduces these limitations.
The 3D-printed models were not sterilized and surgeons couldn’t handle them during surgery. Sterilised 3D-printed models could be beneficial in understanding the fracture morphology for surgeons, especially for the less experienced ones. Approximately 3 to 4 h took to create and print the 3D-printed model, furthermore, surgery simulation for the adequate repositioning of the fracture fragments and plate and screw positioning required an additional investment of the surgeon’s time. The time required was not part of this study as it could potentially decrease significantly with surgeon experience with the use of 3D printed models.
In our study, we provide results after 6 months of follow-up - because participants achieve optimize functional results after 3–6 months. 30 Our study shows that the detected difference in range of motion differs after 6 weeks and 3 months of follow-up, however, after 6 months there is no significant difference, p > .05. Nevertheless, participants will be followed until 1-year follow-up.
In this study, we didn’t evaluate X-rays of distal radius fracture after 6 months, because in both groups all distal radius fractures healed at 6 weeks to 3 months period. Further, we didn’t measure the duration of intraoperative fluoroscopy and a postoperative CT scan was not performed to evaluate distal radius fracture.
Conclusion
Our study suggests that using the 3D-printed models for preoperative planning surgically treating distal radius fracture AO/OTA C type can help minimize the complication rate, however, they can’t improve functional outcomes in the short-term results. Further research is required to determine indications for using 3D-printed models in the treatment of distal radius intra-articular fractures.
Footnotes
Acknowledgements
The authors thank Artur Versocki and Simonas Utkus for their technical support in 3D printing.
Author contributions
A.G, A.M concept of the study. A.G, V.U and A.M designed the study. P.M, G.P and T.S contributed to data collection and analysis. T.S, G.P and P.M contributed to the interpretation of data. A.G, V.U contributed to drafting the article. A.M, G.P, and P.M contributed to a critical review of the article. All authors read and approved the final version of the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Vilnius Regional Biomedical Research Ethics Committee (approval No. 2021/01-1299-777).
Informed consent
Written informed consent was obtained from all subjects before the study.
