Abstract
Background
Muscle injury and concomitant bone injury are important drivers to induce heterotopic ossification (HO). However, the related roles of muscle and concomitant bone injury in HO formation are still unclear. This study aims to develop a mouse model through the combination of hindlimb amputation (Am) and cardiotoxin (CTX) injection to investigate the mechanism of HO formation.
Method
The mice were randomly divided into Am group (Am of right hindlimb, n = 12), CTX group (CTX injection in the calf muscle of left hindlimb, n = 12) and Am + CTX group (the combination of Am of right hindlimb and CTX injection of left hindlimb, n = 18). MicroCT was used to evaluate the incidence of HO. Histology was used to investigate the progression of HO.
Results
The MicroCT showed that only Am or CTX injection failed to induce HO while the combination of Am and CTX injection successfully induced HO. The incidence of HO was significant in Am + CTX group on day 7 (0% vs 0% vs 83.3%, p = 0.001) and day 14 (0% vs 0% vs 83.3%, p = 0.048). HO was located on the left hindlimb where CTX was injected. Moreover, the bone volume and bone density on day 14 were higher than those on day 7 in Am + CTX group. Histology revealed the evidence of calcification and expression of osteogenic markers in calcification sites in Am + CTX group.
Conclusion
In summary, the combination of Am and CTX injection could successfully induce dystrophic calcification/HO, which occurs in the location of muscle injury.
Introduction
Heterotopic ossification (HO), the abnormal formation of bone outside skeletal tissues, could be divided into two broad categories: hereditary and acquired HO. 1 Hereditary HO, known as fibrodysplasia ossificans progressiva, resulted from the gene mutation of the bone morphogenetic protein (BMP) type Ⅰ receptor. 2 Acquired HO was precipitated by blast injury, fracture, joint replacement, direct muscle injury, burns or neural trauma and so on.3–6 HO emerged as a prevalent and persistent complication, which could lead to excision procedures, long rehabilitation and significant long-term pain. 7 However, the effective prevention and treatment strategies for HO still remained limited.
The exact etiology of HO was still poorly understood. More and more studies confirmed that muscle injury was the essential cause to induce HO.8,9 Muscle injury could cause inflammatory response, increase osteogenic potential of muscle progenitors, and provide suitable tissue environment for HO formation.10,11 However, single muscle injury did not seem sufficient to drive HO and other extra factors were necessary, such as concomitant bone injury, injection of BMP-2.9,12 A persuasive evidence was that the incidence of HO after blast injury then amputation (Am) was more than 65% while normal blunt Am or blast overpressure alone failed to result in HO.5,13 Reasonable hypothesis was that HO formation following concomitant bone injury required additional muscle injury and muscle injury would induce HO following concomitant bone injury.
Cardiotoxin (CTX), belonged to the family of snake venom polypeptide toxins that resulted in depolarization and contraction of muscle fibers, could contribute to muscle death through myocyte death. 14 CTX injection is a common method to induce muscle injury and subsequent HO. As reported, only CTX injection could not drive HO formation.9,12 This study aim to create a HO mouse model through the combination CTX injection and hindlimb Am. As an effective vehicle, the HO model can be used to study pathogenesis and evaluate novel treatments. We hypothesize that the combination CTX injection and Am could successfully induce HO.
Materials and methods
The induction of HO
The animal study protocol was approved by Animal Care and Use Committee of our institution (Protocol No. 18,032,493). A total of 42 mice (8–10 weeks old, male C57BL/6J mice), purchased from Jackson laboratory, were randomly divided into three groups: Am group (Am of right hindlimb, n = 12), CTX group (CTX injection in the calf muscle of left hindlimb, n = 12) and Am + CTX group (the combination of Am and CTX injection, first Am of right hindlimb, subsequently CTX injection in the calf muscle of left hindlimb, n = 18). For the mice in Am group, following adequate anesthesia with isoflurane, the right hindlimb was truncated below the knee joint then sutured with silk thread. All mice were fed on standard chow diet. The CTX (Calbiochem, San Diego, CA, USA) was injected with 50 μL of 10 μM. The study was conducted under the protocol approved by the Institutional Animal Care and Use Committee (Protocol No. 19,055,138). The weeks of age was same. The body weight (16.23 ± 0.55 g, 16.09 ± 0.59 g, 16.21 ± 0.56 g) and nutritional status was comparable among the three groups. The arrangement were shown on flow chart (Figure 1). The flow chart for experimental arrangement.
Micro–computed tomography (MicroCT) assay
On day 7 and 14 after operation, all the mice were anaesthetized with isoflurane (1%–3%) or sacrificed for MicroCT (vivaCT 40; Scanco Medical, Bruttisellen, Switzerland) to detect HO formation. The MicroCT was acquired at 45 kVp, 88 mA, 300 ms integration time and at an isotropic voxel size of 35 mm. HO was segmented from soft tissue using a threshold of 212/1000. Bone mineral density was calculated as mg hydroxyapatite (HA)/volume (cubic centimetre, ccm).
Muscle tissue sample collection
The left calf and the muscle of right hindlimb were collected on day 3, 7 and 14 after operation. The samples were fixed in methanol for 24 h, then processed in 100% alcohol for 2 h, cleared in xylene 2 h and embedded in paraffin. 8 μm sections were cut, collected and rehydrated using a Leica microtome (Model RM 2255). The slides were incubated at 60°C for 1–2 h before staining.
Histology
Hematoxylin and Eosin (H&E) staining was performed according to routine Harris H&E protocol. For alizarin red staining, slides were stained with alizarin red solution (Alfa Aesar, Haverhill, MA, USA) for 45 s. The extent of calcification was evaluated by the ratio (%) of the calcium staining area to the total area, which could be calculated with the Image J version 1.5.0 software (National Institutes of Health). There were totally four region of interest every group and blinding was not performed.
For Immunofluorescence (IF) staining, sections were blocked in 10% bovine serum albumin for 30 min and then incubated overnight at 4°C with mixed primary antibodies against ALP (ab83250, Abcam), OSX (ab22552, Abcam) and BMP-7 (ab56023, Abcam). Sections were incubated with fluorescein isothiocyanate-conjugated secondary antibody (ab150077, Abcam) for 1 h, then mounted with 4′, 6-diamidino-2-phenylindole (DAPI) on second day. The images were taken by an Olympus CKX41 microscope with a Leica DFC 3200 camera.
Statistical analyses
Statistical analyses were performed using GraphPad Prism 7.0. Independent t-test and variance analysis were used to analyze continuous variables. Fisher exact test was used for categorical variable. p < 0.05 was considered statistical significance.
Results
Result of radiology
The incidence of HO on day 7.

Am: amputation; CTX: cardiotoxin.
Variables were compared using Fisher exact test.
Significant differecne
The incidence of HO on day 14.

Am: amputation; CTX: cardiotoxin.
Variables were compared using Fisher exact test.
Significant differecne
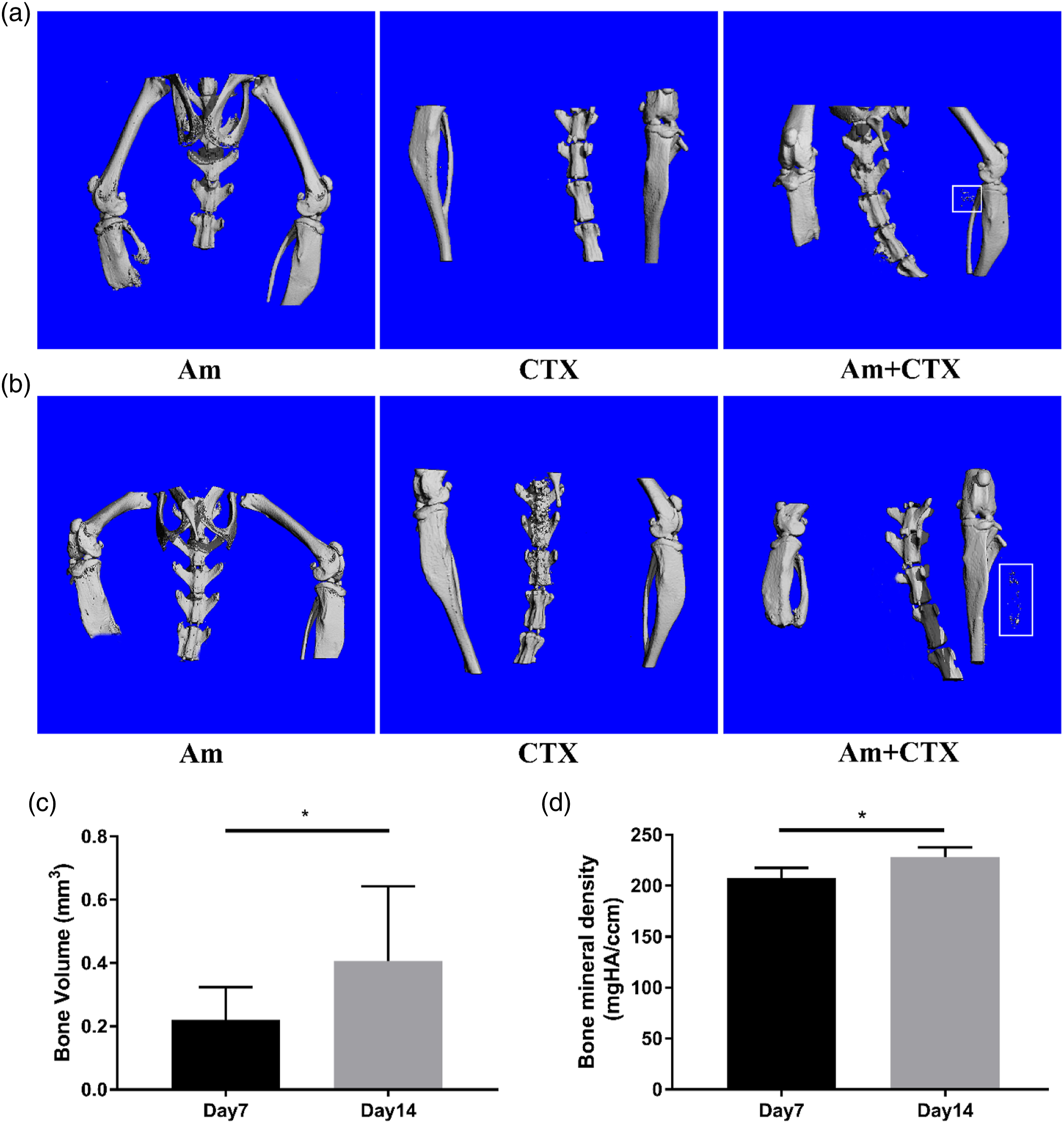
Combination of Am and CTX injection could induce HO compared with only Am or CTX injection. (a) The MicroCT imaging and analysis among the three groups on day 7.
Histology and immunostaining
Histology revealed severe cell infiltration, proliferation and muscle degradation in the muscle tissue of CTX and Am + CTX group on day3. Then, the evidence of early mineralization appeared in the injured muscle tissue in Am + CTX group while the injured muscle restored gradually in CTX group on day7. Moreover, extensive calcification deposition appeared in Am + CTX group while the injured muscle became normal in CTX group on day 14. In addition, there was no evidence of dystrophic calcification (DC)/HO in Am group in different time points (Figure 3). Alizarin red staining was positive on day 7, then enhanced on day 14 in Am + CTX group (Figure 3). The quantitative analysis of alizarin red was 2.724 ± 1.539% on day3, 13.503 ± 1.738% on day 7 and 20.838 ± 2.186% on day 14, the difference is statistically significant among different time points (P < 0.001) (Figure 3). No mature bone tissue was found on day 7 or 14 among the three groups. The combination of Am and CTX injection could induce HO compared with only Am or CTX injection-Histology. H&E staining illustrating the time course of HO progression on day 3, 7 and 14 among the three groups. Alizarin red staining showing the osteogenesis in Am + CTX group. Scale bar = 200 μm.
IF staining demonstrated the evidence of osteogenesis in Am + CTX group, which showed the expression of osteogenic markers (ALP, OSX and BMP7) in the location of muscle injury on day 7 and 14 (Figures 4–6). Immunofluorescent (IF) staining of ALP in muscle tissue harvested on day 3, 7 and 14 in Am + CTX group. Blue: nuclei, green: targeted protein. Scale bar = 50 μm. Immunofluorescent (IF) staining of OSX in muscle tissue harvested on day 3, 7 and 14 in Am + CTX group. Blue: nuclei, green: targeted protein. Scale bar = 50 μm. Immunofluorescent (IF) staining of BMP7 in muscle tissue harvested on day 3, 7 and 14 in Am + CTX group. Blue: nuclei, green: targeted protein. Scale bar = 50 μm.


Discussion
Common animal models of HO included blast injury model, nerve injury model, tenotomy and burn injury model.5,15,16 In this study, we develop a HO mouse model through the combination of Am and CTX injection. The results show that Am + CTX could successfully result in the formation of HO over Am and CTX alone.
The researchers from Pellegrini’s lab established the HO rat model following blast Am procedure, and found that HO demonstrated early fibroproliferative signature.17,18 In addition, the study from Forsberg and Potter’s lab confirmed that the serum from HO patients could activate mitogen activated protein kinase signaling in adipose stem cells. 19 The authors from Levi and Davis’s Lab found that BMP signaling made great contribution to osteogenic differentiation as well as ectopic bone formation and blocking BMP signaling may be an effective prophylactic strategy for HO following blast-related lower extremity trauma.20,21 Above researches mainly focused on the HO following blast related trauma and investigated possible mechanism based on blast injury model. We know that muscle injury and nerve damage are also important drivers for HO except for blast injury. However, there are few researches about other types of HO formation. The establishment of relevant animal model is essential. This study could lay the foundation to study the pathogenesis of HO following muscle injury and concomitant bone injury.
It was worth noting that muscle injury may be the key driving force in HO formation, especially caused by blast injury and orthopaedic surgery. Potter et al. found that the final Am level within the zone of injury was a risk factor for both the development and the grade of HO following trauma and combat-related Am. The more injured muscle was preserved, the higher the incidence of HO was. 22 Anthonissen et al. indicated that significant higher amount of HO was observed in animals that underwent additional muscle trauma in hip surgery. 8 In addition, another study showed that lower rate of clinically significant HO was observed in direct anterior approach (DAA) total hip arthroplasty (THA) than that in posterior approach THA, because DAA approach resulted in less muscle injury than posterior approach. 23 This study found that HO occurred in left hindlimb where CTX was injected, showing muscle injury was a possible inductive reason of HO. However, only muscle injury was not sufficient to induce HO, and concomitant bone injury, the expression of BMPs or cortisol stress response and so on were requisite.9,24 The possible reason was that hindlimb Am (concomitant bone injury) could increase the secretion of related cytokines, providing appropriate microenvironment for HO formation.
It was reported that HO formation required osteogenic cells, microenvironment and related signals. 25 Firstly, muscle injury led to inflammation, which appeared to be the trigger of HO. The level of inflammatory cytokines in HO patients were higher than that in traumatic patients without HO and normal patients.26,27 Especially, as an inflammatory cell, macrophage was thought to be a key regulator of HO. Sorkin et al. investigated the inflammatory response after muscle injury in HO models and identified the cell types at the early stages of inflammation. They found that TGFβ1 expression in macrophage derived HO formation, altering the monocyte/macrophage phenotype could decrease chondrogenic and osteogenic markers, then suppress HO formation. 28 The studies conducted by other authors also supported this conclusion, that was to say, inhibiting or depleting macrophage could contribute to DC/HO formation.12,29 The last but not the least, antiinflammatory was often used as a treatment method for HO, and the application of anti-inflammatory drug would reduce the risk of HO.11,30 Secondly, muscle injury could activate the osteogenic potential of progenitor cells in muscle tissue. Molligan et al. found that the combination of muscle injury and osteotomy of greater trochanter could promote the expression of muscle progenitor’s osteogenic marker. 10 What was more, a new study showed that muscle injury could lead to the accumulation of fibro-adipogenic progenitors in tissue and altering inflammatory environment could activate inherent osteogenic potential of these cells, leading to the formation of HO. 31 Therefore, muscle injury could be essential trigger of HO, however the exact cellular sources as well as the underlying mechanisms were still limitedly understood. This study develops a novel HO animal model, laying the foundation to evaluate the role of muscle injury and concomitant bone injury in HO pathogenesis.
This study showed a possible early pathological progress of HO, including muscle degradation, inflammatory cells infiltration and proliferation, early mineralization and further calcium deposition. The results indicated that the bone volume and density increased gradually from day 7 to day 14. However, the bone density in this study was lower than that of normal bone, 32 showing that the HO in this study was possibly just calcification, not mature HO.
The huge limitation of the study is that only DC is detected, no mature bone tissue. DC is also a pathological stage of HO. Moore et al. found that DC was sufficient to drive HO when macrophage-mediated regression was inhibited. 12 In addition, this study establishes related animal model, but the mechanisms behind need further study. Besides, the animal numbers are small, and a series of critical control arms are missing to draw meaningful conclusions. The follow-up time is only 2–4 weeks. Higher quality researches with larger sample and longer follow-up time are necessary.
In summary, the study successfully establishes a DC/HO mouse model by combining Am and CTX injection. The DC/HO is present in the location of muscle injury. Future studies are necessary using this model to analyze early cellular and molecular pathways, and evaluate new prophylaxis as well as treatment strategies.
Footnotes
Acknowledegment
The authors thank Dr Hongshuai Li (University of Pittsburgh) for assistance with the microCT analyses, Alyssa Falcione (University of Pittsburgh) for advice regarding animal medication treatments and Dr Peter Mittwede (University of Pittsburgh) for manuscript editing.
Author contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Guorui Cao, Fuxing Pei, Rocky Sung Chi Tuan and Hang Lin. Operations were performed by Guorui Cao, La Li, Peter G. Alexander and Shiqi Xiang. The first draft of the manuscript was written by Guoyuan Li and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the from AOTrauma North America and the National Natural Science Foundation of China (82104896).
Ethical approval
This (animal) study was approved by the University of Pittsburgh Institutional Animal Care and Use Committee (IACUC; protocol 18,022,493).
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
