Abstract
Objective
Osteoarthritis (OA) is characterized by synovial cartilage degeneration and is the leading cause of disability and pain worldwide. This study sought to investigate the expression of integrin beta-2 (ITGB2) in synovial fluid of OA patients and its clinical significance.
Methods
A total of 110 OA patients were enrolled, who were classified into grade I (N = 35), II (N = 42), and III (N = 33) according to the Kellgren-Lawrence classification, with 110 healthy subjects as controls, and their clinical data were compared. ITGB2 level was detected by RT-qPCR. The receiver operating characteristic curve was used to analyze the predictive value of ITGB2 on OA occurrence. The correlation between ITGB2 and bone metabolism indexes procollagen type I N-terminal peptide (PINP), bone glaprotein (BGP), bone alkaline phosphatase (BALP), and β-collagen I telopeptide (β-CTX) was analyzed by the Pearson method. Logistic regression model was performed to analyze the influencing factors of OA.
Results
The content of red blood cells, white blood cells, PINP, BGP, and BALP was lowered in OA patients, while β-CTX was elevated. ITGB2 was highly-expressed in OA patients, negatively-correlated with PINP, BGP, and BALP, but positively-correlated with β-CTX. ITGB2 level increased with the elevation of OA grade. The ITGB2 level >1.375 had certain diagnostic values for OA. ITGB2 level is related to OA severity and may be a biomarker for OA classification. ITGB2 was an independent risk factor for OA.
Conclusion
High expression of ITGB2 in synovial fluid can assist OA diagnosis and may be a biomarker for OA grade.
Keywords
Introduction
Osteoarthritis (OA) is the most common degenerative joint disease in adults worldwide, characterized by chronic progressive destruction of articular cartilage and abnormal bone remodeling, resulting in severe pain and disability.1,2 However, the underlying pathogenesis of OA is unknown, and there is no approved treatment to delay its progression.3,4 At present, the gold standard for the clinical diagnosis of OA is X-ray combined with clinical symptoms, but X-ray is not sensitive to the early diagnosis of OA and is easy to lead to missed diagnosis. 5 In addition, molecular biology studies have identified numerous biomarkers and biological processes that contribute to the diagnosis of OA, including integrin ligands.6,7 Accordingly, the search for biomarkers associated with OA progression may have implications for the development of successful treatments for OA in the future.
Synovial fluid is a mixture of hyaluronidase protein complex produced by synovium and leakage liquid of plasma. The synovial fluid of arthritic joints contains the mediators and by-products of inflammation and degradation processes that occur in the affected joints. Hollander et al. published the conventional use of synovial fluid analysis as an auxiliary means of arthritis disease diagnosis and introduced the term “synovial analysis”. 8 At present, the analysis of cytokines,9-11 matrix metalloproteinase (MMP) and other proteases, 12 exogenous and endogenous crystalline substances,13,14 miRNA,15-17 microbubbles and exosomes18,19 in synovial fluid has been reported as one of the important auxiliary diagnostic tools for OA, which provides valuable information for the staging and prognosis of joint abnormalities. Identifying the potential biomarkers in synovial fluid will help further clarify the disease mechanism, and make it possible for patients with knee OA to make early diagnosis, treatment intervention, and more effective clinical treatment monitoring.
Integrin dysfunction has been identified in the pathogenesis of OA. 3 The unique composition and structure of the chondrocyte extracellular matrix (ECM) has been reported to play an important role in joint function, enabling the long-term load-bearing capacity of joints. 20 OA can alter the ECM, leading to increased catabolic activity and inflammatory changes in the mechanical function of the ECM. 21 In the process of OA, abnormal expression of integrin alters ECM signal transduction and changes chondrocyte synthesis, bringing about an imbalance between destructive cytokines and regulatory factors, thus inducing tissue damage. 22 Integrins are a class of heterodimer transmembrane proteins containing α and β subunits, which play essential roles in various physiological functions such as cell adhesion, motility, growth, differentiation, and mechanical signal transduction. 20 Integrin Beta-2 (ITGB2), one of the integrin subunits, is a leukocyte specifically expressed heterodimer surface receptor. 23 Previously, it was disclosed to be expressed only in leukocytes and can act as a cell-connecting skeleton to promote leukocyte adhesion to endothelial cells, thereby participating in intracellular signal transduction. 24 ITGB2 expression is related to a variety of blood cells and may be an important regulator of immune cell infiltration and a therapeutic target for hematological malignancies .25,26 For example, Zhang et al. observed that ITGB2 was visibly overexpressed in cancer-related fibroblasts, and the higher the expression, the more advanced the state of oral squamous cell carcinoma. 23 Among hematological malignancies, ITGB2 is associated with chronic lymphocytic leukemia and myeloma. 26 Recently, ITGB2 has been reported to participate in the progression of OA, and ITGB2 expression is elevated in OA.6,27 In addition, the gene ITGB2 involved in osteoclast function has gender differences in OA bone, and the change of its expression can cause the change of bone remodeled. 28 Nevertheless, the expression of ITGB2 in OA synovial fluid and whether ITGB2 can be acted as a diagnostic marker of OA remains unclear. Moreover, there are few reports about ITGB2 in OA at present. Consequently, the purpose of this study was to analyze the expression, clinical significance, and role of ITGB2 in OA, which helped to further clarify the mechanism of the disease, and provided new clinical information for the continuous study of the biomarkers in synovial fluid in the diagnosis, staging, and treatment of knee OA.
Materials and methods
Ethics statement
This study was approved by the Ethics Committee of Hangzhou Fuyang District Bone Injury Hospital of Traditional Chinese Medicine, and each subject signed the informed consent.
Study subjects and grouping
A total of 110 patients with knee OA admitted to Hangzhou Fuyang District Bone Injury Hospital of Traditional Chinese Medicine from June 2019 to June 2021 were selected, and they were in line with the classification standard for OA of the American College of Rheumatology in 1986. Inclusion criteria were as follows: (1) the duration of knee pain in the 1 month prior to the visit ≥14 days; (2) there was bone abrasion sound during knee joint movement; (3) X-ray showed bone spur hyperplasia or osteophyte formation at the edge of knee joint end; (4) bone swelling of knee joint; (5) the time of knee joint stiffness in the morning ≤30 min; (6) age ≥40 years old. Knee OA can be diagnosed if the patient’s conditions met the criteria (1) and (3) or (1), (2), (4), (5), and (6) above. Kellgren-lawrence (K-L) grading was used to evaluate the clinical conditions of knee OA, including 35 cases of grade I, 42 cases of grade II, and 33 cases of grade III. Exclusion criteria were as follows: (1) with infectious arthritis, knee trauma, surgical history, and diabetes; (2) with autoimmune diseases such as rheumatoid arthritis; (3) used glucocorticoids or non-steroidal anti-inflammatory drugs 2 weeks prior to examination; (4) with other joint diseases such as reactive arthritis. Another 110 healthy subjects were selected as controls. Another 110 patients with ligament or meniscus disease and without cartilage damage were selected as the control group. The relevant clinical indicators of each participant were collected, including gender, age, body mass index (BMI), K-L classification, red blood cells (RBC), white blood cells (WBC), hemoglobin (HB), procollagen type I N-terminal peptide (PINP), β-collagen I telopeptide (β-CTX), bone glaprotein (BGP), and bone alkaline phosphatase (BALP).
Collection and preservation of joint fluid
The subjects were kept in a horizontal position. After local disinfection and anesthesia of the knee joint, the knee joint cavity was punctured through the lateral suprapatellar approach, and 2 mL joint fluid was extracted and left standing at room temperature for 1 h. Then the cells and residual fragments of joint tissue were removed by centrifugation at 3000 r/min for 20 min. The supernatant was collected and placed in a new 1.5 mL microcentrifuge tube and stored in a refrigerator at −80°C for subsequent detection.
RBC and WBC in synovial fluid were measured using ADVIA 120 hematology system (Siemens Healthineers, Erlingen, Germany). The PINP (ML038554), β-CTX (ML038555), BGP (ML058528), and BALP (ML058537) in synovial fluid were determined by enzyme-linked immunosorbent assay (ELISA) kits (Enzyme-linked Biotechnology Co., Ltd, Shanghai, China).
Reverse transcription quantitative polymerase chain reaction (RT-qPCR)
Primer sequences of RT-qPCR.
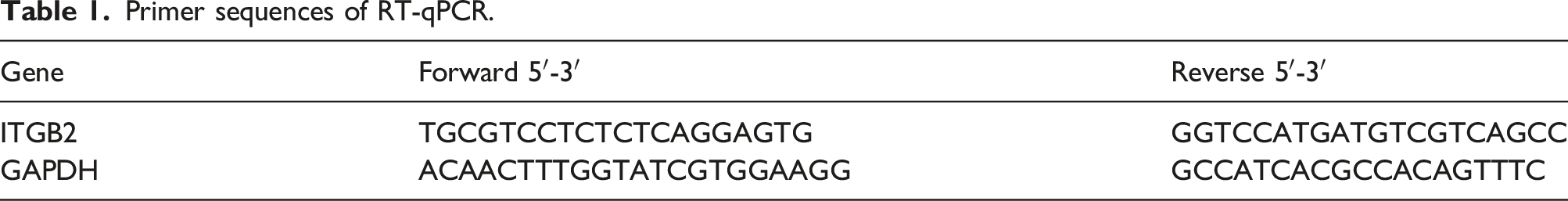
Statistical analysis
Statistical software SPSS 21.0 (IBM Corp. Armonk, NY, USA) and GraphPad Prism 8.01 (GraphPad Software Inc., San Diego, CA, USA) were used for statistical analyses and plotting. Gpower was used to pre-estimate the sample size. The Shapiro-Wilk test was used for the normal distribution test. Data were expressed as mean ± standard deviation. The t test was used for data comparison between two groups, one-way analysis of variance (ANOVA) test was used for data comparison among multiple groups, and Tukey’s test was used for the post hoc test. Receiver operating characteristic (ROC) curve was used to analyze the predictive value of ITGB2 level in synovial fluid on OA. Logistic regression model was used to analyze the independent correlation between ITGB2 and OA. Differences were considered statistically significant at p < 0.05.
Results
Comparative analysis of clinical data between patients with OA and healthy subjects
Comparison of clinical baseline characteristics of OA patients.

Notes: OA (Osteoarthritis); BMI (Body Mass Index); RBC (Red blood cell), WBC (White blood cell), PINP (Procollagen type I N-terminal peptide), β-CTX (β-collagen I telopeptide), BGP (Bone glaprotein), BALP (Bone alkaline phosphatase).
ITGB2 was highly expressed in the joint fluid of OA patients
ITGB2 levels in the synovial fluid of healthy subjects and OA patients were detected by RT-qPCR, which displayed higher levels of ITGB2 in OA patients (1.97 ± 0.72) than that in the controls (1.09 ± 0.47) (p < 0.001, Figure 1). ITGB2 was highly-expressed in the synovial fluid of OA patients. RT-qPCR was used to detect the level of ITGB2 in synovial fluid. The t test was used for data analysis. ***p < 0.001.
High expression of ITGB2 can assist in the diagnosis of OA
To further explore the diagnostic value of ITGB2 level for OA, the ROC curve was drawn. The results illustrated that the area under the curve (AUC) for distinguishing OA patients and healthy subjects was 0.8529 and the cut-off value was 1.375, with a sensitivity of 77.27% and a specificity of 80% (Figure 2), indicating the ITGB2 level >1.375 has certain auxiliary diagnostic values for OA. ITGB2 high expression can assist in the diagnosis of OA. ROC curve was used to analyze the diagnostic efficacy of ITGB2 mRNA level in OA.
Correlation analysis between ITGB2 and bone metabolism in OA patients
To further ascertain whether ITGB2 is associated with bone metabolism indicators PINP, β-CTX, BGP, and BALP, we conducted correlation analysis using the Pearson method. It was discovered that the level of ITGB2 in synovial fluid of OA patients was negatively correlated with PINP, BGP, and BALP, whereas positively correlated with β-CTX (all p < 0.05, Figure 3). Correlation analysis between ITGB2 and bone metabolism indexes in OA patients. A–D: Pearson was adopted to analyze the correlation between ITGB2 and PINP, β-CTX, BGP, and BALP in synovial fluid of OA patients.
ITGB2 is a biomarker for OA severity classification
To further explore whether ITGB2 is related to the grade of OA, OA patients were allocated into the grade I group (N = 35), grade II group (N = 42), and grade III group (N = 33) according to the K-L classification. Subsequently, we compared theITGB2 level in the synovial fluid of each group, followed by ROC curve drawing. RT-qPCR showed that patients in grade I, II, and III groups were found to have higher ITGB2 levels than those in the control group, and the level of ITGB2 increased with the elevation of grading degree (p < 0.001, Figure 4(a)). The ROC curve of ITGB2 level for distinguishing grade I and grade II showed that the AUC was 0.7602 and the cut-off value was 1.885, with a sensitivity of 61.9% and specificity of 97.14% (p < 0.0001, Figure 4(b)). The AUC of ITGB2 level for identifying grade II and grade III was 0.8492 and the cut-off point was 2.670, with a sensitivity of 66.67% and a specificity of 95.24% (p < 0.0001, Figure 4(c)). The above results indicated that ITGB2 amount was correlated with the grade of OA and ITGB2 might be a biomarker for OA grade. ROC curve analysis of ITGB2 in OA grading. (a): The level of ITGB2 in synovial fluid of OA patients was detected by RT-qPCR; (b): ROC curve of ITGB2 level for diagnosing grade I and II of OA in synovial fluid; (c): ROC curve of ITGB2 level in diagnosing grade II and III of OA in synovial fluid. Figure (a) was analyzed by one-way ANOVA, Figure (b) and Figure (c) were analyzed by ROC. ***p < 0.001.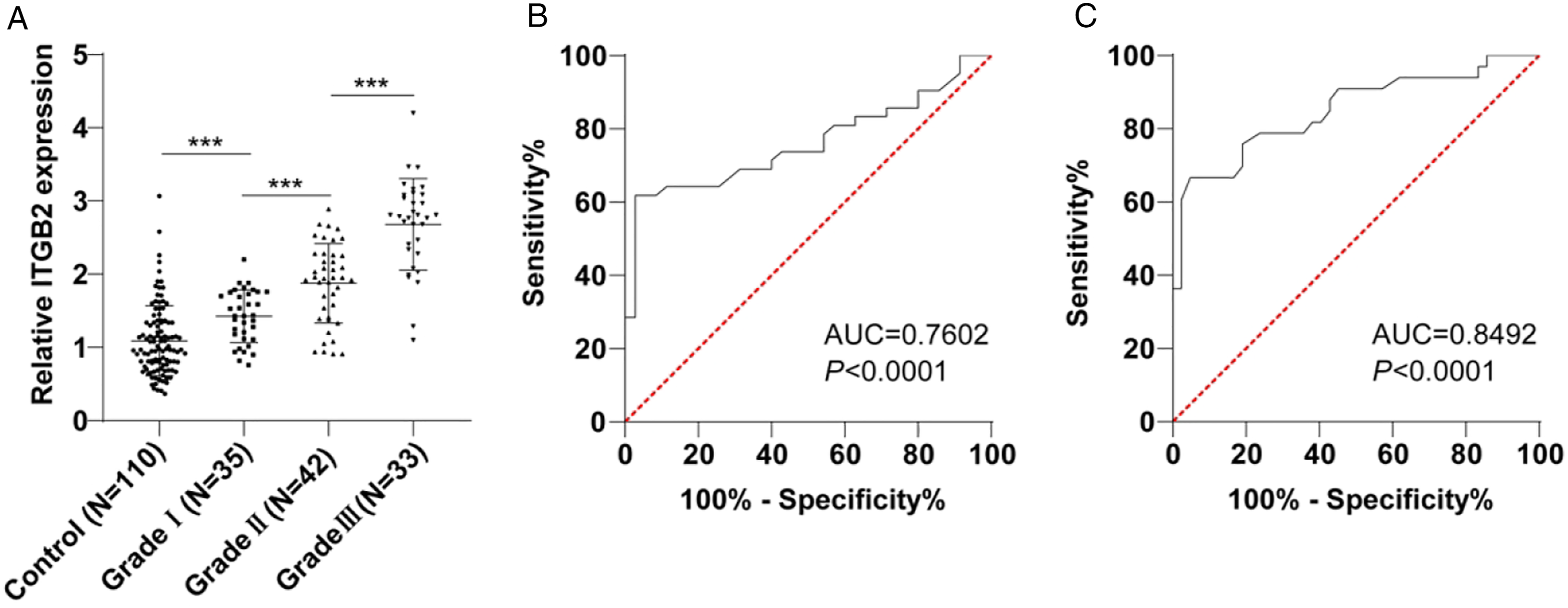
ITGB2 was an independent risk factor for OA
Multivariate logistic regression analysis.

Notes: RBC (Red blood cell), WBC (White blood cell), PINP (Procollagen type I N-terminal peptide), β-CTX (β-collagen I telopeptide), BGP (Bone glaprotein), BALP (Bone alkaline phosphatase).
Discussion
OA is the most common chronic joint disease and its incidence rises with age, predominantly affecting most people over 65 years of age. 4 As the precise molecular mechanism involved in the pathogenesis of OA is still unclear, there is no effective intervention to slow the progression of OA or delay the irreversible degradation of cartilage except total joint replacement. 29 Integrin, a transmembrane receptor that mediates the connection between cells and their external environment (e.g., ECM) plays an important role in OA by coordinating the behavior of chondrocytes. 30 Our finding demonstrated that the high expression of ITGB2 in synovial fluid can assist the diagnosis of OA and its expression may be a biomarker for the grade of OA.
A total of 110 OA patients were included as study subjects, along with healthy subjects (N = 110) as controls. Chondrodegeneration in OA is related to changes in chondrocyte metabolism, and the degradation of type II collagen can lead to cartilage defects in the knee joint and promote the occurrence and development of OA. 31 RBC and WBC in the joint cavity are routine indicators of OA. 32 Metabolism-induced subchondral bone injury, together with bone metabolic markers, is considered to be an important indicator of OA. 33 In our current study, RBC, WBC, PINP, BGP, and BALP of OA patients were reduced, while β-CTX was visibly enhanced compared with healthy controls. Consistent with our findings, it was reported that the bone metabolism indicators PINP, BGP, and BALP decreased in OA patients, while β-CTX increased. 34 The above evidence and findings suggested bone metabolism indicators might potential markers of OA diagnosis.
Under normal conditions, most components in the synovial fluid are lower than those in the blood, but under pathological conditions, many key components with positive predictive values change dramatically. Therefore, the diagnostic value of synovial fluid is no less than that of blood. If interference factors are considered, the diagnostic value of synovial fluid may even be higher than that of blood. Synovial fluid is less interfered with by the external and other factors of the body and can reflect the changes of the microenvironment in the joint, so it is most closely related to the nature and extent of joint diseases. Therefore, the diagnostic value of synovial fluid in joint-related diseases deserves people’s attention. Identifying the potential biomarkers in synovial fluid will help further clarify the disease mechanism, and make it possible for patients with knee OA to make an early diagnosis, treatment intervention, and more effective clinical treatment monitoring. Integrins have been reported to differ in tissue distribution and ligand identification, and their dysfunction has been reported to be associated with OA.20,35 Moreover, a series of experiments have identified that various integrins are abnormally expressed in cartilage tissue, thus affecting OA. 34 For instance, animal experiments showed that the amounts of α4, α5, and α2 integrins were up-regulated, and the content of proteoglycan and fibronectin was changed in cartilage tissue. 36 High levels of α1β1 and α3β1 were detected in OA cartilage tissue, which may help regulate ECM deformation and promote chondrocyte hypertrophy. 37 Besides, Yu Fan et al. revealed in their work that ITGB2 expression was elevated in OA, which may be closely related to the pathogenesis of OA. 27 As expected, our study also uncovered that ITGB2 was highly-expressed in the synovial fluid of OA patients.
Activated cell-specific expression of integrin α5β1 has been reported to contribute to the early detection of OA. 38 To our knowledge, the diagnostic value of ITGB2 level in OA has not been reported to date. Herein, our present study noted that the AUC of ITGB2 level for diagnosing OA was 0.8529 (cut-off value 1.375, sensitivity 77.27%, specificity 80%). These data supported that the ITGB2 level >1.375 has identification values for OA. More importantly, the negative correlation between ITGB2 level and PINP, BGP, BALP, and the positive correlation between ITGB2 level and β-CTX were observed, suggesting that ITGB2 level is closely related to bone metabolism in OA patients. Consistent with our findings, integrin β1 regulates bone formation by influencing osteoblast differentiation by binding to the BK α subunit of osteoblasts. 39 A subset of genes (including ITGB2) involved in osteoclast function was differentially expressed in OA bone between females and males and their altered expression suggests altered bone remodeling. 28 In addition, we found that ITGB2 was associated with the disease severity of OA patients. ITGB2 level increased with the up-regulation of K-L grading. In addition, ITGB2 had better identification abilities in grades I and II, as well as grades II and III of OA, with AUC of 0.7602 and 0.8492, respectively. ITGB2 may be served as a biomarker to assess the prognosis and drug sensitivity in patients with acute myeloid leukemia. 26 However, as far as we knew, via bioinformatics analysis, ITGB2 has only been identified to be abnormally expressed in OA and linked to the progression of OA, and no article has reported the diagnostic value of integrin ITGB2 for OA.6,27 Accordingly, we concluded for the first time that ITGB2 level was correlated with the grade of OA, and it could be a biomarker for the classification of OA. Subsequently, ITGB2 was illustrated to be an independent risk factor for OA after adjustment for BALP. As a whole, ITGB2 was independently correlated with OA. These data provided a valuable additional laboratory test for the diagnosis of arthritis. A high level of ITGB2 indicated the presence of WBC in joints, indicating that the inflammatory process was ongoing, which would eventually lead to the destruction of cartilage homeostasis and the final degradation of matrix.
In conclusion, this study measured the expression of ITGB2 in the synovial fluid of OA patients and the correlation between the ITGBA2 level and the diagnosis of OA, helping to further clarify the mechanism of the disease and providing new clinically relevant information for the continuous study of the biomarkers of synovial fluid in the diagnosis, staging, and treatment of knee OA. However, this study still has some limitations: (1) fewer cases and events included; (2) in terms of achieving a diagnosis of OA, besides demonstrating the presence of inflammation in the joint, one needs to be able to differentiate it from its common differentials. This study included a single control group and didn’t include groups such as pre-radiation arthritis, rheumatoid arthritis, septic arthritis, and post-traumatic arthritis; (3) the changes of ITGB2 during OA treatment are not analyzed; (4) the change in integrin levels in the grade of osteoarthritis also may point towards causation or presence of an unfavorable joint environment, which leads to cartilage damage, and this can be further studied to see if there are any potential therapeutic targets which can modify the progression of OA. In addition, with the further excavation of specific components of synovial fluid, related biomarkers (such as microRNAs, DNA, non-coding RNA, microbubble, and exosome), and the diverse joint fluid-related inspection methods (optical analysis, related proteomic analysis, and deep sequencing), synovial fluid examination and analysis provide the possibility for early and accurate diagnosis of OA. However, while we used synovial fluid specimens to develop more targeted detection projects, we still needed to consider several key issues: (1) the biomarkers in synovial fluid are highly sensitive and specific; (2) non-invasive and simple operation; (3) fast and accurate; (4) target all groups or certain specific groups; (5) good repeatability; (6) reasonable price. Only with the above aspects, the relevant detection of synovial fluid can be transferred from the experimental stage to clinical diagnosis and treatment, providing strong evidence for clinicians to diagnose joint diseases early and accurately.
Footnotes
Authors' contributions
All authors contributed to conceptualization, methodology, validation, formal analysis, investigation, resources, data curation, writing, review and editing, and visualization.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
This study was approved by the Ethics Committee of Hangzhou Fuyang District Bone Injury Hospital of Traditional Chinese Medicine, and each subject signed the informed consent.
Availability of data and materials
The data that support the findings of this study are available from the corresponding author upon reasonable request.
