Abstract
Background
Natural polymer scaffolds used to promote rotator cuff healing have limitations in terms of their mechanical and biochemical properties. This animal study aimed to investigate the effects of combined graphene oxide (GO) and alginate scaffold and the toxicity of GO on rotator cuff healing in a rat model.
Methods
First, the mechanical properties of a GO/alginate scaffold and a pure alginate scaffold were compared. The in vitro cytotoxicity of and proliferation of human tenocytes with the GO/alginate scaffold were evaluated by CCK-8 assay. For the in vivo experiment, 20 male rats were randomly divided into two groups (n = 10 each), and supraspinatus repair was performed: group 1 underwent supraspinatus repair alone, and group 2 underwent supraspinatus repair with the GO/alginate scaffold. Biomechanical and histological analyses were performed to evaluate the quality of tendon-to-bone healing 8 weeks after rotator cuff repair.
Results
The GO/alginate scaffold exhibited an increased maximum load (p = .001) and tensile strength (p = .001). In the cytotoxicity test, the cell survival rate with the GO/alginate scaffold was 102.08%. The proliferation rate of human tenocytes was no significant difference between the GO/alginate and alginate groups for 1, 3, 5, and 7 days. Biomechanically, group 2 exhibited a significantly greater ultimate failure load (p < .001), ultimate stress (p < .001), and stiffness (p < .001) than group 1. The histological analysis revealed that the tendon-to-bone interface in group 2 showed more collagen fibers bridging, tendon-to-bone integration, longitudinally oriented collagen fibers, and fibrocartilage formation than in group 1.
Conclusion
A small amount of GO added to alginate improved the mechanical properties of the scaffold without evidence of cytotoxicity. At 8 weeks after rotator cuff repair, the GO/alginate scaffold improved tendon-to-bone healing without causing any signs of toxicity in a rat model.
Introduction
The incidence of rotator cuff tears has increased rapidly, with rotator cuff repair being increasingly performed worldwide.1–3 To increase the success rate of rotator cuff repair surgery, it is important to minimize the formation of interosseous gaps, maintain mechanical stability during loading, and induce tendon-to-bone healing.2,4 Over the years, various strategies have been developed and tested to improve rotator cuff repair techniques and improve the success rate of rotator cuff repair surgery.5,6 Biodegradable scaffolds, such as alginate, chitosan, and collagen, which are inserted together during suturing in animal models, have been reported to improve tendon healing after rotator cuff repair.7,8 However, natural polymers used to enhance rotator cuff healing have advantages in biocompatibility and biodegradability but often may not provide sufficient strength to serve as supports.9–11
Graphene oxide (GO) has been extensively explored as one of the most promising biomaterials in biomedical applications owing to its inherent properties such as its two-dimensional planar structure, wide surface area, chemical and mechanical stability, and excellent conductivity and biocompatibility.7,12,13 In addition, the integration of GO and a polymer confer electrical conductivity to the scaffold and is ideal for cardiac or neural tissue engineering. 14 GO is not only involved in the migration of osteoblasts and mesenchymal stem cells (MSCs) but also has osteoconductivity.15,16 In particular, several studies have demonstrated the efficacy of GO in animal experiments in the field of bone regeneration.17,18
Biodegradable polymeric alginates are naturally extracted polysaccharides that have been successfully used in tissue engineering for many years and are widely used for cell encapsulation and cell transplantation in experimental and clinical studies.19,20 They can be applied for various purposes, including wound healing, drug delivery, and scaffolding, making them an attractive material for tissue engineering.8,21 We previously used an alginate hydrogel scaffold to improve rotator cuff healing in a rat model of acute rotator cuff tears.4,9 However, as alginate is a biodegradable polymer with relatively low strength and structural integrity, 22 it may not provide sufficient support for reconstructive surgery of the rotator cuff. In addition, biodegradation may occur before the healing of the rotator cuff tear is complete.
To overcome these shortcomings and maximize the advantages of alginates, several studies have explored the use of GO- and alginate-conjugated substances for several years. These GO- and alginate-conjugated substances show significantly improved biomechanical properties 23 compared with alginate and have been reported to have excellent antibacterial activity24,25 and promote cell proliferation26–29 and drug delivery.30–32 Moreover, they have been proven to be safe in terms of cytotoxicity in vitro studies.23,33 However, few studies have examined the efficacy of GO/alginate conjugates in vivo or investigated the efficacy and toxicity of GO/alginate conjugates in vivo in the healing of rotator cuff tears. Hence, we hypothesized that GO/alginate conjugates would play a role in augmenting the healing of rotator cuff tears in a rat model.
The purpose of our study was to (1) investigate the effects of a GO/alginate sheet placed at the supraspinatus bone tendon repair site on the healing of rotator cuff tears through both biomechanical and histological evaluations in a rat model and (2) evaluate the toxicity of the GO/alginate sheet when used for healing in vivo.
Materials and methods
Study approval
All animal procedures were approved by the Institutional Animal Care and Use Committee of the author’s organization. (Kyungpook National University Institutional Animal Care and Use Committee, IACUC NO: KNU 2018 -0123)
Experimental groups
The outcomes of biomechanical and histological analyses at 8 weeks were the primary endpoints for the control and experimental groups after rotator cuff surgery. The ‘resource equation’ approach was used to determine the sample size. 34 The minimum and maximum numbers of animals per group were 6 and 11 animals, respectively. Twenty male Sprague Dawley rats (body weight, 300–350 g; 12 weeks old) were randomly divided into two groups: group 1 (repair only, n = 10) and group 2 (GO/alginate group, n = 10). Each group underwent the same surgery on both shoulders. Eight weeks postoperatively, twenty samples were taken from both shoulders of rats in each group. Ten samples from each group were used for biomechanical evaluation, and 10 samples were used for histological evaluation.
Preparation of the GO/alginate scaffold
All experimental tools were sterilized before use. GO is a lightweight powder that is safest to use in a fume hood. Ten milligrams of GO, weighed with an electronic balance, was dispersed in 100 mL of sterilized water, vigorously stirred for a sufficient period, and sonicated for 3 min to obtain a uniform GO suspension. This GO suspension was mixed with 100 mL of an aqueous solution of sodium alginate (2% w/v). The mixture was vigorously stirred at 400–600 rpm for ∼5 h and sonicated for 1 h to ensure good dispersion of GO in the sodium alginate solution.
35
Ten milliliters of this mixture was dispensed in a culture dish and vacuum-dried for 48 h. When the sample was sufficiently dried, it was crosslinked by treatment with 2.2% CaCl2 and rinsed with sterile water. Before the biomechanical analysis and animal experiments, the washed hydrogels were dehydrated until the water was completely removed.
36
A pure alginate sheet (2% w/v) was also produced by the same method. The above procedure yielded a circular GO/alginate sheet with a diameter of 90 mm and a thickness of ∼0.2 mm (Figure 1); its GO content was ∼7.85 × 10−4 mg/mm3. Preparation of GO/alginate scaffold. (a) The weight of the alginate powder and GO powder was measured. Two grams of alginate was dissolved in 100 mL of sterilized water. (b) GO powder (0.01 g) was dispersed in the solution containing dissolved alginate. (c) The GO/alginate suspension was mixed and ultrasonicated to disperse well for 1 h. (d) This well-dispersed GO/alginate suspension (10 mL) was dispensed in a Petri dish and vacuum-dried for 48 h. (e) The sample was crosslinked by treatment with 2.2% CaCl2 and rinsed with sterile water. (f) The sample was dehydrated until the water was removed. GO; graphene oxide.
Mechanical properties of the GO/alginate scaffold
Ten sections with an area of 10 × 50 mm2 were prepared from the alginate and GO/alginate sheets. The average thickness of the ten GO/alginate scaffolds was 0.19 mm, and the standard deviation was 0.058. The minimum and maximum thickness were 0.1 mm and 0.3 mm, respectively. The tensile strength of both sheets was tested using an OTT-003 Universal Test Machine (OTT-03, Oriental TM, Siheung, South Korea) with a 3-N load cell at a tensile speed of 10 mm/min until failure. The ultimate elongation and tensile strength were measured using a stress-strain analysis. 36 We compared the mechanical properties of the GO/alginate sheet with those of the pure alginate sheet.
In vitro cytotoxicity of the GO/alginate scaffold
To evaluate the cytotoxicity of the GO/alginate sheet and its effects on the metabolic activity of human fibroblasts, a Cell Counting Kit-8 assay (CCK-8) (Cat#ab228554, Abcam, Waltham, MA, USA) was performed according to the manufacturer’s instructions. 37 Human tenocytes (Zen-Bio, Durham, NC, USA) were purchased and used for in vitro cell-based experiments.
The GO/alginate sheet and pure alginate sheet were prepared and sterilized with ultraviolet light. The sheets were incubated with 3 mL of tenocyte growth medium by shaking at 37°C for 24 h at 160 rpm. After 1 day, the extracted solution was filtered to remove insoluble sample residues. Human tenocytes were passaged when they reached 80% confluency and were seeded at a density of 5 × 105 cells/well in 96-well plates. After 24 h, the supernatant was replaced with fresh medium or sample extracts. The cells were then further incubated at 37°C and 5% CO2 for 24 h. On the next day, the supernatant was removed, and 10 μL of WST-8 in 100 μL of fresh tenocyte medium were added to each well and incubated for 3 h at 5% CO2 at 37°C. The absorbance at 450 nm was measured using a microplate reader (SpectraMax 190, Molecular Devices, San Jose, CA, USA). Cell viability (%) was expressed as the relative absorbance of the sample relative to that of the control. Three samples from each group were used for the analysis, and the mean values are presented and compared. Based on the results of a previous study, cytotoxicity was not considered significant when 80% cell survival was observed for treated tenocytes compared with the control. 38
In vitro cell proliferation assay of the GO/alginate scaffold
Human tenocytes were passaged when they reached 80% confluency and seeded at a density of 2 × 104 cells/well in 96-well plates. After 12 h, the supernatant was replaced with a medium mixed with the GO/alginate suspension. Three samples from each group were then incubated for 1, 3, 5, and 7 days under conditions of 37°C and 5% CO2. Tenocyte proliferation was evaluated with a CCK-8 at each time point.
Animal model and surgical procedure
Twenty male Sprague Dawley rats (weight, 300–350 g; 12 weeks old) were divided into 2 groups. Ten rats were randomly assigned to each group: group 1 (repair only, n = 10) and group 2 (GO/alginate group, n = 10). The rats were anesthetized with an intramuscular injection of zolazepam (15 mg/kg, Zoletil, Virbac S.A., Carroscedex, France) and xylazine hydrochloride (5 mg/kg, Rompun, Bayer HealthCare, Leverkusen, Germany). The shoulders of the rats were shaved and sterilized with iodophor. A 3-cm longitudinal incision was made along the scapular spine. The deltoid muscle was split, and the supraspinatus tendon was exposed at the greater tuberosity. The supraspinatus tendon was isolated and transected at the end of the tendon insertion site. Decortication of the footprint was performed with a burr. Two bone tunnels were made using a drill at the greater tuberosity of the humeral head. The supraspinatus tendon was repaired with a single row through the bone tunnel with 3.0 Ethibond. Group 1 (repair only, n = 10) was subjected exclusively to repair. In group 2 (the GO/alginate group, n = 10), the GO/alginate scaffold (5 × 5 mm2) was placed on the detached tendon, which was repaired simultaneously (Figure 2). Rats in both groups were sacrificed at 8 weeks after surgery. Ten samples from each group were used for biomechanical evaluation, and 10 samples were used for histological evaluation. Rats that died or had infections at the surgical site were excluded from the experiment. Animal model and surgical procedure: (a) The deltoid muscle was split, and the supraspinatus tendon (yellow arrow) was exposed. (b) The supraspinatus tendon (yellow arrow) was isolated and transected at the end of the tendon insertion site. (c) Two bone tunnels were made at the greater tuberosity of the humeral head, and a single row repair was performed. (d) Group 1 received repair alone. In group 2, the graphene oxide /alginate scaffold (5 × 5 mm2) was placed on the detached supraspinatus tendon to simultaneously promote the repair of the tendon-to-bone interface.
Biomechanical evaluation
All rats survived after 8 weeks, and there was no evidence of infection at the surgical site. Each rat was euthanized with CO2 gas inhalation. The humeral head and proximal humerus-attached supraspinatus tendons were separated from both shoulders of each rat. Before performing the biomechanical evaluation, the surrounding soft tissues and suture materials from the tendon-to-bone interface were removed as completely as possible. The cross-sectional area of the supraspinatus tendon was measured at the mid-portion of the tendon using a Digimatic Micrometer (MDC25SB, Mitutoyo, Japan), and the width was measured via optical methods.
39
We then evaluated the mode of tear (insertional tear or midsubstance tear) and the load to failure (maximum load that can withstand external forces until failure) at a rate of 10 mm/s with preloading of 3 N using a custom fixture clamping system and a universal test machine (OTT-03, Oriental TM, Siheung, South Korea). We designed and manufactured a custom fixture clamping system for tensile testing of the supraspinatus tendon that consisted of 2 separate fixtures: a humeral head fixation unit, which rigidly fixed the humeral head and permitted the supraspinatus tendon and muscle attached to the humeral head to come out through a hole, and a cryogenic tendon fixation unit, which secured the myotendinous junction of the tendon with a sandpaper-attached clamp and liquid nitrogen to prevent slippage. The supraspinatus tendon was fixed to this system along its anatomic direction to allow tensile loading and to allow the tendon-to-bone interface to form a right angle. The tensile load-to-failure data were automatically collected with a personal computer-based data acquisition system (Figure 3). We distinguished the following modes of failure: insertional tear at the site of the tendon-to-bone interface and midsubstance tendon tear (intratendinous portion, musculotendinous junction) at the part excluding the tendon-to-bone interface. Biomechanical analysis: (a) For accurate measurement, the sample was cleaned leaving only the tendon on the bone. (b) Measurement of the cross-sectional area of the supraspinatus tendon. (c) Tensile strength measurement.
Histological evaluation
All specimens from each group were subjected to histological analysis. The tendon-to-bone samples were placed in 10 mL of sterile 10% formalin in a universal container for transportation to the pathology department. Once fixed in buffered 10% formalin, the pieces were dehydrated, embedded in paraffin, and cut into 4-mm-thick sections. 40 The sections were stained with hematoxylin and eosin (H&E), Masson’s trichrome, picrosirius red, and toluidine blue. H&E staining is often used to identify inflammation or to confirm the integrity of the tissue. Masson’s trichrome staining is also used to confirm inflammation or tissue integrity. Picrosirius red staining was used to analyze collagen organization at the tendon-to-bone interface. Toluidine blue staining was performed to analyze the area of fibrocartilage at the tendon-to-bone insertion site.
The slides were interpreted by scoring to assess various aspects of the tendon tissue. The whole slide was assessed for areas of increased cellularity and vascularity, the proportion of collagen fiber, and the level of maturation of the tendon-to-bone interface structure. The score items were as follows: (1) collagen fiber continuity; (2) collagen fiber orientation; (3) collagen fiber density; (4) maturation of the tendon-to-bone interface structure; (5) vascularity; and (6) cellularity. A four-point scoring system was used. For each item, the histological findings were graded semiquantitatively into 4 stages (grade (G) 0, 1, 2, and 3), where 0 indicates the poorest appearance of the ruptured tendon, 1 indicates a poor appearance, 2 indicates an improved appearance, and 3 indicates marked regeneration. Overall, the total score for a given slide could vary between 0 (a ruptured tendon) and 18 (most marked regeneration). 9 To eliminate observer error, a pathologist with at least 10 years of experience examined all slides in a randomized, blinded fashion 3 times at the same position and area of rotator cuff tissue using a microscope (Leica DM IL LED, Leica Microsystems, Wetzlar, Germany) and image system (LAS V4.8, Leica Microsystems, Wetzlar, Germany) at ×50 magnification. 40
Statistical analysis
All statistical analyses were performed using SPSS 12.0 software (SPSS Inc., Chicago, IL, USA). p < .05 indicated statistical significance. The Shapiro-Wilk test was used for the normality test. The Kruskal-Wallis test and post-hoc Mann-Whitney U-test were performed to evaluate the biomechanical and histological differences based on H&E staining between groups. Data are presented as the mean ± standard deviation (SD).
Results
Mechanical properties of the GO/alginate scaffold
Comparison of the biomechanical characteristics both scaffolds.

GO; graphene oxide.
*Statistically significant. Values expressed as mean ± standard deviation.
In vitro cytotoxicity and cell proliferation
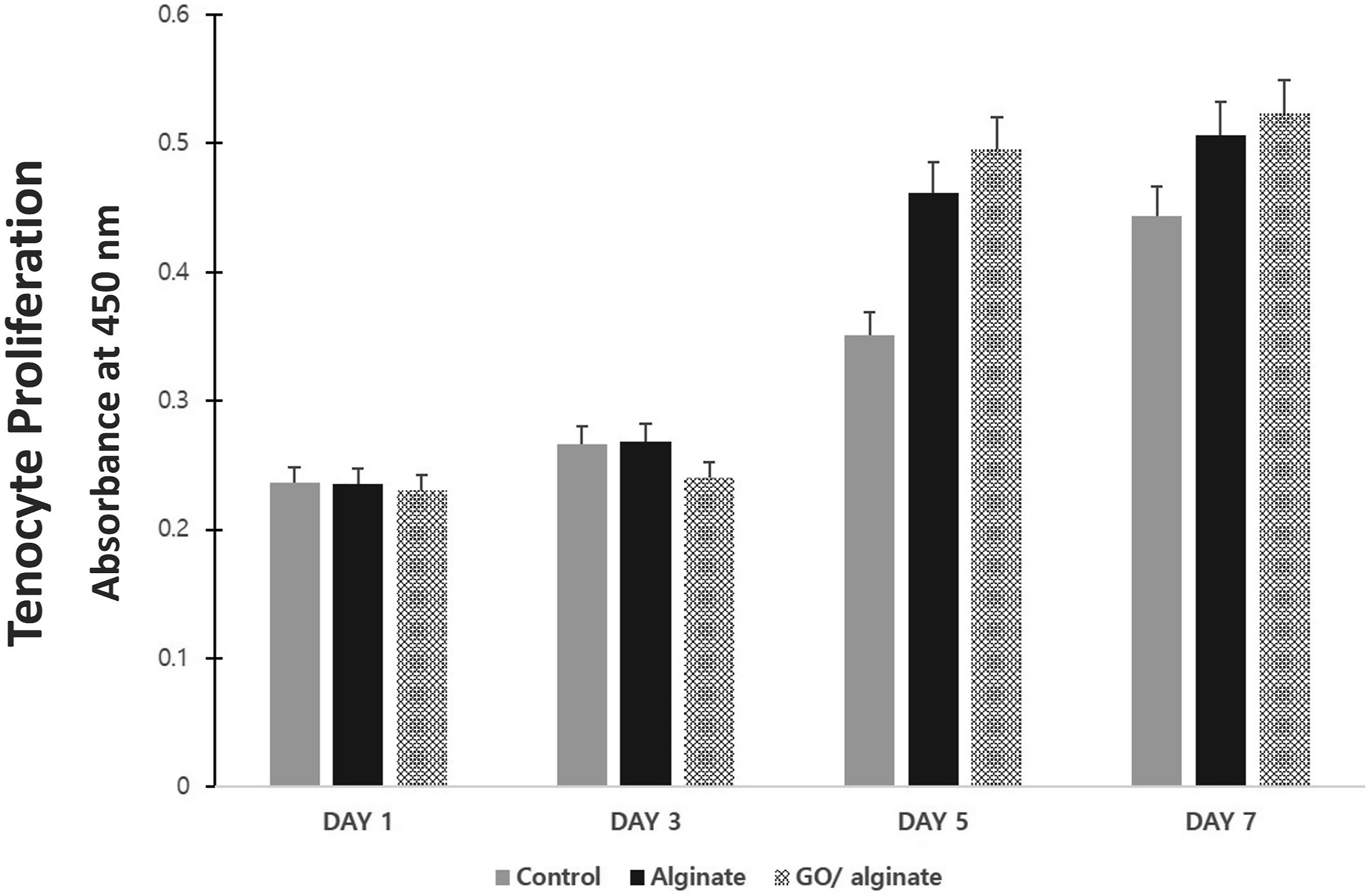
Cytotoxicity was assessed by the CCK-8 assay. The cell survival rate of GO/alginate was 102.08%, and that of pure alginate was 100.76% compared with that of the fresh medium control group (p = .572) (Figure 4). The tenocyte proliferation test showed no significant differences among the three groups until the 3rd day (p = .154 on 1 day, p = .112 on the 3rd day). On the 5th and 7th days, the rate of tenocyte proliferation was significantly increased in the alginate group (p = .001 on the 5th day, p = .008 on the 7th day) and the GO/alginate group (p = .000 on the 5th day, p = .002 on the 7th day) compared with the control group. However, there was no significant difference between the GO/alginate and alginate groups (p = .066 on the 5th day, p = .246 on the 7th day) (Figure 5). Cytotoxicity test: The cell survival rate of graphene oxide /alginate was 102.08%, and that of pure alginate was 100.76% compared with that of the fresh medium control group. Three samples from each group were used for the analysis, and the mean values are presented and compared (p = .572) Cell proliferation assay of GO/alginate: There was no significant difference in cell proliferation rates in the three groups on the 1st and 3rd days. On the 5th and 7th days, the alginate group and the GO/alginate group had significantly higher cell proliferation rates than group 1, but there was no significant difference in both groups (alginate vs control, p = .001 on the 5th day, p = 0.008 on the 7th day; GO/alginate vs control, p= .000 on the 5th day, p = .002 on the 7th day; GO/alginate vs alginate, p = .066 on the 5th day, p = .246 on the 7th day). GO; graphene oxide.
Biomechanical evaluation
Findings on gross inspection and failure mode.

Group 1, supraspinatus repair only; group 2, supraspinatus repair with graphene oxide (GO)/alginate scaffold.
*Statistically significant.
Comparison of the biomechanical characteristics both groups.

Group 1, supraspinatus repair only; group 2, supraspinatus repair with Graphene Oxide (GO)/alginate scaffold.
*Statistically significant. Values expressed as mean ± standard deviation.
When group 2 is compared with each mode of failure in group1, group 2 exhibited a significantly greater ultimate failure load (group 1: 11.44 ± 3.67 N; group 2: 105.34 ± 18.14 N; p < .001), ultimate stress (group 1: 2.81 ±1.45 MPa; group 2: 24.75 ± 8.23 MPa; p < .001), and stiffness (group 1: 1.15 ± 0.41 N/mm; group 2: 10.35 ± 5.05 N/mm; p < .001) than the midsubstance failure of group 1 and significantly greater ultimate failure load (group 1: 55.41 ± 40.91 N; group 2: 105.34 ± 18.14 N; p = .017), ultimate stress (group 1: 12.39 ± 6.29 MPa; group 2: 24.75 ± 8.23 MPa; p = .042) compared to the tendon-to-bone failure of group 1.
Histological evaluation
Scoring of findings on histological analysis*.
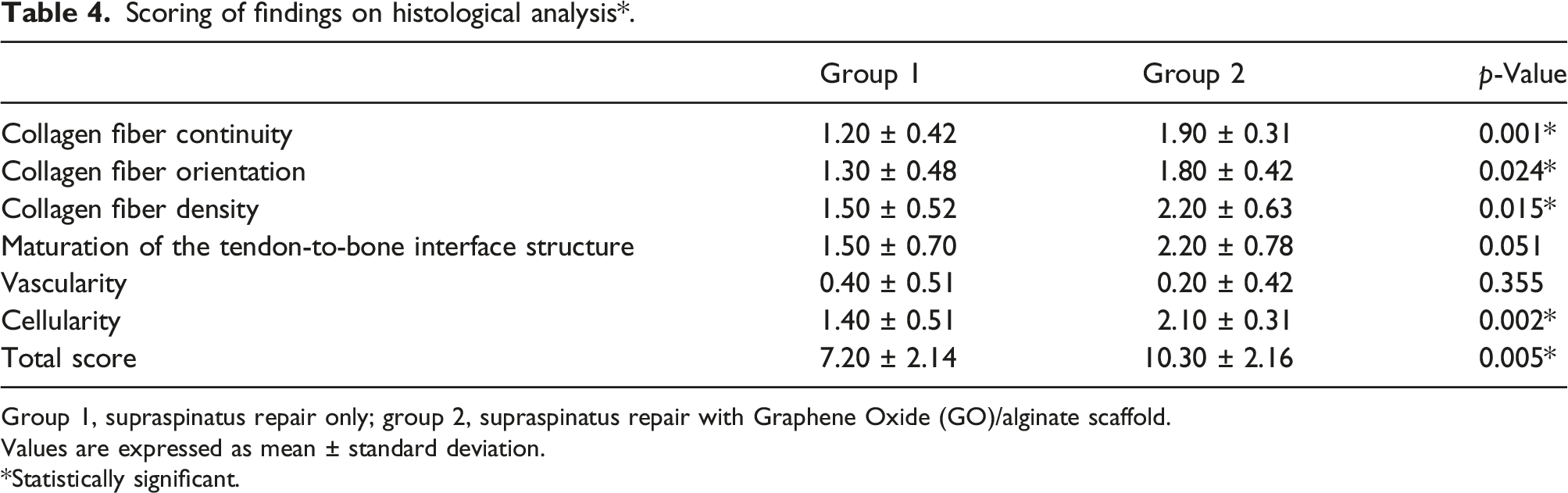
Group 1, supraspinatus repair only; group 2, supraspinatus repair with Graphene Oxide (GO)/alginate scaffold.
Values are expressed as mean ± standard deviation.
*Statistically significant.
Observations of the appearance of collagen fibers at the tendon-to-bone repair site, which are specific to the rotator cuff tendon-to-bone healing evaluation, demonstrated that the collagen fibers were poorly organized and that fiber continuity with bone had not yet been established in group 1. In group 2, however, the tendon-to-bone interface showed more collagen fibers bridging the interface, good tendon-to-bone integration, and longitudinally oriented collagen fibers (Figure 6). Histological evaluation 8 weeks after repair. Group 1 underwent supraspinatus repair alone; group 2 underwent supraspinatus repair with a graphene oxide/alginate scaffold. (a) H&E staining (magnification, ×50). (b) Masson’s trichrome staining (magnification, ×50). (c) Picrosirius red staining (magnification, ×50). The continuity, orientation, and density of collagen fiber showed higher average grades in group 2 than in group 1. Group 2 showed better maturation and density of the tendon-to-bone interface structure (arrow) than group 1.
The area of metachromasia with toluidine blue staining was measured to confirm the presence of fibrocartilage at the tendon-to-bone junction. In group 2, the area of metachromasia appeared stronger than that in group 1 (Figure 7). Histological evaluation 8 weeks after repair. Toluidine blue staining (magnification, ×50). Group 1 underwent supraspinatus repair alone; group 2 underwent supraspinatus repair with a graphene oxide/alginate scaffold. The arrow indicates the tendon-to-bone interface. This area is known as the supraspinatus enthesis—a fibrocartilaginous transition site—which was stained with toluidine blue. The area of metachromasia appeared stronger in the supraspinatus enthesis in group 2 than in group 1.
Discussion
This study investigated the effects of a GO/alginate scaffold placed at the supraspinatus tendon-to-bone repair site on the healing of rotator cuff tears through biomechanical and histological analyses in a rat model and evaluated the toxicity of the GO/alginate scaffold. The GO/alginate group showed improved biomechanical properties and a histologically better healing quality at the tendon-to-bone interface than the repair alone group. In the cytotoxicity and proliferation tests, GO/alginate did not show toxicity to tenocytes and showed a cell proliferation rate equivalent to that of alginate.
When comparing the mechanical properties of the alginate hydrogel sheet and GO/alginate sheet, the maximum load increased by ∼237% when GO was added to alginate, and the tensile strength increased by 430%. Lonita et al. 23 reported that hydrogen bonding and high interfacial adhesions between GO (0.5, 1, 2.5, 6% w/v) and the alginate matrix (1% w/v) significantly changed the thermal stability and mechanical properties of the nanocomposite films. We used 2% w/v sodium alginate, and the content of GO in the GO/alginate sheet was 0.01% w/v. Although the concentrations of alginate and GO in our study were different from those used in previous studies, a significant improvement was observed in the mechanical properties of alginate even with a GO content of 0.01% weight/volume. These findings suggest that the enhanced mechanical properties of GO/alginate may as an internal splint that can reduce the stress on the tendon-to-bone interface that may occur during the healing process after rotator cuff repair.
The biomechanical evaluation 8 weeks after surgery showed a significant difference in the mode of failure between the groups (the ratio of insertional tears to midsubstance tears was 3:7 in group 1 vs 10:0 in group 2 at 6 weeks; p = .003). In group 2, all modes of failure occurred at the tendon-to-bone interface. The GO/alginate group showed a significant difference in approximately 8–9 times in the ultimate failure load, ultimate stress, and stiffness compared with the midsubstance failure in the control group and a significant difference of approximately 2 times in the ultimate failure load and ultimate stress compared with the tendon-to-bone failure in the control group. In a previous study using alginate sheets, the ultimate load and stress values of the alginate scaffold group measured 12 weeks postoperatively in a rat model of acute rotator cuff tears were 61.44 ± 43.67 N and 7.36 ± 2.87 MPa, respectively. 9 The GO/alginate group had 170% and 336% higher values for ultimate load and ultimate stress, respectively, at 8 weeks, than the results of this previous study. In a recent study on GO-doped electrospun poly (lactic-coglycolic acid) nanofibrous membranes (GO-PLGA) in a rabbit rotator cuff tear model, Su et al. 41 also reported that the ultimate load and stress values of the supraspinatus tendon-to-humerus complexes in the GO-PLGA group were significantly higher than those in the control and PLGA groups from 4 to 12 weeks. These findings suggest that the GO/ alginate sheet may indeed possess enhanced biomechanical properties at the tendon-to-bone interface in rotator cuff healing.
The improvement in these biomechanical properties is also related to the improvement in the histological properties during the healing of the rotator cuff. Eight weeks postoperatively, the GO/alginate group showed significantly improved collagen fiber continuity, collagen fiber orientation, collagen fiber density, and cellularity compared with the control group. The semiquantitative evaluation also showed higher scores in the GO/alginate group than in the control group. In a previous study using alginate sheets, the total score of the alginate scaffold group measured 12 weeks postoperatively in a rate model of an acute rotator cuff tear was 5.25 ± 2.62. 9 In this study, this score was 10.30 ± 2.16 at 8 weeks postoperatively. The area of metachromasia in toluidine blue staining was also measured to confirm fibrocartilage at the tendon-to-bone junction. In the GO/alginate group, the area of metachromasia appeared stronger than in the control group. This indicates that GO/alginate, which induces collagen formation and cartilage differentiation, promoted the production of fibrocartilage.
Trudel et al. 42 studied the failure mode using a rabbit model of supraspinatus tears. They reported that early on, suture pullout was the most common mode of failure, whereas, at 6 weeks, midsubstance tears predominated (p < .05). The most common mode of failure in the normal tendon was a midsubstance tear. Kim et al. 9 also reported similar results to the previous study. In our study, only the GO/alginate group showed tendon-to-bone failure, and the biomechanical characteristics were superior to those of the control group. In addition, group 1 also showed better biomechanical characteristics than midsubstance failure in tendon-to-bone failure. In brief, tendon-to-bone failure has higher biomechanical properties, but this fact is in contrast to the findings of previous studies. One hypothesis to explain this phenomenon is that GO/alginate not only promotes healing at the tendon-to-bone interface but also affects the composition of the supraspinatus tendon. In the semi-quantitative histological evaluation, the GO/alginate group showed significantly superior collagen fiber continuity, collagen fiber orientation, and collagen fiber density. Another hypothesis is that the samples were small enough that it was difficult to prove a truly significant difference in the failure mode in the two groups. Of these two hypotheses, we think that the small number of samples is more likely to be the reason for the significant difference in the mode of failure. However, we cannot yet clearly explain these results, and new experiments must be conducted to clarify them.
In this study, at 8 weeks postoperatively, a part of the GO/alginate sheet was not completely biodegraded and was observed around the tendon repair site. The stability of the scaffold is mainly attributed to the extent of biodegradation. GO has a large surface area that enhances the structural integrity of the scaffolds and increases their stability. 43 Previous studies have reported that GO incorporated in a fibrin hydrogel has a controlled degradation rate of 43% on day 14 and 56% on day 28, whereas a scaffold without GO showed a faster degradation rate. A nanocomposite scaffold of gelatin–alginate (GA)–GO was also reported to show 57% degradation of the GA scaffold compared with 28% degradation of the GA-GO scaffold after 28 days. 44 The reduced biodegradability of GO/alginate not only prolongs the promigratory effects of GO on osteoblasts and MSCs but also enhances the drug delivery capacity of alginate. This fact eventually contributes to improved healing at the tendon-to-bone interface.
However, in our study, it was not possible to determine exactly how the GO/alginate sheet affected the tendon-to-bone interface in rotator cuff healing. In previous studies, GO/alginate has been shown to enhance cell attachment and proliferation and induce cell differentiation of osteoblasts and MSCs, indicating osteoinduction.7,12,13,15,16 In previous in vitro studies on GO/alginate composites, MSCs with alginate/GO showed good proliferation and enhanced osteogenic differentiation compared with MSCs with alginate. 45 There have been numerous reports that MSCs promote healing at the tendon-to-bone interface in rotator cuff tears.46,47 The subacromial bursa is a good candidate source of MSCs in rotator cuff tears.48,49 In addition, MSCs were found in bursal tissue infiltrate in repaired rotator cuff tendons after rotator cuff repair in an animal model. 50 Rotator cuff MSCs also express lineage-specific genes, such as Runt-related transcription factor 2 (Runx2) and osteocalcin in osteogenic induction, peroxisome proliferator-activated receptor gamma (PPARγ) and lipoprotein lipase (LPL) in adipogenic differentiation, and aggrecan and collagen type II α1 (ColIIα1) in chondrogenic differentiation. 51 Based on these data, it can be inferred that GO/alginate may promote MSC proliferation and differentiation at the tendon-to-bone interface during rotator cuff healing and enhance fibrocartilage and collagen formation.
In this study, an in vitro CCK-8 assay was used to confirm that GO was not cytotoxic. In another study, cell viability was significantly decreased at GO concentrations > 100 μg/mL but was maintained above 80% at concentrations < 62.5 μg/mL. These results suggest that GO is noncytotoxic at concentrations < 62.5 μg/mL. 17 Another in vitro toxicity study showed that a GO/alginate scaffold did not exhibit cytotoxicity even at relatively high concentrations (>100 μg/mL), with no evident toxic effects on NIH-3T3 cells. 31 In our in vitro analysis, the GO/alginate scaffold had a GO concentration of 98 μg/mL and showed a cell viability of 92.26%. These results indicate that the GO/alginate sheet had no apparent in vitro toxic effects on human tenocytes.
In the in vivo experiment, all rats survived for 8 weeks, and we did not find any obvious side effects indicating the toxicity of the GO/alginate sheet with a GO content of 0.039 mg per rat. However, the data on the toxicity of GO is controversial. A recent study reported that subdermal implantation of chitosan beads incorporating GO/titanium dioxide nanoparticles into Wistar rat tissue for 90 days showed proper and complete healing without any allergenic response to a GO content of 0.01% w/v. 52 This demonstrates that subdermal implantation of low-dose GO did not cause any significant side effects. However, histological evidence showed that when GO was injected into the medial femoral muscle tissues of BALB/c mice, it caused muscle atrophy and necrosis. 53
In our study, although GO did not show any specific side effects in the localized area, the systemic effects of GO are not known. Previous studies that tested the in vivo toxicity of GO at a dose of 0.1 mg or 0.25 mg showed no toxicity when GO was administered into the tail veins of rats; however, 0.4 mg of GO showed toxicity such as death and lung granuloma formation and could not excreted from the body through the kidneys.50,54 A study on another type of graphene—polyethylene, glycosylated (PEG) nanographene—showed accumulation in the reticuloendothelial system, including the liver and spleen, after intravenous administration and gradual clearance, likely by both renal and fecal excretion; there was no appreciable toxicity at 20 mg/kg in the treated mice over a period of 3 months as evidenced by blood biochemistry, hematological analysis, and histological examination. 55 Based on these previous studies, we speculate that the GO concentration used in our experiment would not have any significant systemic effect.
This is the first study to verify the effects of GO-based scaffolds on rotator cuff tendon-to-bone healing. In addition, the results of our study show that the addition of a small amount of nontoxic GO in vivo enhanced the biomechanical properties of natural polymers that have been previously used as scaffolds and can act as a booster to promote healing at the tendon-to-bone interface.
Nevertheless, this study has some limitations. First, this study was an animal study. The differences in the anatomical features and other wound and healing responses between humans and rats would necessitate further studies before using these scaffolds in the human body. Our study showed that the GO/alginate scaffold improved the mechanical properties and tendon-to-bone healing without any signs of toxicity in vitro and in vivo. Although it can be considered as one of the suitable material candidates for application in patients with rotator cuff in clinical settings, the following studies on adverse reactions and efficacy in the human body, including ethical issues, are needed. Second, an 8-week evaluation period may not be sufficient for a conclusion about the safety of GO as GO-induced toxicity and other side effects may still occur after 8 weeks. It is therefore necessary to prolong the study duration. Third, the total amount of GO used and the GO concentration of the scaffold are important. Even if a low concentration of GO support is used, pure GO dissociates due to the decomposition of the base material, alginate, and may accumulate in certain regions in the body. However, the movement and behavior of the GO in the body could not be confirmed, and caution should be exercised if the amount of GO used is greater than that used in this study. Fourth, this study did not analyze the effects of GO/alginate on previously evaluated MSCs and osteoconductivity. Therefore, further research on these is needed to understand the effects of GO/alginate more clearly on tendon-to-bone healing. These limitations should be addressed in subsequent studies. Despite these limitations, this GO/alginate scaffold has various advantages and can function as an amplifying agent for the mechanical and biological factors involved in the healing of rotator cuff tears.
Conclusions
In conclusion, a small amount of GO added to alginate improves the mechanical properties of the scaffold without causing any cytotoxicity. Eight weeks after rotator cuff repair, this GO/alginate scaffold improved tendon-to-bone healing without any signs of toxicity in a rat model.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by the Basic Science Research Program through the National Research Foundation of Korea funded by the Ministry of Science, ICT and Future Planning (NRF-2022R1A2C1005374).
IACUC information
Kyungpook National University Institutional Animal Care and Use Committee IACUC NO: KNU 2018 -0123
