Abstract
Background
Femoral bone deficiency is a challenging problem in revision proximal femoral replacement. The purpose of this study is to evaluate the clinical and radiological outcomes of revision proximal femoral replacement as a salvage treatment for severe bone loss after oncologic proximal replacement surgery in patient with benign giant cell tumor of bone.
Methods
16 patients (6 men and 10 women) were included in this retrospective study, with a mean age of 46.6 year at the time of revision surgery. All patients underwent revision proximal femoral replacement with the use of modular prosthesis and cortical strut allografts. The modified Harris Hip Score, Short Form 36, and musculoskeletal Tumor Society Score were used for patient evaluation. Regular follow-up was performed to evaluate the recurrence and metastases rate, limb function, and long-term complications of patients.
Results
The average follow-up was 46.3 months (range, 26–75 months), during which there was no local recurrence and metastases of patient. At the latest follow-up, the mean modified Harris Hip Score was 70.6 points, which was significantly improved compared with that of preoperative (p < 0.05). The final follow-up results of Short Form 36, Musculoskeletal Tumor Society Score, and limb-length discrepancy were also significantly improved compared to that of preoperative (p < 0.05). At the latest follow-up, the implanted femoral stems were all stable and all cortical strut allografts were also incorporated to their own bone.
Conclusion
Using modular prosthesis and cortical strut allografts in revision, proximal femur replacement is an acceptable procedure for relatively young patient with severe proximal femoral bone loss after oncologic surgery with benign giant cell tumor of bone. More attentions should be paid to reduce the risk of complications in these complex reconstructions.
Keywords
Introduction
Giant cell tumor of bone (GCTB) is an aggressive benign bone tumor, which is prone to local recurrence after resection.1,2 As with malignant tumors, the treatment of GCTB in the proximal femur is a challengeable problem. 3 Many therapeutic methods are associated with high complication rates.4–6 With the improvement in surgical techniques and prosthesis design, limb salvage surgery has achieved unforeseen strides in the treatment of bone tumors.2,7 Meanwhile, with the rising life expectancy of patients, the revision rate of complex femoral bone defects is also expected to increase. 8 The remaining femur often has very thin cortical bone presenting a challenge for fixation during femoral reconstruction, especially for patients with multiple revision replacement.8,9
Proximal femoral allografts 10 or total femoral replacement prosthesis 11 are alternative methods for complex femoral bone defects, but these operations require advanced surgical techniques. Furthermore, poor femoral bone stock prior to revision proximal femoral replacement is related to poor functional outcome and high complication rate. Take all this together, the revision procedure of proximal femoral replacement remains challenging for patient with severe proximal femoral bone loss. Interestingly, the cortical strut allografts were not only used as supplementary augmentation of the femur but also provide axial and torsional stability to the femoral stem.9,12 For non-oncologic patients with severe proximal femoral bone loss, some reports have presented that using proximal femoral replacement and cortical strut allografts for revision total hip arthroplasty (THA) is a useful alternative salvage procedure as alternative way. 13 However, there have been no studies of revision proximal femoral replacement using modular prosthesis and cortical strut allografts as a salvage treatment for severe bone loss after oncologic surgery in patient with GCTB.
Here in, we retrospectively analyzed GCTB patients of revision proximal femoral replacement with complete clinical and radiographic data through a single center. This study aims to study the clinical and radiographic efficacy of revision proximal femoral replacement using modular prosthesis and cortical strut allografts and evaluate whether this method can achieve good therapeutic effect for benign GCTB patient. The purpose is to provide an alternative choice for revision in benign GCTB patient after oncologic proximal replacement surgery.
Patients and methods
Approval for this retrospective study was granted by the Institutional Ethics Review Board, and informed consent was obtained from each patient. The clinical and radiographic data of consecutive patients from December 2013 to February 2018 were retrospectively reviewed. Aseptic loosening was defined based on the clinical and radiologic standards.
4
Inclusion criteria were as below: (Ⅰ) patients who underwent oncologic proximal replacement surgery due to GCTB; (Ⅱ) patients who underwent revision proximal femoral replacement with the use of modular prosthesis and cortical strut allografts. The exclusion criteria were as follows: (Ⅰ) any other causes to undergo revision apart from aseptic loosening; (Ⅱ) cortical strut allografts were not used in the revision surgery; (Ⅲ) less than 2 years follow-up; (Ⅳ) patient with incomplete medical records. According to the above criteria, the patient with incomplete data would be excluded. The clinical and radiographic data (Figure 1) were collected from medical record system under the same criteria. Preoperative anteroposterior (a) and lateral (b) radiograph shown aseptic loosening of a 47-year-old female after proximal femoral replacement with custom-made prosthesis.
All the patients were operated by or under the direct supervision of the senior surgeons (TL and XSZ). All revision surgeries were performed by the part ilioinguinal and Smith-Petersen incision with the patient in a lateral position maintained by bags and rolls, which allowed semi-supine and semi-prone positions.
14
The operative time was defined from incision time to hip dressing completion time. An extensile incision was necessary for the safe extraction or correction of the failed implants (Figure 2). Direct microscopical examination and staining with Gram’s and Acid-fast, rapid frozen pathological diagnosis of multi-point necrotic tissue and routine bacterial culture were performed to identify and exclude infection and tumor recurrence. The severity of femoral bone defects was classified based on the Paprosky classification
15
and confirmed by intra-operative findings after tissue debridement (Figure 3(a)). We began to reconstruct when obtaining negative results of the microscopy and the rapid frozen pathological diagnosis during surgery. After obtaining the desired stable position,
16
we implanted the suitable acetabular cup. Based on the instability of the femoral stem, cortical strut allograft was used to reconstruct the bone volume and repair the intra-operative fracture. Once satisfactory femoral bone stock has been obtained, we would loosely secure the grafts using double-loop cerclage wires (Figure 3(b)) and take an intra-operative radiograph. The modular prosthesis with appropriate length, diameter and offset was knocked into the medullary cavity after stability trials, and complete tightening of the cortical strut allografts was achieved after taking intra-operative radiographs once again (Figure 4 and Figure 5). After soft tissue reconstruction, a trial reduction was carried out to test stability and tension.11,17 Postoperatively, all patients were given oral celecoxib and managed with intravenous broad-spectrum antibiotics. Anti-embolism exercises were routinely started immediately, and chemical anti-embolism drugs were used for prophylactic anticoagulation. Patients were routinely made to stand at bedside, or walk non-weight bearing on the affected side for a minimum of 6 weeks. The patients were then progressed to full weight bearing ambulation as tolerated after 6 to 10 weeks under the supervision of a therapist. The failed custom-made prostheses of the female (Figure 1) have some abrasion at inner liner. (a) Intra-operative photograph shown femoral Paprosky type Ⅲ B bone defects after tissue debridement, and the white arrow indicate the very thin cortical thickness, and (b) shown the revision technique using modular prosthesis and cortical strut allograft. The revision modular prosthesis consists of three components, “a” is the proximal femoral replacement components, “b” is the extension rod, and “c” is distal intramedullary needle. A diagram describing the surgical technique of allografts reconstruction. (a) the host bone was retained as much as possible after removing the previous femur endoprosthesis and bone cement, (b) the cortical strut grafts were placed on top of the host bone and were slightly fixed using double-loop cerclage wires, (c) completely tightening the cortical strut allografts after implanting a new modular prosthesis.



All patients were assessed clinically and radiologically after 6 weeks, 3 months, 6 months, and then every 6 months within 2 years, and then annually during the follow-up period. Functional status was assessed using modified Harris Hip Score (mHHS), 18 Short Form 36 (SF-36), 19 and Musculoskeletal Tumor Society Score (MSTS). 14 Distance from the anterior superior iliac spine to the medial maleolus was measured as the leg-length, and the difference between both lower extremities was calculated and defined as limb-length discrepancy (LLD). 20 Femoral bone defects were classified based on the preoperative radiographs and the records of intra-operative findings using the Paprosky classification system. 12 When comparing with subsequent radiographs, we use the initial postoperative immediate radiograph as the baseline. The acetabular cup abduction angle and anteversion angle were measured or calculated on radiographs. 16 Instability of the femoral stem was defined as subsidence of greater than 5 mm. 9 We evaluated the incorporation of the cortical strut allograft to the host bone and graded the resorption of the allograft according to the technique of Kim et al. 9 The criteria of osseointegration were applied to evaluate the ingrowth of the distal stems, and postoperative complications of loosening, subsidence, heterotopic ossifications, and stress shielding were reviewed and assessed. 21 More importantly, two independent authors retrospectively reviewed and evaluated clinical and radiographic data blindly to minimize bias and the inconsistent evaluation was resolved through discussion and consensus with additional two senior surgeons.
Statistical analysis was performed using SPSS Statistics for Windows, version 21.0 (SPSS Inc., Chicago, IL, USA). We described continuous variables as means ± standard deviations and the categorical variables as count and rate. Differences between preoperative and the latest follow-up results were analyzed using Wilcoxon Mann–Whitney test. A two-tailed p-value < 0.05 was considered significant for all tests.
Results
Patient demographics.

aValues given as mean ± standard deviation (range); BMI: body mass index.
Clinically, all the included patients were alive without local recurrence and metastases at the latest follow-up. No patient had wound problems and nerve palsy after the revision surgery. There were no calf thrombi or pulmonary embolisms. No patient underwent re-revision surgery for any causes except for one patient (6.3%) who underwent an amputation due to repeated infection. There were two patients (12.5%) with postoperative infections. In one of these patients, the organism isolated was Staphylococcus aureus. The early postoperative infection was treated successfully by debridement and prolonged administration of intravenous and oral antibiotics. The microorganism isolated from the other patient was methicillin-resistant staphylococcus aureus. Unfortunately, due to the underlying diseases and poor compliance, the patient developed repeated infection and eventually had to undergo amputation. Three (18.8%) patients had postoperative dislocation within 2 weeks after the surgery, and there were no recurrent dislocations after treating with closed reduction and bracing. The average preoperative mHHS was 37.9 points and it was 70.6 at the latest follow-up with significant difference (p < 0.05). The average SF-36, MSTS score (Figure 6(a)) and LLD at the latest follow-up were improved significantly when compared to the preoperative scores (p < 0.05) (Table 2). (a) In our series, the mean MSTS scores of the latest follow-up were higher than that of preoperative. (b) The survival proportions of the primary and (c) revision implants. Preoperative clinical results versus that of the last follow-up. aValues given as mean ± standard deviation; mHHS: Modified Harris Hip Score; MSTS: Musculoskeletal Tumor Society Score; SF-36: 36-item Short Form Health Survey; LLD: Limb-Length Discrepancy.

Radiographically, all patients have Paprosky grade Ⅲ femoral defect. There was no patient outside the Lewinnek acetabular cup abduction and anteversion safe range. Enhanced bone stock was provided by the cortical strut allografts in all patients (Figure 7). Because of the complete coverage of cortical strut allografts, we did not observe bone resorption of the host femur. Among the implanted 15 femoral stems, all the allografts were incorporated in to the host femur. There were no signs of loosening, subsidence and stress shielding. No patient had Grade Ⅲ or Ⅳ heterotopic ossification which would need advanced treatment. In addition, we had not found statistically significant loss of length of the cortical strut allografts by remodeling at the final follow-up. The results of survivorship of the primary and revision femoral components are shown in Figure 6(b) and (c). Revision components were well fixed in a satisfactory position at 1 month postoperatively (a–c) and at 6 months postoperatively (d, e). Modular prosthesis was embedded with solid bone growth, and the cortical strut allografts were incorporated in the host bone at 2 years postoperatively (f, g).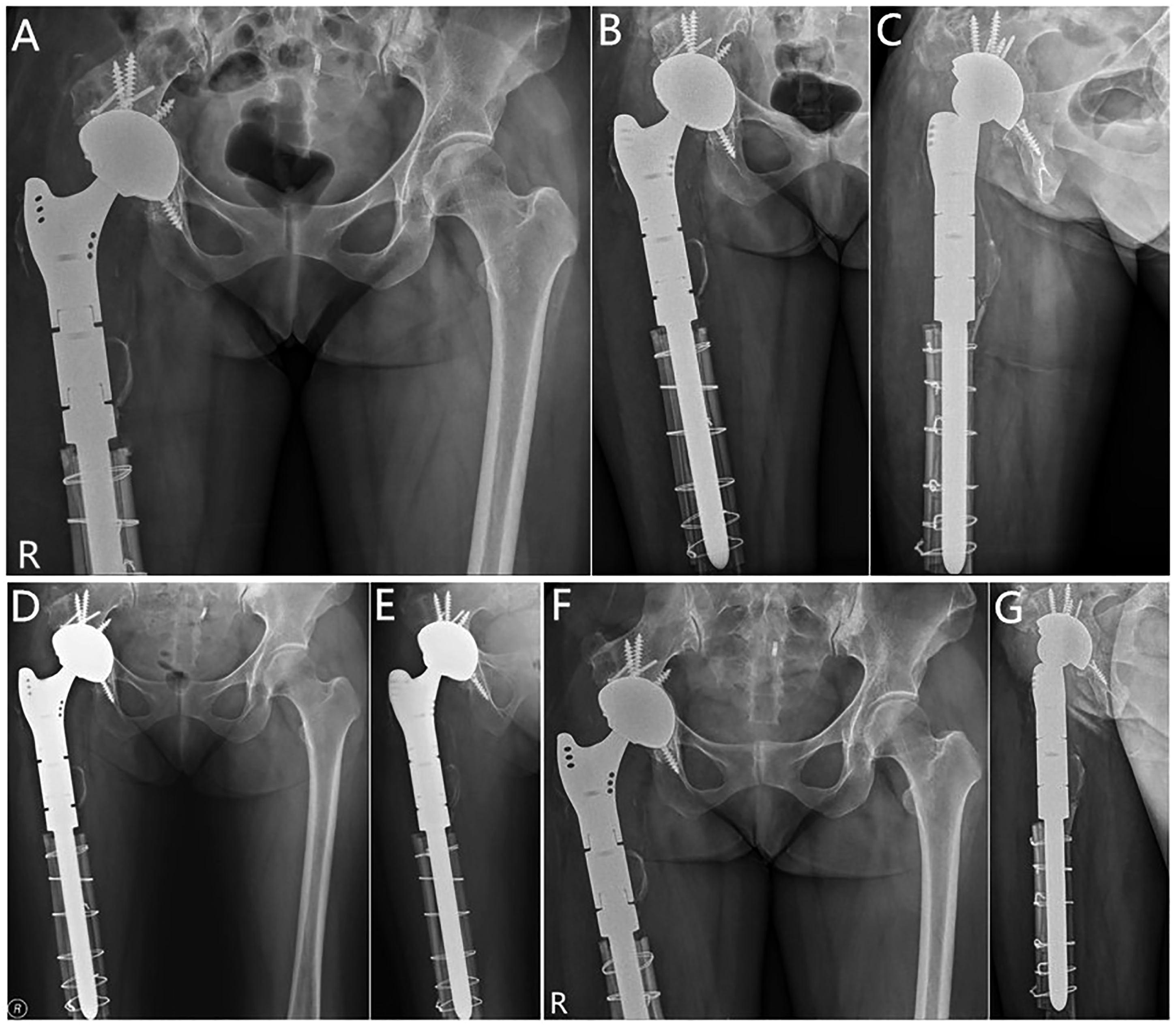
Discussion
As the region of femoral neck and intertrochanteric is an essential mechanical conduction pathway of the human body, the probability of pathological fracture is higher than other regions. Moreover, considering the blood supply, osteonecrosis is more likely to occur after the surgery of proximal femoral GCTB. Therefore, the surgeon must pay more attention to the optimal selection of surgical methods for proximal femoral tumors. 1 Endoprosthesis can provide patients with immediate postoperative stability and rapid recovery, but there is a risk of revision. 7 In the current study, the average time interval from the primary implant surgery to the revision proximal femoral replacement for aseptic loosening was similar to previous report. 5 Due to the complex bone structures, especially in Paprosky type Ⅲ and type Ⅳ bone defects, the revision of proximal femoral replacement is technically challenging. According to the clinical and radiographic outcomes from this retrospective study, we believed that the modular implants provide flexibility for femoral reconstruction, and cortical strut allografts are a good choice for restoring bone stock.
With the changes in major implant design and manufacturing, modular endoprosthesis have the advantages of fewer mechanical complications, and decreased revision and infection rates. 22 It is easy to implant the distal segment of modular endoprosthesis independently and to gain optimal leg-length and offset. 21 Some researchers suggested the cemented component is a better choice for patients with poor bone quality, especially in elderly patients with Parprosky type Ⅳ bone defects. 12 Nevertheless, some others recommended the cementless long-stem prostheses with fluted design is a better choice for patients with femoral distal revision. 23 A recent study found the cemented long-stem can provide adequate stability, but it is bad for bone union when cement was interposed between the fracture fragments. 24 Furthermore, some studies suggested the fluted, tapered modular femoral prostheses have the advantage of achieving long-term biological osteosynthesis even in patients with extensive bone defects.21,25 Therefore, all patients in the current study have used cementless modular prosthesis for proximal femoral reconstruction. After a mean follow-up of 46.3 months, no patient required re-revision surgery for aseptic loosening after revision with the fluted and tapered modular femoral prostheses. When we take into consideration only the re-operation for aseptic loosening, our revision surgery was less than the re-operation frequencies reported by Martino et al. 26 (4.9%) and Viste et al. 12 (2.3%). However, our patients had an average age of 46.6 years and were younger than the patients in their cohorts. Therefore, we believed that the revision of proximal femoral replacement using the modular prosthesis is a reliable method to restore mechanical and functional results for relatively younger patients who have GCTB of proximal femur with a short to middle-term follow-up.
Cancellous allografts were successfully used to reconstruct mild or moderate femoral bone defects, but complications of extensive subsidence and femoral fractures occur frequently for patients with severe bone defects. 27 When the cortical strut allografts are compared with artificial plates, cortical strut allografts are biologically active, and have the ability to increase bone stock and to decrease stress shielding complications. 23 Cortical strut allografts also have the advantage of reattaching soft tissues to the construct. 27 Therefore, to our knowledge, cortical strut allografts are widely used for supplementary augmentation of the femur and provide axial and torsional stability to the femoral stem. 12 Moreover, many studies recommended cables were better than wires in fixation of cortical strut allografts.23,24 However, less was known about the revision of proximal femoral replacement using cortical strut allograft for bone tumor. In the current study, the clinical results showed that the improved mHHS and MSTS scores were similar to previous studies reported.9,28,29 The radiologic outcomes showed all the allografts were united to the host bone at the latest follow-up. Therefore, we believed that the revision technique using cortical strut allografts was also suitable for benign bone tumors patients. However, in order to identify and rule out infection and tumor recurrence, other diagnostics should be done for these patients, which including preoperative imaging assessment, smear microscopy of joint fluid, rapid frozen pathological diagnosis, and routine bacterial cultures. Take all this together, cortical strut allografts are a good alternative to reconstruct the severe bone defects in the revision surgery.
Review the literatures of hip revision using modular prosthesis and cortical strut allograft.

#: Oxford Hip Score.
This study has several limitations. First, the sample size is small because it was hard to obtain a larger number of patients who have undergone the revision of proximal femoral replacement with the use of modular prosthesis and cortical strut allografts in a single center, due to the relative rarity of this condition. However, to our knowledge, the current study is one of the largest series of the revision technique for patients who have aggressive benign bone tumor to date. Second, it was a retrospective study without a control group mainly because of the same reasons above, and thus had all the inherent bias of retrospective studies. Third, the current study had a short-term follow-up study and any conclusion about the clinical and radiographic outcomes of the current study must be viewed in light of this.
Conclusion
Our study supports the use of modular prosthesis and cortical strut allografts for revision of proximal femoral replacement as limb salvage option for benign GCTB with severe bone defects in relatively young patient. The re-operation is acceptable, with a low incidence of aseptic loosening. To avoid major complications, attention should be given to identify and rule out infection and tumor recurrence pre-operatively and intra-operatively. Further studies and even longer follow-up are required to assess the durability of these reconstructions.
Footnotes
Acknowledgements
XHZ as the first author, and CT as the corresponding author. We would like to thank all participating patients, as well as the study nurses, co-investigators, and colleagues who made this trial possible.
Authors’ contributions
XH Z, TL and CT were conceptualized and designed the study, collected the data, analyzed the statistics, wrote and edited the manuscript. ZH L, XS Z, FM L and XZ T were conceptualized and designed the study, supervised the statistical analysis. All authors have read and approved the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
This retrospective study was approved by the Ethics Committee on Human Research of the Second Xiangya hospital of Central South University, and written informed consent was obtained from the patients.
Consent for publication
This retrospective study was well understood and agreed by all the included patients, which was recorded in the consent for publication.
Availability of date and materials
All data generated or analyzed during this study are included in this published article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by National Natural Science Foundation of China (Grant No. 81871783 and 82072441), the Scientific Research Project of Human Health Committee (Grant No.20200357), the Fundamental Research Funds for the Central Universities of Central South University (Grant No. 2020zzts288), the Research Project of Liuzhou People’s Hospital (Grant No. lry202101), and the Natural Science Foundation of Hunan Province, China (Grant No, 2021JJ30954).
