Abstract
Monitoring SARS-CoV-2 antibody levels can provide insights into a person’s immunity to COVID-19 and inform decisions about vaccination and public health measures. Anti-S may be useful as an indicator of an effective immune response. Thus, we conducted this study that aimed to determine the immune response of anti-S antibodies against SARS-CoV-2 for all the vaccine types over time among adult recipients in Malaysia and to determine the associated factors. This study was a cohort that recruited 2513 respondents aged 18 years and above from June to December 2021. Each participant was followed-up for 1-year period from the initial vaccine dose (baseline). We found that the anti-S antibody generally increased for all vaccine types and peaked at two weeks after the second dose vaccination, with Pfizer recipients having the highest median of 100 (100.00-100.00). During the third-month follow-up, the seropositivity of anti-S antibody and the median level decreased for all vaccines. We found that type of vaccines, comorbid status, infection, and booster status were significantly associated with the anti-S antibody level after one year.
What We Already Know
The body will produce SARS-CoV-2 antibody once infected or vaccinated.
The neutralizing antibodies will help to protect host from infections.
What This Article Adds
The trend of anti-antibody S among four administered vaccines in Malaysia.
The associated factors of anti-S antibody level after 1 year of vaccination.
Introduction
The COVID-19 pandemic has caused widespread illness and death, with over 200 million confirmed cases and nearly 15 million deaths reported worldwide. 1 In response, health care professionals have been working tirelessly to understand the virus and develop effective treatments and vaccines. SARS-CoV-2 antibody levels have been a critical area of focus throughout the ongoing pandemic. The level of SARS-CoV-2 antibodies in a person’s blood can vary depending on factors such as the severity of the infection, the person’s age, and their overall health. 2 Some people produce high levels of antibodies in response to infection, while others may not.3,4 In general, people who experience more severe COVID-19 symptoms tend to produce higher levels of antibodies. 5 However, even people with mild or asymptomatic infections can produce significant levels of antibodies. 6
The four primary structural proteins of the coronavirus are the nucleocapsid (N), spike (S), membrane (M), and envelope (E). For serological measurement of SARS-CoV-2 antibodies, tests that detect antibodies to S and N proteins, which are the most immunogenic proteins of SARS-CoV-2, are mainly used. Anti-S may be useful as an indicator of an effective immune response, including vaccine effectiveness as most COVID-19 vaccines target the spike protein. The anti-S bind specifically to the spike protein (S protein) of the SARS-CoV-2 virus. The virus’s S protein is essential for its ability to penetrate mammalian cells. When a virus attempts to infect human cells, the anti-S antibodies bind to the S protein. In the case of the SARS-CoV-2 virus, this binding prevents the virus from attaching to the ACE2 receptor on human cells. Consequently, it cannot penetrate human cells and replicate without the ability to bind to the ACE2 receptor, effectively neutralizing the virus. 7 The anti-S antibodies is an immunoglobulin G (IgG) antibody that has no role once the virus is inside the cells.
Studies have shown that individuals with high levels of anti-S antibodies are less likely to contract SARS-CoV-2 and have milder symptoms if they do become infected. In addition, research has demonstrated that anti-S antibodies can provide long-term immunity against COVID-19, protecting individuals against reinfection for several years.8,9
The duration of SARS-CoV-2 antibody protection is also an area of ongoing research. While antibodies are a critical component of the immune response to COVID-19, they may not provide long-lasting immunity. Some studies have suggested that SARS-CoV-2 antibodies may start to decline within a few months of infection, although other research has found that some people can maintain high levels of antibodies for up to a year or more. 10 The level of SARS-CoV-2 antibodies can also influence decisions about vaccination. People who have already had COVID-19 may have some level of immunity, but it is unclear how long that protection will last. Therefore, health care professionals typically recommend that people who have had COVID-19 still receive the vaccine to boost and extend the immunity against the virus.
Monitoring SARS-CoV-2 antibody levels can provide valuable information about a person’s immunity to SARS-CoV-2 and help inform decisions about vaccination and public health measures. However, more research is needed to fully understand the duration and strength of SARS-CoV-2 antibody protection and how it varies between individuals. Thus, we conducted this study that aims to determine the immune response of the anti-S IgG against SARS-CoV-2 over time among adult vaccinees in Malaysia and to determine the factor associated.
Methods
Study Design and Study Population
This longitudinal study involved 2513 respondents who received COVID-19 vaccine between June 2021 and December 2021. Respondents from selected vaccination centers were followed-up for a period of 12 months. Those who had received vaccination prior to the study period or were contraindicated for COVID-19 vaccination were excluded from this study. The selection of respondents were purposive samples that fulfilled the inclusion criteria and agreeable and consented to be followed-up. The cohort of vaccinees was divided into four main groups depending on their primary vaccinations; Pfizer (PF)—mRNA, Sinovac (SN)—viral vector, AstraZeneca (AZ)—weakened adenovirus, and CanSino (CS)—viral vector. All these of vaccines were targeting the same SARS-CoV-2 variants. All vaccines were developed based on the wild-type SARS-CoV-2. The booster dose was however optional. The booster given mainly was Pfizer.
Study Sites
For each of the four vaccines purchased and administered in Malaysia, multiple sites were selected purposively from across Malaysia. For the Pfizer vaccine, there were three study sites, namely, in Selangor, Terengganu, and Sarawak. For the Sinovac vaccine, the sites were in Selangor and Melaka. AstraZeneca vaccine sites were in Penang and Selangor. While the CanSino vaccine sites were mainly Selangor, Kedah, and Sabah. All the sites were in urban areas due to logistic issues.
Data Collection
Each cohort participant from all vaccine types was followed seven times in one year period, namely, at baseline (initial dose), prior to second dose if any, 14 days after the second dose or 28 days after completed vaccination (single dose vaccine), and subsequently at 3/6/9/12 months from baseline. During the data collection, respondents were recruited during the first visit to the vaccine center. Respondents who agreed to join the study were given questionnaires, taken their height and weight, and their venous blood to check for their antibody levels. The blood was centrifuged to get the serum and was sent to the designated laboratory. The data from the questionnaires that were collected in the mobile tablet was synced at the end of each data collection day with secured servers through the Internet. Laboratory results were traced and merged with the main data set. During follow-ups, respondents were called prior to the appointment date to remind them, and those who were absent from the follow-up appointment were called and home visits were done to trace them back. Those who were not able to come for an appointment after calls and home visits were treated as a loss to follow-up.
Sample Size
The sample size was based on the calculation made to compare the seroconversion rates at any same time points between different vaccines. To compare the herd immunity threshold of 70% and Malaysia’s target COVID-19 vaccination coverage of 80%, 291 participants in each population group receiving a particular vaccine was required to achieve a power of 80% and a two-sided significance of 5%. The sample size was calculated using online Select Statistical Services. 11
Study Tools and Laboratory Test
This study used laboratory test results and structured questionnaires to collect data. The independent study variables were age groups, sex, ethnicity, comorbidity status, BMI, vaccine types, booster vaccine, and history of COVID-19 infection. The comorbidity status was grouped as no comorbid, any one comorbid and two or more comorbid. Having at least one comorbidity (either diabetes, hypertension, heart disease, asthma, chronic pulmonary, liver failure, cancer, rheumatoid arthritis, SLE, kidney disease, stroke, or HIV). The history of COVID-19 infection was self-reported and was asked in every follow-up. During the 6th month of follow-up, some of the respondents received booster vaccination and some did not. The booster dose was optional; thus, some respondents chose not to receive the booster dose. This question was only asked during sixth-month follow-up onwards. The dependent variable of this study was the level of anti-S IgG antibodies. It can be seen in qualitative (seroconversion) or quantitative (the titer of the antibody). The seroconversion is measured in percent and the antibody titer was measured in Index value.
The serum samples were sent to the dedicated laboratory. The ADVIA Centaur SARS-CoV-2 IgG (sCOVG) assay is a chemiluminescent immunoassay intended for qualitative and semi-quantitative detection of IgG antibodies to SARS-CoV-2 in human serum using the using the ADVIA Centaur Immunoassay Systems. This assay requires 40 µL of sample for a single determination. Details procedure can be referred to the ADVIA Centaur SARS-CoV-2 IgG (sCOVG) kits package insert. 12
The system reports ADVIA Centaur sCOVG assay results in Index Values and as Nonreactive or Reactive:
Nonreactive (negative): <1.00 Index. These samples are considered negative for SARS-CoV-2 IgG antibodies. Report nonreactive patient results as <1.00 Index.
Reactive (positive): ≥1.00 Index. These samples are considered positive for SARS-CoV-2 IgG antibodies. Results with the numeric Index Value within the measuring interval for semi-quantitative measurements. Antibody levels were reported with values between 1.00 and 100.00 Index.
Data Analysis and Interpretation of Results
Data were analyzed using descriptive, bivariate, and multivariate analyses. Median (interquartile range), and proportion with their 95% confidence intervals were computed and described. Only those who received complete vaccination was included in the analyses. Factors associated were analyzed at 12th-month follow-up with 1118 respondents. For the association of infection status and booster vaccination with the level of anti-S IgG antibody, the Mann-Whitney test was used. While for the association of types of vaccines with the level of anti-S IgG antibodies, the Kruskal-Wallis test was used. Any value of P < .05 was considered to have a significant association. Multiple linear regression was used to examine the association of age, ethnicity, BMI, comorbid status, booster status, types of vaccine and infection status with the level of anti-S IgG antibody.
Ethical Approval
This study was funded by the Ministry of Health Malaysia and was conducted in accordance with research ethics. Written consent was obtained prior to data collection. All responses and results from this study were kept confidential. This study obtained ethical approval from the Medical and Research Ethics Committee (MREC) Ministry of Health Malaysia with reference number KKM/NIHSEC/P21-573 (12), and registered under National Medical Research Register (NMRR-21-411-58817). All the authors have no conflicts of interest to be declared.
Results
Sociodemographic Characteristics of the Vaccinees
Table 1 described the sociodemographic characteristics of the participants. Most of the respondents were in 18- to 39-years-old group mainly among male and Malay ethnic for all vaccine types. More than 95% of the respondents were Malaysians except in CanSino vaccine with 51.3% of non-Malaysians. The majority of respondents across all vaccine groups have no comorbidities. For the BMI status, Sinovac recipients have the highest percentage of obese individuals (38.1%), while CanSino recipients have the lowest (17.5%). Normal and overweight categories are relatively evenly distributed across vaccine groups. Very few respondents in each vaccine group reported a past COVID-19 infection (ranging from 2.6 to 5.6%). The majority of respondents have not had a past COVID-19 infection. In booster status, Pfizer and AstraZeneca recipients have a higher percentage of respondents who have received a booster dose compared with Sinovac and CanSino recipients. CanSino recipients have the lowest percentage of respondents who received a booster.
Sociodemographic Characteristics of the Respondents at Baseline Based on Vaccine Types.
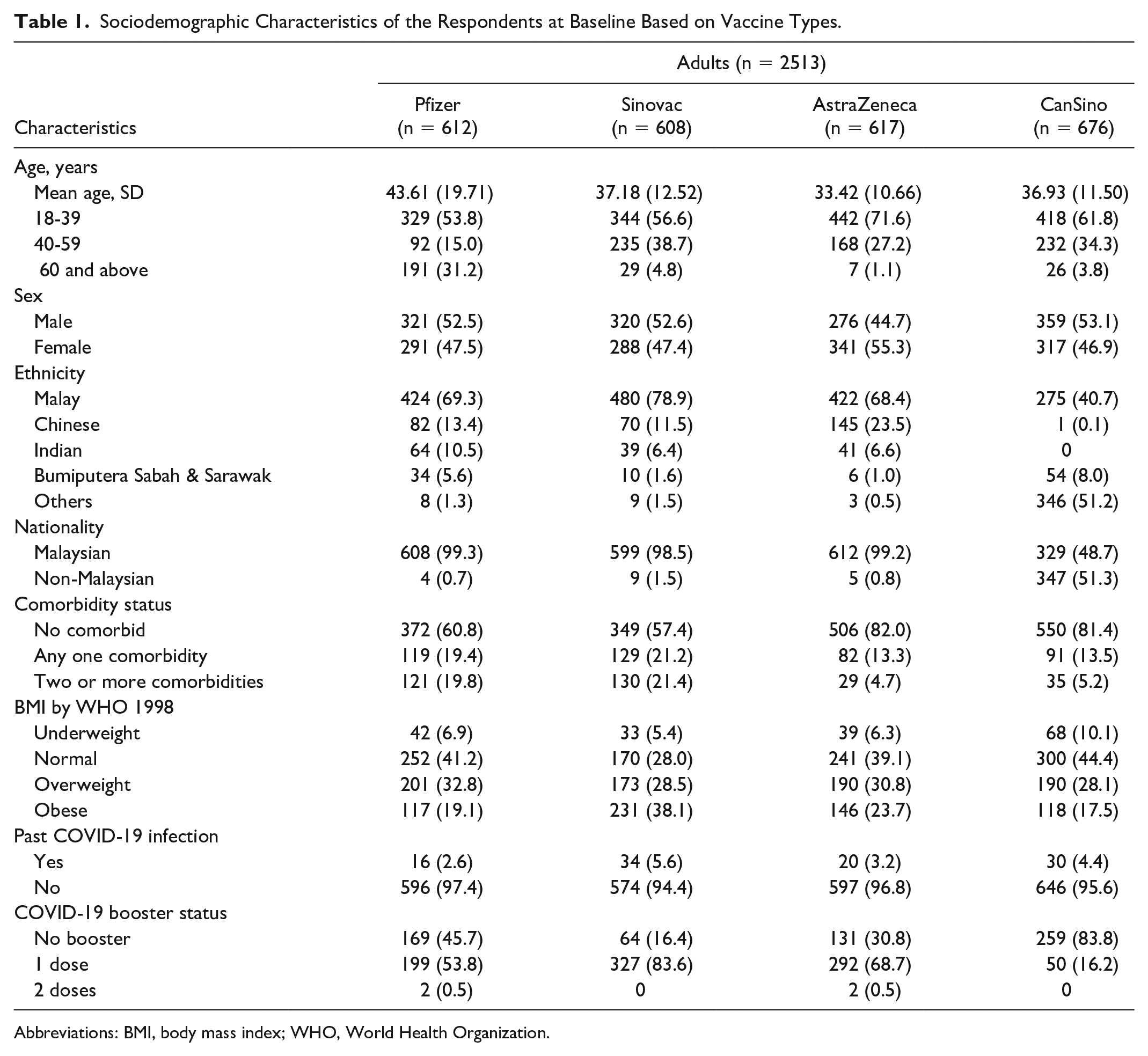
Abbreviations: BMI, body mass index; WHO, World Health Organization.
Antibody Detection by Follow-Ups
We analyzed the variations in antibody titres and seropositivity against the novel coronavirus by detecting the anti-S IgG from respondents in Table 2. A total of 2513 respondents were recruited at baseline, while only 1118 (44.5%) respondents remained in the final 12th-month follow-up. The seroconversion during the baseline follow-up showed the infection status prior to vaccine among all respondents.
S-Antigen Neutralizing Antibody Seroconversion Rate and Titres From Baseline to 12th Months After Vaccination Follow-Up (After 6th Months Based on Booster Dose).
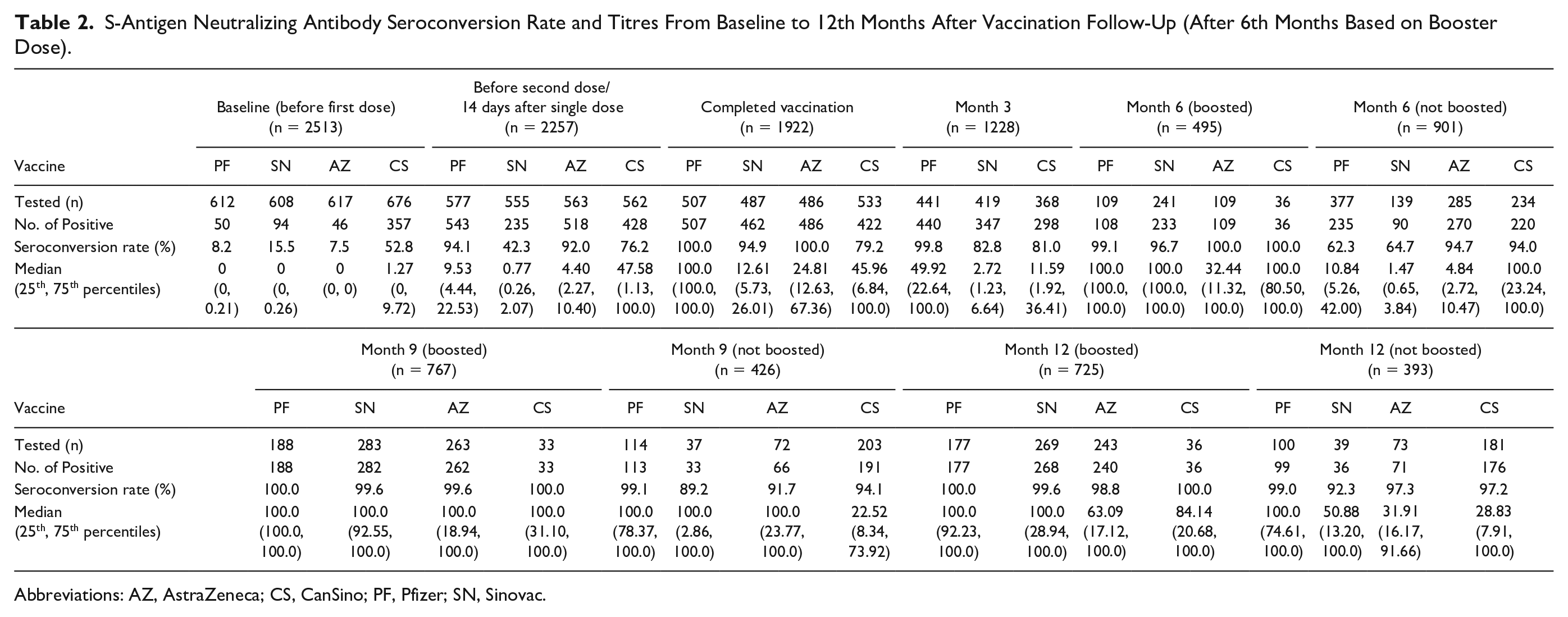
Abbreviations: AZ, AstraZeneca; CS, CanSino; PF, Pfizer; SN, Sinovac.
Associated Factors of SARS-CoV-2 Anti-S-IgG Level at 12th-Month Follow-Up
Table 3 presents the results of the Mann-Whitney U and Kruskal-Wallis test to determine the association of factors and antibody titer, along with simple linear and multiple linear regression to identify the associated factors.
Associated Factors of SARS-CoV-2 Anti-Spike-IgG Level in Index at 1-Year (n = 1118).
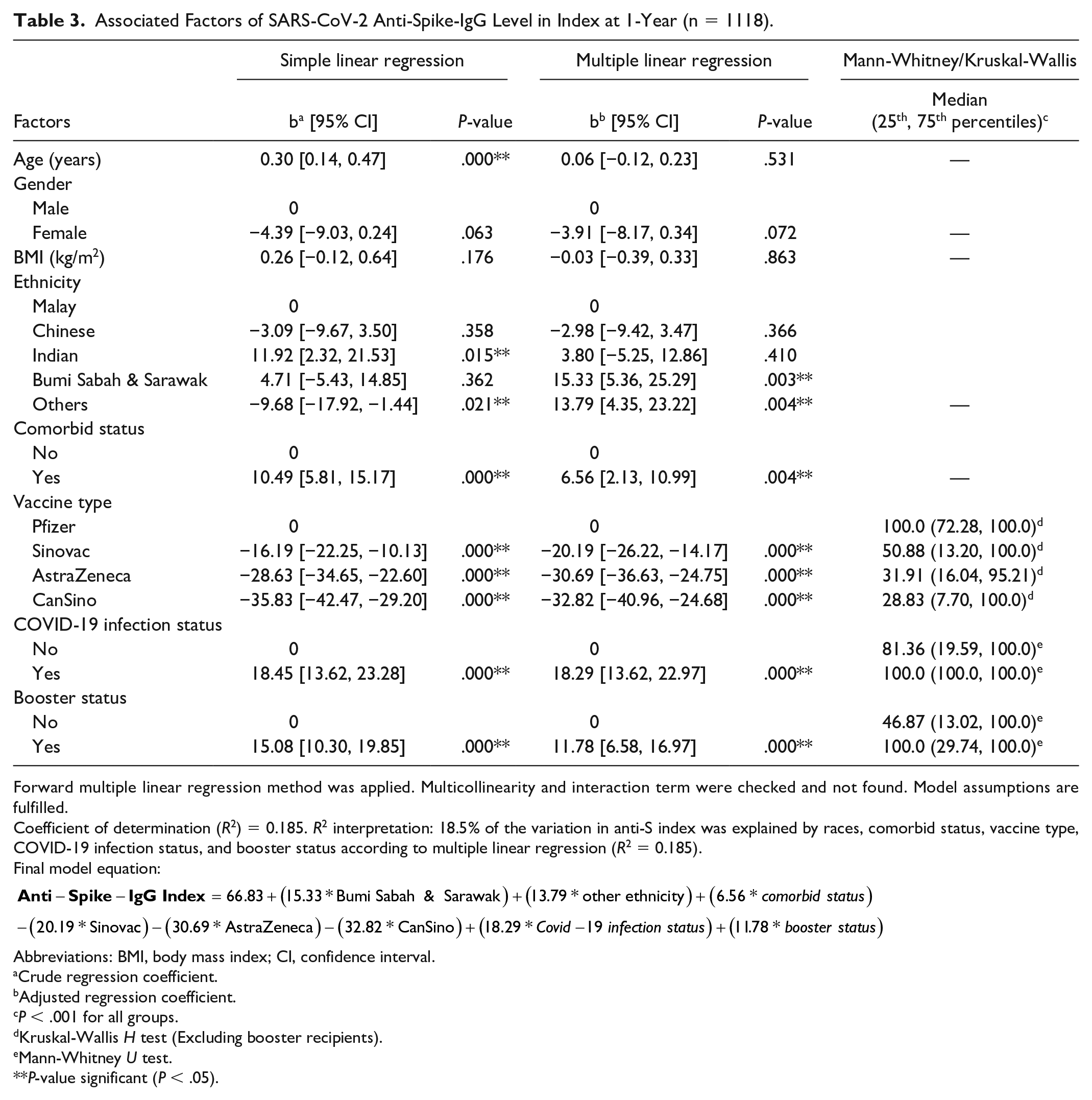
Forward multiple linear regression method was applied. Multicollinearity and interaction term were checked and not found. Model assumptions are fulfilled.
Coefficient of determination (R2) = 0.185. R2 interpretation: 18.5% of the variation in anti-S index was explained by races, comorbid status, vaccine type, COVID-19 infection status, and booster status according to multiple linear regression (R2 = 0.185).
Final model equation:
Abbreviations: BMI, body mass index; CI, confidence interval.
Crude regression coefficient.
Adjusted regression coefficient.
P < .001 for all groups.
Kruskal-Wallis H test (Excluding booster recipients).
Mann-Whitney U test.
P-value significant (P < .05).
When looking at the association between anti-S antibodies and booster status, individuals who received a booster had significantly higher median antibody levels at 12th-month follow-up compared with those who did not receive a booster.
From the simple linear and multiple linear regression, it is shown that Bumi Sabah and Sarawak ethnicity was 15.33 times higher in anti-S index in comparison with Malay, while Others ethnicity reported 13.79 times higher in anti-S index. Those with comorbidity was 6.56 times higher anti-S index compared with those without any comorbidity. Vaccine recipients of Sinovac, AstraZeneca and CanSino reported lower anti-S index in comparison with Pfizer recipients with regression coefficient of −20.19, −30.69 and −32.82, respectively. Those with COVID-19 infection and received booster have higher anti-S index compared with those aren’t.
Overall, these results suggest that anti-S antibody levels are associated with types of vaccine, comorbidity status, infection status, and booster status.
Discussion
In this study, we analyzed 2513 adult vaccine recipients in 12 months of follow-up to see the seropositivity rate and the pattern of anti-S antibodies level in every type of vaccine. After one year, the number of respondents compared to 1118. We found that in general, more than 98% of the respondents were still positive after one year among the boosted recipients and more than 92% of the respondents were positive among those who did not receive the booster. The highest median among those who were boosted was 100.0 (92.23-100.00) and 100.0 (74.61-100.00) among the nonboosted at the 12th-month follow-up.
From the findings, we could see the factors associated with anti-S antibody level were types of vaccine, comorbid status, infection status and booster status. Infection status made a significant difference in the anti-S antibody levels at all time points and across all types of vaccines with higher antibody levels among those who had been infected and respondents who received Pfizer vaccination had a higher level of antibody. This is similar findings found in a study done in Indonesia that showed receiving mRNA vaccine and being infected were associated with the antibody level. 13
The anti-S antibody among all the vaccines seemed to decline starting at third months after vaccination. This is similar to studies done previously that showed the antibody level decreased at 3 months after vaccination in 60% of the sample who received Pfizer vaccination.14,15 In our study, the level of anti-S antibody was increased in sixth-month follow-ups and stable afterward even among the nonboosted recipients. However, the median of anti-S antibody level was higher among the boosted recipients. The possible reasonable explanation for the decline of the anti-S antibody during the third month of follow-ups and increased again is that the immune system is adapting and transitioning from an initial robust response to a more stable and long-term response and memory B cell are generated. 16
In our study, some of the respondents have already been infected prior to their vaccination, and there were some respondents that were infected after one dose and after completing the vaccination. The anti-S antibodies among those who had been infected with COVID-19 regardless of the time points seemed to be higher as compared with those who were never been infected. After a natural infection, the immune system generates memory B cells and memory T cells specific to the virus. Memory B cells remember the virus and can produce antibodies quickly if the person is exposed to the virus again. This memory response contributes to sustained antibody levels.9,17 -19
After some time from the initial COVID-19 vaccination, the levels of antibodies that provide protection against the virus may start to decline. This is according to the studies done during the early phase of the vaccination worldwide.14,15,18,20,21 Thus, the suggestion to take booster vaccination was introduced and implemented.14,15,22 Booster doses of COVID-19 vaccines are designed to provide an additional level of protection against the virus, especially in the face of emerging variants and waning immunity over time. In our study, respondents who received boosters were shown to have higher anti-S antibody levels even after one year. This result was comparable with other studies; for example, a study done among health care workers in Russia showed that the antibody level significantly increased among the health care that received booster vaccination. 23 Booster dose also helped to lessen the complications and infection among young adults during the Omicron variant wave. 24 In general, booster doses can help provide an additional layer of protection against COVID-19 and help reduce the spread of the virus in the community.
The level of anti-S antibodies can also be affected by COVID-19 vaccines types. In this study, it was shown that respondents who received Pfizer tend to have higher antibody levels as compared with other types of vaccine recipients. In one study done in Southern Sarawak (Malaysia) who received Pfizer, Sinovac, and AstraZeneca, the median of anti-S antibody levels was higher among Pfizer recipients too. 25 This is probably caused by several factors such as antigenic effects; the different mechanisms of action and ways of production are likely to introduce additional variation to the characteristics of immune responses and possible adverse reactions. In our study, only the Pfizer vaccine uses mRNA technology. mRNA vaccine teaches our cells to make proteins to trigger immune responses inside of our bodies. 26 AstraZeneca and CanSino vaccines are using the adenovirus vector. Even though the complexity and the production process of those adenovirus-vector and mRNA vaccines are different, the intention of these vaccines is the same in which; to produce the native S proteins from a specific mRNA in the cells of the vaccine recipients.
However, in this study, it was reported that respondents with comorbid have higher anti-S antibody level unlike other studies.27 -29 The difference may be attributed to some residual confounding in the analysis.
The limitations of this study were the selection of respondents which was done purposively and we did not have any unvaccinated respondents to evaluate the efficacy and effectiveness of the vaccines which would add more valuable information on the vaccines. However, this study had a big number of respondents and compare the level of anti-S antibody among the four administered vaccines in Malaysia which has not been done in other studies.
Conclusion
Our study showed the high seropositivity and various level of anti-S antibodies among the four vaccine types after one year of vaccination. Respondents who received Pfizer showed the highest level of anti-S antibodies regardless of their booster status. Even though the level of anti-S antibodies among other vaccines is declining, the exact protective level against the virus is not known.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We would like to thank the Director General of Health Malaysia for his permission to publish this article.
