Abstract
The purpose of our study is to clarify the effect of microRNA-129-5p in the progression of human gastric cancer cells by regulating SPOCK1. The expression of microRNA-129-5p and SPOCK1 was tested by quantitative real-time polymerase chain reaction in tissues and cell lines. We validated the targeted relationship between microRNA-129-5p and SPOCK1 by dual luciferase reporter gene assay. 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide, colony formation, flow cytometry, transwell, and wound scratch assays were used to analyze the effects of microRNA-129-5p on SGC-7901 cell viability, proliferation, cell cycle and apoptosis, invasiveness, and migration. MicroRNA-129-5p was downregulated while SPOCK1 was upregulated in gastric cancer tissues and cell lines. The result of luciferase reporter gene assay demonstrated that microRNA-129-5p can target SPOCK1 by binding to the 3′untranslated region. The overexpression of microRNA-129-5p or the inhibition of SPOCK1 inhibited SGC-7901 viability, proliferation, migration, and invasion while promoted cell cycle arrest in G0/G1 stage and cell apoptosis. Our results suggested that microRNA-129-5p could directly specifically suppress SPOCK1, which might be one of the potential mechanisms in inhibiting cell processes including viability, proliferation, cell mitosis, migration, and invasiveness of gastric cancer cells.
Introduction
Gastric cancer (GC) is one of the most common human cancers featured with leading morbidity and lethality. 1 There are 679,100 new cases and 498,000 deaths per year in China and 26,370 estimated new cases and 10,730 deaths per year in the United States.2,3 Although a small portion of patients could benefit from molecular target therapeutic drugs, poor prognosis and outcomes of GC patients still exist because of the refractory characteristics and acquired drug resistance. 4 Tremendous advancement of GC studies and clinical surveillance has been made in past few decades; however, the underlying molecular mechanism of GC biology involving proliferation and invasion is still beyond thorough understanding. 5
Emerging evidence had shown that abnormal expression of microRNAs (miRNAs) is closely correlated with development and progression of various cancers. For instance, the expression of miR-148a was repressed in both GC cell lines and tissues, resulting from the hypermethylation of its promoter. 6 Moreover, it was unveiled that miR-409 directly targeted RDX and resulted in the inhibition of GC cell invasion and metastasis. 7 In addition, Sun et al. 8 identified miR-196a as a potential prognostic biomarker and therapeutic to stimulate cell cycle progression and proliferation of GC both in vitro and in vivo. Moreover, reports showed that three miR-129 family members exhibited low level in GC cell lines and functioned as tumor suppressors. 9 Du et al. 10 found that miR-129-1-3p might promote proliferation of GC cell BGC-823 cell line by targeting PDCD2 and BDKRB2. 11 It was reported that miR-129-2 led to overexpression of SOX4 and induced apoptosis in GC. 12 Besides, the migration and invasion of GC cells would be affected by miR-129-5p/interleukin 8 (IL-8) axis. 13 The functional pattern and regulatory mechanism of miR-129s in GC proliferation and metastasis still remain to be further studied.
Epithelial–mesenchymal transition (EMT) is characterized by a loss of cell junctions and mesenchymal characteristics instead of epithelial markers, which is related to embryonic development, acquired drug resistance, and is one of the main mechanisms through which tumor invasion and migration are promoted during the tumorigenesis.14–16 Sparc/osteonectin, cwcv and kazal-like domains proteoglycan 1 (SPOCK1), also named testican-1, is an EMT-related gene that encodes a matricellular glycoprotein, which is abundant in extracellular matrix (ECM).17,18 The upregulation of SPOCK1 was reported to promote the invasiveness of cancer cells in esophageal squamous cell carcinoma (ESCC) by inducing EMT signaling. 19 Furthermore, continuous activation of EMT signaling pathway by SPOCK1 was involved in acquired resistance to human epidermal growth factor receptor 2 (HER2)-directed treatment such as lapatinib in GC cell lines. 20 Another report acclaimed that the expression of SPOCK1 increased with the progression of human hepatocellular carcinoma (HCC) and indicated poor outcomes. SPOCK1 could be activated by CHD1L, which could then activate Akt signaling pathway to inhibit HCC cell apoptosis. 21 However, how SPOCK1 influences GC development and progression is beyond understanding.
In this study, we measured the levels of SPOCK1 and miR-129-5p in GC and adjacent tissue samples. Subsequently, we studied the negative effects of miR-129-5p on human GC cell lines. In addition, we examined the oncogenic role of SPOCK1 in migration and invasiveness of GC cells. Our work may render a novel perspective for the function of miR-129-5p/SPOCK1 axis in GC pathogenesis.
Materials and methods
Human tissue samples
In total, 30 paired tumorous and adjacent non-tumorous tissues were obtained from patients with primary GC according to the World Health Organization (WHO) classification of GC. The patients in this study received the surgical treatment in Hongqi Hospital of Mudanjiang Medical University from May 2013 to August 2015. No patients had undergone any pre-operative treatment, such as radiotherapy or chemotherapy. The tissue samples were promptly frozen in liquid nitrogen until subsequent experiments started. The study was approved by the ethics committee, and the informed consents were signed by all the participants.
Cell culture
Normal gastric mucosa epithelial cell line GES-1 and human GC cell lines AGS, SGC-7901, BGC-823, and MKN-45 (American Type Culture Collection (ATCC), Manassas, VA) were initially selected for the experiments. All cells were cultured in RPMI-1640 medium containing 10% fetal bovine serum (FBS; Gibco, Carlsbad, CA) in a humidified chamber with 5% CO2 at 37°C.
RNA isolation and quantitative real-time polymerase chain reaction
Total RNA was isolated from frozen GC tissues or cultured cells using TRIzol reagent (Invitrogen, Carlsbad, CA) following the instructions of manufacturer. The RNAs were then reverse-transcribed to complementary DNAs (cDNAs) using messenger RNA (mRNA) and miRNA Reverse Transcription Kit (TaKaRa, Beijing). The LightCycler 480 (Roche, Basel, Switzerland) was employed to conduct the polymerase chain reaction (PCR) reaction, and the used primers are listed in Table 1. The internal controls were U6 and glyceraldehyde 3-phosphate dehydrogenase (GAPDH), and the experimental data were analyzed by the 2–∆∆Ct method. 22
Primers used in qPCR.
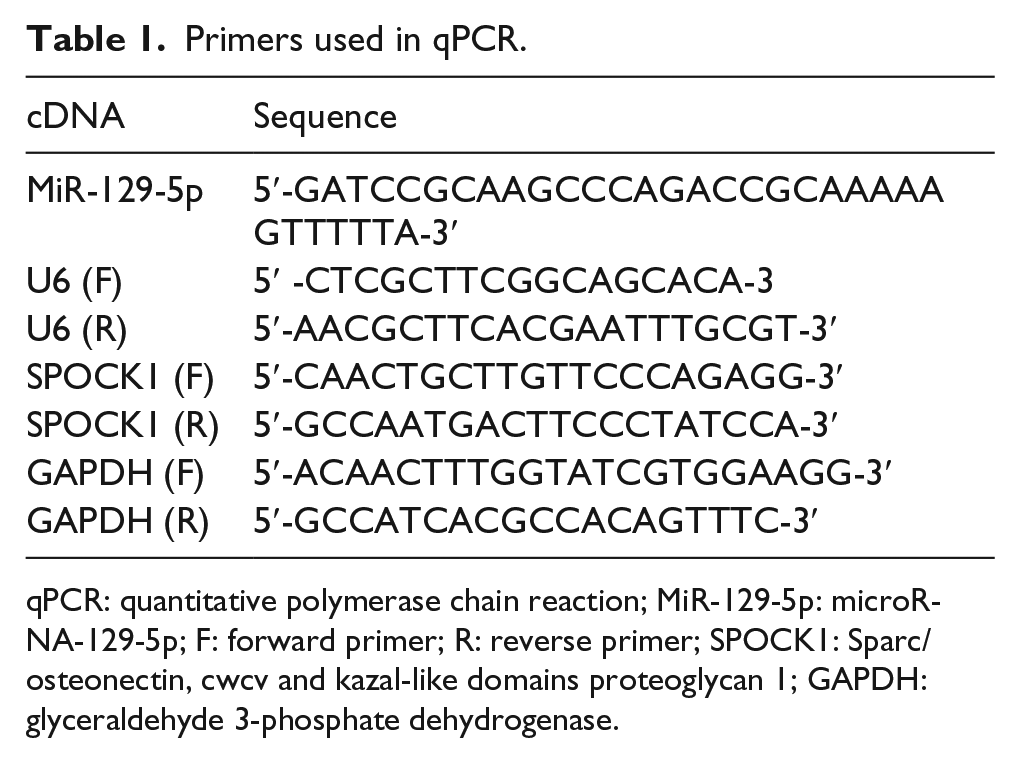
qPCR: quantitative polymerase chain reaction; MiR-129-5p: microRNA-129-5p; F: forward primer; R: reverse primer; SPOCK1: Sparc/osteonectin, cwcv and kazal-like domains proteoglycan 1; GAPDH: glyceraldehyde 3-phosphate dehydrogenase.
Cell transfection
SGC-7901 cells were seeded onto six-well plates at 30% concentration and cultured for 24 h. Then, miR-129-5p mimics (miR-mimics), mimic controls (miR-NC), small-interfering RNAs targeting SPOCK1 (SPOCK1-siRNA), overexpression SPOCK1 (SPOCK1-cDNA), siRNA controls (SPOCK1-NC), or miR-129-5p mimics + SPOCK1-cDNA (miR-mimics + SPOCK1-cDNA) (Shanghai GenePharma Co. Ltd, Shanghai) were transfected into cells with Lipofectamine 2000 following the instructions of the manufacturer (Invitrogen). After 48 h of transfection, the cells were harvested.
Dual luciferase reporter gene assay
Multisite-directed mutagenesis was used to mutate the 3′ untranslated region (3′UTR) of SPOCK1. The wild type and the mutant type of 3′UTRs were cloned in the luciferase reporter vectors. MiR-129-5p mimics or mimic controls were co-transfected with SPOCK1-wt or SPOCK1-mut 3′UTRs into cells using Lipofectamine 2000, respectively. After 48 h of transfection, the cells were assessed with Luciferase Assay Kit (Promega, Madison, USA).
Cell viability assay
SGC-7901 cell viability was tested by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) method according to the standardized protocol (Roche). Cell viability was assessed at 1, 2, 3, 4, and 5 days. Cells (2 × 103 cells/well) were seeded on 96-well plates with a medium of RPMI-1640 medium containing 10% FBS for overnight at 37°C. Then, 20 µL of MTT solution (Roche) was added to each well and incubated with cells for 4 h. Then, the cells were lysed in 150 µL of dimethyl sulfoxide (DMSO) to facilitate the dissolution of crystals. The optical density (OD) was detected every day at the same time using enzyme-linked immunosorbent assay.
Colony formation assay
SGC-7901 cells were trypsinized and suspended. Then, the cells (500 cells/well) were seeded onto 12-well plates and incubated for 2 weeks. The culture surface was washed with poly butylenes succinate (PBS) and stained using 5% crystal violet solution. Cell colonies that contained more than 50 cells were visible to naked eyes and therefore counted. Each experiment was done in triplicate.
Transwell assay
Cells (5 × 104 cells/well) were seeded in the upper transwell chamber (Corning Costar Corp., Corning, NY). The Transwell chamber was 8-µm pore size and pre-coated with 20 µL Matrigel (50 g/mL; BD Biosciences, Bedford, MA). Chambers were placed into 24-well plates, and 600 µL of RPMI-1640 medium containing 10% FBS was added to the lower chambers. After incubating for 36 h, the membrane was fixed in 4% paraformaldehyde and stained with 0.1% crystal violet. Finally, 12 different fields were randomly selected to calculate the number of cells penetrated across membrane under the microscope.
Wound-healing assay
After 48 h of transfection, the cells in each group were seeded onto six-well plates. On the second day, the cell layers in each well were scratched with a sterile pipette when the confluency reached approximately 80%. Then, the cells were cultured in RPMI-1640 medium (Gibco) comprising 2% FBS (Gibco) in a humidified incubator. We monitored the wound closure at 0 and 48 h after scratching.
Cell cycle analysis and apoptosis analysis
In cell cycle analysis, the cells in every group were digested with 0.25% trypsin. The digested cells were washed twice with PBS. Then, the cells were re-suspended in prepared sample buffer containing 20 µL of 5 µg/mL propidium iodide (PI) and 50 µL of 10 mg/mL RNase A. Then, the cells were incubated in the dark for 10 min at 37°C.
In cell apoptosis analysis, the cells were re-suspended in binding buffer (pH 7.4) including 100 mmol/L 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES), 100 mmol/L NaCl, and 25 mmol/L CaCl2 after washing with cold PBS twice. Subsequently, cells were stained with Annexin V–fluorescein isothiocyanate (FITC)/PI at 37°C in the dark for 15 min. Apoptotic cells were evaluated using flow cytometry (BD Biosciences). Each experiment was done in triplicate.
Western blotting analysis
The total protein was extracted, and the cytoplasmic proteins and nuclear proteins were isolated. The protein concentration was determined by bicinchoninic acid (BCA) protein quantitative method. After sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE), proteins were transferred to polyvinylidene difluoride (PVDF) membrane. Then, 5% skim milk was added to block the membrane for 2 h. Subsequently, primary antibodies of SPOCK1 and GAPDH (1:1000; Abcam, Cambridge, UK) were added to the membrane and incubated for overnight at 4°C. Then, horseradish peroxidase (HRP)-conjugated secondary antibodies were added to the membrane and incubated for 2 h. The film was then developed using enhanced chemiluminescence (ECL). The internal control was GAPDH.
Statistical analysis
All statistical analyses were accomplished using SPSS 22.0 and GraphPad prism 6.0 (GraphPad Software Inc., San Diego, CA). Measurement data were recorded as the mean ± standard deviation (SD), which was analyzed by t test or one-way analysis of variance (ANOVA). p < 0.05 was statistically significant.
Results
MiR-129-5p was downregulated in GC tissues and cells
The miR-129-5p levels in GC tumorous tissues and adjacent non-tumorous tissues were detected by quantitative real-time PCR (qRT-PCR), and the results suggested that the expression levels of miR-129-5p in GC tumorous tissues were significantly attenuated compared with adjacent non-tumorous tissues (p < 0.05, Figure 1(a)). MiR-129-5p expression was remarkably downregulated in all GC cancerous cells AGS, SGC-7901, BGC-823, and MKN-45 compared with the normal cell GES-1 (p < 0.05, Figure 1(b)). The succeeding experiments were conducted in SGC-7901 cells in which miR-129-5p expression was the lowest.

The expression of miR-129-5p and SPOCK1 decreased in GC tissues and cells. (a) MiR-129-5p was downregulated in GC tissues in comparison with normal tissues. Data are expressed as mean ± SD (*p < 0.05 compared with adjacent tissues). (b) MiR-129-5p was downregulated in GC cell lines compared with normal gastric cell line GES-1 (*p < 0.05 compared with GSE-1 cell line). (c) SPOCK1 mRNA was upregulated in GC tissues compared with adjacent tissues. Data are expressed as mean ± SD (*p < 0.05 compared with adjacent tissues). (d) SPOCK1 mRNA was upregulated in GC cell lines compared with normal gastric cell line GES-1. Data are expressed as mean ± SD (*p < 0.05 compared with GSE-1 cell line). (e) SPOCK1 protein was upregulated in gastric cancer tissues compared with normal tissues. (f) SPOCK1 protein was upregulated in GC cell lines compared with normal gastric cell line GES-1.
SPOCK1 was upregulated in GC tissues
The expression of SPOCK1 mRNA in GC tumorous tissues and adjacent non-tumorous tissues was detected by qRT-PCR, and the results demonstrated a significant increase of SPOCK1 mRNA in GC tissues compared with adjacent tissues (p < 0.05, Figure 1(c)). SPOCK1 expression was remarkably upregulated in all GC cells AGS, SGC-7901, BGC-823, and MKN-45 compared with normal cell GES-1 (p < 0.05, Figure 1(d)D). The expression level of SPOCK1 protein was measured by western blotting, and the results showed a significant upregulation of SPOCK1 protein in cancer tissues and cells (p < 0.05, Figure 1(e) and (f)).
SPOCK1 was the target gene of miR-129-5p
The online database TargetScanHuman 7.0 predicted that SPOCK1 was a target gene of miR-129-5p with the possible binding sequence GCAAAAA in SPOCK1 3′UTR (Figure 2(a)). The luciferase activity of SPOCK1-wt 3′UTR + miR-129-5p declined substantially (p < 0.05) while that of SPOCK1-mut 3′UTR + miR-129-5p did not show significant difference from the SPOCK1-mut 3′UTR group (p > 0.05, Figure 2(b)).

SPOCK1 was specifically targeted by miR-129-5p. (a) SPOCK1 mRNA is predicted to be a target of miR-129-5p, and the complimentary sequences were illustrated. The SPOCK1-mut 3′UTR was also constructed using site mutation method and the mutated sequence was shown here. (b) Luciferase activity of SGC-7901 cells transfected with miR-129-5p mimics and wild-type SPOCK1 3′UTR was significantly reduced compared with the miR-129-5p control + wild-type SPOCK1 3′UTR group. Whereas the luciferase activity of SPOCK1-mut 3′UTR + miR-129-5p did not show significant difference from the SPOCK1-mut 3′UTR group. Data are expressed as mean ± SD (*p < 0.05 compared with the miR-NC group). (c) SPOCK1 translation was suppressed in the miR-129-5p mimics group.
The depression effect of miR-129-5p on SPOCK1 was evaluated at protein levels. SPOCK1 protein level was downregulated in SGC-7901 cells transfected with miR-129-5p mimics and SPOCK1-siRNA, while the protein level of SPOCK1 was upregulated in the SPOCK1-cDNA group compared with the NC and control groups (p < 0.05). SPOCK1 protein level had no significant difference in SGC-7901 cells transfected with miR-129-5p mimics + SPOCK1-cDNA compared with the miR-NC group, SPOCK1-NC group, and control group (p > 0.05, Figure 2(c)).
MiR-129-5p inhibited the viability and proliferation of GC cells
MTT assay indicated that cells transfected with miR-129-5p mimics and SPOCK1-siRNA had significantly lower viability, whereas cells transfected with SPOCK1-cDNA had higher viability compared with those transfected with corresponding controls (p < 0.05, Figure 3(a)). No significant difference was found among the cells transfected with control, miR-NC, and SPOCK1-NC and co-transfected with miR-129-5p mimics and SPOCK1-cDNAs (p > 0.05).

MiR-129-5p inhibits the viability, propagation, migration, and invasiveness of GC cells. (a) MTT assay demonstrated that the overexpression of miR-129-5p or the inhibition of SPOCK1 inhibited the viability of SGC-7901 cells. (b) Colony formation assay demonstrated that the overexpression of miR-129-5p or the inhibition of SPOCK1 inhibited SGC-7901 cell proliferation. (c) Wound-healing assay demonstrated that the overexpression of miR-129-5p and the inhibition of SPOCK1 both inhibited the migration of SGC-7901 cells. (d) Transwell assay demonstrated that the overexpression of miR-129-5p and the inhibition of SPOCK1 both inhibited the invasiveness of SGC-7901 cells. Data are expressed as mean ± SD (*p < 0.05 compared with the control group).
Similar to MTT results, cells transfected with miR-129-5p mimics and SPOCK1-siRNAs formed fewer colonies, while cells formed more colonies in the SPOCK1-cDNA group compared with those transfected with corresponding controls (p < 0.05, Figure 3(b)), demonstrating that the overexpression of miR-129-5p and the inhibition of SPOCK1 could both inhibit SGC-7901 cell proliferation. No significant difference was found among the control, miR-NC, SPOCK1-NC, and the miR-129-5p mimics + SPOCK1-cDNA groups (p > 0.05).
MiR-129-5p inhibited the migration of GC cells
The wound-healing assay results demonstrated that cells transfected with miR-129-5p mimics and SPOCK1-siRNA had lower degree of wound closure than the control group, the miR-NC group, and the SPOCK1-NC group (p < 0.05, Figure 3(c)), suggesting that the miR-129-5p overexpression and SPOCK1 inhibition could both inhibit SGC-7901 cell migration. Cells in the SPOCK1-cDNA group had higher degree of wound closure in contrast with the corresponding control group. There was no significant difference among the control, miR-NC, SPOCK1-NC, and miR-129-5p mimics + SPOCK1-cDNA groups (p > 0.05).
MiR-129-5p inhibited the invasion of GC cells
The Transwell assay results indicated that cells transfected with miR-129-5p mimics and SPOCK1-siRNA had weaker invasiveness compared with the control cells, whereas SPOCK1-cDNA had stronger invasiveness (p < 0.05, Figure 3(d)). No remarkable difference was observed among the control, the miR-NC, SPOCK1-NC, and miR-129-5p mimics + SPOCK1-cDNA groups (p > 0.05).
The effects of miR-129-5p on cell cycle and apoptosis of GC cells
Results of flow cytometry demonstrated that the proportion of SGC-7901 cells in the G0/G1 phase increased while that in the S phase decreased in the miR-129-5p mimics and SPOCK1-siRNA groups compared with the control group, the miR-NC group, and the SPOCK1-NC group (p < 0.05, Figure 4(a)). The overexpression of SPOCK1 led to more SGC-7901 cell arrest at S and G2/M phase compared with the corresponding control group. However, cells transfected with miR-mimics + SPOCK1-cDNA had no significant effects on cell cycle progression in contrast with the control, the miR-NC, and the SPOCK1-NC groups (p > 0.05).
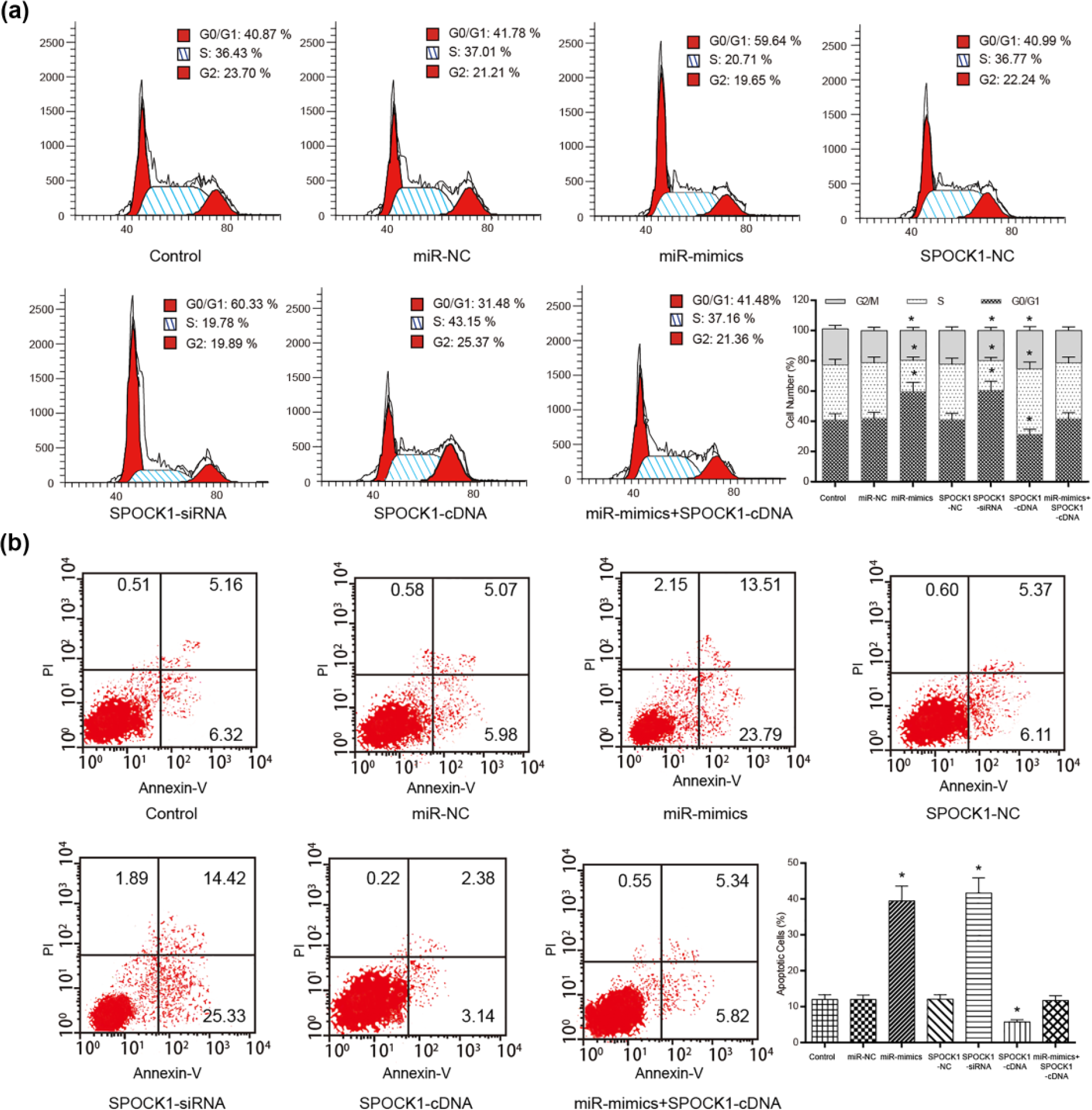
MiR-129-5p affected the mitosis and apoptosis of SGC-7901 cells. (a) The overexpression of miR-129-5p or the inhibition of SPOCK1 increased the proportion of SGC-7901 cells in G0/G1 phase. (b) The results of cell apoptosis assay conducted using flow cytometry suggested that the overexpression of miR-129-5p or the inhibition of SPOCK1 could facilitate cell apoptosis. Data are expressed as mean ± SD (*p < 0.05 compared with the control group).
SGC7-901 cells transfected with miR-129-5p mimics and SPOCK1-siRNA facilitated cell apoptosis, whereas cells transfected with SPOCK1-cDNA suppressed cell apoptosis compared with the cells in the corresponding control group (p < 0.05, Figure 4(b)). No notable altered apoptosis rate was seen after the co-transfection of miR-129-5p mimics + SPOCK1-cDNA in contrast with the control, the miR-NC, and the SPOCK1-NC groups (p > 0.05).
Discussion
Recently, the effects of miRNA dysfunction in modulating progression and prognostic outcome in GC had been discussed extensively.23,24 We herein identified miR-129-5p as a novel molecular marker of GC cancer. A previous study demonstrated that miR-129-1-3p and miR-129-2-3p, members of miR-129 family, were aberrantly expressed in GC patients and can be potential biomarkers for screening GC in a non-invasive manner. 25 MiR-129-5p was found to be downregulated in GC tumorous tissues compared with the adjacent tissues, indicating that miR-129-5p might play as a tumor suppressor in GC. We then further investigated whether the cell abilities of GC cell lines can be modulated by miR-129-5p. Our study discovered that the miR-129-5p mimics repressed the viability, migratory ability, and invasiveness of SGC-7901 cell lines. In addition, a larger proportion of GC cells were arrested in G0/G1 phase and the apoptosis rate increased substantially in the miR-mimics group, compared with the miR-NC group. Consistent with our findings, Yu et al. 9 discovered that miR-129-5p functioned as a tumor suppressor gene in the regulation of GC cells, and evidences were provided that the restoration of miR-129 did induce cell death and cycle arrest.12,26
SPOCK1, an EMT-related gene which is also labeled as testican-1, had been reported to be involved in tumorigenesis and invasiveness.19,21 In the specimens of GC patients, we found that the expression of SPOCK1 was upregulated at both transcriptional and translational levels, indicating that SPOCK1 might function as a tumor facilitator. The knockout of SPOCK1 tremendously inhibited the viability, propagation, migration, and invasiveness of GC cells, while the overexpression of SPOCK1 had the opposite trend compared with the corresponding control groups. These findings were in accordance with the work done by Shu et al., 27 who demonstrated that SPOCK1 could promote the proliferation and migration of gallbladder cancer cells by activating the phosphoinositide 3-kinase (PI3K)/AKT pathway. In addition, the flow cytometry analysis results showed that a large proportion of cells in the SPOCK1-siRNA group were arrested in the G0/G1 phase and the cell apoptosis rate rose significantly in comparison with the SPOCK1-NC group. We thus inferred that the knockout of SPOCK1 could lead to the suppression of GC.
We have confirmed the target relationship between miR-129-5p and SPOCK1 by the dual luciferase reporter gene assay. A series of biological assays in this study were conducted to investigate the effects of miR-mimics + SPOCK1-cDNA on GC cell activities. The results suggested that the co-transfection of miR-mimics and SPOCK1-cDNA demonstrated no significant difference from the miR-NC, control, and the SPOCK1-NC groups in terms of the SPOCK1 expression, cell viability, wound closure, invasiveness, and mitosis but showed substantial difference from the SPOCK1-siRNA and the miR-mimics groups.
Taken together, we hypothesize that miR-129-5p could negatively regulate the progression of GC by suppressing the oncogene SPOCK1. Although several studies argued that the abnormal expression of miRNAs could be either a foe or a friend in regulating cell activities, it mainly depends on the tendency toward oncogenesis of the target genes of these miRNAs in GC.28–32 Therefore, it is imperative to comprehensively illustrate the regulatory effects of miR-129-5p. By knowing that we intended to investigate the interaction between miR-129-5p and its other targets, together with the mechanism through which they regulate the oncogenesis and the progression of GC in our further study. It is notable that numerous available evidence suggested that transforming growth factor beta 1 (TGF-β1)/Smad signaling pathway was the key regulatory mechanism in tumor growth and metastasis by stimulating EMT,33,34 and SPOCK1 is the downstream responsive factor of TGF-β signaling cascades. Thus, we speculated that SPOCK1 might get involved in the GC tumor growth and metastasis by regulating TGF-β1/Smad signaling pathway and regarded this point as our next investigation focus.
Conclusion
In summary, our data demonstrated the significance of miR-129-5p/SPOCK1 activity in GC cells. We illustrated that miR-129-5p could regulate GC cells by targeting SPOCK1. In addition, we speculated that miR-129-5p may serve as a suppressor for GC progression by downregulating the expression of SPOCK1 at both the mRNA and protein levels. Understanding this fact of GC will not only help identify SPOCK1 as a novel GC therapeutic target but also contribute to the acknowledgement of the pathogenesis of a disease that remains aggressive.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
