Abstract
We investigated the expression and function of miR-409-5p in human breast cancer. Quantitative real-time polymerase chain reaction was conducted to evaluate endogenous miR-409-5p expression in breast cancer tumors and breast cancer cell lines. Lentiviral transduction was performed to stably downregulate miR-409-5p in breast cancer cell lines MDA-MB-231 and MCF-7 and cells. The effects of miR-409-5p downregulation on breast cancer proliferation, migration, and xenograft development were then evaluated. Downstream target gene of miR-409-5p, Ras suppressor protein 1, was examined by dual-luciferase activity assay, quantitative real-time polymerase chain reaction, and western blot in lentiviral-transduced breast cancer cells. Ras suppressor protein 1 was also inhibited in miR-409-5p-downregulated breast cancer cells to examine its functional effect on breast cancer proliferation and migration. MiR-409-5p was aberrantly upregulated in both breast cancer tumors and cell lines. Lentiviral transduction successfully downregulated endogenous miR-409-5p expression as well as suppressed proliferation, migration, and xenograft development in MDA-MB-231 and MCF-7 cells. Ras suppressor protein 1 was confirmed to be directly targeted by miR-409-5p in breast cancer cells. Small interfering RNA–mediated Ras suppressor protein 1 inhibition reversely promoted cancer proliferation and migration in miR-409-5p-downregualted breast cancer cells. MiR-409-5p is downregulated in breast cancer and its inhibition has anti-cancer effect on breast cancer development both in vitro and in vivo. The regulatory effect of miR-409-5p inhibition is likely through the inverse upregulation of Ras suppressor protein 1 in breast cancer.
Keywords
Introduction
Breast cancer (BC) is one of the most malignant gynecological cancers in the world. In developed country of United States, BC is the most prevalent form of carcinomas among female patients, accounting for more than three million women who are currently diagnosed with or have had history of BC. 1 In developing countries, such as China, BC is also the most prevalent form of female cancers and the leading candidate for cancer-related mortality among young and middle-aged patients.2,3 Thus, it is important to understand the underlying molecular mechanisms responsible for BC carcinogenesis, maturation, and metastasis in order to provide early diagnosis and efficient treatment strategies for patients with BC.
MicroRNAs (miRNAs) are families of evolutionally conserved non-coding, small size, single-stranded RNAs that post-transcriptionally subdue gene transcription and suppress protein production by deactivating downstream target genes through the binding on their 3′-untranslated regions (3′-UTRs).4–6 Through both clinical and laboratory studies, miRNAs have been demonstrated to play critical roles in regulating pathogenesis, chemo-pharmacology, cancer development, and cancer apoptosis in human BC.7–9 For instance, miR-222 was demonstrated to regulate chemotherapy drug–resistance of BC partly through the inhibition of phosphatase and tensin homolog (PTEN)/Akt/FOXO1 signaling pathway. 10 In addition, miR-31 was shown to suppress triple-negative BC migration and invasion through the inhibition on downstream target gene of special AT-rich sequence-binding protein 2 (SATB2). 11
Among many of the human carcinoma-associated miRNAs, mi-409-3p, the member of the human mature microRNA-409 (miR-409) family, was shown to be highly expressed in plasma of BC patients.12,13 In addition, studies had demonstrated that miR-409-3p was aberrantly downregulated in breast carcinomas of BC patients, acting as BC prognostic biomarker and have tumor-suppressing effect on BC cell progression and invasion through the targeting on Akt1 gene.14,15 Interestingly, another member of miR-409 family, miR-409-5p, was also reported to be present in BC carcinoma and suggested to be negatively associated with BC–specific survival. 16 However, it is still unclear what the exact expression profile of miR-409-5p is or what the functional roles miR-409-5p may have in BC.
Human gene of Ras suppressor protein 1 (RSU1) was originally characterized as a transformation suppressor during the cloning process of Ras oncogene. 17 Subsequent studies demonstrated that overexpression of RSU1 had tumor suppressive effects in glioblastoma and BC by inhibiting anchorage-independent cancer cell proliferation.18,19 Specifically, in human BC, RSU1 was shown to stabilize Rsu1-PINCH1-ILK-parvin complex to suppress cancer cell migration. 20 However, little is known whether there is any upstream activator to initiate the anti-tumor effect of RSU1 in BC.
Materials and methods
Ethical statement
This study was approved by the Clinical Research & Ethic Committees at China-Japan Union Hospital of Jilin University and The First Hospital of Jilin University in Changchun China. All participating patients signed consent forms. All protocols in the study were conducted in accordance with the Declaration of Helsinki and the general principles of good medical practice in China.
BC in situ tumors and in vitro cell lines
From December 2012 to June 2016, in situ epithelial breast carcinoma tissues (carcinoma) were surgically obtained from 113 patients who were diagnosed with BC and underwent segmental mastectomy at China-Japan Union Hospital of Jilin University and The First Hospital of Jilin University in Changchun China. Adjacent non-carcinoma epithelial breast tissues (healthy, at least 3 cm away from the clear edge of carcinoma tissues) were also surgically from those patients. All in situ samples were immediately snap-frozen in liquid nitrogen after surgery and stored in a bio-grated freezer (−80 °C) until RNA extraction.
Also in this study, in vitro breast carcinoma cell lines, MDA-MB-231, MDA-MB-451, MCF-7, CAMA-1, SK-BR-3, MB-157, HCC1806, UACC-732, UACC-3199, and ZF-75-1, and an epithelial non-carcinoma breast cell line, MCF10A, were all commercially obtained from American Type Culture Collection (ATCC, Beijing, China). All in vitro cell lines were maintained in cell culture medium of Dulbecco’s Modified Eagle’s Medium (DMEM; Thermo Fisher Scientific, USA) supplemented with 10% fetal bovine serum (FBS; Thermo Fisher Scientific) and antibiotic cocktail of PenStrep (100 U/mL penicillin + 100 µg/mL streptomycin; Thermo Fisher Scientific). All cell cultures were kept in a bio-grated tissue culture chamber with 5% CO2 at 37°C.
RNA extraction and quantitative real-time polymerase chain reaction
High-quality and purified RNA was isolated from in situ breast carcinoma tumors or in vitro BC cell lines using a PureLink RNA Mini Kit (Thermo Fisher Scientific) according to manufacturer’ recommendation. A volume of 10 µg RNA of each sample was then retrotranscribed into complimentary DNA (cDNA) using a High-Capacity cDNA Reverse Transcription Kit (Applied Biosystems, USA) according to manufacturer’ recommendation. Quantitative real-time polymerase chain reaction (qRT-PCR) was conducted on a 7900HT Fast Real-Time PCR system (Applied Biosystems). For miR-409-5p measurement, a TaqMan microRNA Assay (Applied Biosystems, USA) was performed and a non-coding RNA SNORD66 was used as loading control. For RSU1 gene measurement, a Brilliant SYBR-Green qRT–PCR kit (Stratagene, USA) was used, and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as loading control. Relative fold changes of gene expressions were then quantified using the 2−ΔΔCt method.
Lentivirus transduction of miR-409-5p inhibitor in BC cells
In this study, lentiviral transduction was used to downregulate the endogenous gene expression of miR-409-5p in in vitro BC cell lines. A lentivirus containing the synthetic oligonucleotides of human mature miR-409-5p inhibitor, L/miR409I, and a control lentivirus containing the non-specific inhibitor oligonucleotides, L/I, were both commercially obtained from Shanghai GenePharma (Shanghai, China). MDA-MB-231 and MCF-7 cells were transduced with either L/miR409I or L/I, along with 8 µg/mL polybrene, for 48 h at multiplicity of infection (MOI) of 20–25. Purification was conducted by treating post-transduction MDA-MB-231 and CCF-7 cells with 1 mg/mL blasticidin for 1 week. After that, healthy cell colonies were collected and replated in 24-well plate. MDA-MB-231 and CCF-7 cells were then passaged for at least 3–5 times, followed by qRT-PCR to evaluate transduction efficiency.
Cancer proliferation assay
MDA-MB-231 and MCF-7 cells were replated in 96-well plate (3500 cells/well) and their in vitro proliferation was monitored by a Cell Proliferation Kit I (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT); MilliporeSigma, Shanghai, China) according to manufacturer’s recommendation. Cells were maintained in culture for 5 days. At interval of every 24 h, 15 µL MTT stock solution was diluted into each well (total volume of 100 µL) for 4 h at 37°C. After that, culture medium was removed and MTT-treated cell culture was processed with HCl-SDS to solubilize formazan product for 2 h at room temperature; 96-well plate was then placed in a Synergy 2 Multi-Mode Microplate Reader (BioTek, USA) and luminescence was read at optical density (OD) of 570 nm.
Cancer migration assay
The migrating capability of BC was measured using a QCM 24-Well Colorimetric Cell Migration Assay (MilliporeSigma) according to manufacturer’s recommendation. Briefly, MDA-MB-231 and MCF-7 cells were plated in the upper chamber (1 × 106 cells/mL) in 300 µL serum-deprived culture medium (0% FBS). The lower chamber was filled with 500 µL regular culture medium (10% FBS) as chemoattractant. After 24 h, upper chamber and culture medium were removed. Lower chambers were quickly fixed by 70% ethanol and treated with 0.1% crystal violet for 2 h. MDA-MB-231 and MCF-7 cells migrated into lower chambers were imaged under an inverted fluorescent microscope. In addition, luminescence of each lower chamber was measured as OD of 560 nm. Relative migration was characterized by normalizing lower chambers’ luminescences against the luminescence under control condition.
In situ xenograft assay
In vivo development of BC xenograft was evaluated using the method described in a previous study. 21 Briefly, lentiviral-transduced MDA-MB-231 cells were subcutaneously injected into left flanks of 4-week-old BALB/C nude mice (1 × 106 cells/injection). In situ MDA-MB-231 xenografts were allowed to grow for 5 weeks. At interval of 1 week, length (l, milometer, mm) and width (w, milometer, mm) were measured for the xenograft and its volume (V, milometer, mm) was calculated by the equation V = l × w × w/2. At the end of xenograft assay, mice were sacrificed. MDA-MB-231 xenografts were extracted and their images were taken under an upright broad-band microscope.
Dual-luciferase activity assay
3′-UTR of human wild-type (Wt) RSU1 gene was amplified from a human cDNA library using regular PCR. It was inserted between XhoI and BamHI restriction enzymes in a pcDNA3.1-luc plasmid (Addgene, USA) to create a firefly luciferase vector of RSU1-3′UTR (Wt). The putative miR-409-5p binding site on RSU1 3′-UTR was mutated by a Site-Directed Mutagenesis Kit (SBS Genetech, China). The mutant (Mu) 3′-UTR was also inserted into pcDNA3.1-luc to create another firefly luciferase vector of RSY1-3′-UTR (Mu). Lentiviral-transduced MDA-MB-231 and MCF-7 cells were replated in 24-well plate and then co-transfected with either RSU1-3′-UTR (Wt) or RSU1-3′-UTR (Mu). A control luciferase vector pSV40-Renilla plasmid was also co-transfected into BC cell lines; 48 h after transfection, relative luciferase activities were characterized as the ratio of firefly/Renilla signals using a dual-luciferase reporter system (Promega, USA) according to manufacturer’s recommendation.
Western blot assay
MDA-MB-231 and MCF-7 cells were lysed by a Tris-based lysis buffer (Thermo Fisher Scientific). Equal amount of protein (20 µg) was drawn from each sample cell lines and separated on 12% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) gel (Thermo Fisher Scientific). After transferring proteins into a nitrocellulose membrane, primary antibody against RSU1 (rabbit polyclonal, 1:500; Abcam, USA) was applied to the membrane for 24 h at 4°C. Control membrane was probed by β-actin primary antibody (rabbit polyclonal, 1:2000; Abcam, USA). Then, horseradish peroxidase–conjugated secondary antibody was applied for 2 h at room temperature. Western blot was then visualized using an enhanced chemiluminesence system (Amersham Biosciences, USA).
RSU1 inhibition assay
Human RSU1 gene-specific small interfering RNA (siRNA), Si-RSU1, was commercially purchased from SunBio Technology (Nanjing, China). A scrambled non-specific siRNA, Si/C (SunBio Technology), was used as control siRNA. In L/miR409I-transduced MDA-MB-231 and MCF-7 cells, transfection of si-RSU1 or si/C was performed using Lipofectamine 3000 reagent (Thermo Fisher Scientific, USA) for 24 h. Transfection efficiency was measured by qRT-PCR.
Statistical analysis
In this study, data were averaged from at least three biological repeats and were presented as mean ± standard errors. Statistical analysis was conducted using an unpaired student’s t-test on windows-based SPSS software (version 15.0; SPSS, USA). Significant difference was indicated if
Results
MiR-409-5p is upregulated in both BC tumors and cell lines
In a previous clinical report, miR-409-5p was found to be negatively associated with BC-specific survival.
16
In this study, we first used qRT-PCR to specifically examine the expression of miR-409-5p in in situ BC tumors. Carcinoma epithelial breast tissues and their adjacent healthy epithelial breast tissues were extracted from 113 patients with BC. The result of qRT-PCR showed that miR-409-5p was significantly upregulated in carcinoma breast tissues than in normal breast epithelial tissues (Figure 1(a), *

MiR-409-5p gene level in breast cancer. (a) qRT-PCR was used to compare endogenous miR-409-5p expression between carcinoma breast epithelial tissues (carcinoma) and adjacent healthy epithelial tissues (normal) in 113 patients diagnosed with breast cancer (*
MiR-409-5p downregulation suppressed proliferation and migration in BC cells
In order to elucidate the functional mechanism of miR-409-5p in BC, we transduced BC cell lines, MDA-MB-231 and MCF-7, with lentivirus expressing miR-409-5p inhibitor (L/miR409I) to downregulate their endogenous miR-409-5p expression levels. In control MDA-MB-231 and MCF-7 cells, they were transduced with lentivirus expressing a non-specific miRNA inhibitor (L/I). After transduction was stabilized, transduction efficiency was checked by qRT-PCR, which showed that lentiviral transduction successfully downregulated endogenous miR-409-5p expressions in both MDA-MB-231 and MCF-7 cells (Figure 2(a), *
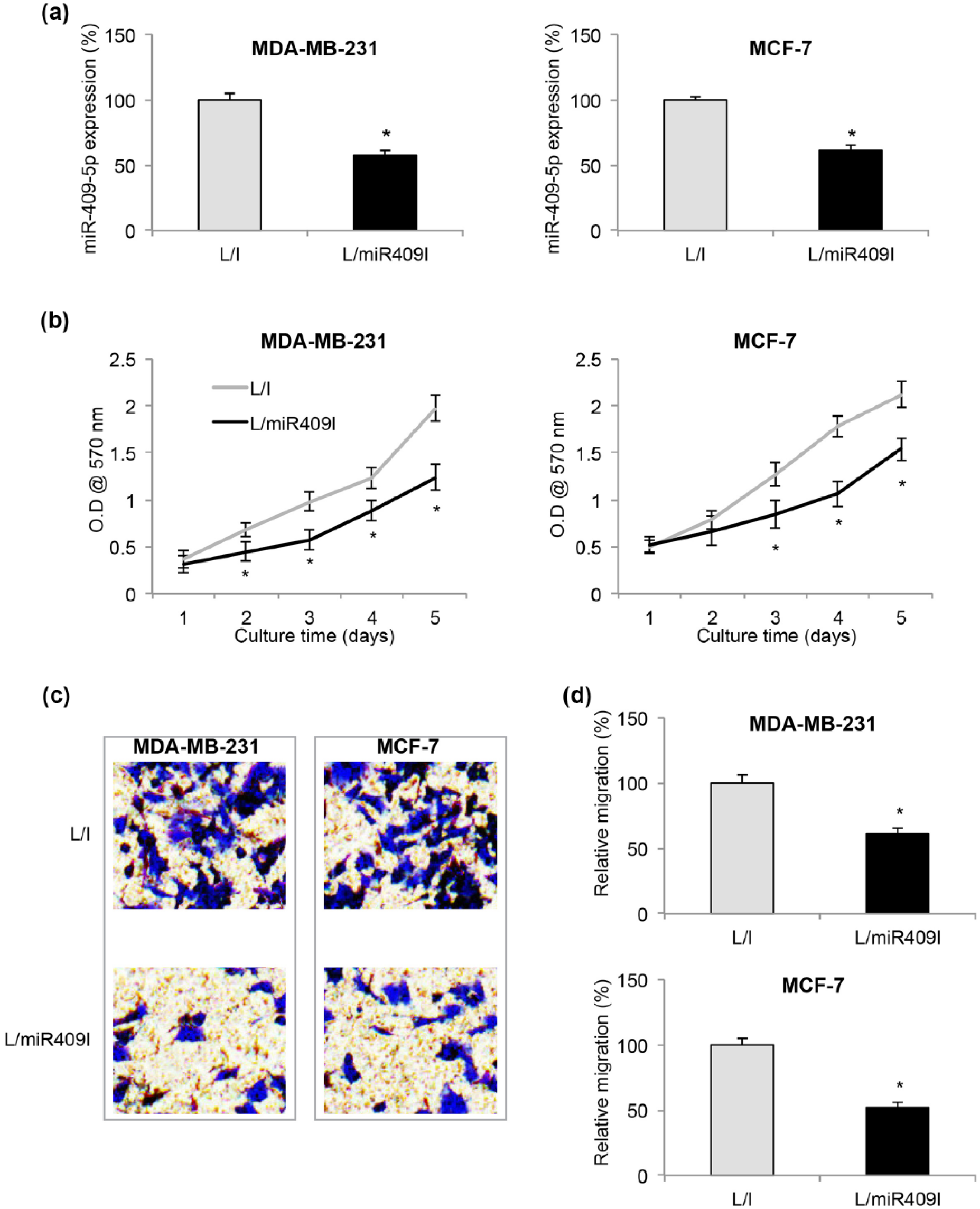
Effects of miR-409-5p downregulation on breast cancer proliferation and migration. (a) MDA-MB-231 and MCF-7 cells were transduced with a lentivirus expressing miR-409-5p inhibitor (L/miR409I) or a lentivirus expressing non-specific miRNA inhibitor (L/I). After cells were purified and stabilized, qRT-PCR was performed to compare endogenous miR-409-5p expressions between L/I-transduced and L/miR409I-transduced breast cancer cells (*
Lentiviral-transduced MDA-MB-231 and MCF-7 cells were then transferred to a 96-well plate to be tested by a 5-day proliferation assay. Every day, cells were stained with MTT reagent and their relative proliferating rates were measured at OD of 570 nm. It showed that BC in vitro proliferation was significantly suppressed by miR-409-5p downregulation in both MDA-MB-231 and MCF-7 cells (Figure 2(b), *
Migrating capability was also examined in lentiviral-transduced MDA-MB-231 and MCF-7 cells by a cell migration assay. After 24 h, cells were plated in upper chambers and chemoattractant (10% FBS) in lower chambers, and MDA-MB-231 and MCF-7 cells successfully migrated into lower chambers were fixed and stained by crystal violet. It showed that significantly less cancer cells migrated into lower chambers with miR-409-5p downregulation (Figure 2(c)). Luminescence was measured for each lower chamber at OD of 560 nm and compared between L/I-transduced and L/miR409I-transduced BC cells. Characterization on relative migration demonstrated that miR-409-5p downregulation significantly reduced BC migration (Figure 2(d), *
MiR-409-5p downregulation inhibited BC xenograft
We then evaluated the in vivo effect of miR-409-5p downregulation on BC. L/miR409I- or L/I-transduced MDA-MB-231 cells were subcutaneously injected into left flanks of 4-week-old BALB/C nude mice. In situ MDA-MB-231 xenografts were allowed to develop for 5 weeks. Weekly live measurements on xenograft volumes indicated that in vivo development of MDA-MB-231 xenograft was significantly inhibited by miR-409-5p downregulation (Figure 3(a), *
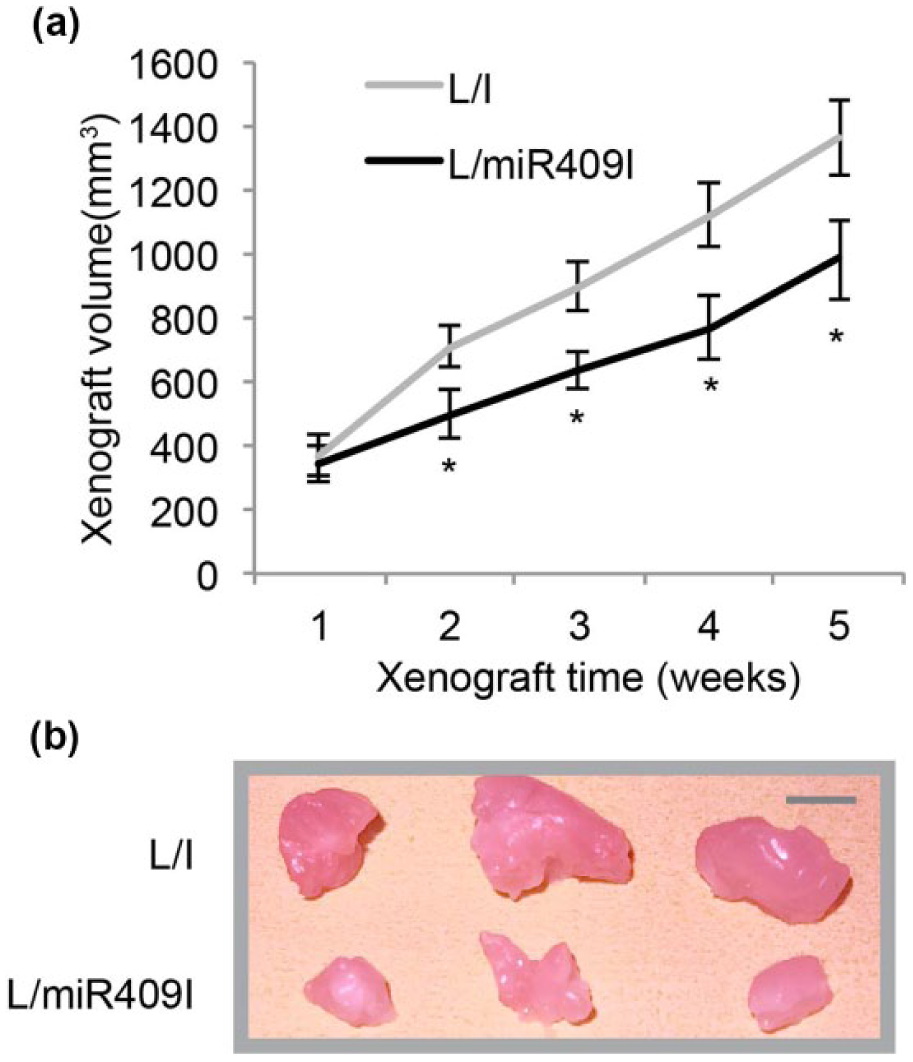
Effects of miR-409-5p downregulation on breast cancer xenograft. (a) L/I- or L/miR409I-transduced MDA-MB-231 cells were subcutaneously injected into left flanks of 4-week-old BALB/C nude mice. An in situ xenograft assay was carried out by comparing xenograft volumes at weekly base for five consecutive weeks (*
RSU1 is downstream target gene of miR-409-5p in BC
After functional role of miR-409-5p (downregulation) was revealed by this study, we sought the downstream target gene of miR-409-5p in BC. Using online miRNA binding algorithms such as miRDB (www.miRDB.org) and TargetScan (www.targetscan.org), we took notice that a tumor suppressor gene RSU1 might be the candidate, since miR-409-5p can bind the cDNA sequence on human RSU1 3′-UTR (Figure 4(a)). Thus, in lentiviral-transduced MDA-MB-231 and MCF-7 cells, we further transfected them with firefly luciferase vectors containing wild-type RSU1 3′-UTR (Wt) or mutated RSU1 3′-UTR (Mu, without miR-409-5p binding site). After 48 h, analysis by a dual-luciferase activity assay showed that in L/I-transduced BC cells, co-transfection of RSU1 3′-UTR (Wt) and RSU1 3′-UTR (Mu) did not result in difference in their luciferase activities (Figure 4(b), Δ

Association of RSU1 gene and miR-409-5p in breast cancer. (a) The wild-type (Wt) human RSU1 3′-UTR was shown to be bound by human miR-409-5p. In a mutant (Mu) RSU1 3′-UTR, its miR-409-5p binding DNAs were point-mutated. (b) Lentiviral-transduced MDA-MB-231 and MCF-7 cells were and then co-transfected with either RSU1-3′-UTR (Wt) or RSU1-3′-UTR (Mu) firefly luciferase vectors and a pSV40-Renilla control luciferase vector. After 48 h, relative luciferase activities were characterized as the ratio of firefly/Renilla signals for co-transfected MDA-MB-231 and MCF-7 cells (*
To further verify the result of our dual-luciferase activity assay, we used qRT-PCR to evaluate the endogenous expression level of RSU1 in lentiviral-transduced MDA-MB-231 and MCF-7 cells. It showed that RSU1 was indeed upregulated in L/miR409I-transduced cancer cells rather than in L/I-transduced cancer cells (Figure 4(c), *
RSU1 inhibition had opposite effect as miR-409-5p downregulation in BC
Finally, we sought the possible functional role of RSU1 in miR-409-5p downregulation–mediated BC inhibition. In L/miR409I-transduced MDA-MB-231 and MCF-7 cells, we transfected them with RSU1-specific siRNA (Si-RSU1) or a scrambled control siRNA (si/C); 24 h after transfection, qRT-PCR demonstrated that endogenous RSU1 expression was significantly downregulated by Si-RSU1 in L/miR409I-transduced MDA-MB-231 and MCF-7 cells (Figure 5(a), *
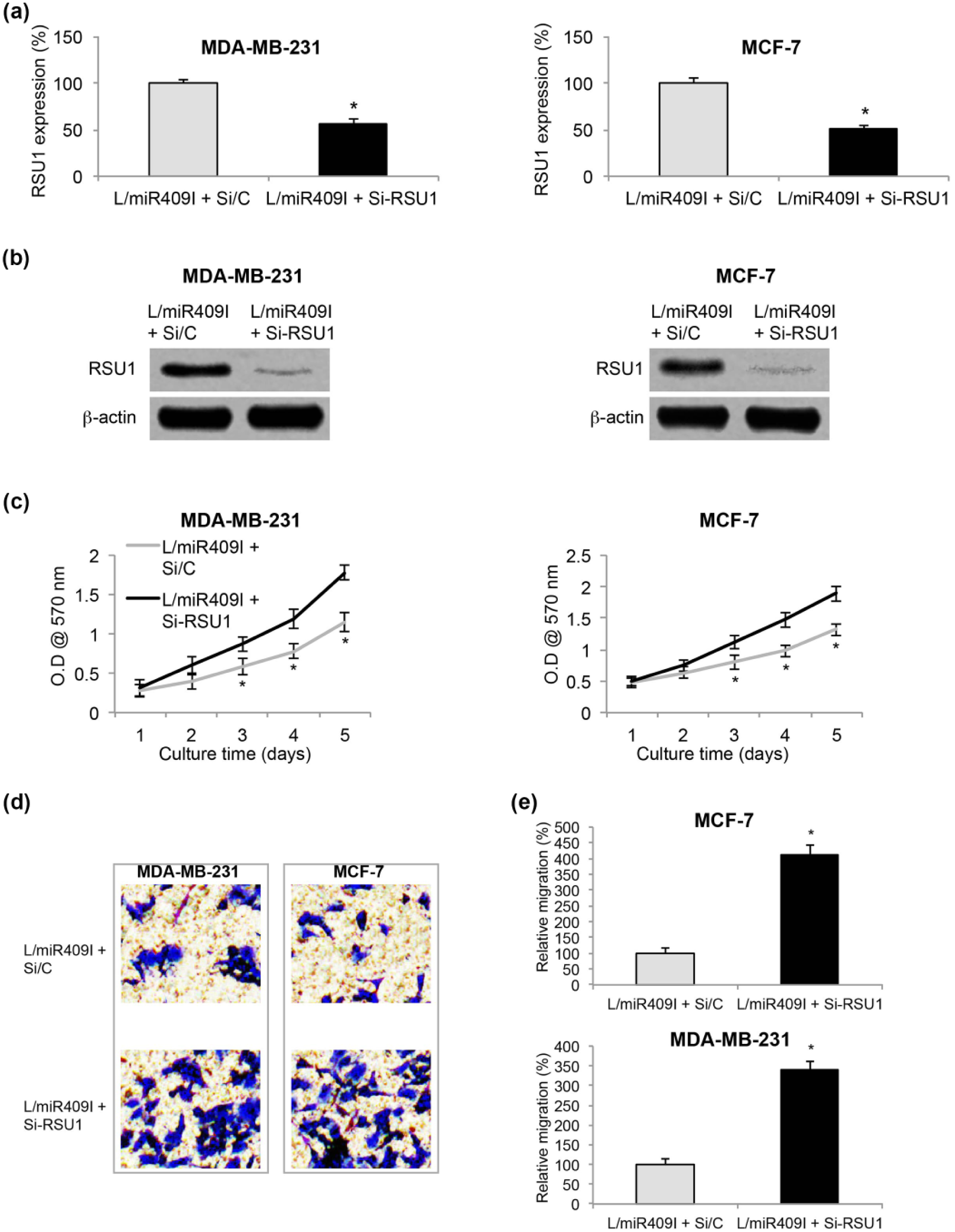
Effects of RSU1 inhibition on miR-409-5p-downregualted breast cancer. (a) L/miR409I-transduced MDA-MB-231 and MCF-7 cells were transfected with Si-RSU1 or Si/C siRNAs for 24 h. qRT-PCR was performed to examine transfection efficiency (*
Transfected MDA-MB-231 and MCF-7 cells were then replated in 96-well plate and tested by the proliferation assay for 5 days. It showed that BC in vitro proliferation was significantly enhanced by RSU1 downregulation in L/miR409I-transduced MDA-MB-231 and MCF-7 cells (Figure 5(c), *
In addition, migration was evaluated in transfected BC cells; 24 h after the cell migration assay, MDA-MB-231 and MCF-7 cells in the lower chambers were stained and pictured. It showed that significantly more L/miR409I-transduced cells migrated into lower chambers while they were transfected with Si-RSU1 (Figure 5(d)). Quantitative measurement on BC relative migration confirmed our immunohistochemical results, showing that RSU1 downregulation significantly promoted migrating capability in L/miR409I-transduced BC cells (Figure 5(e), *
Thus, these results indicated that RSU1 downregulation had opposite effect as miR-409-5p downregulation in BC by reversing its inhibition on cancer in vitro proliferation and migration.
Discussions
In this study, we first explored the expression profile of miR-409-5p in BC. The analysis of qRT-PCR demonstrated that miR-409-5p was aberrantly upregulated in both BC human tumors and BC cell lines. This is an interesting observation as the other member of miR-409 family, miR-409-3p, was shown to be downregulated in BC tumors and cell lines. 15 This is different than the cohesive expression of miR-405-3p and miR-405-5p in other cancer forms, such as prostate cancer, 22 thus suggesting that miR-409-5p and miR-409-3p might have distinctly different functions or associated signaling pathways in BC.
To explore the functional mechanism of miR-409-5p in BC, we used lentiviral technology to stably downregulate miR-409-5p in two of the BC cell lines MDA-MB-231 and MCF-7 cells, which was confirmed by qRT-PCR after transduction. We then compared cancer proliferation, migration, and in vivo xenografts between miR-409-5p-downregulated BC cells and BC cells with normal miR-409-5p expression. The results of our functional assays showed that miR-409-5p downregulation had profound anti-tumor effect by inhibiting cancer proliferation, migration, and in vivo xenografts in BC cells. To our knowledge, this is the first-ever report demonstrating functional regulation of miR-409-5p, probably acting as oncogene, in BC. Interestingly, a very recent study showed that the other member of miR-409 family, miR-409-3p, had opposite mechanism as miR-409-5p in BC. 15 In that report, Zhang et al. 15 showed that in MDA-MB-231 and MDA-MB-468 cells, overexpression of miR-409-3p inhibited cancer proliferation and migration, thus acting as tumor suppressor, in BC. The discrepancy in tumor regulatory functions between miR-409-5p and miR-409-3p may be explained by their different expression profiles in BC tumors (or cell lines) as miR-409-5p was upregulated (demonstrated by our study), whereas miR-409-3p was downregulated 15 in BC. In addition, it is possible that the intrinsic signaling pathways associated with miR-409-3p or miR-409-5p may be different, thus deciding their either oncogenic or tumor-suppressing role in BC.
In order to further explore the molecular signaling pathways associated with miR-409-5p in BC, we performed dual-luciferase activity assay in lentiviral-transduced MDA-MB-231 and MCF-7 cells. By further transfecting those BC cells with firefly luciferase vectors containing either wild type or mutant (without miR-409-5p binding site) RSU1 3′-UTR, we demonstrated that RSU1 was indeed the downstream target gene of miR-409-5p in BC. This finding was further supported by our qRT-PCR and western blot assays in lentiviral-transduced MDA-MB-231 and MCF-7 cells, showing both gene and protein expression levels of RSU1 were significantly upregulated in L/miR409I-transduced BC cells, presumably induced by miR-409-5p downregulation. Furthermore, we investigated the functional mechanisms of RSU1 in miR-409-4p-mediated tumor regulation in BC cells. Through siRNA transfection, we discovered that inhibiting RSU1 could reverse the tumor-suppressive effect of miR-409-5p downregulation on BC cell proliferation and migration. The functional mechanisms of RSU1, presumably acting as oncogene in BC, had been demonstrated previously.19,20 However, our study is the first report to show that RSU1 could be regulated by upstream modulator of miR-409-5p in BC. Moreover, given the fact that miR-409-3p and miR-409-5p had different downstream target genes in BC, as our study shows RSU1 being downstream target of miR-409-5p, whereas Zhang et al.’s 15 report shows Akt1 being downstream target of miR-409-3p, it may further explain the functional discrepancy between miR-409-3p and miR-409-5p, as one is acting as tumor suppressor gene and the other acting as tumor oncogene in BC.
Conclusion
Overall, in this study, we demonstrated that miR-409-5p is highly expressed in BC and its downregulation has anti-cancer effect on BC development both in vitro and in vivo. In addition, we demonstrated that the regulatory effect of miR-409-5p inhibition is likely through the inverse upregulation on its downstream target gene of RSU1 in BC.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
