Abstract
Neoadjuvant chemotherapy has been established as standard treatments for advanced breast cancer among multidisciplinary therapies. A simple and instructive biomarker for the postoperative recurrence and metastasis is needed to evaluate the therapeutic effect. Plasma fibrinogen level has been shown to be associated with tumor progression and poor outcomes in breast cancer patients. This study aims to further evaluate the clinical and prognostic value of plasma fibrinogen level as a biomarker in breast cancer patients receiving neoadjuvant chemotherapy. In this study, data of 67 patients were retrospectively collected and analyzed to identify the relationship between the plasma fibrinogen level and the clinical progression and outcome of these patients. Patients with increased plasma fibrinogen level after neoadjuvant chemotherapy had significantly shorter disease-free survival and overall survival (p < 0.001 and p = 0.001, respectively). In a univariate survival analysis, molecular type (p = 0.0004/p = 0.005), clinical response (p = 0.008/p = 0.015), and changes in plasma fibrinogen level (p = 0.012/p = 0.007) were associated with disease-free survival and overall survival, and all of them, molecular type (p = 0.0003/p = 0.005), clinical response (p = 0.027/p = 0.021), and changes in plasma fibrinogen level (p = 0.035/p = 0.025), were associated with disease-free survival and overall survival in a multivariate survival analysis, respectively. The plasma fibrinogen level was found to be a possible biomarker for clinical response to chemotherapy and postoperative metastasis or death in advanced breast cancer patients who received neoadjuvant chemotherapy.
Introduction
Breast cancer is a major health threat among females, as being the most frequently diagnosed cancer (25% of all cancer cases) and carries the highest mortality rate among females worldwide (15% of cancer death), with an estimated 1.7 million cases and 521,900 deaths in 2012. 1 Although stable or even decreasing in developed countries, the incidence rates and mortality rates are shown to be increasing in developing countries.
Among patients with recurrence or metastatic advanced breast cancer, the lack of optimal therapy is still one of the main contributing factors to death. Neoadjuvant chemotherapy implies initiation of systemic therapy prior to definitive local-regional management. Also referred to as primary systemic therapy or preoperative chemotherapy, it has been established as a standard treatment for advanced breast cancer. Although neoadjuvant chemotherapy demonstrates tumor response as a strong predictor of outcome as evidenced by the association of pathologic complete response and survival, changes of the postoperative clinicopathological parameters influence the judgment of real prognosis strongly. So, a simple and instructive biomarker for classification needs to be identified to monitor high-risk patients and detect postoperative metastasis earlier in patients receiving neoadjuvant chemotherapy.
The malignancy-related hypercoagulable state is shown to be present in 50% of cancer patients.2,3 Fibrinogen is an important coagulation factor. It is a 340-kDa glycoprotein synthesized by liver upon stimulation of interleukin 6 (IL-6) and IL-1β4,5 and is converted to fibrin by activated thrombin. Fibrinogen is also a modest acute-phase response protein.4,6 Fibrinogen has recently been shown to play vital roles in tumorigenesis and contributed to angiogenesis, stroma formation, and hematogenous metastasis of tumor cells.7–10 Elevated plasma fibrinogen level is related with tumor progression and other poor outcomes in several types of malignancies, for example, covered esophageal cancer, 11 gastric cancer, 12 pancreatic cancer, 13 colon cancer, 14 lung cancer, 15 hepatocellular cancer, 16 gallbladder cancer, 17 and gynecological cancer.18–21 For operable breast cancer patients, elevated preoperative plasma fibrinogen level is demonstrated to be associated with cancer progression and is independently associated with a poor prognosis in patients. 22 With regard to chemotherapy, treatment response and the plasma fibrinogen level were shown to correlate with each other in lung cancer and esophageal cancer.23,24 There has been very few studies on the correlation between the plasma fibrinogen level and breast cancer patients receiving neoadjuvant chemotherapy followed by radical mastectomy.
In this study, we investigated the clinical value of the plasma fibrinogen level and its correlation with clinicopathological parameters, the disease-free survival (DFS) rates, and overall survival (OS) rates in patients with advanced breast cancer.
Materials and methods
Patients
Medical records were reviewed for all 92 patients with invasive breast cancer, all the patients received neoadjuvant chemotherapy and followed radical mastectomy at Qilu Hospital of Shandong University from January 2010 to January 2013. Patients were included only if they met the following criteria: (1) newly diagnosed breast cancer, (2) histological confirmation of the diagnosis, and (3) neoadjuvant chemotherapy for at least four cycles. Exclusion criteria for all participants were as follows: (1) overt bacterial or viral infection during the course of treatment, (2) concurrent liver disease, (3) coagulation disorders, (4) taking anticoagulants or acetylsalicylic acids within 3 months before the treatment, and (5) no adequate clinical data or loss of follow-up. In total, 67 patients were included in this study.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee of the Qilu Hospital of Shandong University and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Clinical treatment and patient follow-up
Neoadjuvant chemotherapy with the combination of epirubicin 60 mg/m2 and docetaxel 75 mg/m2 was repeated twice every 3 weeks for 4–6 cycles. Epirubicin and docetaxel were administered by intravenous drip infusion on day 1. Before the third cycle, we evaluated treatment response use Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1 for the first time and divided patients into four groups (complete response (CR), partial response (PR), stable disease (SD) and progressive disease (PD)) before surgery.
All patients were treated by either modified radical mastectomy or radical mastectomy after neoadjuvant chemotherapy, together with standard systemic therapy in the adjuvant setting. As standard protocol, all patients have been followed up carefully after the initial treatment and reassessed every 3 months for 3 years and once a year thereafter. The start of the follow-up was the date of the pathologic diagnosis of breast cancer. The end of the follow-up was the time of death or the last follow-up time as part of standard treatment follow-up protocol and before March 2016. The end points of the analyses were DFS and OS. DFS was defined as the period from the date of initial diagnosis to the date of recurrence, metastasis, or censoring. OS was calculated as the time from initial diagnosis to death or censoring.
Plasma fibrinogen level
In the medical records of patients, plasma fibrinogen concentrations of peripheral venous blood samples taken before breakfast were examined for less than 7 days before the first chemotherapy course and surgery. For assaying fibrinogen, we used the clotting method of Clauss with the STA®-Fibrinogen kit (Diagnostica Stago, Asnières-sur-Seine, France). The intraassay and interassay coefficient of fibrinogen were both defined as less than 5%. The reference ranges for plasma fibrinogen levels were set between 2.0 g/L and 4.0 g/L. Plasma fibrinogen levels higher than 4.0 g/l indicated hyperfibrinogenaemia, and the changes more than 0.2 g/L before and after chemotherapy was meaningful.
Statistical analysis
Plasma fibrinogen levels were presented as mean ± standard deviation (SD). Comparisons between groups were performed using the Kruskal–Wallis test and the Wilcoxon rank-sum test for continuous variables. The DFS and OS rates were calculated using the Kaplan–Meier method, and differences in survival rates between the groups were compared using the log-rank test. Both univariate and multivariate analyses were performed for the prognostic factors using the Cox proportional hazard model. A two-sided p < 0.05 was considered to be statistically significant. Data analysis was performed using SAS version 9.4(SAS Institute, Inc., Cary, NC, USA).
Results
Patient characteristics and the changes in plasma fibrinogen levels
The median age of all the patients was 47 years (range: 29–66), all female. A total of 37 patients (55.2%) were experiencing menopause during treatment. Based on the tumor staging system, most patients were diagnosed at stage II and IIIA (42, 62.69%) and only 5 patients (7.46%) were classified as stage IIIC. A total of 56 patients (83.58%) had lymph node metastasis. And 30 (44.78%) patients had clinical response CR or PR and 34 (50.75%) patients’ fibrinogen levels decreased after neoadjuvant chemotherapy. Building on the immunohistochemical analysis for estrogen receptor (ER), PR, HER2, and Ki-67, we calculated molecular subtypes: luminal A ((ER+ and/or PR+, HER2−, Ki67 ≤ 20%) (18, 26.87%)), luminal B ((ER+ and/or PR+, HER2−, Ki67 > 20%) (28, 41.79%)), basal-like ((ER− and PR−, HER2−) (11, 16.42%)), and HER2+ ((ER+/−, PR+/−, HER2+) (10, 14.92%)). None of the patients had local relapse or died from causes unrelated to breast cancer. There were 26 (38.81%) patients with distant metastasis and 16 (23.88%) died from breast cancer during follow-up period. The baseline patient characteristics are shown in Table 1.
Baseline characteristics of the patients.

CR: complete response; PR: partial response; SD: stable disease; PD: progressive disease.
Correlation between plasma fibrinogen levels and clinicopathological parameters
The mean pretreatment plasma fibrinogen level was 3.26 ± 0.81 g/L, and the mean preoperative level was 3.28 ± 0.59 g/L. We had investigated the correlation between pretreatment plasma fibrinogen levels and clinicopathological parameters in our previous study. 23 The preoperative plasma fibrinogen levels had no association with any clinicopathological parameters (Table 2), may be limited by the size of the sample and short-time follow-up. So, we focused on the changes in plasma fibrinogen levels after neoadjuvant chemotherapy. Changes in fibrinogen plasma levels were associated with menopause (p = 0.022), clinical response (p < 0.001), and metastasis (p < 0.001; Table 3). Among the fibrinogen levels decrease group, more patients responded to neoadjuvant chemotherapy and showed a significantly lower metastasis rate. The levels of fibrinogen in 21 patients increased after neoadjuvant chemotherapy in all 26 patients with metastasis.
Correlation between preoperative plasma fibrinogen levels and clinicopathological parameters.
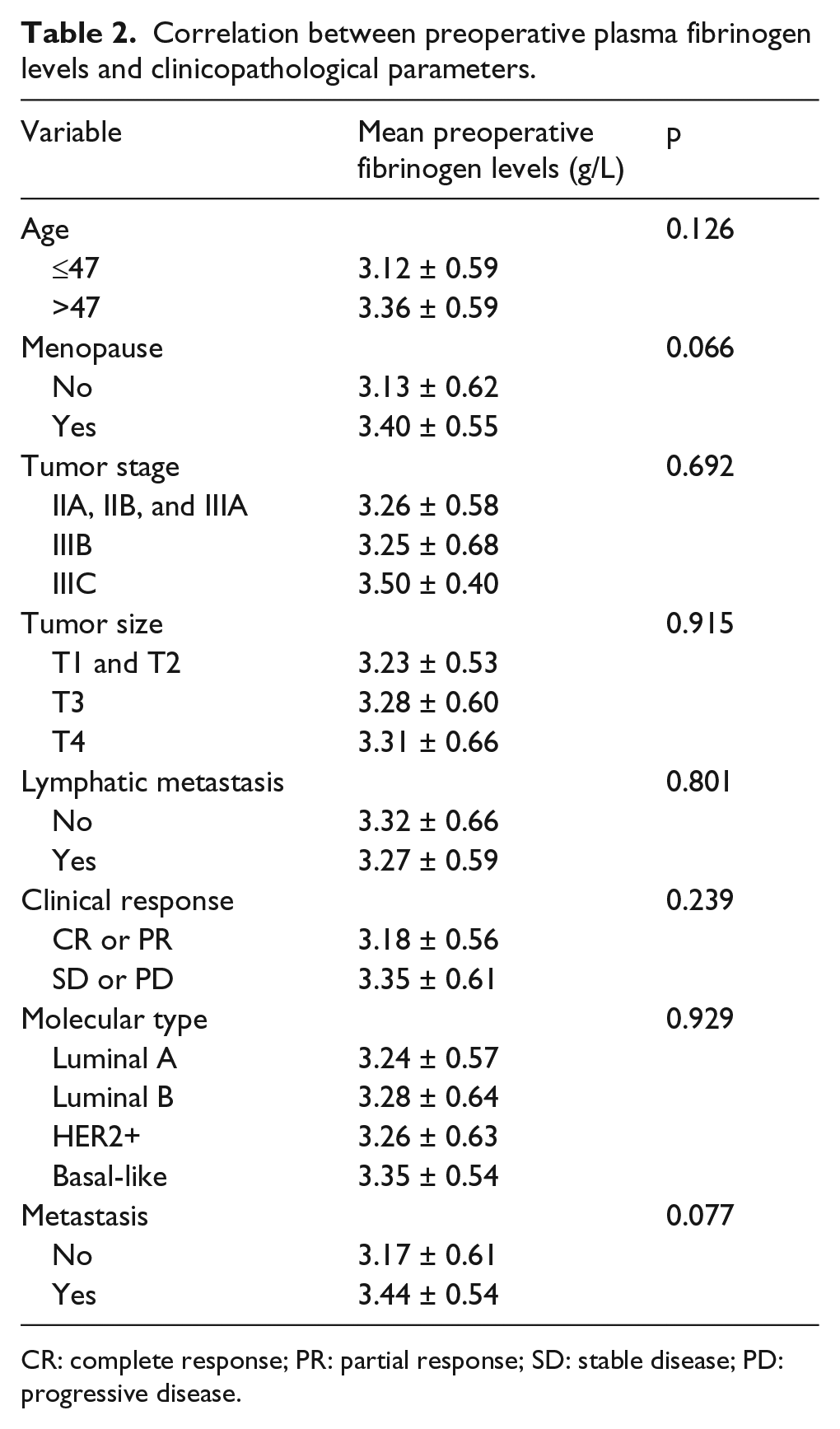
CR: complete response; PR: partial response; SD: stable disease; PD: progressive disease.
Correlation between changes in plasma fibrinogen levels and clinicopathological parameters.
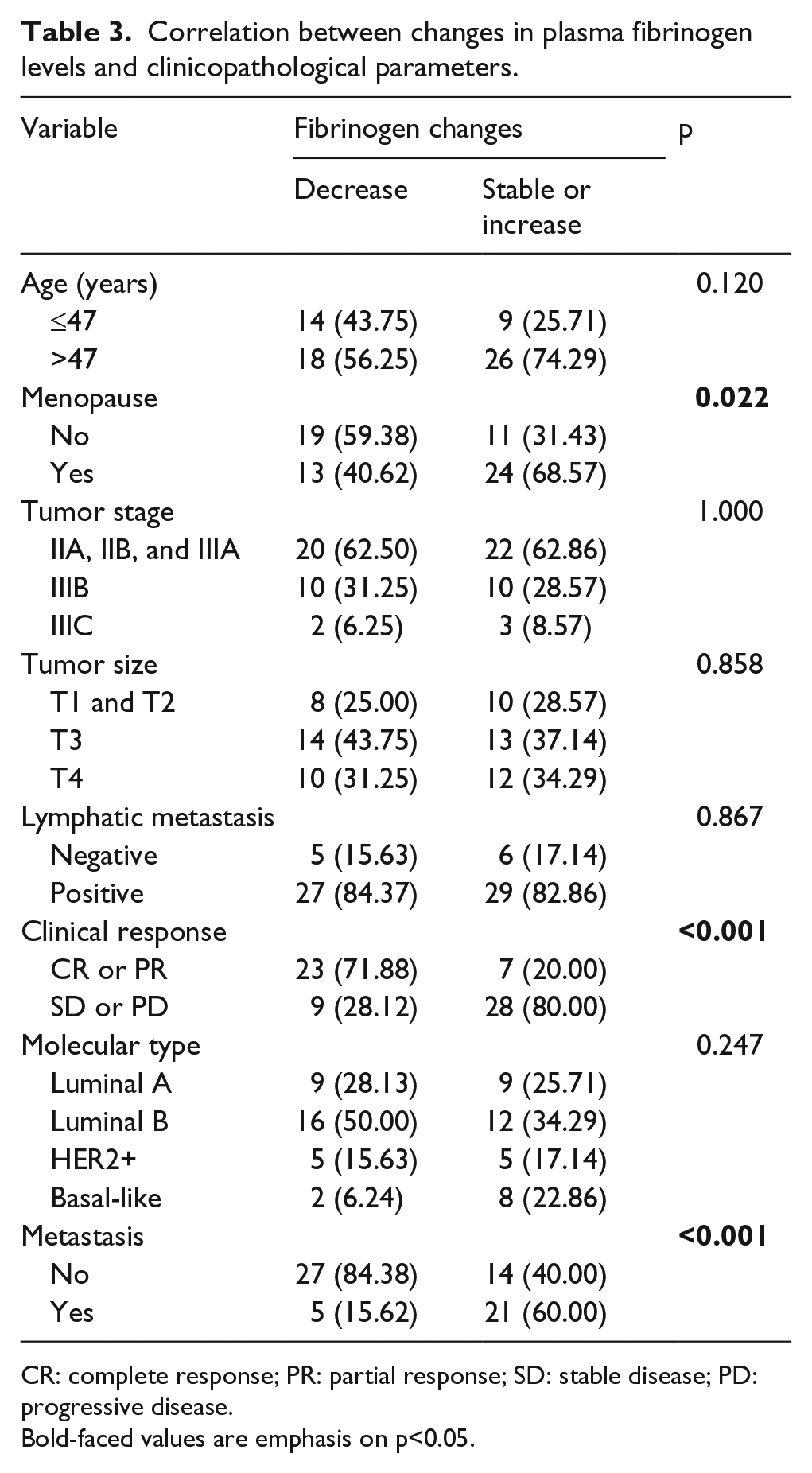
CR: complete response; PR: partial response; SD: stable disease; PD: progressive disease.
Bold-faced values are emphasis on p<0.05.
Survival analysis
The median follow-up duration was 45 months (DFS; range: 11–82) and 46 months (OS; range: 20–82). To assess the prognostic impact of the changes in plasma fibrinogen levels, we performed Kaplan–Meier analysis to compare patients grouped according to changes in plasma fibrinogen levels; the 3-year DFS and OS rates of patients in the fibrinogen levels decrease group were 90.6% and 93.8% and in the other group were 51.4% and 77.1%, respectively. Patients in the fibrinogen levels decrease group had longer DFS and OS than the patients in the other group (p < 0.001, p = 0.001; Figure 1).

Kaplan–Meier curves for DFS and OS according to the changes in plasma fibrinogen levels.
Prognostic factor
In the univariate analysis, molecular type, clinical response, and changes in plasma fibrinogen levels were associated with both DFS (Table 4) and OS (Table 5). Then, when we used the multivariate analysis to verify the potential prognostic factors, all the above factors were found to be independently associated with DFS (Table 4) and OS (Table 5).
Univariate and multivariate survival analyses of disease-free survival in patients with advanced breast cancer.

HR: hazard ratio; CI: confidence interval.
Bold-faced values are emphasis on p<0.05.
Univariate and multivariate survival analyses of overall survival in patients with advanced breast cancer.
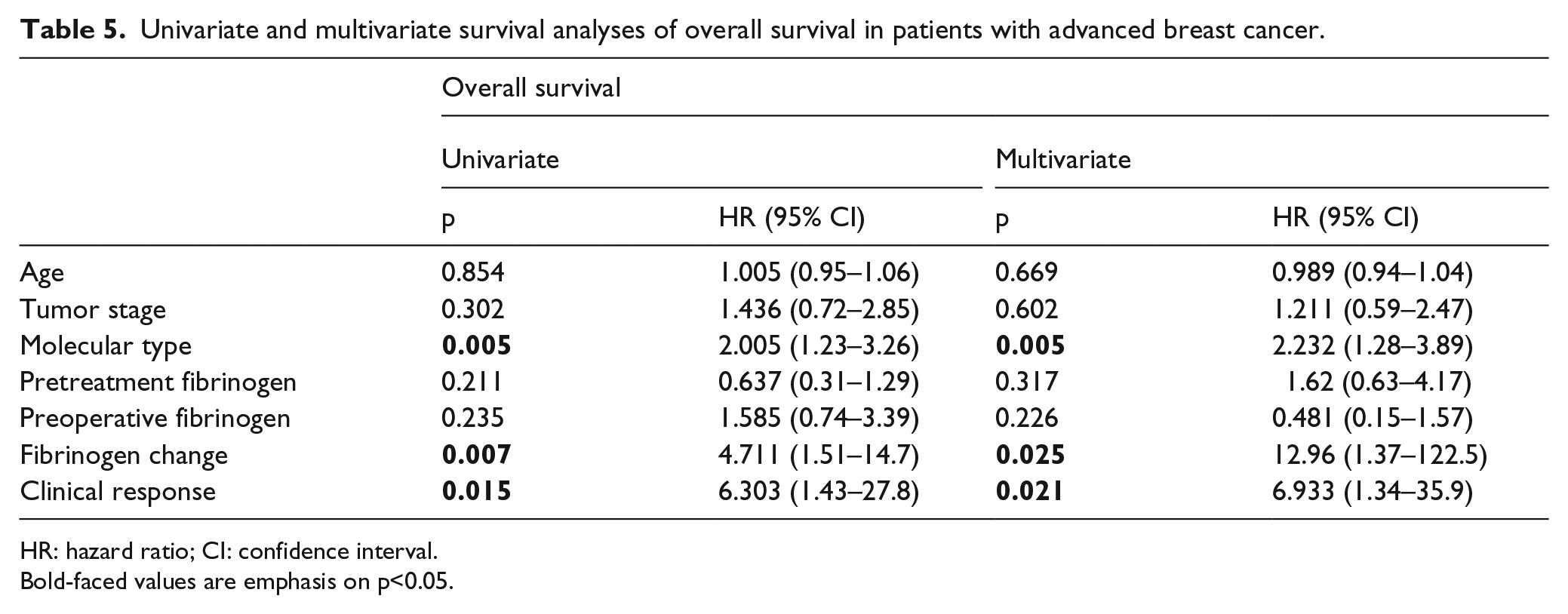
HR: hazard ratio; CI: confidence interval.
Bold-faced values are emphasis on p<0.05.
Discussion
In recent years, a number of studies revealed an association between elevated plasma fibrinogen level and tumor progression/poor prognosis in several types of cancer. In addition, with regard to chemotherapy, treatment response and the plasma fibrinogen levels were shown to correlate with each other in lung cancer and esophageal cancer. 24 In our previous study, we demonstrated that elevated preoperative plasma fibrinogen level is associated with cancer progression and is independently associated with a poor prognosis in patients with operable breast cancer. This study on breast cancer patients receiving neoadjuvant chemotherapy reveals that the changes in plasma fibrinogen level during neoadjuvant chemotherapy predict the DFS and OS; patients who had decreased fibrinogen level had longer DFS and OS than patients who had stable or increased fibrinogen level. In addition, multivariate survival analyses showed that the changes in plasma fibrinogen level and response to neoadjuvant chemotherapy and molecular type are independent prognostic factors for poor survival in patients receiving neoadjuvant chemotherapy. Plasma fibrinogen level increased in neoadjuvant chemotherapy treatment can indicate poor response to chemotherapy and earlier metastasis or death. All our studies on the correlation between plasma fibrinogen level and patients with breast cancer showed that plasma fibrinogen level is associated with local cancer progression and hematogenous and lymphatic metastasis of cancer, 22 although the reason for why the change of plasma fibrinogen levels correlates with tumor cell growth and metastasis remains to be elucidated.
Fibrinogen was shown to be produced mainly from the liver upon the inflammatory stimulation by IL-6 and IL-1β.4,5 The overproduction of inflammatory cytokines by cancer cells, such as IL-6, may stimulate increased of fibrinogen production. 5 Other studies show that fibrinogen can be endogenously synthesized by tumor cells themselves, and it functions as a mediator of cancer cell proliferation and metastasis.7,25,26 Fibrinogen can regulate cellular adhesion, proliferation, and migration through interactions with fibroblast growth factor 2 (FGF-2) and vascular endothelial growth factor (VEGF). 27 By increasing the expression of vimentin and reducing the expression of E-cadherin, Shu et al. 17 demonstrated that highly concentrated fibrinogen induced epithelial–mesenchymal transition (EMT), which confers migration, invasion, and metastatic capacity and multidrug resistance to tumor cells.28,29 Fibrinogen may act as ligands for integrin and non-integrin receptors, which were found on tumor cells, stromal cells, and inflammatory cells. Such cellular interactions could result in changes in cellular behaviors, including cell proliferation and migration by causing conformational changes of integrin, and transduce intracellular signals.7,30 In fibrinogen-deficient mice with melanoma, Palumbo et al. 9,10,27 demonstrated that spontaneous hematogenous and lymphatic metastasis was diminished through a series of studies with evidence that plasma fibrinogen significantly improved the survival of circulating tumor cells in the vasculature. Other studies from the same group showed that the increase in metastatic success was linked to natural killer cell functions; specifically, the platelet–fibrinogen axis was shown to contribute to metastatic potential by impeding natural killer cell–mediated elimination of tumor cells. The above evidence could support some results of this study that operable breast cancer patients with different plasma fibrinogen levels before surgery or advanced breast cancer patients with different changes of plasma fibrinogen levels after neoadjuvant chemotherapy have different hematogenous metastasis incidence.
In this study, we reproduced similar conclusion as previous studies that the responders to neoadjuvant chemotherapy, including CR and PR patients, tended to have decreased fibrinogen level during treatment. Therefore, fibrinogen changes appear to indicate the response to neoadjuvant chemotherapy. In addition, the changes of fibrinogen level were also found to be an independent determining factor for DFS and OS. With the use of the changes in fibrinogen level during neoadjuvant chemotherapy, we could classify advanced breast cancer patients into good and poor prognosis groups, select patients who have a high risk of postoperative metastasis, and need strict follow-up and intense treatment. So, if the plasma fibrinogen level in patients increases in neoadjuvant chemotherapy procedure, we may change the plan of chemotherapy or add new drugs that are not previously used and search any other method to be followed after surgery.
This study has several limitations. First, as a retrospective study, data collection bias is unavoidable. Second, small sample size may lead to an inappropriate conclusion. Last but not the least, the follow-up period was short. Therefore, large-scale multi-center prospective validation study with longer follow-up period to further determine the results is required.
Conclusion
With the validity of the changes in fibrinogen level during neoadjuvant chemotherapy, plasma fibrinogen level was found to be a possible biomarker for postoperative metastasis and death in advanced breast cancer patients who received neoadjuvant chemotherapy. Moreover, the plasma fibrinogen level could be a simple and valuable predictive indicator for postoperative follow-up.
Footnotes
Acknowledgements
The authors thank members of Department of Breast Surgery, Qilu Hospital of Shandong University, for their advice on the research: Dr Kai Zhang, Jiang Zhu.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
