Abstract
NIMA-related kinase 2B has been known to be an important centrosome regulatory factor. The aim of this study was to investigate the effect of NIMA-related kinase 2B on the sensitivity of breast cancer to paclitaxel. We detected the expression of NIMA-related kinase 2B messenger RNA in MCF-10 cells, including MCF-10A, MCF-10AT, MCF-10DCIS.com, and MCF-10CA1a. The influence of NIMA-related kinase 2B in nude mouse was also detected. The association between NIMA-related kinase 2B and clinicopathological factors was explored in invasive ductal carcinoma tissues. NIMA-related kinase 2B was lowly expressed in the precancerous cells, MCF-10A and MCF-10AT, and it was highly expressed in carcinomatous cells, MCF-10DCIS.com and MCF-10CA1a. The upregulation of NIMA-related kinase 2B can introduce the growth of MCF-10AT cells, knockdown of NIMA-related kinase 2B could remarkably inhibit cell proliferation in MCF-10DCIS.com and MCF-10 CA1a cells. Comparing the volume of the xenografts in nude mouse, we found that the tumors treated by NIMA-related kinase 2B small interfering RNA associated with paclitaxel were the smallest among all the groups. Expression of NIMA-related kinase 2B messenger RNA was associated with higher histological grades, positive lymph node, and high Ki67 index (>20%). The partial response rates were 75.0% in NIMA-related kinase 2B negative (NIMA-related kinase 2B−) patients and 15.8% in NIMA-related kinase 2B++ patients. The progressive disease rates were 10.0% in NIMA-related kinase 2B− patients and 52.6% in NIMA-related kinase 2B++ patients (p = 0.002). Our findings suggested that NIMA-related kinase 2B could play a role in the development and progression of breast cancer. Combination treatment using NIMA-related kinase 2B small interfering RNA and paclitaxel might be a novel potential therapy method for breast cancer.
Introduction
NIMA-related kinase 2 (Nek2) is a cell cycle–regulated serine/threonine kinase that localizes to the centrosome and is upregulated in human cancers. 1 Functionally, it is implicated in control of centrosome separation and bipolar spindle formation in mitotic cells and chromatin condensation in meiotic cells. Three splice variants of Nek2 have been described differently in vertebrates, the non-catalytic C-termini differs in three splice variants of Nek2A, Nek2B, and Nek2C. For its distinct C-terminus, the degradation of Nek2B does not occur during M/G1 phase and the splice variant of Nek2B persisted throughout all the mitosis.2,3 As reported previously, Nek2B is not capable of stimulating centrosome splitting with the same efficiency as Nek2A. 4 One speculation is that Nek2B helps to maintain the integrity of spindle poles in Xenopus embryos throughout mitosis. 3 The downregulation of Nek2B leads to a mitotic delay in the majority of cells, upon exiting mitosis, cells exhibit mitotic defects such as the formation of multinucleated cells. 4 The studies suggest that Nek2B may be required for the execution of mitotic exit.
The centrosome is the primary organizer of the microtubule network in animal cells.5,6 Some studies have shown that 60%–80% invasive breast cancers are aneuploid and approximately 80% exhibit amplified centrosomes. Our previous study 7 also demonstrated that centrosomal aberrations might play key role in breast tumorigenesis. Centrosome amplification and the associated chromosomal instability may greatly contribute to breast cancer progression.8,9 Guo et al 10 suggested that Nek2B is one of the first proteins recruited during centrosome assembly. Therefore, we assumed that Nek2B depletion would block the form of a functional centrosome as measured by microtubule aster formation, and the abnormal expression of Nek2B might influence the sensitivity of cancer cells to chemotherapy which disrupts the therapeutic targets of spindle poisons, such as paclitaxel.
Here, we report the analysis of Nek2B in the MCF-10 isogenic series of breast cancer cell lines. Specifically, we have analyzed Nek2B expression from MCF-10A (non-tumorigenic), MCF-10AT (premalignant and tumorigenic), MCF-10DCIS.com (tumorigenic and locally invasive), and MCF-10CA1a (tumorigenic and metastatic). These cell lines represent a stepwise progression of breast epithelial cells toward a metastatic phenotype.11,12 The cytogenetic and molecular variation may help to reveal genetic changes relevant to breast cancer. Hence, identification of Nek2B in these cells may yield insights into the development and progression of breast carcinoma. In addition, we showed that subcutaneous injection of Nek2B small interfering RNA (siRNA) in tumor nodules can suppress tumor growth in a xenograft nude mouse model. Moreover, the combined administration of both Nek2B siRNA and paclitaxel results in the suppression of tumor growth compared to the single administration of Nek2B siRNA or paclitaxel.
Chemotherapy is well established as one of the major treatment modalities for metastatic breast cancer patients. 13 Response rates to chemotherapy with anthracyclines, taxanes, capecitabine, vinorelbine, and gemcitabine range from 25%–60%, and the median time to progression was approximately 6 months. However, the puzzle is that tumor cells may become resistant to the chemotherapy agents. 14 By now, few studies have conducted Nek2B in human breast carcinoma tissues. We detected the messenger RNA (mRNA) expression of Nek2B in 62 cases of invasive ductal carcinoma (IDC) tissues obtained from patients who were treated with paclitaxel neoadjuvant chemotherapy, and a close relationship was found between the overexpression of Nek2B and the resistance to paclitaxel. Our study provided the clue to explore using Nek2B siRNA as a novel potential combination treatment for breast cancer especially for those who are resistance to paclitaxel.
Materials and methods
Cell culture
MCF-10A (non-tumorigenic and non-metastatic), MCF-10AT (tumorigenic and non-metastatic), and MCF-10CAla (metastatic) were obtained from the Barbara Ann Karmanos Cancer Institute of Wayne State University (Detroit, MI, USA). MCF-10DCIS.com (tumorigenic, locally invasive, and non-metastatic) was purchased from an Asterand Business Development Representative (Detroit, Michigan, USA). MCF-10DCIS.com and MCF-10CA1a were grown in Dulbecco’s Modified Eagle’s Medium/nutrient mixture F-12 (DMEM/F12; 1:1; Invitrogen, Carlsbad, CA, USA) with 5% horse serum, 0.029 M sodium bicarbonate, 10mM HEPES (4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid), penicillin, and streptomycin. MCF-10A and MCF-10AT were cultured in DMEM/F12 (Invitrogen) supplemented with 5% horse serum, 20 ng/mL of epidermal growth factor, 100 ng/mL of cholera toxin, 10 µg/mL of insulin, and 0.5 µg/mL of hydrocortisone.11–13
Reverse transcription polymerase chain reaction
Reverse transcription polymerase chain reaction (RT-PCR) array was used to analyze the Nek2B expression or the efficiency of transfection. Total RNA was isolated from cultured cells using TRI reagent (Invitrogen), according to the manufacturer’s instructions. RNA was treated with DNase I (RNase-free; Roche; Mannheim, Germany) and purified over an RNeasy column. The first-strand complementary DNA (cDNA) was synthesized with oligo(dT) primer using the Reverse Transcription System (Promega, Beijing, China). PCR amplification of Nek2B-specific fragments of 497 bp was performed in a 20 µL reaction using TaqDNA polymerase (Invitrogen), with 1 µL of the first-strand cDNA synthesis mixture as the template. The primers set was 5′-CTAGCTAGCTAGCCGTCACGGGTCGAG-3′ (forward) and 5′-CGGGATCCTTAGAATTTGCTCCATTCATTCC-3′ (reverse) for Nek2B. The PCR reaction condition was as follows: 95°C for 5 min, 95°C for 15 sec, 62.8°C for 15 sec, 72°C for 20 sec, and 72°C for 5 min, performed for 35 cycles. The expression of β-actin was used as the internal control.
Western blotting
Cells were grown in 100-mm dishes and lysed in radioimmunoprecipitation assay (RIPA) buffer and 1 mM phenylmethylsulfonyl fluoride (PMSF). The extracts were centrifuged and the supernatant fractions were collected for western blot analysis. Total cell lysates containing 30 µg of protein were electrophoresed on a sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) using precast 4%–20% gradient Tris–glycine gels (Invitrogen) and then transferred to polyvinylidene fluoride (PVDF) membranes (Millipore, MA, USA). The membranes were blocked with 5% skim milk for 1 h at room temperature and then incubated with primary antibodies: anti-Nek2B (1:500; Sigma, St Louis, USA), anti-γ-tubulin (1:1000; Sigma, St Louis, USA), and anti-β-actin (1:2000; Santa Cruz Biotechnology, USA). The membranes were then incubated with Tris buffered saline with Tween 20 (TBST) containing horseradish peroxidase–labeled anti-rabbit IgG (1:5000) or horseradish peroxidase–labeled anti-mouse IgG (1:5000) and the chemiluminescence was detected by electrochemiluminescence (ECL, Millipore, USA).
Transfection of plasmid and siRNA
Nek2B expression plasmid (pcDNA-mycNek2B) was generated by cloning its coding region into the pcDNA3.0-myc vector (Invitrogen, Beijing, China). And the pcDNA3.0-myc vector was used as control. For plasmid DNA transfections, MCF-10A and MCF-10AT cells were plated on six-well plates 2 days prior to transfection and cultured to 70% confluence the following day in 5% horse serum. Cells were transfected with pcDNA3.1-Nek2B or with the empty vector as the control according to the manufacturer’s instructions using Lipofectamine 2000 (Invitrogen). After 48 h of transfection, the cells were harvested and used for further experiments. Nek2B (mRNA target: CTT-GGT-CAA-CAG-AAG-CTG-G) siRNA was obtained from Invitrogen, together with a commercially available control siRNA (Cat. No. 12935–300; Invitrogen). After 72 h of transfection, MCF-10DCIS.com and MCF-10CA1a cells were harvested and used for further experiments.
Colony formation assay
For colony formation assay, 5000 MCF-10AT-Nek2B or MCF-10AT-control cells in complete DMEM/F12 medium were seeded onto 10-cm culture dish and allowed to grow for 14 days to form colonies. Results were observed and photographed after Giemsa staining.
Immunofluorescence
Cells grown on glass coverslips were fixed with cold methanol for 5 min. Cells were then blocked by 2% bovine serum albumin in phosphate-buffered saline (PBS) for 30 min. Cells were then fixed and double-stained with anti-Nek2B (1:200; Sigma) or anti-γ-tubulin (1:1000; Sigma). The cells were then incubated with Alexa Fluor 594 goat anti-mouse or Alexa Fluor 488 goat anti-rabbit (1 g/mL; Invitrogen) as the secondary antibodies. After washing with PBS, cells were mounted with 4′,6-diamidino-2-phenylindole (DAPI; Thermo Fisher Scientific, Waltham, MA, USA) away from light. Images were acquired using an Olympus IX71 fluorescence microscope (Olympus, Tokyo, Japan) and color images were generated using the LuminaVision software (Mitani Sangyo Co., Tokyo, Japan).
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay
MCF-10CA1a-Nek2B siRNA and MCF-10CA1a-control siRNA cells (1 × 103) were seeded on a 96-well microtiter plate and incubated overnight. Paclitaxel with different concentrations (0–20 nmol/L) were added to each cell culture for 72 h. The cells were incubated with 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT; 0.5 mg/mL) for 4 h. The formazan precipitate was dissolved in 200 µl dimethylsulfoxide (DMSO), and the spectrometric absorbance at wavelength of 570 nm was measured on a benchmark microplate reader (Bio-Rad Laboratories,Tianjin, China). At least three independent experiments were done and the averages were calculated.
Detection of apoptosis by flow cytometry
The Annexin V-fluorescein isothiocyanate (FITC) binding assay was performed to determine the apoptosis rate of cells in vitro. Briefly, cells were seeded on a six-well plate overnight, harvested after 48 h of treatment, and incubated with 5 µl of Annexin V-FITC and 5 µl of propidium iodide (PI) for 15 min at room temperature in the dark. Apoptosis was detected within 1 h in a flow cytometer. Each experiment was performed three times.
In vivo experiments
Female athymic nude mice (aged 7–8 weeks, 20–25 g in weight) were purchased from Tianjin Medical University Cancer Institute. MCF-10CA1a cells (1 × 106) were suspended in 100 µL of Hank’s balanced salt solution (Gibco; Invitrogen, China) and injected into the right femoral area of each mouse. To confirm the amount of paclitaxel that could inhibit tumor growth, we administered intraperitoneally different concentrations and found that the efficacy of tumor suppression reached plateau at a dose of 30 mg/kg. After 7 days of tumor inoculation, paclitaxel was given at 30 mg/kg once a week for 4 weeks in the abdominal cavity of the mice with the concentration of 0.6 mg/mL, while Nek2B siRNA was administered directly around the tumor at 80 µL with the concentration of 20 µM. PBS or control siRNA was used as controls. Tumor dimensions were measured by a vernier caliper twice a week and the tumor volume was calculated using the following formula (length (cm) × width (cm) × width (cm) × 0.5326).
Tissue specimens
A total of 62 cases invasive breast cancer (IDC) specimens based on core-biopsy before operation were collected in this study, with a median age of 53.3 years. All patients underwent neoadjuvant chemotherapy and treated with paclitaxel, epirubicin, and cyclophosphamide (TEC) regimen for 2–3 cycles at Tianjin Medical University Cancer Institute and Hospital from January 2010 to January 2012. According to the different response to neoadjuvant chemotherapy TEC, the patients were divided into four groups: pathology complete response (pCR), partial response (PR), stable disease (SD), progressive disease (PD).
Immunohistochemistry
The expression status of estrogen receptor/progesterone receptor (ER/PR), c-erbB-2, and Ki67 was determined by immunohistochemistry (IHC). Normal breast tissues as control were obtained from the quadrant far away from the original foci of IDC. The pathologic diagnosis was confirmed by two senior pathologists.
Statistical analysis
The SPSS 17.0 software package was used for statistical analysis. Mann–Whitney U test and Kruskal–Wallis test were performed for group comparisons. Correlations between two variables were evaluated by Spearman’s rank correlation. The statistical differences of continuous variable were analyzed by Student’s t test or repeated measures of analysis of variance (ANOVA). For all statistical analyses, the level of significance was set at p < 0.05.
Results
Nek2B mRNA and Nek2 protein expression in MCF-10 cells
The protein expression of Nek2 was examined in the breast cancer cell lines of MCF-10A, MCF-10AT, MCF-10DCIS.com, and MCF-10CA1a by western blotting, and the increased expression of Nek2 was observed in MCF-10DCIS.com and MCF-10CA1a, whereas it was hardly detectable in MCF-10A and MCF-10AT (Figure 1(a)). To examine the expressions of Nek2B mRNA, plasmid pcDNA3.0-mycNek2B or pcDNA3.0-myc vector was transfected into MCF-10A (Figure 1(b)) and MCF-10AT (Figure 1(c)) cell lines. The expression of Nek2B mRNA in Nek2B-transfected groups was significantly increased compared to the control groups (Figure 1(b) and (c)). MCF-10DCIS.com and MCF-10CA1a were transfected with Nek2B siRNA or the control siRNA. The reduced expression of Nek2B was observed in the MCF-10DCIS.com (Figure 1(d)) and MCF-10CA1a cells (Figure 1(e)), which were transfected with Nek2B siRNA. As shown by Western blot analysis, the expression levels of Nek2B protein were significantly increased in Nek2B-transfected MCF-10A and MCF-10AT groups compared with the control groups, while decreased in Nek2B siRNA–transfected MCF-10DCIS.com and Nek2B siRNA–transfected MCF-10CA1groups (Figure 1(f))

The expression of Nek2 and Nek2B in MCF-10 cell lines. Western blotting showed Nek2 protein expression was higher in MCF-10DCIS.com and MCF-10CA1a than MCF-10A and MCF-10AT cell lines (a). As shown by RT-PCR analysis, the expression levels of Nek2B mRNA were significantly increased in (b) Nek2B-transfected MCF-10A and (c) MCF-10AT groups compared with the control groups, while decreased in (d) Nek2B siRNA–transfected MCF-10DCIS.com and (e) Nek2B siRNA–transfected MCF-10CA1 groups. As shown by Western blot analysis, the expression levels of Nek2B protein were significantly increased (f) in Nek2B-transfected MCF-10A and MCF-10AT groups compared with the control groups, while decreased in Nek2B siRNA–transfected MCF-10DCIS.com and Nek2B siRNA–transfected MCF-10CA1groups.
Effect of Nek2B in MCF-10AT cell line
We subsequently examined the effect of Nek2B transfection on the growth of MCF-10AT cell by colony formation assay. The number of colonies formed by transfection with Nek2B MCF-10AT cell was increased compared with control group (Figure 2(a), left). The clones in Nek2B siRNA–transfected group were decreased compared with control group in MCF-10CA1 cell line (Figure 2(a), right). The data indicated that Nek2B may increase the proliferation of MCF-10AT cells.
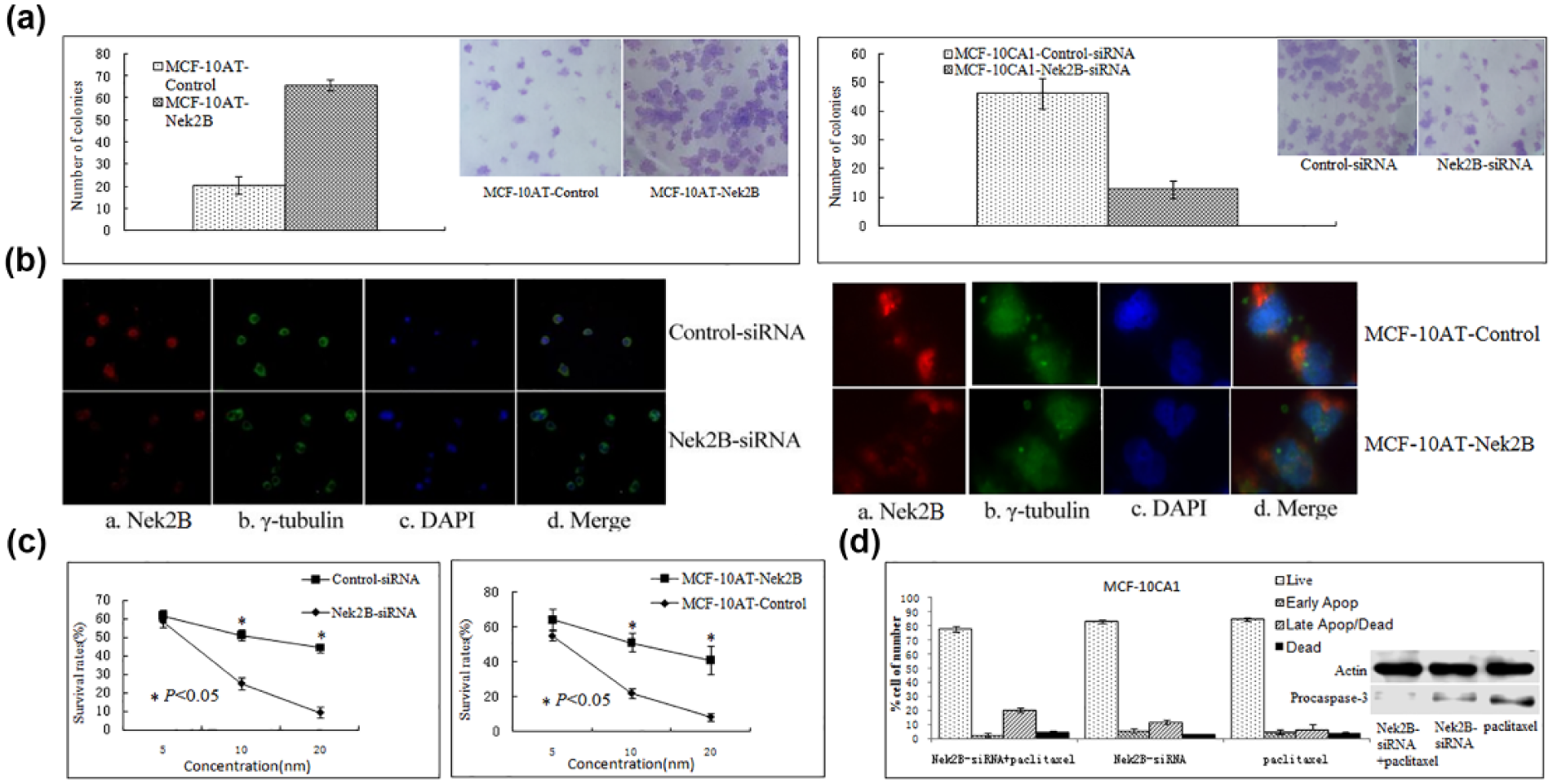
Colony formation assay showed that (a: left) the clones in Nek2B-transfected group were increased compared with control group in MCF-10AT cell line. (a: right) The clones in Nek2B siRNA–transfected group were decreased compared with control group in MCF-10CA1 cell line. (b: left) Polykaryocytes were found in Nek2B siRNA–transfected group in MCF-10DCIS.com cell line by immunofluorescence. (b: right) There were no polykaryocytes found in Nek2B-transfected group and control group in MCF-10AT cell line. (c: left) MTT showed that the survival rate of the cells in Nek2B siRNA–transfected group was reduced compared to control group in MCF-10CA1a cell line treated with paclitaxel. (c: right) The survival rate of the cells in Nek2B-transfected group was increased compared to control group in MCF-10AT cell line treated with paclitaxel. (d) Flow cytometry was performed to analyze cell apoptosis in MCF-10CA1 cell. The percentage of apoptotic cells was higher in the Nek2B siRNA combining with paclitaxel group compared to Nek2B siRNA group and paclitaxel group, respectively, and procaspase-3 were significantly decreased in Nek2B siRNA group compared with Nek2B siRNA group and paclitaxel group.
Effect of Nek2B in MCF-10DCIS.com and MCF-10AT cell line
After transfection with Nek2B siRNA and control siRNA in MCF-10DCIS.com cell, immunofluorescence was conducted to determine whether the altered expression of Nek2B affected the subcellular structure of MCF-10DCIS.com cells. As shown in Figure 2(b), staining by antibodies Nek2B (Figure 2(b), left) and γ-tubulin (Figure 2(b), right), reduced expression of Nek2B did not appear to cause a significant abnormal centrosome; however, polykaryocytes were found more in Nek2B siRNA–transfected group than in control group in MCF-10DCIS.com cell line (left). While there were no polykaryocytes found in Nek2B-transfected group and control group in MCF-10AT cell line (right). These data indicated that knockdown of Nek2B gene can decrease the proliferation by suppressing the activity of microtubule polymerization in MCF-10DCIS.com cell line.
Effect of Nek2B and Paclitaxel in MCF-10CA1and MCF-10AT cell line
Paclitaxel with different concentrations was administered in Nek2B siRNA–transfected group and control in MCF-10CA1 cell line for 72 h. As shown in Figure 2(c), Nek2B siRNA–transfected group can reduce the survival rate of the cells more efficiently compared to control group (left). The survival rate of the cells in control group was decreased more quickly compared to Nek2B group in MCF-10AT cell line treated with paclitaxel (right). It showed that Nek2B may facilitate resistance to paclitaxel. To further demonstrate the correlation between Nek2B and paclitaxel resistance in breast cancer cells, we performed flow cytometry to analyze cell apoptosis in MCF-10CA1 cell (Figure 2(d)). Data showed that the percentage of apoptotic cells was higher in the Nek2B siRNA combining with paclitaxel group compared to Nek2B siRNA group and paclitaxel group, respectively, and the levels of proapoptotic marker procaspase-3 were significantly decreased in Nek2B siRNA group compared with Nek2B siRNA group and paclitaxel group.
Combination treatment using Nek2B siRNA and paclitaxel in nude mice
To further detect the effect of Nek2B in breast cancer, we performed a combination treatment using Nek2B siRNA and paclitaxel which disrupted the therapeutic targets of spindle poisons in nude mouse model. As shown in Figure 3(A) and (B), the tumor volume in Nek2B siRNA combining with paclitaxel group was decreased compared with Nek2B siRNA group (p < 0.01) and paclitaxel group (p < 0.05), respectively. The data demonstrated that combination treatment using Nek2B siRNA and paclitaxel may suppress breast tumor growth rate more efficiently than paclitaxel alone or Nek2B siRNA alone.
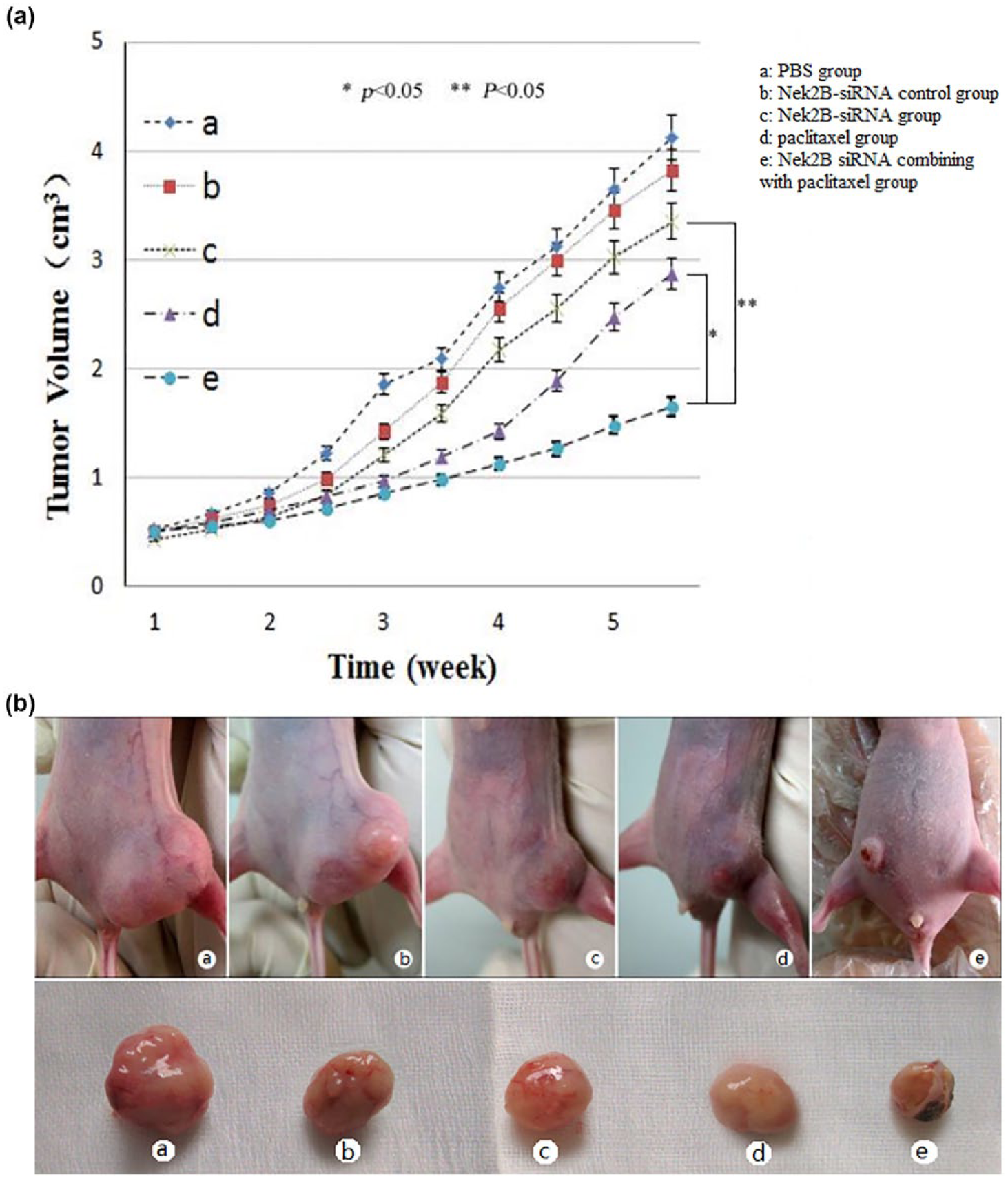
(A) Combination treatment using Nek2B siRNA and paclitaxel in nude mouse model. The nude mice were divided into five subgroups (a–e) by different treatment, (a) PBS group, (b) Nek2B siRNA–control group, (c) Nek2B siRNA group, (d) paclitaxel group, (e) Nek2B siRNA combining with paclitaxel group. The tumor volume in Nek2B siRNA combining with paclitaxel group was decreased compared with Nek2B siRNA group (p < 0.05) and paclitaxel group (p < 0.05), respectively (*p < 0.05; **p < 0.05). (B) Representative photographs of tumors isolated from each group after inoculation with different treatment MCF-10CA1a cells.
Expression of Nek2B mRNA in human breast cancer tissues
The IHC images of Nek2B, ER, c-erbB-2, and Ki67 were shown in Figure 4. According to the different response to neoadjuvant chemotherapy TEC, the 62 patients were divided into four groups, 12 cases were pCR, 25 cases were PR, 11 cases were SD, and 14 cases were PD. As shown in Table 1, the PR rates were 75.0% in Nek2B negative (Nek2B−) patients and 15.8% in Nek2B++ patients. Inversely, the PD rates were 10.0% in Nek2B− patients and 52.6% in Nek2B++ patients (p = 0.002). It indicated that higher Nek2B expression was correlated with lower sensitivity for paclitaxel chemotherapy.

Serial slices with immunohistochemical analysis of Nek2B, ER, c-erbB-2, and Ki67 expression in breast cancer specimens: (a) Nek2B positive stained (200×), (b) ER positive stained (200×), (c) c-erbB-2 positive stained (200×), and (d) Ki67 positive stained (200×).
Correlations between the expressions of Nek2B mRNA and paclitaxel sensitivity.
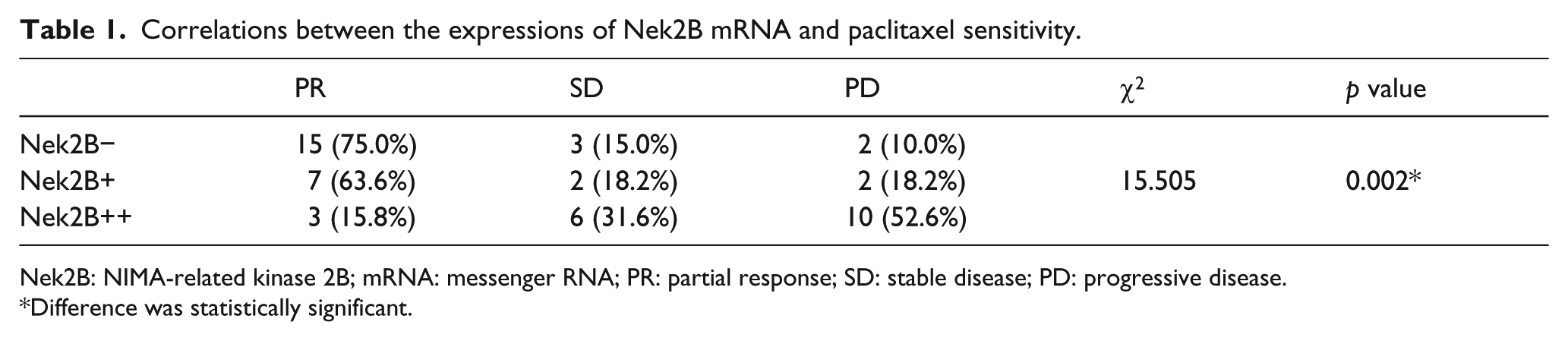
Nek2B: NIMA-related kinase 2B; mRNA: messenger RNA; PR: partial response; SD: stable disease; PD: progressive disease.
Difference was statistically significant.
In order to explore the correlations between expression of Nek2B mRNA and other prognostic factors in IDC patients, tumor size, lymph node status, histological grades, c-erbB-2, Ki67, and ER/PR status were analyzed. As shown in Table 2, there were correlations between expression of Nek2B mRNA and lymph node status (p = 0.016), histological grades (p = 0.000), and Ki67 index (p = 0.006).
Correlations between the expressions of Nek2B mRNA and other prognostic factors.

Nek2B: NIMA-related kinase 2B; mRNA: messenger RNA; ER/PR: estrogen receptor/progesterone receptor.
Difference was statistically significant.
Discussion
Centrosomes play a dominant role in bipolar spindle formation during mitosis in most animal cells. 15 Centrosome aberrations were believed to be correlated with the generation of aneuploidy and the acquisition of a chromosome instability phenotype in breast tumors. Deregulation of cell cycle regulators, including p53, BRCA1, Aurora-A, Nek2, Plk1, and Cyclin B1, are closely associated with tumorigenesis and clinical therapeutic sensitivity as well. 16 As the important centrosome regulatory factor, Nek2 is transported into the centrosome and aligns along microtubules. 17 Of the three splice variants (Nek2A, Nek2B, and Nek2C), Nek2B is the most significant one for the efficient assembly or maintenance of centrosomes.3,18 To examine whether Nek2B can influence bipolar spindle formation, Sato et al. 19 found when immunostained with anti-tubulin antibody, embryos injected with dominant-negative Nek2B mRNA exhibited numerous dot-like structures instead of a bipolar spindle, each of the dot-like structures had an associated microtubule aster, indicating that it acted as a microtubule-organizing center (MTOC). In most animal cells, the MTOC is equivalent to the centrosome, duplication and separation of intact centrosomes are fundamental for bipolar spindle formation, 20 and the abnormal expression of Nek2B might influence the sensitivity of cancer cells to paclitaxel which disrupts the therapeutic targets of spindle poisons. The appearance of numerous MTOCs in Nek2B-inhibited embryos could have been either due to a failure to assemble or maintain the centrosome structure, or due to a spontaneous development of MTOCs. 10 It strongly suggested that Nek2B was recruited to the sperm centrosome upon fertilization and was a component of the centrosome. Similar results 21 demonstrating that the defect in bipolar spindle formation was the primary and direct cause of the abortive cleavage of Nek2B-inhibited embryos were obtained. Thus, these results do suggest that Nek2B localizes to the centrosome and is essential for its assembly and/or maintenance.
In this study, we detected the Nek2B protein or mRNA expression in MCF-10 cell lines, including MCF-10A, MCF-10AT MCF-10DCIS.com, and MCF-10CA1a. Data showed that Nek2B mRNA expression was significantly upregulated in MCF-10DCIS.com and MCF-10CA1a cell lines than in MCF-10A and MCF-10AT. So we inferred that Nek2B may introduce tumorigenesis. Myc-tagged Nek2B plasmid DNA was transfected into MCF-10A and MCF-10AT breast cancer cell lines. Clone formation test showed that the clones in Nek2B-transfected group were increased compared with control group in MCF-10AT cell line. It indicated that the expression of Nek2B may stimulate the growth of the premalignant immortalized cells. In MCF-10DCIS.com cell lines, we demonstrate that the downregulation of Nek2B results in a mitotic delay that can lead to mitotic defects, such as the formation of multinucleated cells. It may indicate that the overexpression of Nek2B in breast tumors did not reveal the formation of bipolar spindle and the stabilization of microtubule as in normal tissues but might change microtubule structure or dynamics and then alter formation of spindle through demolished function of cell cycle checkpoints. Nevertheless, the precise mechanisms by which centrosomes are assembled and nucleate microtubules are not known. 22
It is well-known that paclitaxel can stabilize microtubules and also interacts directly with microtubules by enhancing the polymerization of tubulin. 23 Paclitaxel is widely used for chemotherapy in solid tumors, such as breast tumors, ovarian, and lung cancers. However, the clinical use of this drug is limited due to the emergence of intrinsic and acquired resistance and severe side effects. 24 In our study, paclitaxel with different concentrations was administered in Nek2B siRNA–transfected group and control group in MCF-10CA1a cell line. MTT results showed that the survival rate of the cells in Nek2B siRNA–transfected group was reduced compared to control group. It seemed that overexpression of Nek2B may resist the apoptosis in vitro breast cancer cells treated by paclitaxel. Several studies indicated molecular-target therapy using siRNA combined with chemotherapy drugs was effective in cancers in vitro.25,26 Karasawa 15 reported that cellular inhibitor of apoptosis protein-2 siRNA efficiently enhanced tumor cell sensitivity to 5-flurouracil (5-FU). Guo 11 found that nuclear factor-kappa B siRNA inhibited chemotherapy resistance. Tsunoda et al. 27 showed that Nek2 siRNA induced growth suppression of breast cancer cells in vitro and significantly inhibited tumor growth in a xenograft nude mouse model. To investigate the potential utility of Nek2B siRNA for clinical cancer therapy, we examined the effect of Nek2B in nude mice. Tumor cells treated by Nek2B siRNA showed significantly smaller tumor volume. Furthermore, the combination of Nek2B siRNA and paclitaxel achieved better tumor suppression in MCF-10CA1a cells than administration of either Nek2 siRNA alone or paclitaxel alone. Nek2B siRNA might lessen the ability of paclitaxel depressing depolymerization of microtubule and resist apoptosis induced by paclitaxel. Thus, combination treatment using Nek2B siRNA and paclitaxel may be a useful therapeutic option for breast cancer.
To corroborate and extend our findings in cell lines, the expression and association of Nek2B with clinicopathological factors were explored in 62 cases of IDC tissues obtained from the patients who were treated by neoadjuvant chemotherapy (TEC). It showed that the overexpression of Nek2B mRNA might be associated with the high invasion or bad prognosis in breast cancer. And a close relationship was found between the overexpression of Nek2B and the resistance to neoadjuvant chemotherapy. We assumed that Nek2B siRNA was a novel potential treatment for those being confirmed to have a tendency to resist paclitaxel. It will be especially significant for the patients who were treated by neoadjuvant chemotherapy and/or resistant to paclitaxel.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the Tianjin Medical University Cancer Institute and Hospital, China and has been performed in accordance with the ethical standards laid down in the 1964 Helsinki declaration and its later amendments.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by National Science Foundation (81172532 and 81470119).
