Abstract
Increasing evidence supports that the dysregulation of microRNA expression plays an important role in the process of tumor occurrence and development. Studies have found that mir-125a-5p expression was downregulated in a variety of tumors, but the effects and mechanism of mir-125a-5p in lung cancer are still unclear. The aim of this study is to detect the expression of mir-125a-5p in lung cancer tissues and lung cancer cell lines and to explore the effects of mir-125a-5p on the biological characteristics of lung cancer cells; thus, this study aims to provide new methods and new strategies for the treatment of lung cancer. The result from quantitative reverse transcription polymerase chain reaction showed that the expression of miR-125a-5p was significantly lower in lung cancer tissues and lung cancer cell lines (95-D, A549, HCC827, and NCI-H1299) than that in normal tissue adjacent to lung cancer or normal human bronchial epithelial cells. In order to explore the function and mechanism of mir-125a-5p in lung cancer cells, miR-125a-5p mimic or mir-125a-5p inhibitor was transfected into A549 cells. Mir-125a-5p displayed an obvious upregulation in A549 cells transfected with miR-125a-5p and an obvious downregulation in A549 cells transfected with mir-125a-5p inhibitor compared to that in A549 cells transfected with control miRNA. 3-(4,5-Dimethyl-2-thiazolyl)-2,5-diphenyl-2-H-tetrazolium bromide, BrdU staining, flow cytometry, and Transwell assay showed that the upregulation of miR-125a-5p could significantly decrease the cell viability, proliferation, and invasion of lung cancer cells and increase apoptosis of lung cancer cells. The downregulation of miR-125a-5p provided very contrasting results. Computational algorithms predicted that the STAT3 is a target of miR-125a-5p. Here, we validated that miR-125a-5p could directly bind to the 3′-untranslated region of STAT3, and miR-125a-5p overexpression could significantly inhibit the protein expression of STAT3. These results suggested that mir-125a-5p can regulate the expression of STAT3 in lung cancer cells. To further verify whether mir-125a-5p can play a biological role through regulating STAT3, 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2-H-tetrazolium bromide, flow cytometry, and Transwell analysis demonstrated that overexpression of STAT3 can reverse the cells’ biological effects induced by mir-125a-5p overexpression. Mir-125a-5p downregulated in lung cancer tissue and cell lines can negatively regulate STAT3 protein expression. Taken together, mir-125a-5p inhibited the proliferation and invasion of lung cancer cells and facilitated lung cancer cell apoptosis through suppressing STAT3. Enhancing the expression of miR-125a-5p is expected to benefit the therapy for the patients with lung cancer.
Lung cancer is one of the main malignant tumors which seriously threaten human health. Despite a lot of labor, materials, and capitals invested into the diagnosis and treatment of lung cancer every year, due to strong latency of lung cancer and a higher degree of malignancy, the disease would be in the later stage when it is found, and the optimal treatment opportunity would always be missing.1–3 In recent years, although great progress has been made in the treatment of lung cancer, the survival rate of patients with lung cancer is still not satisfactory. 4 We have not made great breakthrough for lung cancer and lack understanding for its mechanism of occurrence and development. 5 Therefore, elucidating the pathogenesis and searching for new treatments are very urgent to improve the survival rate of patients with lung cancer.6,7 It is now believed that the occurrence of lung cancer is a multi-factor, multi-stage, and multi-gene change process. The dysregulation of oncogenes and tumor suppressor genes is the molecular basis of the lung cancer occurrence.8,9
In recent years, microRNA (miRNA) is a kind of non-coding small RNA which widely exists in animals and plants. Its length is about 20–24 nucleotides, which resulted from long transcript primary miRNA (pri-miRNA) and precursor-miRNAs (pre-miRNA).10–12 The miRNAs regulate post-transcriptional gene expression by base pairing with 3′-untranslated region (3′-UTR) sequences of target messenger RNAs (mRNAs). In multi-cellular organisms, post-transcriptional gene expression regulation affects almost all the signaling pathways, which is involved in a variety of physiological and pathological processes, and plays an important role in the occurrence and development of tumors.13–16
The invasion and metastasis of tumors are the most common reason for the death of patients with cancer in the world. 17 Recently, more and more studies have reported that miRNA is related to the invasion and metastasis of tumor cells. Its mechanism of action to regulate tumor cell invasion and metastasis has become a focus in the research field.18,19 And a large number of studies confirmed that tumor invasion and metastasis are related to miRNA.20,21
Yanaihara et al.22,23 found that miR-125a was located in 19q13.41 and analyzed the lung cancer tissues and the corresponding adjacent lung tissues with core chip, which displayed that expression of miR-125a in lung cancer tissues was lower than that in the adjacent lung tissues. So far, the mature miR-125a family has two members, miR-125a-3p and miR-125a-5p.24,25 In 2007, Scott et al. 26 found that mir-125a acted as a potential tumor suppressor miRNA in breast tumorigenesis via downregulating the expression of ERBB2 and ERBB3 in breast cancer cells; thus, it inhibits the proliferation and metastasis of breast cancer cells. Some research groups reported that mir-125a could inhibit breast cancer cell proliferation by suppressing the expression of human antigen R (HuR) in breast cancer cell. 27 However, the effect and mechanism of miR-125a-5p on lung cancer cell biological characteristics are not very clear.
Therefore, in this study, we first examined the expression of mir-125a-5p in human lung cancer tissues and lung cancer cells. Due to mir-125a-5p ectopic expression in lung cancer cells, 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2-H-tetrazolium bromide (MTT) assay, flow cytometry analysis and invasion assay were used to examine the influence of mir-125a-5p on the biological characteristics of lung cancer cells. Online software was used to analyze the possible target genes of mir-125a-5p, which was validated through the luciferase reporter gene assay. Rescue experiment was used to explore the action and mechanism of mir-125a-5p in lung cancer. This study may provide experimental basis for application of miRNA in the lung cancer treatment.
Materials and methods
Sample
In all, 18 fresh lung cancer tissues and adjacent normal lung tissues were collected from patients with lung cancer during the surgery in our hospital and were immediately frozen in liquid nitrogen after surgery for subsequent experiments. Before surgery, all the patients had no radiotherapy and chemotherapy, no other treatment history, and no inflammatory diseases. The study protocol was approved by the Ethics Committee of The Affiliated Hospital of Nantong University, Nantong, China.
Methods
Cell treatment
95-D and human bronchial epithelial (HBE) cells were cultured in RPMI 1640 medium containing 10% fetal bovine serum (FBS). NCI-H1299 cells were cultured in RPMI 1640 medium containing 1.0 mM sodium pyruvate, 2.0 mM
The above-mentioned cells were cultured under the temperature of 37°C and 5% CO2 saturated humidity conditions. And then, they were placed under the inverted microscope to observe the growth. Once cells reached 70%–80% confluence, cells were treated with 0.25% trypsin (Invitrogen) for digestion and passage. When the cells were in the logarithmic growth phase, they could be collected for further experiment.
Construction of STAT3 recombinant expression plasmid
According to human STAT3 mRNA sequence in GenBank (NM_139276) and enzyme cleavage site analysis, premier primer 5 software was used to design a pair of primers on both sides of STAT3 open reading frame (ORF). STAT3 upstream primer P1: 5′-CGGGATCCATGGCCCAATGGAATCAGCT-3′, cleavage sites containing BamH I; STAT3 downstream primer P2: 5′-CCCTCGAGCATGGGGGAGGTAGCGCACT-3′, cleavage sites containing enzyme Xho I. Primers were provided by Invitrogen Biotech Corp (Shanghai, China).
Normal cultured lung cancer A549 cells were collected and treated with 1 mL TRIzol reagent to extract the total RNA of A549 cells according to the product specification operation steps. And then, reverse transcription kit (Qiagen, Hilden, Germany) was used for reverse reaction, and thus, the first strand was synthesized according to the manufacturer’s instructions. STAT3 primer was dissolved in ddH2O and packed at −20°C for preservation. A volume of 1 µL of complementary DNA (cDNA) was added into 25 µL polymerase chain reaction (PCR) reaction system, and the amplification conditions were set: pre-denatured DNA at 94°C for 5 min, the degeneration of 30 s at 94°C, annealing of 30 s at 55°C, and extension of 60 s at 72°C. Such actions were repeated for 30 times. Finally, extension of 10 min at 72°C was done, and 1.5% agarose gel electrophoresis was used to identify PCR products. PCR product was connected with pGEM-T vector to select the positive clones. After correct sequencing, expression vector was sub-cloned to reconstruct expression vector pcDNA3.1-STAT3. The TRIzol reagent required for the above-mentioned procedure and pGEM-t and pcDNA3.1 vectors were purchased from Invitrogen. Restrictive endonuclease BamH I and Xho I and DNA marker were purchased from TaKaRa (Dalian, China). T4DNA Ligation was purchased from Promega (Madison, WI, USA). Taq DNA polymerase and pre-staining protein molecular weight standards were purchased from Fermentas (Thermo Fisher Scientific, Waltham, MA, USA).
Cell transfection
A549 cells were uniformly inoculated in a 6 well cell culture. After cell adherence, miR-125a-5p mimic, mir-125a-5p inhibitor, corresponding negative control (GenePharma, Shanghai, China), restructuring plasmid pcDNA3.1-STAT3, and empty vector control pcDNA3.1 were transfected according to Lipofectamine 2000 (Invitrogen) transfection instructions. At the same time, normal control group was set up. Minimum essential medium (MEM) culture medium without serum was used to dilute miR-125a-5p mimic, pre-miR-125a-5p inhibitor, corresponding negative control, pcDNA3.1-STAT3, and pcDNA3.1. Then, Lipofectamine 2000 was collected and diluted in MEM culture medium and then gently mixed and incubated for 5 min at room temperature. The diluted Lipofectamine 2000 was mixed with the previous diluted mixture (miR-125a-5p mimic, miR-125a-5p inhibitor, corresponding negative control, pcDNA3.1-STAT3, and pcDNA3.1) and then gently mixed and incubated for 20 min at room temperature to form a compound. The compound was added into the culture plate containing A549 cells and was placed in a culture incubator with the 5% CO2 at 37°C. After 5 h, the transfection culture medium was replaced by the MEM culture medium containing 10% FBS for 48 h.
Examination of the expression of miR-125a-5p
The quantitative real-time polymerase chain reaction technology (qRT-PCR) method was used to detect the expression of mir-125a-5p in lung cancer tissues, adjacent normal lung tissues, lung cancer cells (95-D, A549, HCC827, and NCI-H1299), and HBE. Lung cancer cells and tissues were collected. TaqMan miRNA isolation kit was used to extract RNA. TaqMan miRNA assay and TaqMan Universal PCR Master Mix (Applied Biosystems, Foster City, CA, USA) were used to detect the expression of mature mir-125a-5p. Meanwhile, U6 was used as internal reference. The experimental result was analyzed by qRT-PCR relative quantitative method.
After 48 h of transfection of A549 cells for miR-125a-5p mimic, mir-125a-5p inhibitor, and the negative control (NC), TaqMan miRNA isolation kit was used to extract RNA from A549 cells of each group. The qRT-PCR was used to detect the change in expression of mir-125a-5p in A549 cells of different groups.
Detecting the effect of miR-125a-5p on cell viability
MTT analysis method was used to detect the effect of miR-125a-5p ectopic expression on A549 cell viability. After 48 h of transfection of A549 cells for miR-125a-5p mimic, mir-125a-5p inhibitor, and the corresponding negative control, 100 µL MTT (0.5 mg/mL) solution was added into each hole and then placed in 5% CO2 culture box at 37°C for 4 h. A volume of 100 µL of 20% sodium dodecyl sulfate (SDS; cosolvent 50% dimethylformamide) solution was added into each hole at 37°C for 24 h. Optical density (OD) value was measured at 570 nm. In each experimental group, 10 complex holes were set up. Experiments were repeated for three times.
Detecting the effect of miR-125a-5p on cell proliferation
BrdU staining was used to detect the effect of miR-125a-5p ectopic expression on proliferation. After 48 h of transfection of A549 cells for miR-125a-5p mimic, miR-125a-5p inhibitor, and negative control, the culture medium was removed. BrdU-labeled liquid (Roche, Alameda, CA, USA) was added for 30 min. Cells were fixed in ethanol solution at −20°C for 30 min and blocked at 37°C for 30 min. Then, cells were incubated with BrdU primary antibodies (Roche) at 37°C for 30 min and with BrdU secondary antibodies (Roche) at 37°C for 30 min. After that, cells were labeled with 0.5 g/mL Hoechst 33342 (Sigma-Aldrich, St. Louis, MO, USA) for 20 min. Finally, the sample was placed under fluorescence microscope for observation; 12 view fields were randomly selected from each group and the experiment was repeated for three times.
Detecting the effect of miR-125a-5p on cell apoptosis
Flow cytometry analysis was used to detect the effect of miR-125a-5p ectopic expression on cell apoptosis. After 48 h of transfection of A549 cells for miR-125a-5p mimic, miR-125a-5p inhibitor, and negative control, A549 cells were washed with phosphate-buffered saline (PBS) and incubated with Annexin V-fluorescein isothiocyanate (FITC; Roche) and propidium iodide (PI) staining solution (Roche) in dark at room temperature for 15 min. After the sieve screening, A549 cells were analyzed by flow cytometry. FCM CellQuest software and Macquit software (BD Biosciences, San Jose, CA, USA) were used to analyze the data.
Detecting the effect of miR-125a-5p on cell invasion
Transwell invasive analysis was used to detect the effect of miR-125a-5p ectopic expression on cell invasion. A549 cells were transfected with miR-125a-5p mimic, miR-125a-5p inhibitor, and corresponding negative control. After 24 h, A549 cells were inoculated in Transwell invasion chamber (Costar, Cambridge, MA, USA) pre-covered with a layer of Matrigel (BD Biosciences) and cultured under the normal condition for 24 h beforehand. A549 cells were washed and stained with Hoechst 33342 (Sigma-Aldrich). Eight view fields were observed randomly, and the number of cells passing through the Transwell polycarbonate membrane was counted.
STAT3 is a target gene of miR-125a-5p
Online software TargetScan (http://www.targetscan.org) was used to analyze and predict that STAT3 might be the target gene of miR-125a-5p. Luciferase reporter gene system was used to validate whether STAT3 was the target gene of miR-125a-5p. The sequences of STAT3 3′-UTR, which may be combined with miR-125a-5p, were cloned into pMIR-REPORTER luciferase vector (Applied Biosystems); therefore, the wild type (WT) of STAT3 luciferase reporter plasmid (STAT3WT-luc) could be constructed. Then, point mutation method was used to make a section of nucleotide sequences contained in STAT3 3′-UTR which could combine with miR-125a-5p, which were cloned into pMIR-REPORTER luciferase vector; therefore, the mutation (MUT) type of STAT3 luciferase reporter plasmid (STAT3MUT-luc) could be constructed. STAT3WT-luc or STAT3MUT-luc and miR-125a-5p mimic or negative control were transfected into HEK293 cells. After 48 h of transfection, the cells were collected. Dual-luciferase reporter gene system was adopted in this experiment. Renilla luciferase was used as the reference to detect Firefly luciferase activity. Finally, the ratio of the two was used as the relative expression value (Firefly/Renilla).
Western blot was used to determine the effect of miR-125a-5p on the protein expression of STAT3. After 48 h of A549 cell transfection with miR-125a-5p mimic or negative control, the cells of each group were collected. Radioimmunoprecipitation assay (RIPA) lysate (Beyotime Institute of Biotechnology, Shanghai, China) was added, and bicinchoninic acid (BCA) method was used to detect the supernatant protein concentration; 5% of stacking gel and 15% of separating gel were made, and 50 µg of total protein was added into each lane for electrophoresis, and then, they were transferred to polyvinylidene fluoride (PVDF) membrane (Bio-Rad, Richmond, CA, USA) and blocked with Tris-buffered saline with Tween 20 (TBST) solution with 5% skim milk (10 mM Tris-HCl, pH7.5 mM, 150 NaCl, 0.1% Tween-20) at room temperature for 1 h. And then, rabbit anti-STAT3 polyclonal antibodies (1:500 dilution; Abcam, Cambridge, MA, USA) and mouse anti-beta-actin monoclonal antibody (1:1000 dilution; Sigma-Aldrich) were added and incubated overnight at 4°C. The horseradish peroxidase (HRP)-labeled goat anti-rabbit IgG or HRP-labeled goat anti-mouse IgG secondary antibodies (Sigma-Aldrich) were added and incubated at 37°C for 1 h. Enhanced chemiluminescence (ECL) test was used to detect the image. The relative content of STAT3 was expressed by STAT3/beta-actin gray ratio, and its relative expression change was analyzed by PDQuest software (Bio-Rad).
Statistical analysis
SPSS17.0 statistical analysis software (International Business Machines, Armonk, NY, USA) was used for statistical treatment of the experimental data by t-test or one-way analysis of variance. The p < 0.05 showed that the difference was statistically significant.
Results
MiR-125a-5p displayed downregulation in lung cancer
The results from qRT-PCR showed that the expression of mir-125a-5p in lung cancer tissues was significantly lower than that in adjacent normal lung tissues (p < 0.01; Figure 1(a)). The expression of miR-125a-5p in lung cancer cell lines (95-D, A549, HCC827, and NCI-H1299) was significantly lower than that in HBE (p < 0.01; Figure 1(b)). These data showed that the downregulation of mir-125a-5p might play an important role in lung tumorigenesis.

Expression of miR-125a-5p in lung cancer tissues and lung cancer cell lines. (a) Relative expression of miR-125a-5p in lung cancer tissue. (b) Relative expression of miR-125a-5p in lung cancer cell lines (*p < 0.01 vs adjacent tissues or HBE).
MiR-125a-5p inhibited cell viability
First of all, miR-125a-5p mimic, mir-125a-5p inhibitor, and the respective negative control were transfected into A549 cells, and qRT-PCR results showed that the expression of miR-125a-5p in miR-125a-5p mimic transfection group was significantly higher than that in normal control group (p < 0.01) and negative control group (p < 0.01), while the expression of miR-125a-5p in miR-125a-5p inhibitor transfection group was significantly lower than that in normal control group (p < 0.01) and negative control group (p < 0.01; Figure 2(a)).
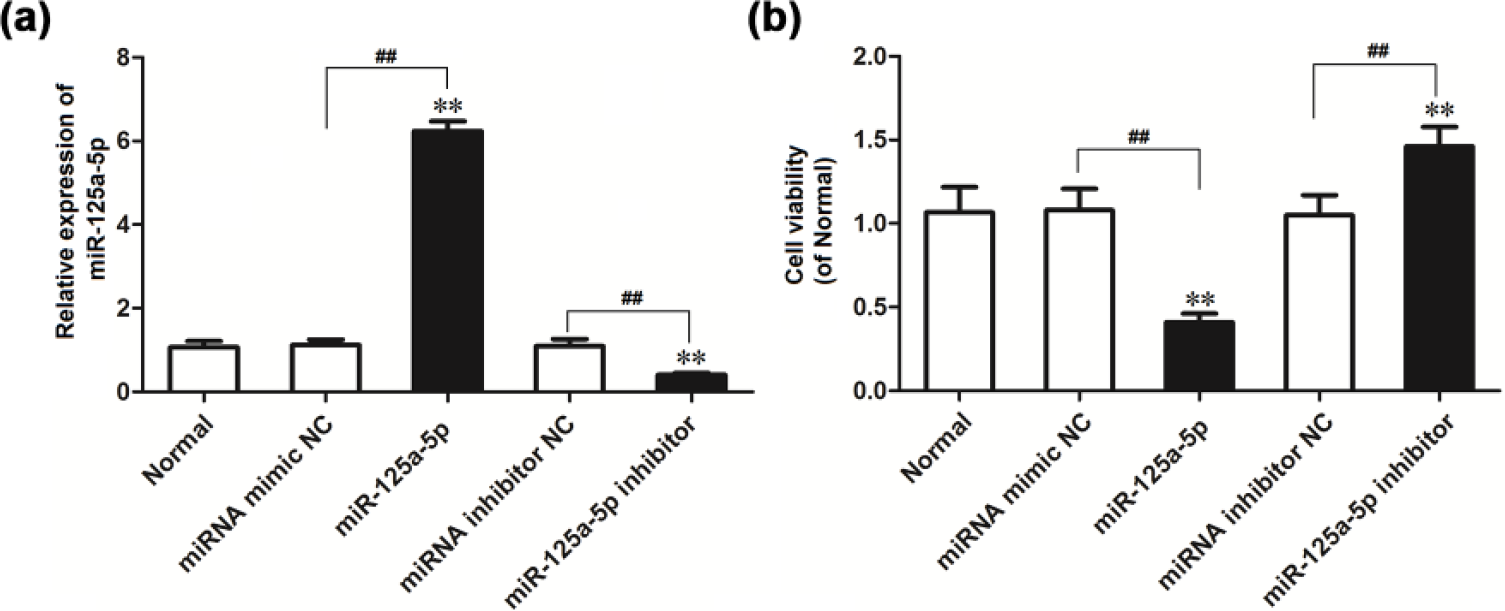
Effect of ectopic expression of miR-125a-5p on cell viability (**p < 0.01 vs normal bone tissue). (a) Relative expression of miR-125a-5p in A549 transfected with miR-125a-5p mimic or inhibitor. (b) The effect of ectopic expression of miR-125a-5p on the viability of A549 cells (**p < 0.01 vs normal control group; ##p < 0.01 vs negative control group).
MTT analysis results showed that cell viability in miR-125a-5p mimic transfection group was significantly lower than that in normal control group (p < 0.01) and negative control group (p < 0.01). But after transfection of miR-125a-5p inhibitor, the results were in contrast to the previous findings (Figure 2(b)). These results showed that miR-125a-5p overexpression inhibits the A549 cell viability; the inhibition of miR-125a-5p expression could promote A549 cell viability.
MiR-125a-5p inhibited cell proliferation
BrdU staining method was used to detect the effect of miR-125a-5p on A549 cell proliferation. BrdU staining results indicated that the number of BrdU positive cells in miR-125a-5p mimic transfection group was significantly lower than that in the normal control group (p < 0.05) and negative control group (p < 0.05). And after transfection of mir-125a-5p inhibitor, the results were in contrast to the previous findings (Figures 3 and 4). These results showed that miR-125a-5p overexpression can inhibit the proliferation of A549 cells, while inhibited expression of miR-125a-5p can promote the proliferation of A549 cells.

The effect of ectopic expression of miR-125a-5p on the proliferation of A549 cells was detected by BrdU staining method.

The effect of ectopic expression of miR-125a-5p on the proliferation of A549 cells (*p < 0.01 vs normal control group; #p < 0.01 vs negative control group).
MiR-125a-5p induced cell apoptosis
Flow cytometry analysis results demonstrated that the number of apoptotic cells in miR-125a-5p mimic transfection group was significantly higher than that in the normal control and negative control groups (p < 0.01). And the number of apoptotic cells in miR-125a-5p inhibitor transfection group was lower than that in the normal control and negative control groups (p < 0.01; Figure 5). These results showed that miR-125a-5p overexpression can promote the apoptosis of A549 cells, and miR-125a-5p downregulation can suppress the apoptosis of A549 cells.
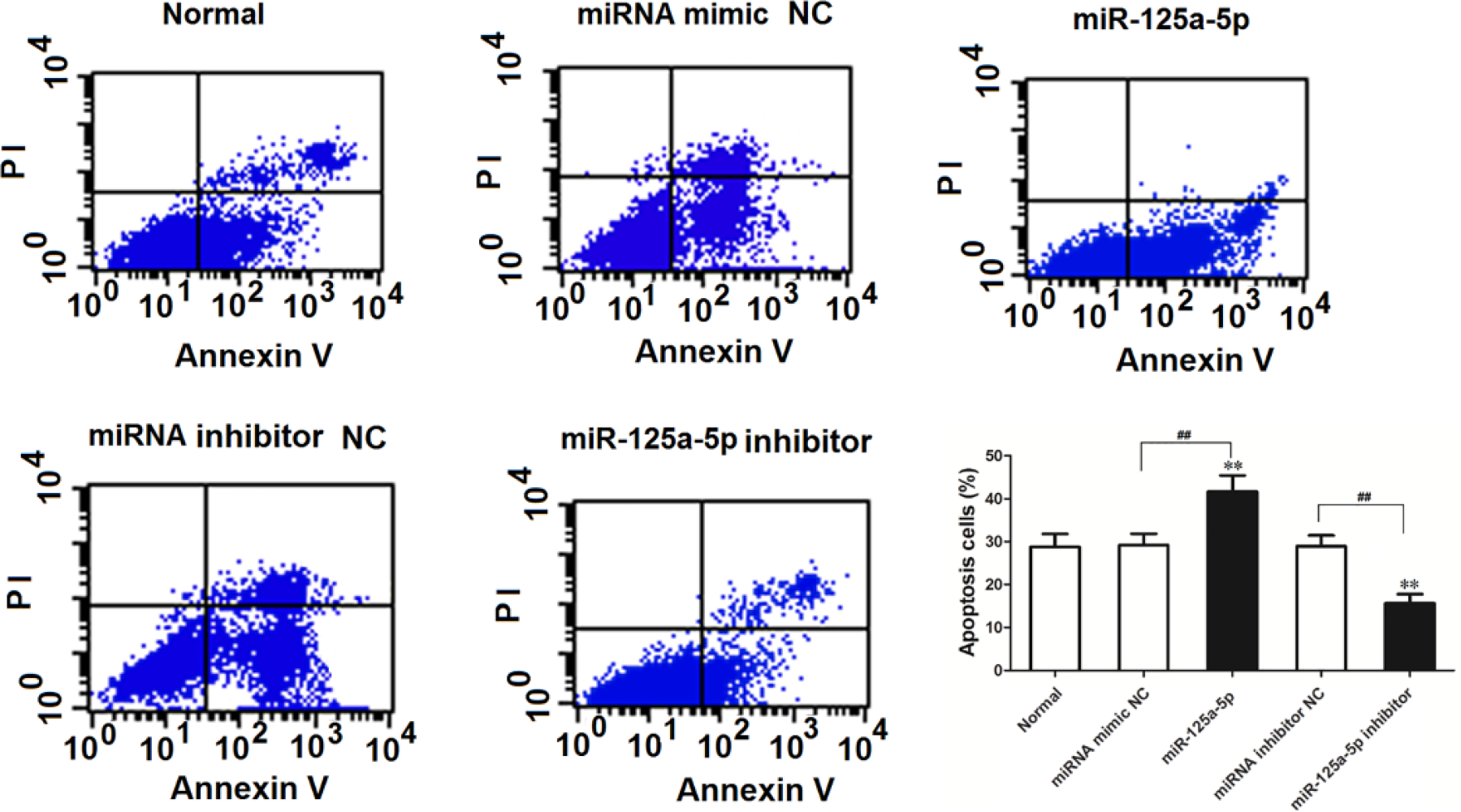
The effect of miR-125a-5p on cell apoptosis was analyzed by flow cytometry (**p < 0.01 vs normal control group, ##p < 0.01 vs negative control group).
MiR-125a-5p inhibited cell invasion
Transwell analysis results showed that the number of cells passing through the polycarbonate membrane in miR-125a-5p mimic transfection group was significantly lower than that in the normal control and negative control groups (p < 0.01). And the number of cells passing through the polycarbonate membrane in miR-125a-5p inhibitor transfection group was higher than that in normal control and negative control groups (p < 0.01; Figure 6). These data showed that miR-125a-5p overexpression inhibited the invasion of A549 cells.

Transwell analysis of the impact of miR-125a-5p on cell invasion (**p < 0.01 vs normal control group; ##p < 0.01 vs negative control group).
STAT3 was a target gene of miR-125a-5p
Luciferase reporter gene detection results suggested that the luciferase activity of miR-125a-5p transfected group was significantly lower than that of the negative control group (p < 0.01; Figure 7). Results demonstrated that mir-125a-5p might directly bind the target sites of STAT3 3′-UTR. Western blot analysis was used to detect the effects of mir-125a-5p on STAT3 expression. The results showed that the expression of STAT3 protein in miR-125a-5p transfection group was significantly lower than that in the normal control and negative control groups (p < 0.01). Result was shown in Figure 7. The result showed that mir-125a-5p overexpression inhibited the expression of STAT3 proteins.

STAT3 is the target gene of miR-125a-5p. (a) Schematic representation of STAT3 3′-UTR showing putative miR-125a-5p target site. (b) Statistical results of relative activity of luciferase in each group. (c) STAT3 expression was regulated by miR-125a-5p (**p < 0.01 vs normal control group or negative control group).
MiR-125a-5p played an important role in lung cancer through regulating STAT3
STAT3 recombinant expression vector pcDNA3.1-STAT3 was constructed to further verify whether mir-125a-5p played an important role in lung cancer through regulating STAT3. By the similar method, miR-125a-5p and recombinant expression vector pcDNA3.1-STAT3 were transfected into A549 cells. MTT, flow cytometry, and Transwell analysis were used to further verify whether mir-125a-5p can play an important role in lung cancer through regulating STAT3. First, the expression change of STAT3 in cell lines was detected by western blot. The result showed that transfection of pcDNA3.1-STAT3 into A549 cells significantly promoted the STAT3 expression (p < 0.01). STAT3 expression in miR-125a-5p + pcDNA3.1-STAT3 transfection group was significantly lower than that in pcDNA3.1-STAT3 transfection group (p < 0.01), while it had similar expression level with the normal control group and empty vector control group (pcDNA3.1; p > 0.05; Figure 8(a)).

Validation of miR-125a-5p playing a role in lung cancer through the regulation of STAT3. (a) The expression of STAT3 in the cells was detected by western blot. (b) The viability of A549 cells was detected by MTT method. (c) The apoptosis of A549 cells was detected by flow cytometry. (d) The invasion of MG-63 cells was detected by Transwell invasion chamber (**p < 0.01 vs normal group, pcDNA3.1 control group, and miR-125a-5p + pcDNA3.1-STAT3 group).
MTT and Transwell analysis methods showed that cell viability and the number of cells passing through the polycarbonate membrane in pcDNA3.1-STAT3 transfection group were significantly higher than that in the normal control and empty control vector groups (p < 0.01). The cell viability and the number of cells passing through the polycarbonate membrane in miR-125a-5p + pcDNA3.1-STAT3 transfection group were significantly lower than that in the pcDNA3.1-STAT3 transfection group (p < 0.01), but they had no differences compared with normal control group and empty vector control group (p > 0.05; Figure 8(b) and (d)). Flow cytometry analysis results showed that the number of apoptotic cells in pcDNA3.1-STAT3 transfection group was significantly lower than that in the normal control group and empty vector control group (p < 0.01). The number of apoptosis cells in miR-125a-5p + pcDNA3.1-STAT3 transfection group was significantly greater than that in pcDNA3.1-STAT3 transfection group (p < 0.01), but it had no differences compared with normal control group and empty vector control group (p > 0.05; Figure 8(c)). According to the above results and the rescuing experimental results, overexpression of STAT3 could reverse the biological characteristics of lung carcinoma cell induced by miR-125a-5p overexpression.
Discussion
Lung cancer is a disease seriously threatening human health. With the increase in incidence and mortality rates of lung cancer, it has attracted many researchers’ attention.28,29 Lung cancer of bronchial mucosa and alveolar is one of the most common malignant tumors. 30 Its morbidity and mortality topped all malignant tumors. 31 A large number of studies have proved that human miRNA genes frequently positioned in genomic regions were associated with cancer, and the disordered miRNA expression was found in many malignant tumors.32,33 Therefore, further elaborating the biological functions of miRNAs in lung cancer is particularly important.
One miRNA may have hundreds of targets, which allows these small RNA to significantly or slightly reduce a whole range of gene expression. This process depends on the feature of their 3′-UTR of target sites.34,35 The small RNA molecules are related to biological process of tumor. 36 The researchers found that many miRNAs are coded by cancer relevant gene region, which suggested that the expression of miRNAs may influence the occurrence of tumors.37,38 Reports confirmed that mir-125a was the potential inhibitor miRNA of many tumors.26,27,39 But the effects and mechanism of mir-125a-5p on the biological characteristics of lung cancer cells are unclear. In this study, we first used qRT-PCR to detect the expression of mir-125a-5p in lung cancer tissues and lung cancer cell lines. The results showed that mir-125a-5p in lung cancer tissues and cell lines was significantly reduced, which suggested that miR-125a-5p may also inhibit cancer gene in lung cancer. In order to explore the role of miR-125a-5p in lung cancer, A549 cells were chosen and transfected with miR-125a-5p mimic or mir-125a-5p inhibitor. A series of methods were used to detect the effect of mir-125a-5p on A549 cell proliferation, apoptosis, and invasion. The results suggested that mir-125a-5p overexpression reduced cell viability and inhibited cell proliferation and invasion, but promoted cell apoptosis. Results reproved that mir-125a-5p played a role as the tumor suppressor gene in lung cancer cells.
The signal transducer and activator of transcription (STAT) is a latent transcription factor activated by polypeptide ligands, such as a variety of cytokines and growth factor.40,41 STAT is a unique protein family combined with DNA, which is composed of 750–900 amino acids. At present, a number of members encoded by different genes have been found in human bodies. They have similar amino acid sequences and play an independent regulation of cell signal transduction in human body.42,43 Among them, STAT3 is an important signaling cascade component in STAT family and is widely involved in the response of a variety of cytokines.44,45 Activation of STAT3 signal transduction pathway is closely related to cell proliferation, differentiation, apoptosis, angiogenesis, invasion, metastasis, and immune escape. 46 The continuous activation of STAT3 signal transduction pathway can lead to abnormal cell proliferation and malignant transformation. Therefore, STAT3 has been defined as a cancer gene. 47 Under normal physiological condition, activation of STAT3 lasted from only a few minutes to several hours. But STAT3 expression or activity was enhanced in a large number of human tumor tissues and cell lines; for example, STAT3 abnormal expressions existed in human leukemia, multiple myeloma, head and neck squamous cell carcinoma, breast cancer, prostate cancer and lung cancer and its activity was enhanced.48,49 Researchers found that the positive rate of STAT3 protein in lung cancer was significantly increased, and STAT3 was involved in the proliferation, differentiation, and anti-apoptosis of lung adenocarcinoma, which suggested that abnormal activation of STAT3 signaling pathway can promote the lung tumorigenesis.50,51
The online software TargetScan (http://www.targetscan.org) was chosen to analyze and predict the target genes of mir-125a-5p. The results showed that STAT3 might be the target gene of mir-125a-5p. So, luciferase reporter gene system was chosen to verify whether STAT3 was the target of mir-125a-5p. The results showed that mir-125a-5p could interact with 3′-UTR of STAT3. Again western blot was used to verify the effects of mir-125a-5p on the expression of STAT3. The results showed that overexpression of mir-125a-5p inhibited the expression of STAT3. In order to further verify whether miR-125a-5p can play a role in cell biology through regulating STAT3, we constructed STAT3 recombinant expression vector pcDNA3.1-STAT3. MTT, flow cytometry, and Transwell analysis methods were used to further verify whether mir-125a-5p played a biological role through regulating STAT3. The results suggested that overexpression of STAT3 could reverse the lung cancer cell proliferation, invasion, and apoptosis induced by overexpression of miR-125a-5p. These data indicated that mir-125a-5p could regulate the expression of STAT3 in lung cells.
In summary, mir-125a-5p displayed a downregulation in lung cancer tissue and cell lines. Mir-125a-5p can inhibit the proliferation and invasion of lung cancer cells and promote the cell apoptosis through negatively regulating STAT3 expression. Enhancing the expression of mir-125a-5p is expected to benefit treatment strategy for the patients with lung cancer.
Footnotes
Acknowledgements
L.Z. and S.S. have contributed equally to this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (grant no. 81501967).
