Abstract
The aim of the study was to evaluate the effectiveness of placental-specific markers, extracellular fetal DNA (sex-determining region Y and hypermethylated
Keywords
Introduction
Since the placenta is being continuously remodeled during normal placental development, extracellular nucleic acids of both fetal and placental origins, packed into either trophoblast-derived apoptotic bodies or shedding syncytiotrophoblast microparticles, may be detected in maternal circulation during the course of normal gestation.1–5
Initially, researchers focused on the detection of male fetal-derived DNA in maternal circulation; most often, this was done using the single-copy sex-determining region Y (
Subsequently, Chan et al.
9
introduced the
Latest findings revealed that microRNAs are also packed within exosomal nanoparticles released into the blood and extracellular compartment mediating the communication between diverse types of neighboring or distant cells.10–13
MicroRNAs belong to a family of small noncoding RNAs that regulate gene expression at the posttranscriptional level by degrading or blocking translation of messenger RNA (mRNA) targets.14,15
There has been a trend over the last 10 years to develop non-invasive methods utilizing quantification of cell-free nucleic acids in maternal circulation.5,16–33 The diagnostic potential of particular molecular biomarkers and their implementation in the current predictive and diagnostic algorithms for pregnancy-related complications are subjects of interest. 5
Gestational trophoblastic disease (GTD) encompasses a spectrum of pregnancy-related disorders. This heterogeneous group can be divided into (1) pre-malignant forms: partial hydatidiform mole (PHM) and complete hydatidiform mole (CHM) and (2) malignant forms: represented by invasive mole (IM), choriocarcinoma (CH), and very rare trophoblastic tumors such as epithelioid trophoblastic tumor (ETT) and placental site trophoblastic tumor (PSTT). Malignant forms of disease are collectively named gestational trophoblastic neoplasia (GTN).34,35
A progression to GTN, which develops in approximately 15%–20% with complete mole and 1%–5% with partial mole, is signified by plateau or rising human chorionic gonadotropin (hCG) concentrations. 36
Recent study of Hasegawa et al. 37 has shown that the plasma concentrations of selected C19MC microRNAs (miR-520b, miR-520f, and miR-520c-3p) were significantly higher in patients with CHM than in the control group consisting of gestational age–matched uncomplicated pregnancies. Furthermore, they demonstrated significant decrease in circulating microRNAs after the evacuation of molar pregnancy. Additional study of the same group demonstrated a decreasing tendency of the plasma microRNA concentrations similar to the pattern for serum hCG concentration in two cases of CHM resulting in later development of GTN, who underwent the chemotherapy. 38
We have previously identified C19MC microRNAs (miR-516-5p, miR-517-5p, miR-518b, miR-520a-5p, miR-520h, miR-525, and miR-526a) present in maternal plasma differentiating between normal pregnancies and non-pregnant individuals. 39 Significant increases in extracellular C19MC microRNAs levels (miR-516-5p, miR-517-5p, miR-518b, miR-520a-5p, miR-520h, miR-525, and miR-526a) over time in normally progressing pregnancies were observed.39,40
Initially, we tested in maternal plasma 16 C19MC microRNAs, which according to the miRNAmap database 41 have been reported to be placenta specific. Those C19MC microRNAs, which were clearly positive in placenta and maternal plasma samples throughout gestation (from the first trimester till term pregnancy) and concurrently negative in whole blood and plasma samples of non-pregnant controls, were selected for further research. Finally, we identified seven C19MC microRNAs (miR-516-5p, miR-517-5p, miR-518b, miR-520a-5p, miR-520h, miR-525, and miR-526a) as pregnancy-associated microRNAs with diagnostic potential. 39 They fulfilled our established criteria for the selection of an optimal microRNA marker for potential diagnostic and/or prognostic purposes (100% specificity: three negative wells out of three tested wells in non-pregnant controls, high detection rate in plasma samples of pregnant women: at least four positive wells out of six tested wells during the first trimester of gestation, low intra-assay variability: below 2%, and low inter-assay variability: below 10%) and were used in all our consecutive studies. Low abundant microRNAs with higher variation between intra-assay and inter-assay replicates were excluded from further research at the beginning of the study.
To our knowledge, no prospective study to describe the profile of extracellular fetal DNA (
Extracellular fetal DNA and appropriate circulating C19MC microRNAs were monitored in patients’ plasma samples weekly for 6 weeks post-chemotherapy together with serum hCG levels, and then biweekly until 6 months, continuing initially with monthly follow-up and decreasing gradually to just six times monthly monitoring over a long period. 42 An hCG level of less than 5 IU/L was considered negative for pregnancy (American Pregnancy Association recommendation; available at http://americanpregnancy.org). Levels of extracellular fetal DNA and C19MC microRNAs in plasma samples were compared between groups (serum hCG-positive cases, women with GTD/GTN with positive hCG levels during diagnosis, treatment or follow-up versus serum hCG-negative cases, women already known to have GTD/GTN, whose hCG levels dropped to normal during the treatment). In total, 38 hCG-positive samples and 60 hCG-negative samples derived from 16 patients with GTD/GTN were examined.
Materials and methods
Patients’ characteristics
The study was designed in a prospective manner. The study cohort consisted of 16 Caucasian women (mean age: 34.0 years, range: 17–49 years) with diagnosis of CHM (n = 10), PHM (n = 4), invasive/proliferative hydatidiform mole (n = 1), and CH (n = 1) dispensarized in the Trophoblastic Disease Center at the Institute for the Care of the Mother and Child in Prague. GTN subsequently developed in 10 patients with complete mole and three patients with partial mole. In one patient with PHM, metastatic GTN was diagnosed. All patients provided written informed consent. The study was approved by the Ethics Committees of the Third Faculty of Medicine, Charles University in Prague and the Institute for the Care of the Mother and Child in Prague.
The pathologic pattern of CHMs is characterized by presence of enlarged, edematous villi (usually generalized hydrops and cistern formation), surrounded by diffuse abnormal trophoblast hyperplasia and presence of nuclear debris in villous stroma. Many of these characteristics may not be fully developed in early moles. Also, absence of embryo is typical. The diagnosis was established by pathologic examination (performed by expert pathologist), immunostaining with absence of p57 positivity and genetical analysis—karyotyping; all cases of complete moles were diploid (46,XX or 46,XY).
The same diagnostic criteria were applied for cases of PHMs. All the pathological features are less distinctive in comparison with complete moles. Trophoblast proliferation is less marked, usually only focal. Also, hydropic changes and cistern formation are only focal involving only some of the villi. Stromal blood vessels are common finding and the villi have a specific scalloped shape. Embryo is usually present, but its absence does not exclude the diagnosis. The main aim of the diagnostic algorithm is to distinguish partial moles from hydropic abortions. Immunostaining for p57 is not effective (positive in both cases) and genetical analysis is the most helpful method (triploid character—69, XXY; 69, XXX; 69, XYY).
An invasive/proliferative mole is an aggressive form of complete mole (rarely of partial mole) with tendency to invade the myometrium. Histologically, the appearance of chorionic villi is similar to that of complete moles. The diagnosis is defined by excessive trophoblastic proliferation and irregular-shaped villi.
The diagnostics criteria of GTN include the following: plateau or rising levels of hCG after evacuation, massive bleeding, or histological diagnosis of CH with or without metastatic spread to other organs.
Patients who developed GTN were divided in two groups according to the risk score (FIGO 2000 scoring system). 43 The prognostic score predicts the potential risk for developing resistance to single-drug chemotherapy (methotrexate or actinomycin D). A score of 0–6 indicates a low risk, and a score ≥7 indicates high risk of developing resistance.
Low-risk patients were treated by methotrexate (n = 1) or act D (n = 10), both applied intravenously. High-risk patients were treated by multi-agent therapy including etoposide, methotrexate, and dactinomycin alternating with cyclophosphamide and vincristine (EMA/CO regimen, n = 1). Remission was considered to be achieved when the hCG level was undetectable for 3 consecutive weeks.
hCG (intact hCG + free hCG β subunit) measurement
Intact hCG + β subunit level measurement was routinely performed in serum samples using hCG + beta kit (Roche Diagnostics, Basel, Switzerland) on COBAS 6000 equipment.
Processing of samples
A total volume of 9 mL of anticoagulated peripheral blood were collected by standard antecubital vein phlebotomy into ethylenediaminetetraacetic acid (EDTA)–containing tubes. All specimens were processed on the same day of blood draw, almost immediately after collection. Blood samples were aliquoted into 2.0 mL microcentrifuge tubes and centrifuged for 10 min at 1200
RNA extraction from plasma samples
Total RNA was extracted from 1 mL of plasma and 25 mg of normal placental tissue preserved in RNAlater (Ambion, Austin, TX, USA) followed by an enrichment procedure for small RNAs using a mirVana microRNA Isolation Kit (Ambion). TRIzol LS Reagent was used in plasma samples for total RNA extraction from biological fluids (Invitrogen, Carlsbad, CA, USA) and preceded the small RNAs’ enrichment procedure. To minimize DNA contamination, we treated the eluted RNA with 5 µL of DNase I (Fermentas International, Burlington, ON, Canada) for 30 min at 37°C.
Reverse transcriptase reaction
Each microRNA was reverse transcribed into complementary DNA (cDNA) using TaqMan MicroRNA Assay, containing microRNA-specific stem-loop reverse transcription (RT) primers, and TaqMan MicroRNA Reverse Transcription Kit (Applied Biosystems, Branchburg, NJ, USA) in a total reaction volume of 32 µL on a 7500 Real-Time PCR system (Applied Biosystems) with the following thermal cycling parameters: 30 min at 16°C, 30 min at 42°C, 5 min at 85°C, and then held at 4°C.
Quantification of microRNAs
The characteristics of studied C19MC microRNAs are outlined in Table 1.
Characteristics of PCR assays.

PCR: polymerase chain reaction;
A total volume of 15 µL of cDNA corresponding to each microRNAs was mixed with components of TaqMan MicroRNA Assay and the ingredients of the TaqMan Universal PCR Master Mix (Applied Biosystems) in a total reaction volume of 35 µL. TaqMan PCR conditions were set as described in the TaqMan guidelines with the exception of an increase to 50 cycles. The analysis was performed using a 7500 Real-Time PCR System. All PCRs were performed in duplicates. A sample was considered positive if the amplification signal occurred before the 40th threshold cycle.
Fourfold diluted RNA derived from the fetal part of the placenta (the part of the placenta derived from the chorionic sac that encloses the embryo, consisting of the chorionic plate and villi) was used to create the standard curves. The two-step real-time polymerase chain reaction (RT-PCR) with TaqMan MicroRNA assays showed reliable and reproducible results (slope and R2 coefficient of determination) for all tested microRNAs in placental tissues and were also applied to plasma samples. Concentrations of individual microRNAs were expressed as pg of total RNA enriched for small RNAs per milliliter of plasma.
The expression of particular microRNA in maternal plasma was determined using the comparative Ct method 46 relative to the expression of the same microRNA in the reference sample. RNA fraction highly enriched for small RNA isolated from the fetal part of one randomly selected placenta derived from gestation with normal course (the part of the placenta derived from the chorionic sac that encloses the embryo, consisting of the chorionic plate and villi) was used as a reference sample for relative quantification throughout the study.
In addition, each RNA sample was assayed for spike-in

In future experiments, other spike-in oligonucleotides may be added to each reaction. The expression of particular C19MC microRNA present in plasma samples may be determined using the comparative Ct method relative to normalization factor (geometric mean of all spike-in oligonucleotides) to enhance minimization for variations during the preparation of RNA, cDNA synthesis, and RT-PCR.
DNA extraction from plasma samples, digestion by methylation-sensitive restriction enzyme
DNA was extracted from plasma samples (1 mL) using a QIAamp DSP Virus Kit (Qiagen) according to a modified version of the manufacturer’s instructions for the vacuum protocol. DNA was eluted in buffer AVE (60 µL). 47
Plasma DNA (44 µL) was digested with 12.8 U of
Quantification of fetal and total extracellular DNA in maternal plasma samples using SRY , RASSF1 , and β-actin as markers
Enzyme-digested plasma DNA (15 µL) (hypermethylated
A calibration curve (logarithmic scale), plotting the threshold cycle (Ct) against known concentrations of a serially diluted DNA reference sample was run in parallel with each analysis. Enzyme-untreated DNA extracted from the fetal part of the male placenta was used as a reference sample for quantification of
Statistical analysis
Levels of extracellular fetal DNA, extracellular total DNA (total
Receiver operating characteristic (ROC) curves were constructed to calculate the area under the curve (AUC), and the best cut-off point for particular extracellular nucleic acid marker was used in order to calculate the respective sensitivity, specificity, predictive values, and likelihood ratios for GTD/GTN.
Correlations between variables including serum hCG levels (IU/L) and circulating fetal DNA levels (copies/mL) or circulating C19MC microRNA levels (pg/mL) were calculated using Spearman’s rank correlation coefficient (ρ), a non-parametric measure of statistical dependence between two variables.
If the correlation coefficient value is −1 or 1, there is a perfect negative or positive correlation. If it ranges within <−1; 0.5> or <0.5; 1>, there is a strong negative or positive correlation. If it varies from −0.5 to 0 and from 0 to 0.5, there is a weak negative or positive correlation. If the R value reaches 0, there is no correlation. Significant levels of correlation were set at a p value of <0.05.
Results
Quantification of total extracellular DNA in plasma samples using total RASSF1A and GLO sequences as markers
The levels of extracellular total DNA determined using
Detection of fetal extracellular DNA in plasma samples of patients with GTD/GTN using SRY and hypermethylated RASSF1A sequences
Fetal extracellular DNA (
Hypermethylated
The concentration of extracellular fetal DNA determined using hypermethylated

Extracellular fetal DNA in patients with gestational trophoblastic disease (GTD)/neoplasia (GTN). (a) The levels of extracellular fetal DNA determined using hypermethylated
Nevertheless, in the same cohort of patients, quantities of male fetal extracellular DNA, determined using the
Increased levels of circulating C19MC microRNAs in hCG-positive patients with active GTD/GTN
The comparative analysis within the same group of patients with GTD/GTN revealed significantly increased plasma levels of

Increased levels of circulating C19MC microRNAs in hCG-positive patients with active GTD/GTN. Comparison between patients with GTD/GTN revealed significantly increased plasma levels of (a) miR-516-5p, (b) miR-517-5p, (c) miR-518b, (d) miR-520a-5p, (e) miR-520h, (f) miR-525, and (g) miR-526a in hCG-positive cases (serum hCG levels above 5 IU/L) compared to hCG-negative cases (serum hCG levels below 5 IU/L).

Increased gene expression of circulating C19MC microRNAs in hCG-positive patients with active GTD/GTN. The difference in gene expression of circulating (a) miR-516-5p, (b) miR-517-5p, (c) miR-518b, (d) miR-520a-5p, (e) miR-520h, (f) miR-525, and (g) miR-526a between the hCG-positive group with active form of the disease (serum hCG levels above 5 IU/L) and the hCG-negative group of patients in remission (serum hCG levels below 5 IU/L) was identified.
Association between plasma C19MC microRNA and serum hCG levels in patients with GTD/GTN
Additionally, the association between plasma levels or gene expression of particular C19MC microRNAs and serum hCG levels was analyzed in the cohort of patients complicated with GTD/GTN.
The serum hCG levels showed a positive correlation or even a strong positive correlation with C19MC microRNA plasma levels in women with GTD/GTN (

Association between plasma C19MC microRNA and serum hCG levels in patients with GTD/GTN. The serum hCG levels in women with GTD/GTN showed a positive correlation or even a strong positive correlation with plasma levels of (a) miR-516-5p, (b) miR-517-5p, (c) miR-518b, (d) miR-520a-5p, (e) miR-520h, (f) miR-525, and (g) miR-526a.
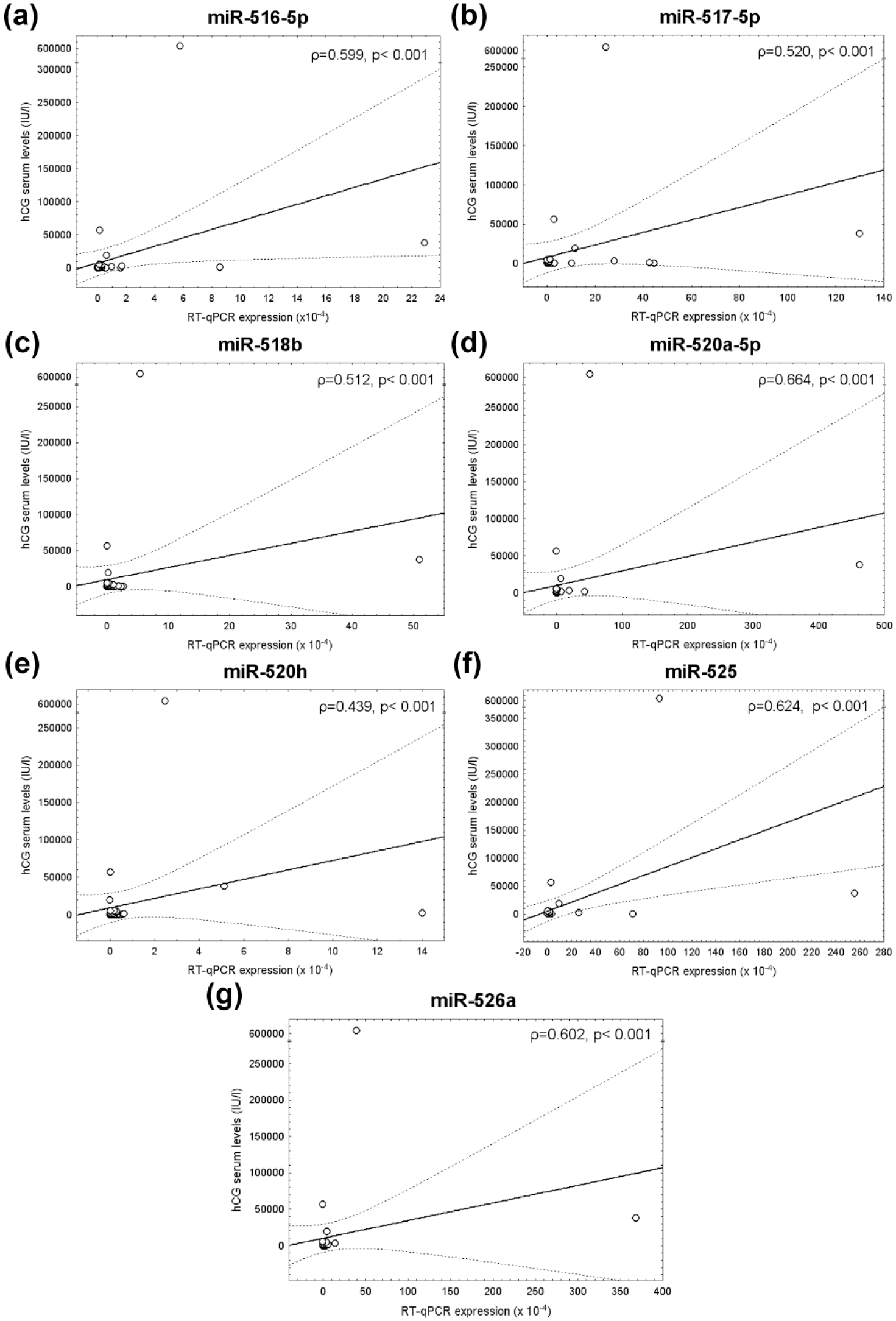
Correlation between plasma C19MC microRNA gene expression and serum hCG levels in patients with GTD/GTN. Positive correlation or a strong positive correlation between serum hCG levels and plasma placental-specific C19MC microRNA gene expression in patients with GTD/GTN was detected ((a) miR-516-5p, (b) miR-517-5p, (c) miR-518b, (d) miR-520a-5p, (e) miR-520h, (f) miR-525, and (g) miR-526a).
Circulating C19MC microRNAs are able to distinguish between patients with active and non-active GTD/GTN
Table 2 displays the diagnostic accuracy of plasma concentrations of placental-specific microRNAs in the identification of patients with active and non-active GTD/GTN (serum hCG levels above or below 5 IU/L) using cut-offs derived from the ROC curves. The experimental data were compared between the groups of identical patients (the group of patients with active disease before suction evacuation of molar pregnancy, the hysterectomy, and/or initiation of chemotherapy with serum hCG levels above 5 IU/L vs the group of patients after suction evacuation of molar pregnancy, the hysterectomy, and/or termination of chemotherapy with serum hCG levels below 5 IU/L).
Diagnostic accuracy of plasma concentrations of placental-specific microRNAs in patients with gestational trophoblastic disease or persisting trophoblastic disease.

PPV: positive predictive value; NPV: negative predictive value; PLR: positive likelihood ratio; NLR: negative likelihood ratio; AQ: absolute quantification; RQ: relative quantification; miRNA: microRNA; CI: confidence interval; AUC: area under the curve; ROC: receiver operating characteristic.
The predictive accuracy of single plasma microRNA markers was assessed. The largest AUC was observed for miR-520a-5p (1.0), miR-516-5p (0.989), and miR-525 (0.989). Using miR-517-5p, miR-526a, miR-518b, and miR-520h, prediction rules for the presence or the absence of GTD/GTN had a bit lower AUC 0.913/0.895/0.871/0.860, respectively (Figure 6, Table 2).

Circulating C19MC microRNAs in the identification of patients with active gestational trophoblastic disease/neoplasia (GTD/GTN). The predictive accuracy of single plasma microRNA marker was assessed. The largest area under the curve was observed for (a) miR-520a-5p, (b) miR-516-5p, and (c) miR-525. Using (d) miR-517-5p, (e) miR-526a, (f) miR-518b, and (g) miR-520h prediction rules for the occurrence of GTD/GTN had a bit lower area under the curve 0.913/0.895/0.871/0.860, respectively.
The best positive predictive value (PPV) (100%) and negative predictive value (NPV) (100%) were observed for miR-520a-5p. MiR-516-5p predicted the occurrence of GTD/GTN with a sensitivity of 100.0%, a specificity of 96.15%, a PPV of 96.29%, and a NPV of 100.0%. MiR-525, which had the same AUC as miR-516-5p, finally showed a sensitivity of 100.0%, a specificity of 92.31%, a PPV of 92.86%, and a NPV of 100.0%. Although miR-517-5p had significantly higher AUC than miR-526a, finally, miR-526a showed better PPV (100%) and NPV (84.62%) than miR-517-5p.
Overall, the likelihood ratios for a positive test for these three best placental-specific microRNA markers were large, ranging between 13.00 and 25.97. Similar results were achieved in case of relative quantification of C19MC microRNA (Figure 7, Table 2).
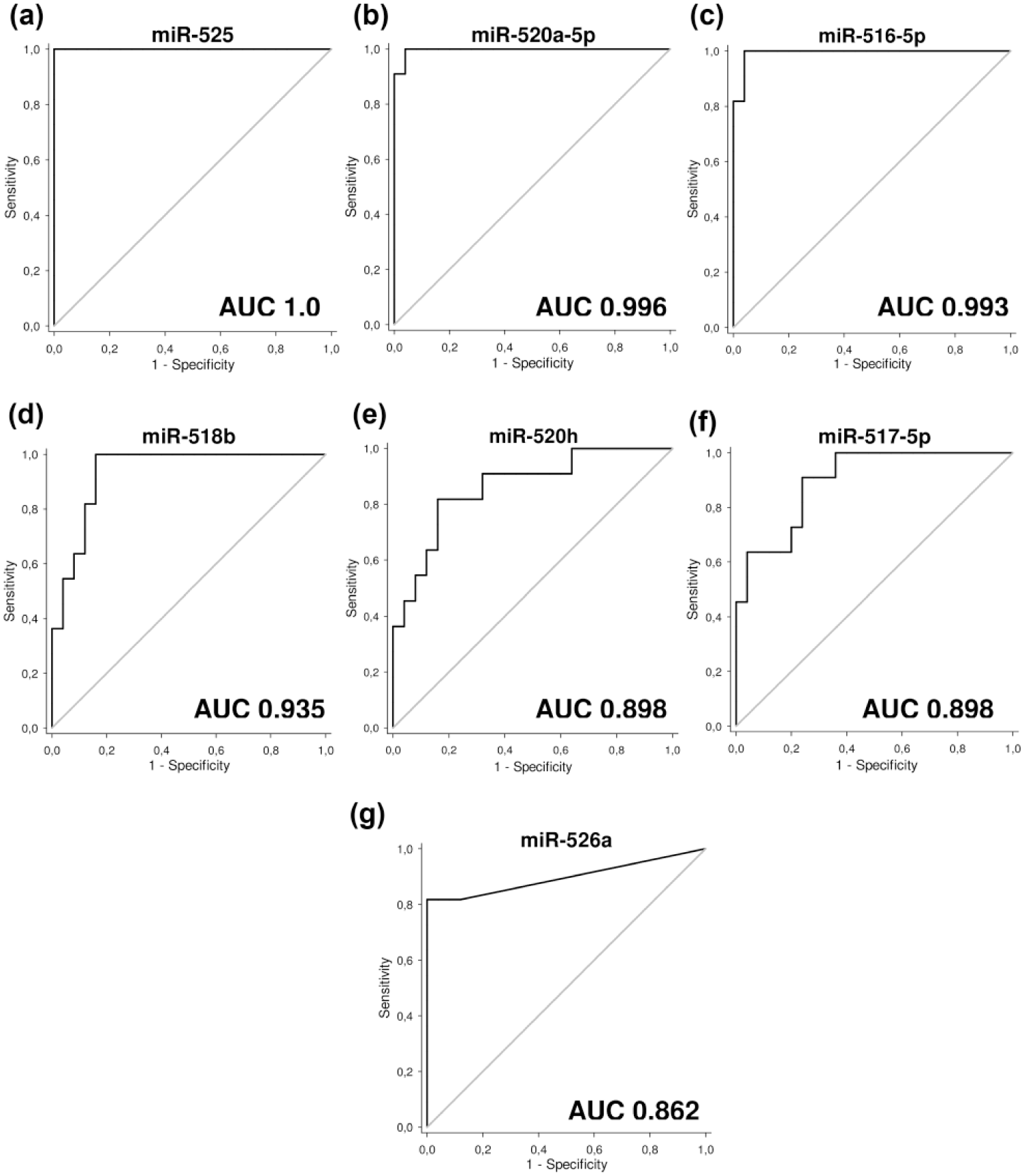
Receiver operating curve (ROC) analysis of circulating C19MC microRNA gene expression—differentiation between patients with active GTD/GTN and patients in remission of the disease. ROC curves for the gene expression of (a) miR-525, (b) miR-520a-5p, (c) miR-516-5p, (d) miR-518b, (e) miR-526a, (f) miR-517-5p, and (g) miR-520h are demonstrated. Similarly, as in absolute quantification, relative quantification showed the largest area under the curve for (a) miR-525, (b) miR-520a-5p, and (c) miR-516-5p.
However, while raised plasma levels of miR-517-5p, miR-526a, miR-518b, and miR-520h have been observed in patients with GTD/GTN, the overall predictive capacity for GTD/GTN was a little bit lower (miR-517-5p: PPV 85.53%, NPV 90.3%; miR-526a: PPV 100.0%, NPV 84.62%; miR-518b: PPV 72.22%, NPV 100.0%; and miR-520h: PPV 77.15%, NPV 88.94%) (Table 2).
Circulating fetal DNA (hypermethylated RASSF1A sequence) and C19MC microRNAs are able to distinguish between patients with regard to the severity of GTD/GTN
We compared the levels of extracellular fetal DNA and placental-specific C19MC microRNAs between the groups of patients with regard to the severity of the disease. Since the number of patients in particular groups is low, statistical analysis was not performed. Preliminary data indicated that the highest levels of extracellular fetal DNA (hypermethylated
In non-metastatic GTN which subsequently developed from complete mole (n = 10) or partial mole (n = 3) where serum hCG levels ranged from 617 to 4618 IU/L (median: 901 IU/L) before beginning of chemotherapy, the levels of extracellular fetal DNA and circulating C19MC microRNAs were much lower than in patients with metastatic GTN (CH) before start of chemotherapy (hypermethylated
A case report of a patient with GTN developed after PHM
The clinical characteristics and experimental data of a 20-year-old patient with partial molar pregnancy who developed GTN are presented. The patient achieved complete remission after the application of five cycles of methotrexate chemotherapy (each cycle lasted 5 days). The continuous intense decline of hCG, all examined circulating placental-specific C19MC microRNAs and circulating fetal DNA (hypermethylated

Case report of a patient with gestational trophoblastic neoplasia developed after partial hydatidiform mole. A 20-year-old Caucasian primigravida was hospitalized for bleeding in seventh week of pregnancy. Evacuation of the uterine cavity was performed while diagnosis of missed abortion was established. The material was sent for histological examination which raised suspicion of molar pregnancy. After dilatation and curettage, hCG was monitored weekly. The level dropped from more than 200,000–40,000 IU/L when re-elevation occurred. At this time, the patient was sent to the Trophoblastic Disease Center. Ultrasound examination of the uterus revealed a hypervasculated mass in the anterior uterine wall 54 × 61 × 31 mm in diameter. The uterine cavity was without presence of residual tissue. Also, no evidence of metastatic disease in other organs was present. The second histological examination by expert pathologist confirmed the suspicion of molar pregnancy. The FIGO score of the patient was 4 (largest tumor mass more than 5 cm in diameter and hCG levels between 104 and 105 IU/L). The patient was treated with monotherapy (low-risk disease) methotrexate 0.4 mg/kg i.v. in 5-day regiment every 2 weeks. Continuous decline of hCG (a) circulating placental-specific C19MC microRNAs, (b-h) and extracellular fetal DNA, (i, j) during and after methotrexate therapy of the patient was observed. After application of five cycles of therapy, hCG reached undetectable levels. The treatment continued for two additional courses. The patient is now 10 months after the last chemotherapy with no evidence of disease and negative hCG.
A case report of a patient with GTN developed after CHM
The clinical characteristics and experimental data of a 35-year-old patient with complete molar pregnancy who developed GTN are presented. The patient achieved complete remission after the application of five cycles of actinomycin D chemotherapy. The patient showed negative amplification of

Case report of a patient with gestational trophoblastic neoplasia developed after complete hydatidiform mole. A 35-year-old Caucasian primigravida was sent to the Trophoblastic Disease Center after instrumental evacuation of molar pregnancy in local hospital. Trophoblastic hyperplasia, cellular atypia, and diffuse swelling of chorionic villi were revealed by histological examination with conclusion of complete molar pregnancy. After dilatation and curretage the hCG level was monitored every week. Initial decline of hCG level was followed by progressive re-elevation. Second dilatation and curretage was performed, but histological examination did not reveal any pathology and the progressive trend of hCG level continued. Patient was sent to the Trophoblastic Disease Center. Here, the second histological examination was performed and the diagnosis of complete molar pregnancy was confirmed. Staging was completed with no evidence of metastatic disease. hCG reached nearly 4600 IU/L. The diagnosis of gestational trophoblastic neoplasia was confirmed, and the treatment with actinomycin D 0.5 mg/kg intravenously in 5-day regimen every 2 weeks was started. The patient’s FIGO score was 1 (hCG level 103 and 104 IU/L). Continuous decline of hCG with advancing cycles of chemotherapy was observed (a) Although significant rise in circulating placental-specific C19MC microRNAs 1 week after the end of the first cycle of chemotherapy was detected, it was followed by rapid decline of C19MC microRNA plasma levels, (b-h) Extracellular fetal DNA was detected in very low levels, and (i) before the start and during chemotherapy. The treatment response was very good, and after five cycles of chemotherapy (actinomycin D), hCG levels reached negative values. Two additional courses of actinomycin D were applied as a consolidation therapy to minimize the risk of disease relapse. Now the patient is 12 months after the termination of the treatment with no evidence of disease. hCG level is negative for all the time.
A case report of a patient with metastatic GTN developed after PHM
The clinical characteristics and experimental data of the 38-year-old patient with partial molar pregnancy who developed metastatic GTN are presented. The patient received nine cycles of EMA/CO chemotherapy. The patient showed negative amplification of

Case report of a patient with metastatic gestational trophoblastic neoplasia developed after partial hydatidiform mole. A 38-year-old Caucasian secundipara after two previous missed abortions in early pregnancy and cesarean section in 2012 was sent to the Trophoblastic Disease Center because of rising hCG levels after two previous evacuations of uterine cavity. First dilatation and curettage for suspicion on molar pregnancy was performed at the University Hospital. Histological examination confirmed partial mole. After 1 month, the second curettage and hysteroscopic resection of residual tissue in the uterine cavity were done at the same hospital. The hCG level before the second curettage was more than 16,000 IU/L. The rising trend of hCG level continued after the second evacuation to 53,300 IU/L. The pelvic ultrasound revealed hypervascular tumor in the uterine fundus with maximal diameter of 42 mm. Chest X-ray and ultrasound of liver and kidney were done with positive finding of multiple metastatic lesions in both lungs (seven lesions). The largest metastasis was 10 mm large. Because of the positive finding, PET-CT was performed (positron emission tomography combined with whole body computer scanning). Up to 20 focuses with elevated FDG (fluoro-deoxy-glucose) consumption were found in both lungs. The FIGO score was 7 and the patient was included in high risk disease group (two points for hCG level higher than 104 IU/L, one point for the largest tumor diameter exceeding 3 cm, and three points for number of metastases exceeding eight in total), and chemotherapy in regimen EMA-CO (etoposide, methotrexate, and actinomycin D alternating with cyclophosphamide and vincristine weekly) was started immediately. Patient suffered from selective IgG deficiency with continuous substitution with human gammaglobulins for last 3 years. This event was a treatment-complicating factor. Because of severe change in blood pattern during the chemotherapy, patient was repetitively administered CSF (colony stimulating factor, Neulasta). After application of seven cycles of chemotherapy in EMA-CO regimen, patient reached the levels of hCG 2.9 IU/L. After that a significant leukopenia with febrile neutropenia led to the interruption of treatment. After 2 weeks, after normalization, additional two courses of chemotherapy were administered as a consolidation therapy to minimize the risk of disease relapse. In the last cycle of chemotherapy, we omitted the dose of etoposide on the second day of the regimen to minimize the side effects. The chemotherapy was finished after the ninth cycle. Significant rise in circulating C19MC microRNAs was observed 1 week after the end of the first cycle of chemotherapy (b-h). Thereafter, decline of hCG (a) circulating C19MC microRNAs, (b-h) and extracellular fetal DNA, and (i) continued. Now, several months after termination of the treatment, the patient is regularly monitored and the hCG levels are negative.
Discussion
The results of our pilot study strongly supported the need for a more detailed exploration of extracellular fetal DNA and placental-specific C19MC microRNAs in circulation with the view toward their recognition as potential biomarkers for diagnosis and consecutive follow-up of GTD/GTN. It is suggested that qRT-PCR is still the gold standard for expression validation.50–52 Stem-loop RT-PCR is highly sensitive and can detect an expression range of at least 7 logs. 53
Using both absolute and relative quantification approa-ches, the ability of extracellular fetal DNA (hypermethylated
Effective monitoring of GTD/GTN can be achieved using a single C19MC placental-specific microRNA biomarker (miR-520a-5p). Alternatively, other three placental-specific C19MC microRNA biomarkers (miR-516-5p, miR-525, and miR-526a) may be used for the follow-up of GTD/GTN.
Our preliminary data also indicated that the highest levels of extracellular fetal DNA and placental-specific C19MC microRNAs are present in patients at time of diagnosis MHC, MHP, or IM before evacuation of the uterine cavity or before the performance of hysterectomy. Furthermore, metastatic GTN (CH) is associated not only with higher levels of serum hCG but also with higher levels of extracellular fetal DNA (hypermethylated
The establishment of the balance between trophoblast proliferation and apoptosis is crucial during normal placental development. 1 Both aging syncytiotrophoblasts and extravillous cytotrophoblasts undergo apoptosis.2–4 It is obvious that both pre-malignant and malignant molar tissues are also able to release to the circulation apoptotic bodies derived from placental trophoblasts containing extracellular nucleic acids. In addition, a strong positive or a positive correlation between the plasma levels of C19MC microRNAs and serum levels of hCG (intact hCG + free hCG β subunit) was found. hCG is mainly secreted by the syncytiotrophoblast into maternal blood where it peaks around 8–10 weeks of gestation. The invasive extravillous trophoblast also secretes hCG, and in particular, like CH cells, hyperglycosylated forms of hCG. After delivery, miscarriage, or pregnancy termination, hCG falls with a half-life of 24–36 h, until pre-pregnancy levels are reached.54–56
MicroRNAs have been shown to be very stable in the plasma samples for up to 24 h at room temperature57,58 and rapidly cleared after the delivery, usually within 24 h after the delivery.39,40,59,60 In this study, we showed that the degradation of the residual pre-malignant or malignant tissues is accompanied by the continuous decline of circulating C19MC microRNAs, and in case of complete remission of GTD/GTN, extracellular C19MC microRNAs are undetectable in patients’ circulation similar as in the circulation of non-pregnant individuals.39,40
Many cancer treatments work by causing cancer cells to enter an advanced stage of a process known as programmed cell death or apoptosis. Many studies have demonstrated that there are two major cell-intrinsic pathways for inducing apoptosis, one that begins with ligation of cell surface death receptors and another that involves mitochondrial release of cytochrome c. 61 When a cell begins apoptosis, it takes a series of metabolic steps such as fragmenting its DNA or reducing its volume that eventually kills it. It seems that the chemotherapy may induce excessive trophoblast cell death and increased shedding of residual placenta debris into the circulation; thereby, the initial cycle/cycles of chemotherapy are associated with abnormal levels of circulating C19MC microRNAs in plasma samples, at least in some examined patients. Increased levels of extracellular nucleic acids of placental origin, packed into either trophoblast-derived apoptotic bodies or shedding syncytiotrophoblast microparticles, may be detected in circulation of patients after the initial cycle/cycles of chemotherapy.
Therefore, the measurement of extracellular placental-specific C19MC microRNA biomarkers may enable hCG independent non-invasive monitoring of the growth/proliferation and apoptosis of the placental trophoblasts in patients with GTD/GTN. It is obvious that the levels of circulating nucleic acids, mainly the levels of circulating placental-specific C19MC microRNAs, present in the circulation of patients with these particular pregnancy-related disorders reflect the overall status of the residual placental tissue. Besides, this might be useful mainly in patients with hCG false-positive and hCG-negative results.
Outside of pregnancy, hCG may be secreted by abnormal germ cell, or embryonal tissues, in particular, ovarian germ cell tumors and benign or malignant nontesticular teratomas. Rarely, other tumors including hepatic, neuroendocrine, breast, ovarian, pancreatic, cervical, and gastric cancers may secrete hCG, usually in relatively modest quantities. 62
Quiescent GTD is a form of GTD that is completely inactive, benign, or quiescent. Hyperglycosylated hCG is the molecule made by cytotrophoblast cells that drives invasion or implantation and growth during pregnancy. When a pinhead mass remains after removal of hydatidiform mole, a spontaneous abortion due to hydatidiform mole, or after CH or gestational trophoblastic neoplasm chemotherapy, the small mass may lack cytotrophoblast cells and it lacks the hyperglycosylated hCG growth and invasion signal, as such, it is inactive, benign, or quiescent. 63
C19MC is the largest human microRNA gene cluster discovered to date. This 100-kb-long cluster consists of 46 tandemly repeated, primate-specific pre-miRNA genes that are flanked by
By convention, upstream and downstream relate to the 5′–3′ direction in which RNA transcription takes place. Upstream is toward the 5′ end of the RNA molecule, and downstream is toward the 3′ end. Borchert et al.
64
have proposed that upstream
The C19MC cluster is imprinted and exclusively expressed in the placenta from the paternally inherited allele. 66 C19MC microRNAs are expressed predominantly in placental trophoblasts during pregnancy, although they have also been detected in the testis, embryonic stem cells, and specific tumors.45,65,67–73 While the full repertoire of the biological action of C19MC microRNAs remains to be established, data from the various expression studies of C19MC microRNAs imply a role for them in cell proliferation, self-renewal, and in resistance to viral infections.13,72,74
Consecutive large-scale studies are needed to assess sensitivity, specificity, positive, and negative predictive values of circulating hypermethylated
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The work was supported by the Charles University research program PRVOUK P32.
