Abstract
This study aimed to evaluate survival trends for patients with gastric cancer in northeast China in the most recent three decades and analyze the applicability of the UICC tumor-node-metastasis (TNM) classification 7th edition for Chinese patients with gastric cancer. A review of all inpatient and outpatient records of patients with gastric cancer was conducted in the first hospital of China Medical University and the Liaoning Cancer Hospital and Institute. All patients who met the inclusion criteria and were seen from January 1980 through December 2009 were included in the study. The primary outcome was 5-year survival, which was analyzed according to decade of diagnosis and TNM classifications. From 1980 through 2009, the 5-year survival rates for patients with gastric cancer (n=2414) increased from 39.1% to 57.3%. Decade of diagnosis was significantly associated with patient survival (p = 0.013), and the 5-year survival rate in the 2000s was remarkably higher than that in the 1980s and 1990s (p = 0.004 and 0.049, respectively). When classified according to the UICC TNM classification of gastric cancer 7th edition, the prognoses of stage IIIA and stage IIIB patients were not significantly different (p = 0.077). However, if stage T4b and stage N0 patients were classified as stage IIIA, the prognoses of stage IIIA and stage IIIB patients were significantly different (p < 0.001). Hence, there was a significant difference in survival during the three time periods in Northeast China. Classifying stage T4b and stage N0 patients as stage IIIA according to the 7th edition of UICC gastric cancer TNM classifications better stratified Chinese patients and predicted prognoses.
Background
Various surgical approaches, including conventional surgery, function-preserving surgery, minimally invasive surgery, and less extensive lymph node dissection, are used to treat gastric cancer. 1 Previously, surgical interventions were associated with significant perioperative risk, but recently, this risk appears to be decreasing. 2 In Asia, especially in Japan, extended lymph node dissection in gastric surgery is usually not associated with a significant increase in postoperative complications. However, in many Western countries, such as the United States, lymph node dissection is often accompanied with increased postoperative complications and affects patient survival. 3 This could be because Asians generally have less abdominal fat. Thus, organ gaps are more obvious after laparotomy, and vital organs will not be easily damaged during lymph node dissection.
Our previous reports suggested that in the 2000s, survival of patients with gastric cancer was significantly prolonged. In the most recent three decades, and particularly in the last 10 years, remarkable improvements have been made in the comprehensive treatment of gastric cancer. Outcomes for patients with gastric cancer have improved due to early diagnosis, radical operation, and the development of adjuvant therapy. However, most patients with gastric cancer still have poor prognoses.4–6
In 2010, the Union for International Cancer Control (UICC) released the 7th edition of the tumor–node–metastasis (TNM) classification of gastric cancer. 7 Our previous studies followed the standards of the 6th edition. Thus, we retrospectively analyzed the survival trends of patients with gastric cancer who were seen in our hospital from 1980 to 2003. 8 Since some patients seen in 2000s had not met the benchmark of 5-year follow-up, we have only selected 435 patients who met the 5-year follow-up requirement for analysis. Currently, all patients treated in 2000s have met the 5-year follow-up requirement. We analyzed survival rates for eligible patients who met the inclusion criteria, systematically reviewed the patients’ clinical data, and amended incorrectly documented figures in previous research, particularly for data associated with T and N categories, which were classified based on postoperative pathological classification records. Hence, we reclassified TNM stages for all patients according to the 7th edition to determine whether the new classification standards are fully applicable to Chinese patients. Since a lot of time has elapsed since patients from the 1980s and 1990s underwent surgeries, even patients that were cured after the surgery might have died from various nongastric cancer conditions. Therefore, we used postoperative 5-year overall survival rate as the comparison standard.
This study aimed to conduct a systematic and comprehensive review of Chinese patients with gastric cancer in recent three decades and to evaluate changes in survival of patients with gastric cancer in Northeast China. We compared differences in survival rates of patients with gastric cancer between the three 10-year periods. Due to the improvement of healthcare conditions and enhancement of gastric cancer comprehensive treatment, the survival of patients with gastric cancer is significantly improved in the 2000s compared with the previous two decades. We also determined the applicability of the 7th edition UICC TNM classification of gastric cancer to Chinese patients, aiming to provide a reference for more accurate staging and more effective treatment in the future.
Methods
Patients
We enrolled 2414 patients with histologically confirmed gastric cancer who underwent an operation at the First Hospital of China Medical University and the Liaoning Cancer Hospital and Institute between 1980 and 2009. Of these patients, 496, 673, and 1245 patients were allocated to the 1980, 1990, and 2000 groups, respectively. All patients who had histologically confirmed gastric cancer underwent an operation and had complete medical records available.
All patients were followed up by postal letters or telephone interviews. The last follow-up was in December 2015, with a total follow-up rate of 91%. Clinical findings, surgical findings, pathological findings, and all follow-up data were collected and recorded in the database. The study protocol was approved by the Ethics Committee of the First Hospital of China Medical University and the Liaoning Cancer Hospital and Institute.
Endpoints and follow-up
The primary endpoint was 5-year survival. Overall survival was calculated from the day of surgery until death or the last follow-up contact. Data for patients were censored at last follow-up when they were alive. Follow-up assessments were done every 6 months for the first 5 years after surgery and then every 12 months thereafter until death.
Statistical analysis
Kaplan–Meier survival curves were used to estimate patient survival. Cox proportional hazards regression models were used to assess associations of risk factors with survival. For univariate analyses, the prognostic factor of interest and the diagnosis period were covariates in the Cox regression model. Two-sided p values were calculated for all tests and are reported here. p values <0.05 were considered statistically significant. Analyses were performed using SPSS software, version 17.0.
Results
Survival trends in the past 30 years
During the 30-year study period, the median survival of patients with gastric cancer increased from 33 months in the 1980s and 39 months in the 1990s to 49 months in the 2000s. Five-year survival rate estimates were 39.1% (95% confidence interval (CI), 34.8%–43.4%), 38.9% (95% CI, 35.2%–42.6%), and 57.3% (95% CI, 54.2%–60.4%) for the 1980s, 1990s, and 2000s, respectively. There was a significant difference in survival during the three time periods (p = 0.013). Kaplan–Meier survival curves for patients with gastric cancer, by decade, are shown in Figure 1.

Kaplan–Meier survival curves for patients with gastric cancer by three decades from 1980 to 2009.
Patient characteristics by decade are shown in Table 1. No significant changes were detected in the type of gastrectomy or hepatic metastasis during the 30 years studied (p = 0.072 and p = 0.126, respectively). The median age was 56 in the first decade, 60 in the second decade, and 59 in the third decade. Male predominance also decreased from a ratio of 4:1 to 7:3. Among tumor factors, whole stomach tumors decreased from 12% to 5%. However, fewer stage T1 tumors were found in the later decades (Table 1). Most cases were diagnosed at advanced stages throughout the three-decade period.
Characteristics of population from the three periods (N = 2414).

TNM: tumor–node–metastasis.
Multivariate Cox proportional hazards regression models for gastric cancer are shown in Table 2. In the Cox model for gastric cancer, after adjusting for 16 variables, there was a significant association between decade of diagnosis and patient survival (p = 0.007). Patient survival was significantly associated with surgical extent (p < 0.001; Figure 2), and cases involving curative surgery were associated with prolonged survival. Figure 3 displays the Kaplan–Meier survival curves of patients with gastric cancer based on surgical intervention (absolutely curative, relatively curative, or palliative). Factors such as tumor position and Borrmann classification were also key factors affecting prognosis (p < 0.001 and 0.001, respectively; Figures 4 and 5). Tumor size is closely correlated with T and N categories (R = 0.401 and 0.268, p < 0.001 and 0.001). Therefore, tumor size affected patient prognosis. Furthermore, factors like adjuvant chemotherapy also affected patient 5-year survival rates (p < 0.001; Figure 6).
HR for death in population (N = 2414): univariable and multivariable analyses.

HR: hazard ratio; CI: confidence interval; Ref.: reference category; TNM: tumor–node–metastasis.
Derived from tests of HR for prognostic factors in univariate model adjusted for treatment group in Cox proportional hazards model.
Cox regression analysis, controlling for prognostic factors listed in the table.

Kaplan–Meier survival curves for patients under different surgical extents.

Kaplan–Meier survival curves for patients under different surgical intervention.

Kaplan–Meier survival curves for patients with different tumor positions.

Kaplan–Meier survival curves for patients with different Borrmann classification.

Kaplan–Meier survival curves for patients with or without adjuvant chemotherapy.
A 30-year investigation of TNM classification standards
Before 2010, the UICC TNM classification of gastric cancer differed from the gastric cancer classification standard in Japan. The differences are not only in the standard of lymph node staging but also in specific stage assignment. The most significant difference is that T4N1 disease is classified as stage IV using the UICC standard, but it is still considered as stage IIIB disease in Japan. Based on the 6th edition UICC classification, the survival curves of stage IIIA and stage IIIB patients were not significantly different (p = 0.561). However, according to the Japanese standard, the survival rates were significantly different between stage IIIA patients and stage IIIB patients (p < 0.001; Figure 7). With the 7th edition UICC TNM classification of gastric cancer, the severity of the tumor was accurately reflected not only by the T and N categories but also by the overall TNM staging, and the differences for each stage become more obvious. Survival differences between stage IIIA and stage IIIB patients increased compared with the previous staging practices, but the difference did not reach statistical significance (p = 0.077). When previous staging standards in Japan were used to classify T4bN0 disease as stage IIIA, the survival difference between stage IIIA and stage IIIB patients became larger and reached statistical significance (p < 0.001; Figure 8).
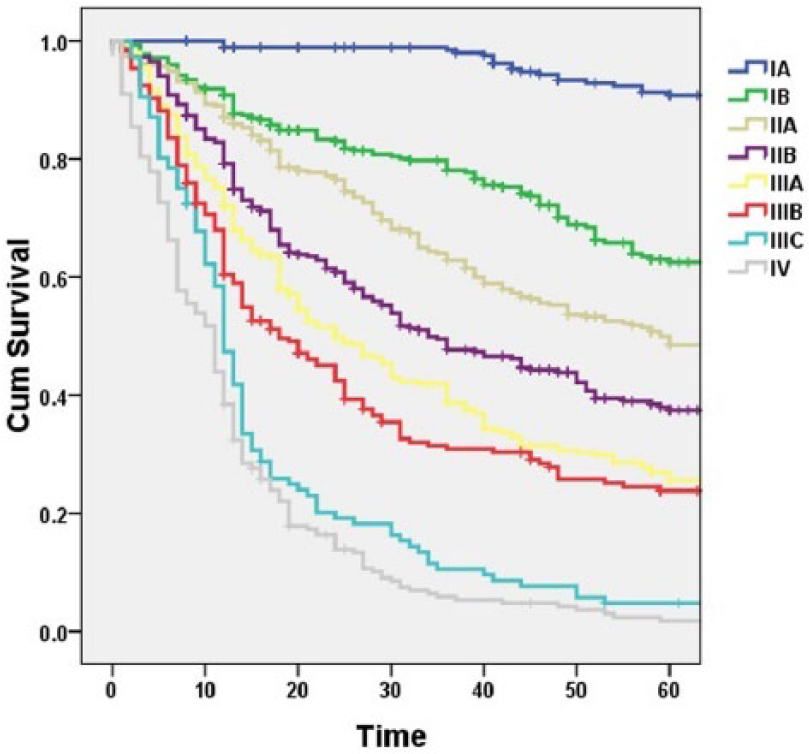
Kaplan–Meier survival curves for patients according to the 7th edition UICC classification.

Kaplan–Meier survival curves for patients after classifying T4bN0 as stage IIIA in the 7th edition UICC classification.
Discussion
This study demonstrated that since 2000, there has been a significant increase in the 5-year survival rate of patients with gastric cancer, which may be closely associated with factors, such as increased physical examination, screening, and improvements in comprehensive treatment. 9 However, the majority of patients with gastric cancer are currently elderly and from rural areas, and the lack of timely and standardized treatments as well as poor compliance are still severe issues with treatment. 10
Unfortunately, a complete cancer registration and follow-up system has not yet been established. Therefore, it is difficult to collect complete treatment information and comprehensive postoperative treatment details. In the future, a complete cancer registration and follow-up system should be established, and randomized controlled clinical trials should be conducted. In general, we believe that with screening and physical examination becoming more and more common, more patients could be identified at early and intermediate stages. 10 According to the T category data in Table 1, the proportion of stage T2 patients indeed increase over time, but the proportion of stage T1 patients decreased significantly. We think that although selection bias in the retrospective study cannot be excluded, we should also consider the possibility of biological behavior changes in gastric cancer over the three decades, that is, whether gastric cancer is developing more rapidly to intermediate-stage disease right after initiation.
The postoperative survival rate of patients who underwent surgery in the 1990s was higher than the survival rate of patients who underwent surgery in 1980s. However, the survival rate in the 1990s declined gradually over time, and the final 5-year survival rate for patients in 1990s was not significantly different from the rate in 1980s. Currently, the cause of this phenomenon is still unclear. We hypothesize that the unstandardized comprehensive treatments in the 1990s may have shortened postoperative survival. The 5-year survival rate in 2000s was significantly higher than that in the 1980s and 1990s, and it is especially higher than that in the 1990s, which may be attributed to improvements in the comprehensive treatment for gastric cancers.
In the most recent three decades, both the Japanese TNM classification of gastric cancer and the UICC TNM classification of gastric cancer have undergone several major changes. 7 The two standards were different in the classification of the N category until 2010, when UICC released the 7th edition of TNM classifications of gastric cancer. Now, the number of metastatic lymph nodes is used for N classification, and this standard was eventually accepted by Japan. 11 Here, we used the 7th edition of UICC TNM classification of gastric cancer as staging criteria for all patients and simply examined its applicability to Chinese patients. Meanwhile, we also reclassified all patients according to the 6th edition of the UICC classification and the concurrent gastric cancer classification standard in Japan, and we reviewed the applicability of these two classification standards.
Statistical analysis showed that many factors, including tumor location, tumor size, and Borrmann classification, can affect the prognoses of patients with gastric cancer. In the future, all of these factors should be considered for gastric cancer classifications, although these factors all significantly correlate with the T and N categories.12,13 In addition, 5-year survival rates are influenced by other factors, such as adjuvant chemotherapy, organ resection, and postoperative complications.5,14,15
By comparing the applicability of previous classification standards, we hope to provide a reference and ideas for improving current staging criteria. Using this comparison, we found that prior to publication of the 7th edition UICC TNM classification of gastric cancer, the Japanese gastric cancer TNM staging was more suitable for Chinese patients, especially for the two late stages, IIIA and IIIB, due to poor prognosis. Objectively, the difference in survival rate may not be significant. However, compared to the UICC standard, the Japanese standard could distinguish patient survival rates between those two stages more clearly. With the 7th edition of UICC classification released in 2010, due to the improvement in T category and N category classifications, 16 survival differences became more obvious between stage IIIA and stage IIIB, but they did not reach statistical significance. We also considered previous gastric cancer classification standards in Japan and creatively classified T4bN0 as stage IIIA to find an increased survival difference between stage IIIA and stage IIIB patients, which was statistically significant.
Since we conducted a retrospective study, the data reliability is lower than that of clinical trials. More appropriate TNM classification guidelines for gastric cancer, specifically in the Chinese population, need to be further studied.
Conclusion
China has a huge population of people with gastric cancer. Unfortunately, accurate patient registration and follow-up information are not always available, and standardized diagnosis and treatment are not performed, likely because scientifically strict large multicenter randomized controlled clinical trials have not been performed. Presently, patients in Chinese hospitals are usually transferred to an oncology ward for continuous treatment after surgeries, making it difficult for the surgical wards to obtain relevant treatment information. In our study, some comprehensive treatment information was hard to obtain, and all patient information was obtained retrospectively, leading to a lower reliability of the data than that of clinical trials. Thus, our discussion of categorizing T4b and N0 disease as stage IIIA in the 7th edition UICC TNM classification of gastric cancer is only of reference value. More accurate classification standards need to be based on scientific data from clinical trials. We hope our study can provide a reference for conducting more scientifically accurate clinical trials and completing more standardized and precise diagnoses and treatments.
Footnotes
Acknowledgements
H.G., D.L., and X.L. participated in the design of the study. Y.Y., H.Z., H.Z., and D.Q. performed the statistical analysis and drafted the manuscript. P.L. and M.Y. participated in the coordination of the study. All authors read and approved the final manuscript.
Availability of data and materials
The data will only be shared to the editors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and informed consent
The study was given ethical approval with Ethical Committee of the First Hospital of China Medical University and the Liaoning Cancer Hospital and Institute, and all the patients had given written informed consent.
Funding
This work was supported in part by China National Natural Science Foundation (81402384).
