Abstract
Background
Prescriptions are medication plans communicated between prescribers and pharmacists for patients. An ideal prescription must be rational, evidence-based, complete, and clear, and enhance the therapeutic outcome of the patient. Elderly patients are vulnerable to multiple illnesses and thereby prone to polypharmacy and drug related problems.
Objectives
Prescription appropriateness and quality of prescribing are a serious concern in geriatric care. This study aims to develop and validate a clinical tool to assess the quality and suitability of geriatric prescriptions.
Materials and Methods
The study population consisted of elderly inpatients. The tool development followed item selection, defining the need for each item, drafting, Delphi analysis, pre-testing, and validation. The tool was tested for feasibility, validity, and reliability during validation.
Results
The tool comprised assessing prescribing information, quality indicators, drug use indicators, cost indicators, and morbidity indicators. The Prescription Quality Assessment Tool in Geriatrics (PQATG) was found to be feasible after pre-testing. The panel achieved consensus on 20 items. The content validity ratio = 0.6, with the sum of content validity index being 16.082. The construct validity test produced a significant correlation, and the intra-class correlation coefficient indicated moderate reliability. The variables PQATG score and number of drugs per prescription were found to be negatively correlated, r (92) = –0.2886, p = .0005371. Pilot studies proved that 86.6% (n = 79) of prescriptions had good quality, 44% (n = 4) rated moderate quality, and 8.8% (n = 8) were poor.
Conclusion
The PQATG is a feasible, reliable, valid, and novel tool for evaluating the quality of geriatric prescriptions.
Keywords
Introduction
According to the United Nations, an “older person” is 60 years or older, with persons aged ≥80 years or older being referred to as “oldest old.” The global population is aging, with the number of people aged over 65 years expected to reach 71 million by 2030. 1 Pharmacokinetic parameters may be altered in the elderly owing to age-related physiologic changes. 2
Despite the age-related changes, comorbidity is a significant factor leading to polypharmacy and drug-related problems. Multi-morbid patients are prescribed more medications, which increases their risk of inappropriate prescribing (IP), drug–drug interactions, drug–disease interactions, adverse drug events, and medication errors. 3
Prescriptions are medication plans communicated between prescribers and pharmacists for patients. An ideal prescription must be rational, evidence-based, complete, and clear, and enhance the therapeutic outcome of the patient. Prescription appropriateness and quality of prescription have been serious concerns ever since the beginning of pharmaceutical care. 4
The term “potentially inappropriate medications (PIMs)” began gaining attention after Mark H. Beers developed Beers’ criteria in 1991. 5 Globally, there was also the development of other criteria/lists of medications stating inappropriateness. For the most part, these are broadly divided into implicit and explicit prescribing criteria. 6 The best-known implicit prescribing criteria set is the Medication Appropriateness Index (MAI), which addresses 10 aspects of each drug prescription, aimed at identifying a variety of potential prescribing errors. However, its use remained in the realm of research rather than routine clinical practice. 1
In India, observational studies evaluating the prevalence of inappropriate medication in geriatric settings were able to unveil enormous data. But these studies were done using explicit and implicit criteria developed and validated in the foreign elderly population. No single criterion or list has its development conducted exclusively in Indian elderly settings. The development of an easy-to-handle, quick-reference single tool may help to address the issues of primary and tertiary care clinicians and thereby meet the quality demand of prescription appropriateness.
Thus, this study aims to develop and validate a tool to assess the quality and appropriateness of geriatric prescriptions, thereby designing a quality checker to address and satisfy prescriber and clinical pharmacist requirements for geriatric care in their routine use.
Materials and Methods
The study was conducted in four different phases, on a prospective observational framework. Multiple rounds of the Delphi method and statistical techniques validated the developed tool. The inpatient setting of a 650-bed tertiary care hospital with clinical pharmacy services in Kerala, India, was sought as the study site. The study extended from January 2020 to June 2024.
Sampling
The study population comprised elderly people under geriatric care in the study settings. Simple random sampling was followed to pre-test the developed draft at the study site. The multi-round Delphi method was used for the validation process. The expert panel selection criteria followed a convenience sampling technique for the selection of respondents. A panel of 15 experts was created for the Delphi analysis, consisting of five doctors, five clinical pharmacists, and five academicians with research expertise in geriatrics. Pilot studies were carried out with a simple random sampling technique. The estimated sample size for pilot studies was 92.
The prescriptions of all inpatients above 60 years of age were included in the study during pilot testing, follow-up, and evaluation of the developed tool. Outpatients were included only during the application of the tool in clinical settings.
Prescriptions with incomplete data and prescriptions of patients in the casualty and on ventilators were excluded.
Study Method
The construction of the prescription quality assessment tool followed several steps.
Item Selection
The identification of items relevant to prescription quality was carried out through an extensive literature review. Literature in the English language, resourced through bibliographic databases like PubMed, Cochrane Library, MEDLINE, and DOAJ, aided the review. A broad range of items significant for prescription appropriateness and prescription quality was studied. The selection of appropriate items also required discussions and verification with researchers and expert clinicians.
Defining the Need for a Selected Item
After the inclusion of a selected item, variable, or question, the relevance of the inclusion of the particular item needed to be well-defined, which necessitated further literature review.
Drafting the Tool
The selected items were then organized and drafted, along with scores, into a tool. Pre-testing of the tool was done in a selected sample of subjects at the study site. The tool was subjected to revision following the pre-testing results.
Content Validation
The revised draft of the tool was subjected to validation. For an expert consensus process, a three-round Delphi method was adopted as the study design. This was materialized with the expert panel. Each panel member was presented with the complete Prescription Quality Assessment Tool in Geriatrics (PQATG). The experts evaluated the developed draft, and their agreements and disagreements were analyzed using a five-point Likert scale. The agreement for each item was rated on the five-point Likert scale of 0 (strongly disagree) to 4 (strongly agree).
Following the responses of the expert panel after the first Delphi method, if any item/indicator needed to be altered/added/removed, changes were made accordingly and redrafting was done. The re-drafted tool was subjected to a second round of the Delphi survey method and revised again. The third round of Delphi included a qualitative assessment of the draft by the experts. The final draft was thus developed and validated.
Pilot Studies
Pilot studies were done among 92 elderly patients to evaluate prescription quality and appropriateness using the PQATG at the study site. The sample size for pilot studies was calculated using the formula sample size (n) = Z1– α /2 2 p (1 – p)/d2, where Z1– α /2 2 is the standard normal variance at 5% type 1 error and is 1.96, p is the expected prevalence, and d is the margin of error. The average time taken for the extraction of a medical chart with PQATG was 8–10 min. The well-defined prospective approach of data collection helped avoid confounding and information bias during pilot studies.
The data obtained were statistically analyzed to frame the results for report writing. Data analysis was carried out using Statistical Package for the Social Sciences (SPSS) version 29.01.0. The tool was tested for feasibility, reliability, and validity during validation. When feasibility was tested through pre-testing, logical and apparent validity was analyzed from expert consensus. Content validity was tested using the content validity ratio (CVR) and content validity index (CVI). Construct validity was measured using the Pearson correlation coefficient. Factor analysis provided a measure of content validity and construct validity. Cronbach’s alpha and intra-class correlation coefficient (ICC) were used to test reliability.
Theoretical Framework of PQATG
The theoretical framework on which the PQATG development was based was the structure–process–outcome (SPO) model, substantiated with an extensive literature review. Donabedian’s (2005) three-component approach for evaluating the quality of care underpins measurement for improvement. The three components are structure, process, and outcomes. According to Donabedian, quality is a measure of an organization’s effectiveness, assessed through quality indicators categorized into structure, process, and outcome variables. These variables are interconnected, where a strong structure creates favorable conditions for an effective method, ultimately leading to better outcomes. 1 The most comprehensive quality assessment tool incorporates all three categories; however, in developing the new tool, factors such as simplicity, practicality, feasibility, available manpower, time constraints, and resource limitations were carefully considered to ensure its usability and implementation in real-world settings.7, 8 The framework elaborates the principles, criteria, indicators, and items in the tool. The quality assessment tool is developed on the basic principle of implicit measurement and a specific approach.9–11 The implicitness and specificity are monitored through four criteria, namely, prescription quality, patient safety, economic burden, and general health.12, 13 Each criterion is represented by relevant indicators, which consist of a set of different patient data elements with certain established relationships. 14 The indicators include the prescribing indicators,15, 16 quality indicators, 17 drug use indicators, 18 cost indicators, 19 and morbidity indicators. 20 Figure 1 depicts the theoretical framework of the development of PQATG. The elements representing the indicators can be used for quantitative or qualitative measurement of patient data.
Theoretical Framework of Prescription Quality Assessment Tool in Geriatrics (PQATG) Development Using Structure–Process–Outcome (SPO) Model.
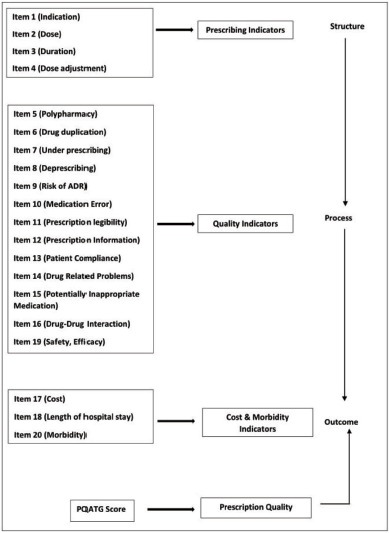
Results
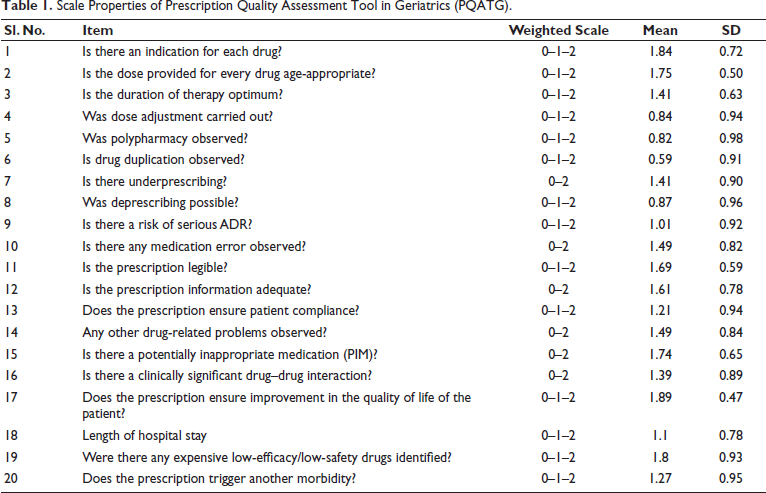
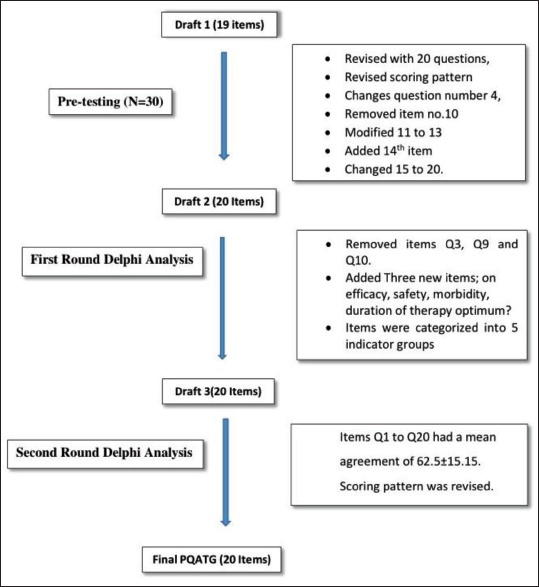
The first draft of PQATG consisted of 19 questions with respective ratings and scores such that the 19 questions cover all 5 indicators mentioned in the theoretical framework (Figure 1). Pre-testing was done on 30 prescriptions of geriatric patients. Inferences drawn from pre-testing among 30 prescriptions demanded a revision of scoring, revision of questions (Q) 4, 2, 8, 9, 10, and 11, and modification in Q1. The results of the internal consistency test using Cronbach’s alpha equation were reported as excellent, with Cronbach’s alpha = 1. Based on the inferences drawn from the pre-testing, the first draft was revised to draft 2 with 20 questions, revised scoring pattern, changed question number 4, removed item no. 10, modified items 11–13, added the 14th item, and changed items from 15 to 20.
Draft 2 was subjected to the first-round Delphi analysis using a five-point Likert scale assessment. The expert panel in the first round of Delphi provided disagreements for items Q2, Q3, Q4, Q6, Q9, Q10, Q11, Q15, and Q17.
Revision of draft 2 was done with the removal of items Q3, Q9, and Q10. Three new items were added, namely, (a) Was there any expensive low-efficacy/low-safety drug identified? (Q19), (b) Does the prescription trigger another morbidity? (Q20), and (c) Is the duration of therapy optimum? (Q3). Also, the items were categorized into prescribing information (Q1–Q4), quality indicators (Q5–Q14), appropriateness indicators (Q15–Q16), cost indicators (Q17–Q19), and morbidity indicators (Q20). Hence, draft 3 was developed. Draft 3 was subjected to a second-round Delphi analysis. The results of the five-point Likert scale analysis of the second round of Delphi were interpreted. Items Q1–Q20 had a mean agreement of 62.5 ± 15.15. Scale properties of the final draft of PQATG, following expert consensus, are outlined in Table 1. Hence, all factors were subjected to validation pilot testing. The scoring pattern was revised as well. Figure 2 depicts the sequential development process of the tool.
Scale Properties of Prescription Quality Assessment Tool in Geriatrics (PQATG).
Detailed Development of Prescription Quality Assessment Tool in Geriatrics (PQATG).
Validation of the Prescription Quality Assessment Tool
The PAQTG was tested for three aspects: feasibility, validity, and reliability. 21
Feasibility
The method used for testing feasibility was a pre-testing study in a group of 30 subjects’ prescriptions. Hence, 30 prescriptions were assessed. Table 1 shows the results of the feasibility tests. The prescription quality assessment tool was found to be feasible.
Validity
A validity test was done to measure the capacity for which the tool was designed. Logical and apparent validity measures the degree of appearance with intention or checks the logic of questions before they are finalized through consulting experts. The content validity test measured the dimensions of the concept. The methods used were CVR and I-CVI.
CVR = Ne-N/2/N/2, where Ne is the number of panelists indicating essential; N is the total number of panelists. CVR ranges from –1 to +1; positive values indicate that at least half of the content is essential.
For the PQATG, CVR = 12-15/2/15/2 = 0.6.
I-CVI = The CVI was measured for each item to assess relevancy and clarity in the tool. Tables 2 and 3 show the calculated CVI for each item and the interpretation, respectively.
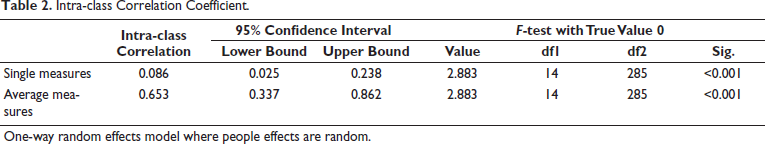
Intra-class Correlation Coefficient.
One-way random effects model where people effects are random.
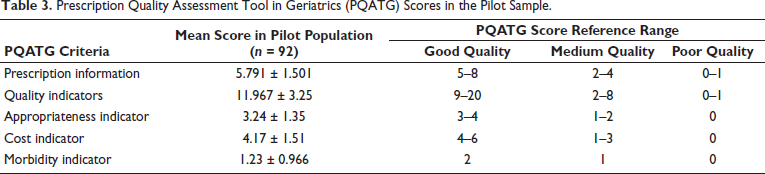
Prescription Quality Assessment Tool in Geriatrics (PQATG) Scores in the Pilot Sample.
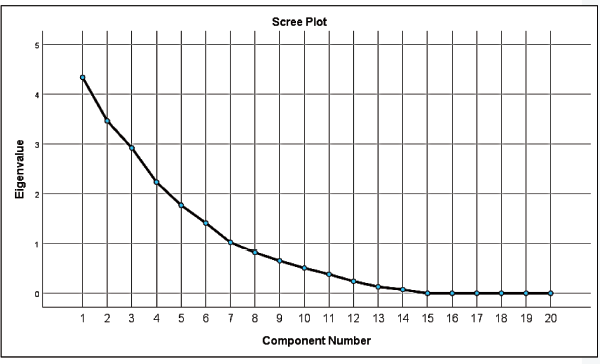
Construct validity measures the capacity of the instrument to measure the theoretical concept. Factor analysis was done since criterion validation was not possible. It is a measure of content validity as well as construct validity. The extraction method used was principal component analysis. The matrix was not positive definite, and hence the curve is concave down. The scree plot shown in Figure 3 flattens from Q15 to Q20. The elbow of the scree plot lies on Q15, indicating that factors Q1 to Q15 attain significant importance, and lesser variance being observed in Q16–Q20.
Scree Plot. X-axis: Principal Component Number; Y-axis: Percentage of Variance Observ
Scale level index of an instrument (SCVI) is the sum of CVI/total number of items in the tool. SCVI of PQATG 16.082/20 = 0.8041, which indicated an appropriate scale level index for PQATG.
Reliability
Reliability measures the magnitude of precision or the tool’s freedom from errors. Methods used include internal consistency to estimate the number of items composing the instrument and the mean correlation between them. The statistical method used was Cronbach’s alpha, with values between 0 and 1 (Table 2). In the PQATG, all items were found to be reliable.
To analyze the test-retest or intra-observer reliability, the statistical method used was the ICC as shown in Table 2. The PQATG observed an ICC of 0.653, indicative of moderate reliability for the PQATG.
The PQATG is thus feasible, reliable, and valid to be used in clinical settings. Figures 4(a) and 4(b) show the final draft of PQATG.
The Validated Final Prescription Quality Assessment Tool in Geriatrics (PQATG).

The Validated Final Prescription Quality Assessment Tool in Geriatrics (PQATG).
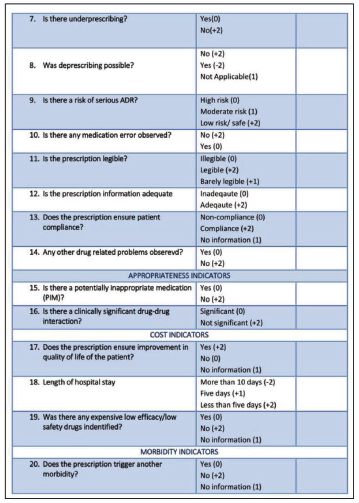
Scoring Scale and Interpretation of PQATG
The scoring of the PQATG is based on interval scales. Evaluators were asked to use the numerical scale to make a rating. The scoring scale is as depicted in Table 3.
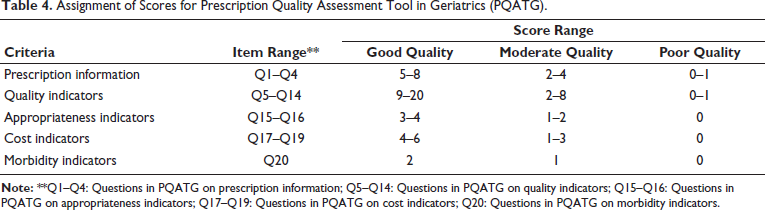
A medication chart with a total score of 40 and a quality indicator score of 20 was rated as an optimum quality prescription, while those scored between 19 and 40 were rated as good, 6–18 as moderate quality, and 0–5 as poor quality. The assignment of scores is depicted in Table 4.
Assignment of Scores for Prescription Quality Assessment Tool in Geriatrics (PQATG).
Pilot Study of PQATG in the Elderly
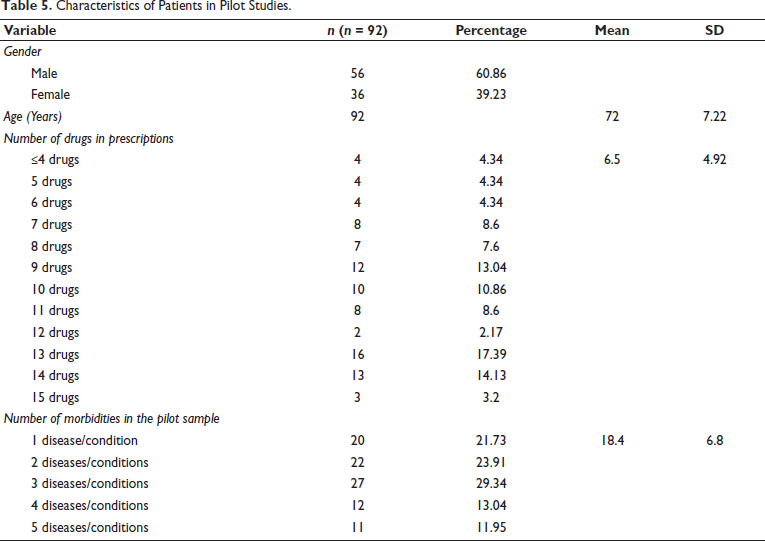
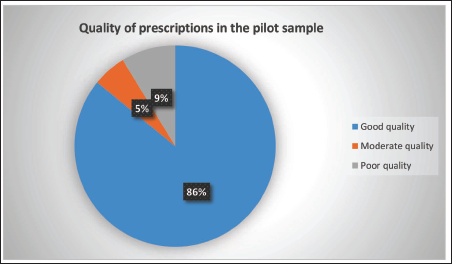
The application of the PQATG in the study settings was carried out as pilot studies. The characteristics of the patients in the pilot sample are outlined in Table 5. The mean age of the participants was 72 ± 7.22 years. The mean score obtained after the evaluation of prescriptions with the PQATG was 25.71 ± 7.39. Table 6 explains the distribution of mean scores among all indicators. It was observed that 86.6% (n = 79) of prescriptions were rated as good quality, 4.44% (n = 4) were rated as moderate quality, 8.8% (n = 8) were poor, and zero prescriptions met the optimum quality rating (Table 6 and Figure 5). The variables PQATG score and number of drugs per prescription were found to be negatively correlated, r (92) = –0.2886, p = .0005371. The result was significant at p < .05. The average time taken for evaluating the prescription was 10.07 ± 3.36. Polypharmacy was observed in n = 83 (90.02%) of prescriptions, while n = 9 (9.7%) had no polypharmacy. It was also observed that an average of 10.71 drugs were prescribed per patient during the pilot studies.
Characteristics of Patients in Pilot Studies.
Distribution of Quality of Prescriptions in the Pilot Sample.

Distribution of Quality of Prescriptions in the Pilot Sample.
Discussion
The prescription quality assessment tool was developed through an extensive review of literature, along with assorted input from the population of interest. The tool development process pre-requisite involves peer review and expert judgment during validation stages. Also, the advantages of adopting an expert-oriented approach strengthen the selection of elements of the tool, as experts provide the most recent thinking in the area of geriatrics and prescription quality. The panel for the Delphi technique was not statistically selected but based on expertise, knowledge, and experience, which were deeply incorporated in the PQATG.
The initial concept of tool development was set out through the SPO model. The Donabedian model is used for tool development and evaluation of established tools, as improvements in the structure of care shall lead to enhanced clinical outcomes and thereby refine patient care. 22 Tossaint-Schoenmakers et al. used the SPO framework for the development of a quality management model and self-questionnaire for hybrid healthcare in 2022. 23 The SPO concept was used by Ameh et al. for the evaluation of the quality of integrated chronic disease management in South Africa in 2017. 24
Implicit approach and patient specificity are the mainstays of the tool. The MAI was used as the implicit criterion in geriatrics to assess the appropriateness of medication prescribed by healthcare providers and to evaluate the self-medication practices of patients. 11 When explicit tools ease the process of medication optimization through interventions, implicit tools like MAI and Assessment of Underutilization (AOU) assess inappropriate medication use effectively. 10 The ability to assess quality depends on the scale of measurement used. Developing scales and seeking quality assessment were judgmental initially. The responses of the expert panel contributed to the choice of elements in the PQATG. The judgments analyzed using the Likert scale made psychometric evaluations easier. Each disagreed element was subjected to an extensive literature review and brainstorming discussions. The weighted agreements were later subjected to statistical assessment. However, the initial draft of the tool was subjected to pre-testing in 30 prescriptions so as to explore further room for improvement.25, 26 Despite being feasible, pre-testing results disclosed the matter of time consumption, demanding minor changes. Pre-testing has been demonstrated to be effective in similar tool development studies, such as those conducted by Hassan et al. and Hanlol et al.17, 19
A good quality assessment tool must be able to differentiate between good prescriptions, prescriptions necessitating changes, and prescriptions with problems. To effectuate the goal, the identification of criteria that reinforce the greatest quality was performed through further extensive study. Clinical experience of healthcare professionals was equally prioritized. Hence, prescription information and quality indicators in the tool inclined toward clinical expertise were allocated with a weightage of scores in PQATG. Consensus of the panel and healthcare professional team expertise were used equally during the development of the Medication Regimen Complexity Index (MRCI) by George et al. in 2004. 27
MAI could be considered the currently available standard implicit criteria for geriatric prescription quality assessment.9, 19 The PQATG offers a more comprehensive, systematic, and user-friendly approach, hence advantageous than MAI. Unlike MAI, PQATG is specifically designed for routine prescription auditing, quality control, and regulatory compliance in hospitals and pharmacies. It provides a structured, quick, and standardized evaluation of prescriptions, incorporating factors such as dose optimization, therapeutic duplication, adherence to clinical guidelines, and overall prescription integrity. This makes PQATG more practical for real-time clinical decision-making, particularly in geriatric care, where polypharmacy and deprescribing are critical concerns. While MAI is useful for assessing medication appropriateness, PQATG enhances overall prescription quality and patient safety, making it a valuable tool for improving geriatric healthcare standards.
During the validation, feasibility, validity, and reliability tests analyzed the clarity, measuring capacity, and magnitude of precision of PQATG. The contents and scoring evolved further during the process of validation. During the measurement of content validity, three items in the PQATG were eliminated, and five items required revision. Moderate reliability was obtained with internal consistency and the ICC. As Cronbach’s alpha yielded moderate reliability, the F-test was used to confirm whether biases were present.28, 29 The moderate reliability obtained in the study may be attributed to the differences in the experience and perspectives of the raters. These results were consistent with the results of other similar studies.17, 30, 31 Establishing high-quality measures of the construct must be prioritized during tool development. 23 Factor analysis extracted seven factors. 32
While PQATG was applied to geriatric patients, the PQATG scores displayed normal distribution with no ceiling effects during normality tests. This, in turn, allows differentiation between prescriptions of varying quality. It is an accepted fact that age-related changes in the elderly affect pharmacodynamics and pharmacokinetics. However, the lifespan, clinical practice requirements of patients, and guidelines for pharmacotherapy vary according to different countries. The PQATG and its implicit nature make the tool easily adaptable to various clinical environments, suiting different requirements. Yet, modifications in the tool and scoring may demand revalidation.
The PQATG may take 10 min or longer for an evaluation, depending upon the number of drugs in the prescriptions and the clinician’s experience. The tool could be used for written and computer-generated prescriptions. An instant quality check with e-prescriptions is a hallmark of advanced geriatric care. The PQATG is thus appropriate for use in most geriatric clinical settings and research.
The PQATG was developed based on strong theoretical as well as clinical information. The extensive literature review and testing enabled the clinical, legal, and clerical requirements for prescription quality. The multiple rounds of psychometric testing with experts demonstrated acceptable feasibility, reliability, and validity for the tool. The PQATG enabled the differentiation of optimum-, good-, moderate-, and poor-quality prescriptions and met all standards of validation. The implicitness and patient specificity of PQATG make it a quick quality checker in the routine practice of clinicians.
The tool development study was subject to certain limitations. The PQATG was completely developed from the perspectives of physicians, clinical pharmacists, researchers, and patients. However, the magnitude of quality could also be measured from the dimensions of dispensing pharmacists, technicians, nurses, and other supporting healthcare professionals. Appropriateness with drug dilutions and dosage adjustment may vary according to patient requirements and mostly depends upon the discretion of the physicians, which is seldom negotiable. Additionally, healthcare infrastructure and digital integration vary globally, affecting the feasibility of implementing PQATG as a routine prescription auditing tool in low-resource settings.
Inculcating organizational factors influencing prescription quality in the tool found restrictions. Prescription quality may be affected by the resources in the healthcare facility, administrative management, policies and procedures, work schedule, and time. Variations in the time taken for assessment and scores were observed during pre-testing and pilot studies, necessitating sufficient practice and training to use the tool.
Lastly, validation studies across diverse healthcare systems are essential to ensure the tool’s adaptability and effectiveness in different clinical environments. Addressing these limitations through cross-cultural validation, integration with international prescribing standards, and customization for specific healthcare settings will enhance the broader applicability of the tool.
The advancements and future of prescription quality rely on the execution of technology-integrated quality assessment strategies and artificial intelligence-assisted prescription audit systems. Rational drug therapy, evidence-based approach, and drug use considerations of the elderly culminate in promising geriatric care outcomes that satisfy the expanding lifespan.
Evaluation of every single geriatric prescription using implicit PQATG materialized in the current research remained a fantasy earlier in routine clinical practice, particularly in a country with expansive population statistics like India. Thus, the availability of a desktop usable tool turned beneficial for clinicians so that elite care was provided to the older adults even at the study settings.
Conclusion
The PQATG is a promising novel instrument for evaluating the quality of geriatric prescriptions. The tool consolidated multiple criteria of quality assessment. Assessment of quality was benchmarked through prescription information, quality indicators, appropriateness indicators, cost indicators, and morbidity indicators, followed by scoring to rate quality. Extensive psychometric assessments enabled the generation of expert ideas throughout the development of PQATG. The PQATG has been observed to be a valid, reliable, feasible, and responsive tool to assess the quality of geriatric prescriptions. The study also set forth the opportunities and applications of geriatric pharmacy practice in accrediting the quality of healthcare management of the elderly.
Footnotes
Abbreviations
CVI: Content validity index; IP: Inappropriate prescribing; MAI: Medication Appropriateness Index; PIM: Potentially inappropriate medication; PQATG: Prescription Quality Assessment Tool in Geriatrics; SPO: Structure–process–outcome; SPSS: Statistical Package for the Social Sciences.
Acknowledgments
The authors acknowledge all physicians, clinical pharmacists, researchers, and academicians of KIMS Al Shifa Hospital, Perinthalmanna, Kerala, India and Al Shifa College of Pharmacy, Kerala, India, who extended their support during the study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Institutional Ethical Committee Approval was obtained prior to the commencement of the study. The IEC number is KAS:ADMN:IEC:60:21.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Informed consent from the clinical pharmacist, researchers and academicians were obtained prior to the study.
