Abstract
Background
As traditional laboratory animal use faces ethical and logistical constraints, the chick ileum has been experimented with as an alternate tissue for bioassays, which is crucial for drug discovery and pharmacology education. However, the absence of uniform bioassay protocols for chick ileum presents a significant challenge, impacting the accuracy and reliability of results in pharmacological studies.
Objectives
The objective of this review was to compile existing data on chick ileum bioassays from published sources, aiming to enhance future experiments and contribute to the establishment of standardized methods for conducting these bioassays.
Methodology
We conducted a systematic literature review, utilizing keywords like “Chick ileum,” “Bioassay,” “Acetylcholine,” and “Histamine” across databases such as PubMed, Google Scholar, and ScienceDirect. The study selection adhered to specified inclusion and exclusion criteria, focusing on chick ileum research. Data extraction and subsequent analysis were performed to retrieve data for the experiment.
Results
Eleven articles were selected after applying strict inclusion criteria. Studies ranged from 1967 to 2019, with diverse physiological salt solutions and varied responses to essential chemicals like acetylcholine and histamine. Assay methods varied, but three-point assays were common. Commonly employed tissue lengths and temperatures were identified, and additional factors like organ bath volume and aeration were crucial for consistent results.
Conclusion
The review highlights the feasibility and ethical advantages of using chick ileum for bioassays. Despite challenges like variable tissue responses and methodological diversity, the review suggests these can be navigated with careful optimization, making chick ileum a valuable model for pharmacological research and education.
Introduction
Animal experiments have been indispensable for instructing students in fundamental medical sciences for numerous years. They have significantly contributed to biomedical research by elucidating the mechanisms of human organ function and uncovering countless principles underlying bodily processes. Incorporating animal experimentation into medical education fosters a scientific mindset among students. 1 Various rodents like mice, rats, rabbits, guinea pigs, hamsters, and primates are commonly used in lab experiments. Mice, favored for their small size and quick breeding, are especially preferred in pharmacological studies. Other species, like rats, guinea pigs, rabbits, and dogs, have unique qualities suited for specific experiments.2, 3
Guinea pigs, rabbits, and rats are common sources of isolated tissue, often used to study drug effects on specific receptors. This method aids in drug bioassays, receptor characterization, and discovery of new drugs. 4 Isolated tissue offers advantages like testing multiple preparations from one animal, requiring small test quantities, and evaluating drug effects directly without absorption, metabolism, or nerve reflex interference. 5 However, the utilization of such experiments in teaching undergraduate and postgraduate students has been contentious globally in recent years. 6 The ecosystem has been significantly impacted by the indiscriminate utilization of these animals, prompting numerous countries and scientists to seek alternatives to animal testing. 7 In India, the Committee for the Control and Supervision of Experiments on Animals (CCSEA) sets guidelines for lab animal facilities to enhance animal welfare and advance biological knowledge. 8 This fosters the exploration of alternative methods for biological testing.
Alternatives to animal experiments include using tissues, bodily fluids, microorganisms, primary cell cultures, and established cell lines. While models and computer programs offer insights, they are costly and may not fully replicate animal responses. Bioassays remain crucial in drug discovery and pharmacology education.9, 10 Bioassays can be conducted through interpolation, matching, bracketing, three-point assay, and four-point assay.9, 11 Bioassays often require sacrificing whole animals for small tissue samples. An alternative suggests using tissues from commonly consumed animals like poultry, sheep, goats, cattle, and fish.
Chick ileum tissue is easily accessible, eliminating the need for further animal sacrifice. Its use offers benefits like cost-effectiveness, ease of mounting, and reliable responsiveness. 9 Utilizing chicken intestines is ethical and cost-effective, sourced as waste from slaughterhouses. 12 Studies reveal the chicken small intestine’s uniform diameter and thick circular muscles compared to longitudinal ones. 13 The chick ileum contains various receptors like cholinergic muscarinic, serotonergic, histaminic, prostaglandin, progesterone, tachykinin, and motilin receptors.14–18 Experiments involving isolated chick ileum primarily aim to elucidate drug receptor actions in vitro, focusing on grasping concepts such as graded responses, antagonism nature, potentiation, and dose ratios. 19
The limited scope of experimentation on chick ileum under various conditions has yielded inconsistent and non-reproducible results. A significant challenge is the lack of standardized optimal experimental conditions, which impairs the reliability of bioassays and contributes to inaccuracies in data interpretation. This systematic review aims to consolidate and compare the existing body of research, providing a comprehensive overview of the most frequently investigated experimental parameters. The goal is to facilitate future studies by offering insights into commonly used conditions, thereby enabling more precise and standardized experimental designs in chick ileum research. Additionally, ethical considerations surrounding the use of animal models must be addressed, ensuring that future experiments adhere to strict guidelines for animal welfare and the minimization of unnecessary suffering.
Methodology
Study Setting and Design
A systematic literature review was carried out following the guidelines outlined in the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA).
Search Strategy and Study Selection
Search queries were formulated and executed on the designated electronic databases: PubMed, Google Scholar, and ScienceDirect, spanning from inception to June 30, 2024. Studies were retrieved using the following keywords: “Chick ileum,” “Chicken tissue,” “Bioassay,” “Acetylcholine,” and “Histamine.”
Inclusion and Exclusion Criteria
All research conducted globally on the chick ileum, irrespective of evidence levels, and published as full-text articles in indexed journals from the inception of the studies, were included. Inclusion criteria comprised articles published in English with accessible abstracts, without restrictions based on publication dates. Studies utilizing plant extracts for bioassay on chick ileum were not considered for inclusion. Review articles, book chapters, conference abstracts, short communications, correspondences, expert opinions, and unpublished reports were excluded from this systematic review.
Data Extraction and Analysis
The data extracted were independently scrutinized from the selected studies by evaluating the abstracts. After excluding ineligible studies due to duplicates and adherence to exclusion criteria, the full text of the remaining articles underwent eligibility assessment. To mitigate potential bias, the authors meticulously reviewed and deliberated on all selected documents, references, and excluded articles. Any discrepancies were resolved through consensus among authors. As a final step, additional studies possibly overlooked were manually searched for in the reference lists of the included papers.
For every study included in this investigation, the following data points were collected: author name, country of origin, publication year/period, type of chicken tissue used, name and composition of the solution, chemicals utilized, results, outcomes, and conclusions. Additionally, details regarding the experimental conditions employed in the bioassays conducted on chick ileum in these studies were compiled, including author name, three- or four-point assay, tissue length, temperature, relaxation time, dose cycle, contact time, washing interval, organ bath volume, tension weight, aeration, liver type, magnification, and other pertinent information.
Data Synthesis
The quality of the study and relevant characteristics were documented and described narratively.
Results
Literature Search
According to the eligibility criteria, 3,261 studies were initially identified from three databases during the initial search. After removing duplicate studies (1,286), a total of 1,975 articles were initially available for review. Of these, 865 articles were selected for assessment following the application of inclusion and exclusion criteria. Ultimately, 11 articles were included in this systematic review, excluding papers based on title/abstract and full text. The PRISMA flow diagram shows the process of study selection (Figure 1).
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Flowchart of Study Selection.
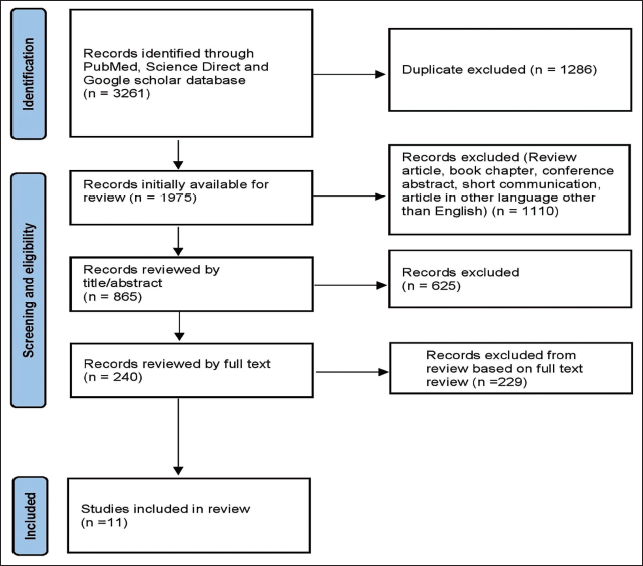
Data Retrieved
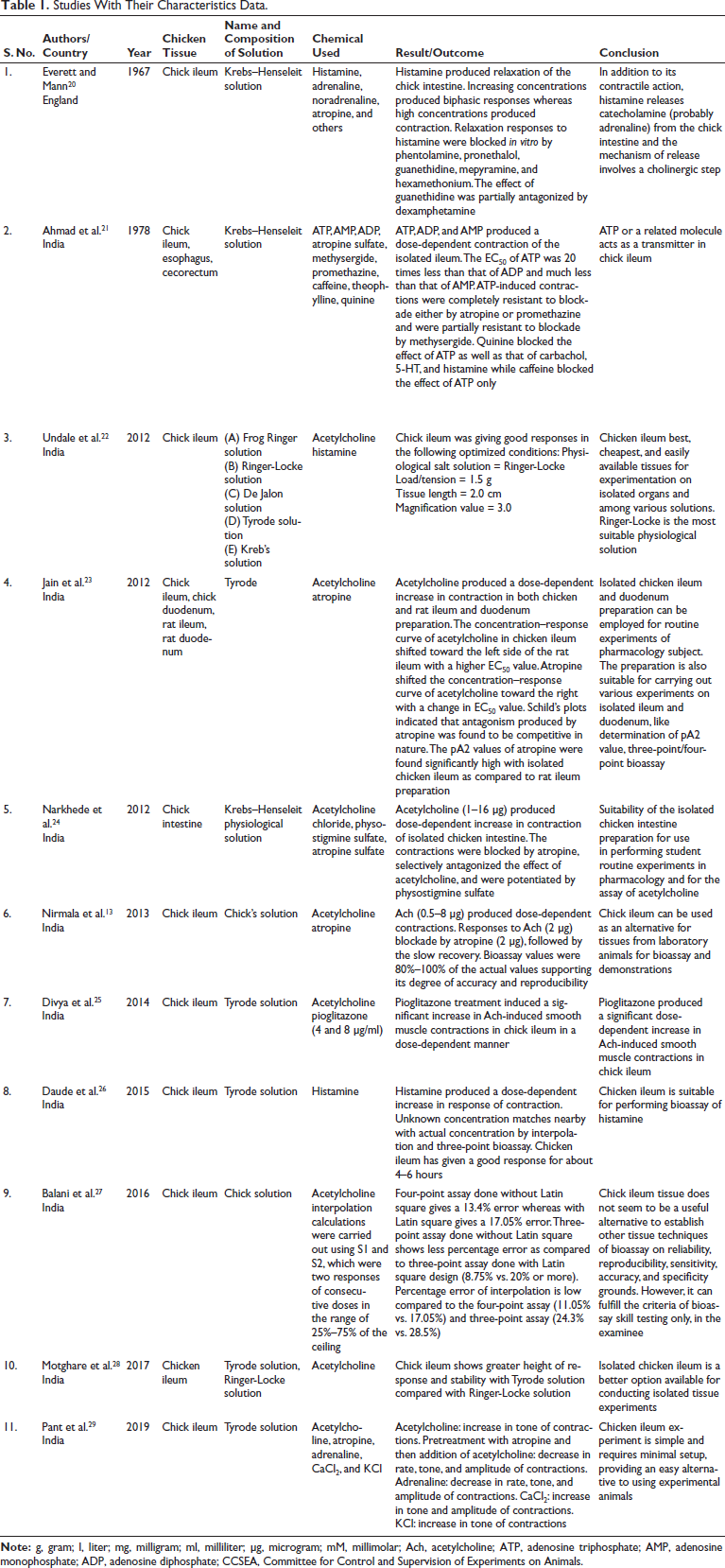
The results obtained in the reviewed studies are presented in Tables 1 and 2.13, 20–29
Studies With Their Characteristics Data.
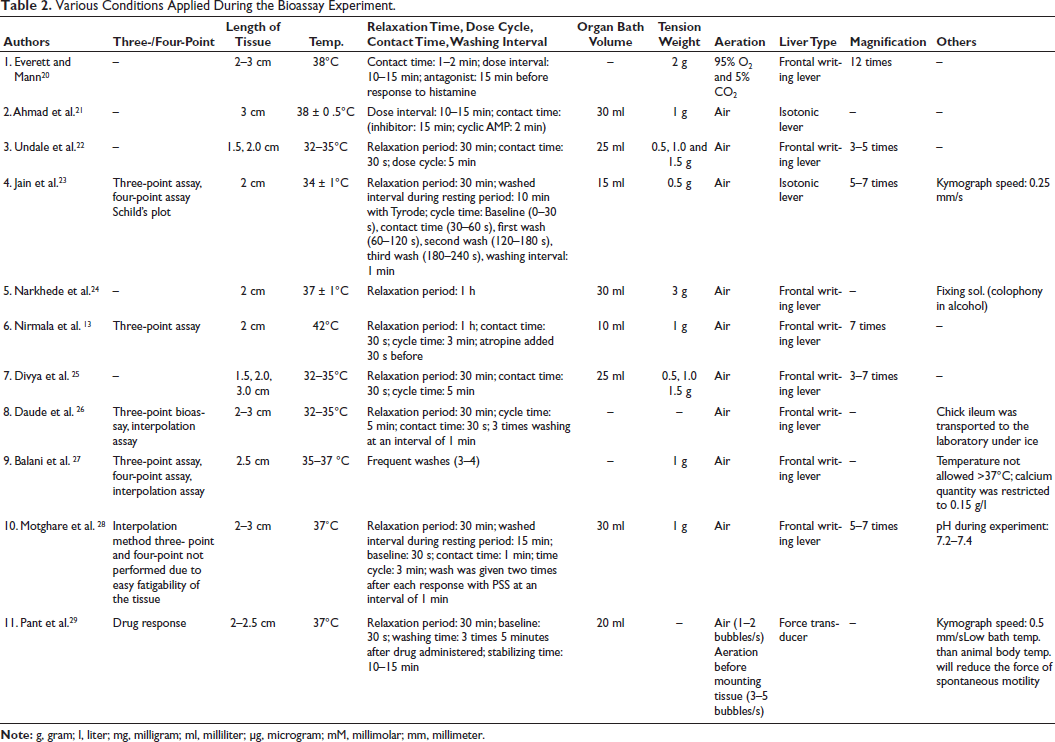
Various Conditions Applied During the Bioassay Experiment.
Acetylcholine consistently caused dose-dependent increases in contractions across various studies, with responses modified by atropine and other agents. Histamine-induced biphasic responses in chick ileum, with low doses causing relaxation and high doses causing contraction. Adenosine triphosphate (ATP), adenosine diphosphate (ADP), and adenosine monophosphate (AMP) induced dose-dependent contractions, unaffected by certain blockers but inhibited by quinine. Pioglitazone enhanced acetylcholine-induced contractions, indicating its effect on smooth muscle responses (Table 1).
Authors and Country
The studies span several decades (1967–2019) and are predominantly conducted in India, with one early study from England (Table 1).
Chick Tissue
All studies utilized chick ileum, with some extending to other parts of the gastrointestinal tract, such as the esophagus and cecorectum 21 and chick duodenum (Table 1). 23
Physiological Salt Solution (PSS)
Various PSSs have been employed across the reviewed studies, including Krebs–Henseleit, Frog Ringer, Ringer-Locke, De Jalon, and Tyrode solutions. Tyrode solution stands out as the predominant choice, appearing in over half of the studies (6 out of 11). The Krebs–Henseleit solution also features significantly, being the selected PSS in roughly one-third of the studies (4 out of 11). The chick solution has been used in a minority of the studies (2 out of 11), while others such as Frog Ringer, Ringer-Locke, and De Jalon are utilized to a lesser extent, each appearing in one or two studies, respectively. Several studies (Motghare et al. 28 and Undale et al. 22) compared multiple PSSs for bioassay. In all the studies where it was utilized as the PSS, a consistent formulation of Tyrode, Krebs–Henseleit, chick solution, Frog Ringer, Ringer-Locke, and De Jalon was employed (Table 1).
Chemicals Used
The chemicals tested encompass a broad spectrum of pharmacological agents, including neurotransmitters (acetylcholine, histamine, adrenaline, noradrenaline), antagonists (atropine, mepyramine, phentolamine, pronethalol), and other compounds (ATP, ADP, AMP, pioglitazone). Acetylcholine featured in 8 of the 11 experiments, with atropine in 6, histamine in 3, and adrenaline in 2, highlighting their prevalence in the studies. ATP, AMP, and ADP, along with a diverse range of other chemicals (reserpine, guanethidine sulfate, phentolamine, mepyramine maleate, hexamethonium bromide, pronethalol hydrochloride, beta hydroxyphenethyl guanidine sulfate, methysergide, promethazine, caffeine, theophylline, cyclic AMP, quinine, physostigmine sulfate, pioglitazone, noradrenaline, CaCl2, and KCl), were each investigated in one of the eleven studies (Table 1).
Assay Methods
The articles reviewed do not uniformly report on assay methods. Nonetheless, of those that do, four studies employed three-point assays, two utilized four-point assays, and three conducted interpolation methods. Additionally, one study included the generation of a Schild’s plot as part of its analysis. Several researchers, including Jain et al., 23 Balani et al., 27 and Daude et al.26, employed a combination of assay techniques, specifically three-point, four-point, and interpolation methods, in their studies (Table 2).
Tissue Length
Tissue lengths ranged from 1.5 to 3 cm. A tissue length of 2 cm was the standard measurement most frequently utilized in the studies. Undale et al. 7 and Divya et al. 25 conducted their bioassays using a range of tissue lengths, specifically 1.5, 2, and 2.5 cm (Table 2).
Temperature
Temperatures were maintained between 32°C and 42°C (minimum: 32°C, maximum: 42°C, range: 10°C), close to physiological conditions to simulate in vivo environments accurately. Three studies mentioned setting the temperature between 32°C and 35°C, and three others mentioned a temperature of 37°C for the bioassay. Only one study experimented at 42°C (Table 2).
Relaxation Time, Dose Cycle, and Contact Time
Relaxation periods varied from 30 minutes to 1 hour across the studies, indicating a standard approach to allowing tissues to stabilize. A few studies mentioned that the washing interval during the relaxation phase ranged from 10 to 15 minutes. Wherever mentioned, the cycle time ranged from 3 to 5 minutes. All studies did not mention the baseline duration, but wherever it was mentioned, the baseline lasted for 30 seconds. The contact time varied from 30 seconds to 15 minutes. Depending on the specific agent used (antagonist/inhibitor—15 minutes). Most studies employed a contact time of 30 seconds. The washing interval after contact time was 1 minute, and the frequency of wash ranged from 2 to 4 times. The dose interval mentioned in studies was around 10–15 minutes (Table 2).
Organ Bath Volume
Organ bath volumes ranged from 10 to 30 ml (minimum: 10 ml, maximum: 30 ml, range: 20 ml). An organ bath of 30 ml was the most commonly set volume level (Table 2).
Tension Weight
Tension weights varied from 0.5 to 3 g (minimum: 0.5 g, maximum: 3 g, range: 2.5 g). Most studies tried experimenting using weights of 0.5–1 g (Table 2).
Aeration
Most experiments utilized air for aeration, with a specific mixture of 95% O2 and 5% CO2 in one study (Table 2).
Lever Type and Magnification
The frontal writing lever was commonly employed for recording, with magnification levels used to enhance visualization ranging from 3 to 12 times (minimum: 3 times, maximum: 12 times, range: 9 times). A magnification of 5–7 was most frequently mentioned across studies. Only 1 study mentioned a magnification of 12 (Table 2).
Additional Conditions
Two studies specified the transport of chick ileum from the bitcher shop to the lab in 0–4°C of PSS (Narkhede et al.24) and ice (Daude et al.26), respectively. Additional experimental conditions such as kymograph speed (0.25–0.55 mm/s), aeration speed (1–2 bubbles/s), lab temperature (20–25oC), fixing solution (colophony in alcohol), and pH (7.2–7.4) were specified in a few studies (Table 2).
Discussion
Exploring alternative tissues for biological assays, such as poultry, circumvents sacrificing traditional lab animals. Isolated chicken ileum offers a humane, cost-effective, and efficient research option, reducing animal use in educational experiments. 26 However, the variability in gut tissue responses to different agents necessitates optimizing experimental conditions for accurate bioassay execution. Studies have shown that specific conditions, including a tissue length of 2.0 cm, a tension of 1.5 g, a magnification value of 3.0, and the use of Ringer-Locke solution, can yield results comparable to those from isolated rat ileum. 22 Other studies have identified Tyrode as a superior alternative in enhancing tissue stability, attributed to the presence of MgCl2. 28 It is clear that standardized parameters are missing, which are crucial for obtaining accurate, straightforward, and consistently reproducible results in chick ileum bioassays. Literature on chick ileum bioassay methods is limited, hindering definitive conclusions. However, this systematic review aggregates available data to aid future researchers, offering informed insights rather than explicit recommendations, enabling navigation of chick ileum bioassays with awareness of capabilities and limitations.
The timeframe of reviewed publications in our systematic review (1967–2019) reflects a longstanding interest in chick ileum pharmacological properties, particularly in India, indicating its significant applicability in research settings.
The choice of a suitable PSS for bioassay is crucial. The development of a specific chick solution for chick ileum bioassays, as opposed to traditionally using Tyrode solution for gut tissue assays, may be attributed to the need for a more tailored physiological environment that closely mimics the chick ileum’s natural conditions. In the studies reviewed, PSSs like Krebs–Henseleit, Frog Ringer, Ringer-Locke, De Jalon, chick solution, and Tyrode have all been experimented with bioassays on chick ileum. Initial experiments faced inconsistent responses with various PSSs. A study by Undale et al. indicated that the Ringer-Locke solution is the most suitable PSS for chick ileum bioassays. 7 It was found to provide optimized conditions that correlated well with results from isolated rat ileum when using acetylcholine and histamine, suggesting that Ringer-Locke is a preferred choice over other solutions for consistent bioassay results. However, Motghare et al. 28 noted that the Tyrode solution yields greater response height and stability than the Ringer-Locke solution, potentially due to Tyrode’s MgCl2 content, which enhances tissue stability. In the study by Daude et al., Tyrode was also selected for experimentation because initial trials with different PSSs led to non-uniform and slow histamine responses in chicken ileum tissues. Switching to the Tyrode solution improved the uniformity and speed of histamine responses, suggesting that this solution provides better results for bioassay experiments using isolated chicken ileum. In contrast, Nirmala et al. reported that using chick solution at 42°C instead of Tyrode or chick solution at 37°C improved tissue response uniformity and relaxation times for bioassays. 13
Temperature plays a crucial role in achieving optimal bioassay responses. However, reported chick ileum studies have shown different temperature requirements, possibly based on factors such as receptor sensitivity and enzyme activity. Nirmala et al. adapted their experiments to use chick solution at 42°C after initial attempts at 37°C, which resulted in uneven acetylcholine responses. 13 In another study, to reduce inherent activity, a strategy was employed that involved maintaining the temperature at 37°C and adjusting the calcium concentration to 0.15 g/l. 28
The chick ileum’s sensitivity to acetylcholine, histamine, and other agents like pioglitazone has been a focal point in these studies, highlighting its utility in pharmacological research. The study by Everett and Mann evaluates how substances like reserpine and noradrenaline, alongside various antagonists, influence histamine action, aiming to determine optimal bioassay conditions. This emphasizes the significance of understanding interactions between histamine, catecholamines, and pharmacological agents for effective and precise bioassay results on chick ileum.20, 26 On the other hand, the study by Divya et al. contributes to the pharmacological understanding of pioglitazone’s effects beyond its known insulin-sensitizing actions, particularly highlighting its potential to increase calcium-mediated smooth muscle contractions in the chick ileum. 25 This could be relevant for further investigations into the drug’s comprehensive physiological effects and mechanisms of action.
Despite potential advantages, concerns over the tissue’s rapid fatigability and the perceived lower precision of interpolation methods compared to three-point and four-point assays have been noted. 28 The paper by Balani et al. 27 evaluates chick ileum’s bioassay in pharmacology practical exams, comparing interpolation and three-point or four-point assays. They find interpolation often more accurate, suggesting a combined mean error approach from two methods can enhance result accuracy. Despite this, the tissue’s drawbacks, like sensitivity and easy fatigability, limit its reliability. Consequently, they imply that chick ileum is good for skill demonstration but not a reliable substitute for established bioassay techniques when accurate result measurement is needed. 27
The use of carbogen (a mixture of carbon dioxide and oxygen) versus air in bioassays can significantly affect tissue sample responses, with carbogen favoring physiological condition simulation and enhancing tissue responsiveness. However, access to carbogen has been a challenge for some researchers.
Based on the analysis of the provided studies investigating the use of chick ileum for bioassay, it can be concluded that this model offers advantages and limitations. One clear benefit is the ethical aspect; utilizing chick ileum, which can be sourced from slaughterhouses, may reduce the necessity for live animal experimentation, thus aligning with the ethical principles of reducing animal usage in scientific research. This supports animal welfare and offers a cost-effective alternative for laboratories.
From a scientific standpoint, chick ileum has shown a reliable response to various neurotransmitters and drugs, demonstrating dose-dependent contractions with compounds like Ach, which suggests its potential as an effective tool for studying drug interactions and receptor activity. The ability of chick ileum to resist blockade by specific antagonists, like atropine, and its response to purinergic agents provide a unique avenue for exploring non-cholinergic pathways.
However, there are constraints to the chick ileum model. The specificity of responses can be challenging, as evidenced by the partial blockade by agents such as methysergide and the potential for non-specific blockade by quinine. Furthermore, the model may not fully replicate the complexity of mammalian gut physiology, possibly limiting the extrapolation of results to human medical applications. Additionally, maintaining consistent experimental conditions, such as temperature and calcium concentrations, is crucial to minimize spontaneous activity and enhance the reproducibility of results.
Conclusion
In conclusion, while the chick ileum preparation is a valuable model for bioassay due to its ethical and practical benefits, careful consideration must be given to its pharmacological limitations, potential differences in response compared to mammalian systems, and the need for standardization of optimal conditions for accurate and reproducible tissue response.
Footnotes
Abbreviations
Ach: Acetylcholine; ATP: Adenosine triphosphate; AMP: Adenosine monophosphate; ADP: Adenosine diphosphate; PSS: Physiological salt solution; g: Gram; l: Liter; mg: Milligram; ml: Milliliter.
Acknowledgments
The authors have nothing to report.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The Ethical approval has been obtained for the study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
