Abstract
Background
Periodontal disease is a chronic inflammatory disease that activates the innate immune response. Activation of the TREM-1 signaling pathway promotes the synthesis of inflammatory cytokines like interleukin-1 beta (IL-1β), tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6) and interleukin-8 (IL-8). Hyaluronic acid (HA) can be used to treat periodontitis as an adjunct to mechanical debridement due to its healing properties.
Objectives
To evaluate and compare soluble triggering receptor expressed on myeloid cells (sTREM-1) and IL-1β in periodontitis patients with and without diabetes mellitus before and after nonsurgical periodontal therapy using conventional root planing and adjuncts like HA (Gengigel®) and diode LASER (940 nm).
Materials and Methods
Chronic periodontitis patients were allocated to groups based on with and without diabetes mellitus (groups I and II), respectively. Assessment of periodontal parameters: periodontal probing depth (PPD), clinical attachment loss (CAL), plaque index (PI), gingival bleeding index (GBI), and periodontal inflammatory surface area (PISA) index were done. About 5 mL of unstimulated whole saliva was collected from every patient at baseline and 1 month. Patients were subgrouped based on the treatment protocol (subgroup A: conventional scaling and root planing (SRP), subgroup B: SRP + HA (0.8% Gengigel®), and subgroup C: SRP + 940 nm diode LASER). The primary and secondary outcomes before and after therapy were determined.
Results
Intragroup comparison of group I showed the statistical significance of parameters in subgroups B and C. Intragroup comparison of group II in all three subgroups. Intergroup comparison of groups II and III showed the statistical significance of all periodontal parameters and salivary biomarker levels in A–C subgroups.
Conclusion
Therefore, within the study’s limitations, the use of adjuncts to nonsurgical periodontal therapy in chronic periodontitis had an additional improvement in all clinical parameters and salivary biomarkers over conventional root planing.
Introduction
Periodontal disease is a persistent inflammatory condition that arises from an imbalanced microbial community within the gingival sulcus. This microbial imbalance triggers the innate immune response through an array of receptors found on host cells, setting off a cascade of intra and intercellular signaling events that ultimately activate the host’s immune-inflammatory response.1–3
Soluble triggering receptor expressed on myeloid cells (sTREM-1), a novel biomarker identified in systemic inflammatory conditions, is a receptor of the immune-inflammatory process. sTREM-1 is expressed on various immune and nonimmune cells. Activation of the sTREM-1/DAP12 (transmembrane adapter) signaling pathway plays a pivotal role in stimulating the production of inflammatory cytokines such as interleukin-1 beta (IL-1β), tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6) and interleukin-8 (IL-8). 4
Periodontitis, recognized as a complex inflammatory condition, was identified as the sixth complication of diabetes mellitus. Multiple pathomechanistic connections have been suggested to reinforce the correlation between diabetes and periodontitis. 5
Non-surgical periodontal therapy aims to attain optimal gingival health by comprehensively eliminating factors accountable for gingival inflammation within the oral milieu, specifically plaque, calculus, and endotoxins. Various modalities such as manual instruments, ultrasonic devices, and light amplification by stimulated emission of radiation (LASER) curettage exhibit significant efficacy in substantially diminishing the subgingival microbial population. 6
Hyaluronic acid (HA) actively participates in tissue repair and wound healing, making it a valuable topical agent with anti-inflammatory and antiedematous properties. Anti-inflammatory properties can be attributed to its ability to act as an exogenous hyaluronan scavenger, effectively removing prostaglandins, metalloproteinases, and other bioactive molecules. 7
LASER technology exhibits exceptional tissue ablation and potent bactericidal properties, rendering it a highly promising novel modality for nonsurgical periodontal interventions. 8
This study compares and estimates salivary sTREM-1 and IL-1β in periodontitis patients with and without diabetes mellitus, before and after nonsurgical periodontal therapy using conventional root planing and adjuncts like HA (Gengigel®) and diode LASER (940 nm).
Materials and Methods
Study Population
Subjects were recruited from the Department of Periodontology at Sri Ramachandra Dental College. Patients were selected based on specific inclusion and exclusion criteria after obtaining informed consent. Subjects who were diagnosed with severe periodontitis according to CDC 2012. Probing depth >5 mm, at least one tooth in all four quadrants were included, and patients under any antimicrobial therapy within the last 3 months, smokers, tobacco chewers, pregnant, lactating patients, and immunocompromised patients were excluded from this study. Ethical approval was obtained from the Institutional Ethical Committee, SRIHER (Sri Ramachandra Institute of Higher Education and Research) (Ethics No: CSP/23/JUL/131/608).
Sample Size Calculation
Sample size calculation was done based on the study by Sigrun Eric et al. A sample size of 9 in each group (18) was determined with 80% power.
Methodology
Patients with chronic periodontitis were allotted to groups based on with and without diabetes mellitus (group I: chronic periodontitis without diabetes and group II: chronic periodontitis with diabetes). Biochemical parameters such as blood glucose level—fasting blood sugar (FBS), postprandial blood sugar (PPBS), and glycated hemoglobin (HbA1c) were investigated in patients with diabetes. At baseline, an assessment of periodontal parameters: periodontal probing depth (PPD), clinical attachment loss (CAL), plaque index (PI), gingival bleeding index (GBI), and periodontal inflammatory surface area (PISA) index was made. After periodontal diagnosis, a sterile Eppendorf container was used to collect 5 mL of unstimulated whole saliva from every patient at baseline and 1 month by spitting method. The subjects were instructed to pool the saliva on the floor of the mouth and spit every 60 s. The collected saliva samples were centrifuged at 4,200 rpm for 15 min.
Random allocation to interventions was done using a computer-generated table of numbers. The randomization was received by the fourth author, and screening and selection were performed by the corresponding author. The operator was blinded regarding the allocation of the subjects into the treatment group. Patients were subgrouped based on the treatment protocol (subgroup A: conventional scaling and root planing (SRP), subgroup B: SRP + HA (0.8% Gengigel®), and subgroup C: SRP + 940 nm diode LASER). All patients underwent complete scaling, and standardized oral hygiene instructions, namely, tooth brushing with modified Bass technique and flossing, were given. Patients were advised to rinse their mouth twice daily for 14 days using 10 mL of 0.2% chlorhexidine gluconate mouthwash. Group A patients underwent root surface debridement at sites with PPD ≥ 5 mm using an ultrasonic scaler and Gracey curettes (Hu Friedy, Chicago, USA) (Figure 1A–F). Group B patients, after thorough SRP, were administered 1 mL of 0.8% HA using Gengigel® Prof syringe subgingivally at sites with PPD ≥ 5 mm, followed by placement of COE-PAK. Patients were recalled after 1 week for removal of COE-PAK (Figure 2A–F). For group C patients, after thorough scaling and root planing, a 940 nm diode LASER at a power of 1.5 W in contact mode was used to remove the inner epithelial wall of the periodontal pocket. The uninitiated laser tip was inserted into the depth of the pocket and placed at an angle against the root surface. Then, the laser was activated, and slow movement was made in the apical direction until the base of the pocket was reached. The fiber was inspected frequently, and any accumulated tissue and debris were wiped off. Following this, the site was irrigated with normal saline. All patients were reviewed after 1 month (Figure 3A–D). Quantitative determination of salivary sTREM-1 and IL-1β levels were according to the manufacturer’s instructions done using GENLISA™ enzyme-linked immunosorbent assay (ELISA), Krishgen Biosystems. The primary outcomes (changes in salivary sTREM-1, IL-1β levels, and PISA and secondary outcomes (PI, GBI, probing depth, and CAL) before and after therapy were determined.
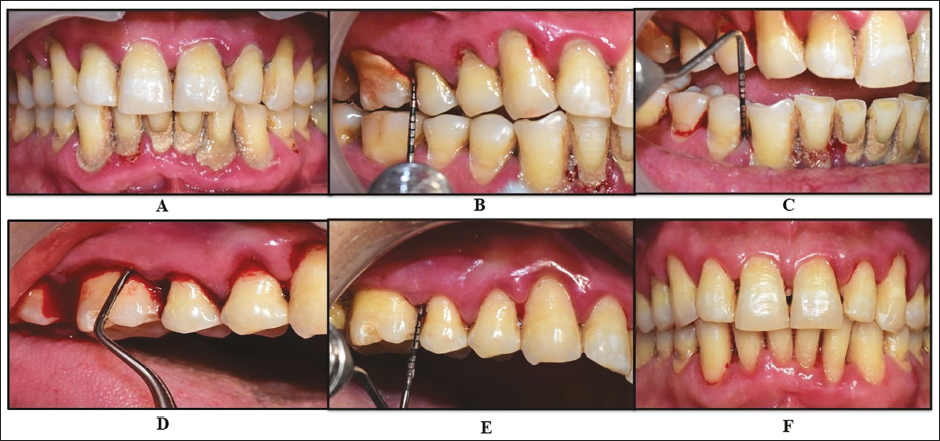
Conventional Scaling and Root Planing. A–C: Preoperative Periodontal Probing Depth (PPD) ≥5 mm; D: Conventional Scaling and Root Planing using Gracey Curettes; E and F: Postoperative Parameters.
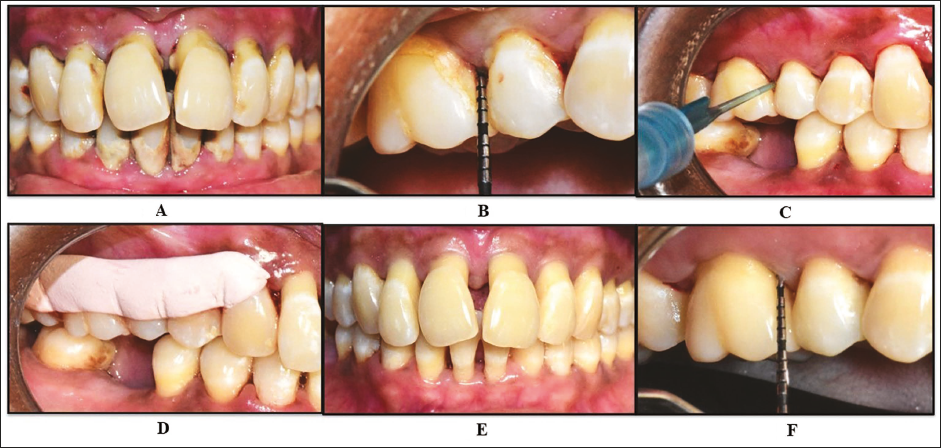
Scaling and Root Planing (SRP) with Hyaluronic Acid (0.8% Gengigel®). A and B: Preoperative Periodontal Probing Depth (PPD) ≥5 mm; C: SRP Followed by Subgingival Administration of 0.8% Gengigel®; D: Placement of COE-PAK; E and F: Postoperative Parameters.
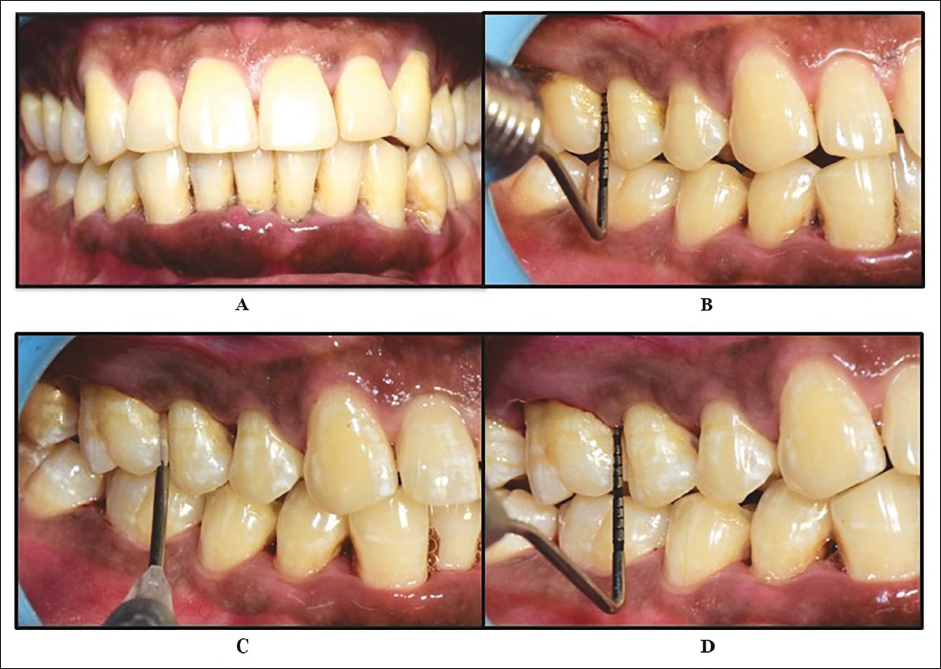
Scaling and Root Planing (SRP) + 940 nm Diode LASER. A and B: Preoperative Periodontal Probing Depth (PPD) ≥5 mm; C: SRP Followed by Application of 940 nm Diode LASER at 1.5 W; D: Postoperative Parameters.
Statistical Analysis
Descriptive and inferential statistics were analyzed by IBM SPSS version 20.0 (IBM Corp. Released 2011. IBM SPSS Statistics for Windows, Version 20.0. Armonk, NY: IBM Corp). All of the data were normally distributed, as shown by their mean and SD range distribution. Mean and SD were used to summarize the quantitative data. Independent samples t-test was used for intergroup comparison of values between groups I and II for each subgroups A–C. The paired t-test was used to compare changes from pre to postoperative intragroup values. Bar graphs were used to represent all the comparisons as mentioned earlier. A “p” value of <.05 was considered a statistically significant difference.
Results
Intragroup Comparison of Group I
sTREM-1, IL-1β, PI, probing pocket depth, and PISA index were significant in the I–B subgroup (SRP + 0.8% Gengigel®) and subgroup I-C (SRP + 940 nm diode LASER) showed statistical significance in GBI, probing pocket depth and PISA (Table 1).
Intragroup Comparison of Group I.
Primary and Secondary Outcome Variables of Group I
Considering the p value less than .05 as statistical significance, it was found that primary and secondary outcome variables were not significant in subgroup I–A. In subgroup I–B, primary outcomes such as sTREM-1 (p value = .040), IL-1β (p value = .013), and PISA index (p value = .040) were found to be statistically significant. Secondary outcomes such as the PI (p value = .035), and probing pocket depth (p value = .040) were statistically significant. In subgroup I–C, primary outcome variables such as the PISA index (p value = .019) were significant, and secondary outcomes such as probing pocket depth (p value = .015) and GBI (p value = .040) were statistically significant.
Intragroup Comparison of Group II
Considering a p value of less than .05 as statistical significance, it was found that the PI, GBI, probing pocket depth, CAL, and PISA index were significant in the conventional root planing II-A subgroup. sTREM-1, IL-1β, PI, GBI, CAL, and PISA index in II-B subgroup (SRP + 0.8% Gengigel®). Group II-C (SRP + 940 nm diode LASER) showed statistical significance in sTREM-1, IL-1β, PI, GBI, probing pocket depth, CAL, and PISA (Table 2).
Intragroup Comparison of Group II.
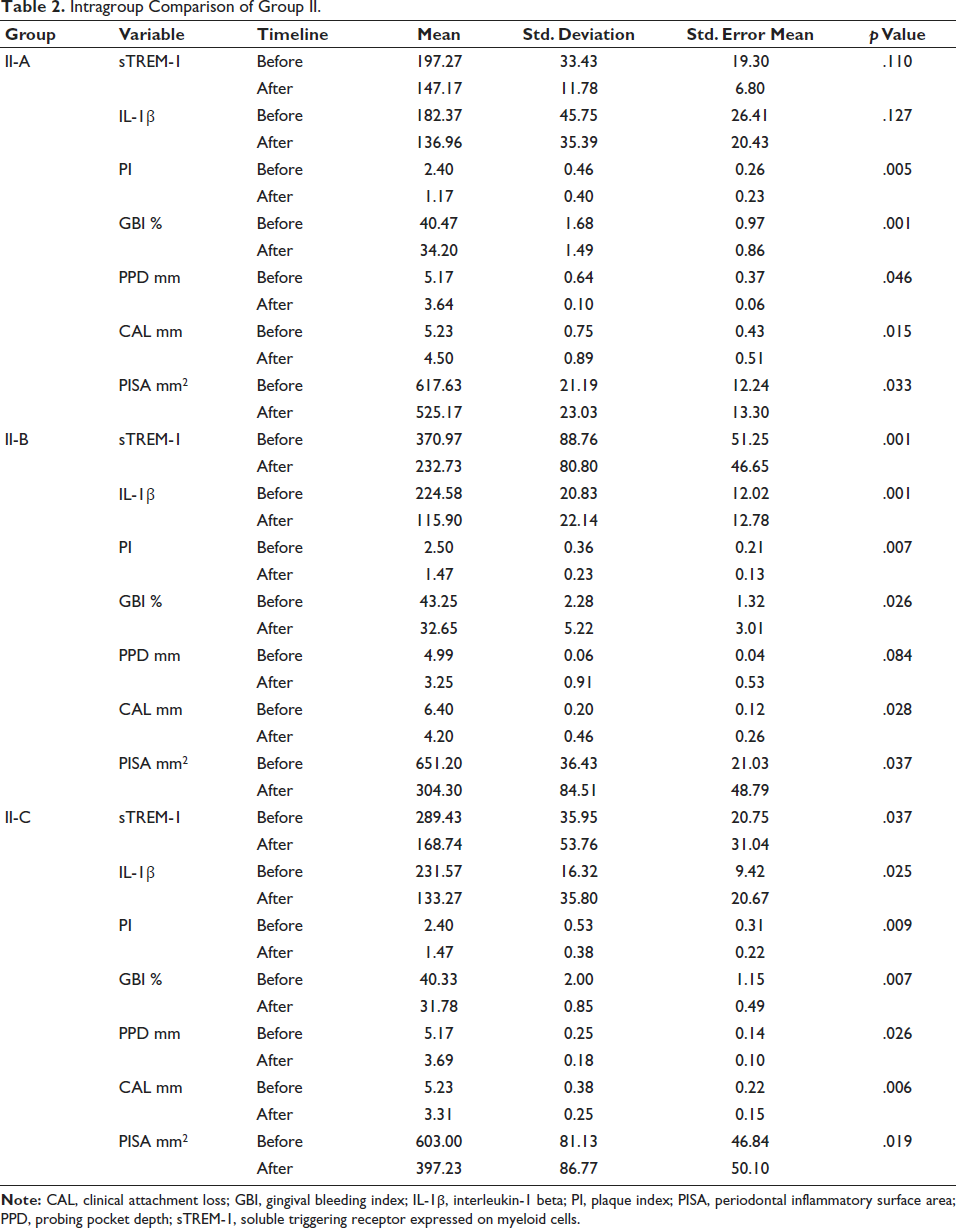
Primary and Secondary Outcome Variables of Group II
Considering p value less than .05 as statistical significance, it was found that the primary outcome: PISA index (p value = .033), was statistically significant, and sTREM-1, IL-1β was not statistically significant in subgroup II-A. In the secondary outcome of subgroup II-A, the PI (p value = .005), GBI (p value = .001), probing pocket depth (p value = .046), and CAL (p value = .015) were statistically significant. The primary outcome variables of subgroup II-B, such as PISA index (p value = .037) were statistically significant. In the secondary outcome variables: PI (p value = .007), GBI (p value = .026), and GBI (p value = .028) were statistically significant, and probing pocket depth (p value = .084) was not statistically significant. The primary outcome variables of subgroup II-C were not significant. In the secondary outcome variables: PI (p value = .009), GBI (p value = .007), probing pocket depth (p value = .26), and CAL (p value = .006) subgroups were statistically significant.
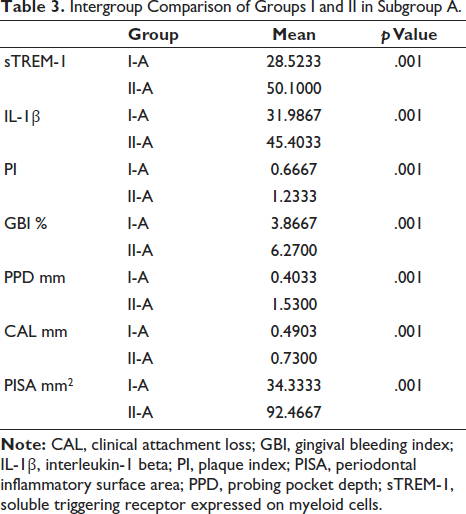
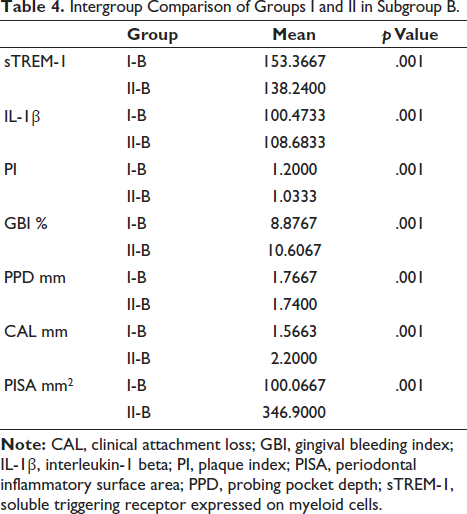
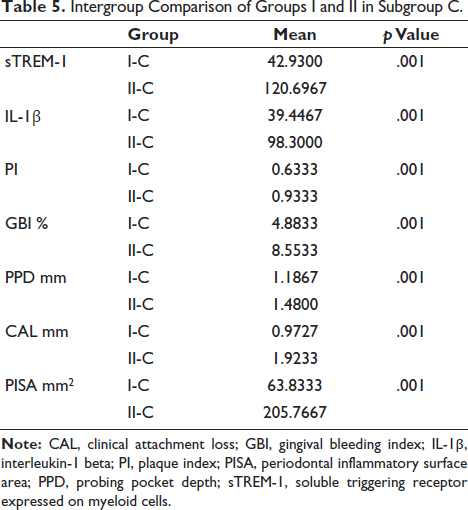
Intergroup Comparison for Groups I and II
An independent samples t-test was used to compare the two groups in terms of intergroups. Considering p value less than .05 as statistical significance, it was found that there is a significant difference before and after nonsurgical periodontal therapy between groups I and II (A–C) in all periodontal parameters and salivary biomarker levels. In the intergroup comparisons for groups I and II in subgroup A, sTREM-1, IL-1β, PISA, PI, GBI, probing pocket depth, CAL was statistically significant with a p value = .001 (Table 3). From the intergroup comparisons for groups I and II in subgroup B, sTREM-1, IL-1β, PISA, PI, GBI, probing pocket depth, CAL was statistically significant with a p value = .001 (Table 4). In the intergroup comparisons for groups I and II in subgroup C, sTREM-1, IL-1β, PISA, PI, GBI, probing pocket depth, CAL was found to be statistically significant with a p value = .001 (Table 5).
Intergroup Comparison of Groups I and II in Subgroup A.
Intergroup Comparison of Groups I and II in Subgroup B.
Intergroup Comparison of Groups I and II in Subgroup C.
Discussion
The primary objective of nonsurgical therapy is to target and remove living bacteria residing within microbial biofilms, reinstate a harmonious relationship, mitigate inflammation within the periodontium, and eliminate calcified biofilm microorganisms from tooth surfaces and surrounding soft tissues. Achieving absolute eradication of these pathogenic microorganisms may be an ambitious goal. However, striving for significantly reducing inflammation within the periodontium through a reduced bacterial presence can result in positive clinical improvements. 9 Various adjuncts to SRP have been proposed to minimize the necessity for invasive surgical procedures that demand technical proficiency. These adjunctive therapies encompass the utilization of antibiotics or anti-inflammatory agents, which have demonstrated notable improvements in reducing periodontal probing pocket depth (PPD) and gaining clinical attachment level (CAL). Systemic administration necessitates the use of elevated drug doses to attain effective concentrations at the target site, demands patient adherence to prescribed administration regimens, and may be accompanied by the risk of adverse effects. Consequently, several localized treatment modalities, including gels, fibers, or chips loaded with diverse bioactive compounds or pharmaceutical agents, have been devised and assessed in clinical settings. The primary advantages associated with these treatments are their ability to deliver active agents precisely to the site of the lesions and the concomitant reduction in the risk of adverse effects. 10
This study aimed to compare and evaluate the changes in salivary sTREM-1 and IL-1β levels and periodontal parameters before and after conventional scaling and root planing, and the use of adjuncts to nonsurgical periodontal therapy using HA (0.8% Gengigel®) and 940 nm diode LASER in chronic periodontitis patients with and without type II diabetes mellitus. The study observations revealed that gold standard root planing showed a reduction in periodontal parameters, but a more significant reduction was seen in the groups that used adjuncts.
In the present study, chronic periodontitis with and without diabetes showed the statistical significance of all clinical parameters such as PPD, CAL, GBI%, PI, and PISA with a p value of <.001. Various studies from the literature support our findings; a systematic review and meta-analysis by Baima et al., in 2021 amounted to a 15.50% reduction of PD ≥ 5 mm after nonsurgical periodontal therapy (p < .001). 11
A systematic review by Eliezer et al., in 2019, showed improvement in periodontal parameters following the use of HA during surgical and nonsurgical periodontal therapy. 12 These findings support the results of our study, which showed statistically significant improvement in all clinical periodontal parameters.
The use of diode LASER as an adjunct to root planing showed a significant reduction of clinical parameters such as GBI%, PPD, and PISA index in the chronic periodontitis without diabetes mellitus, whereas chronic periodontitis with diabetes mellitus showed the statistical significance of all clinical parameters. Coluzzi et al., conducted a systematic review and evaluated different LASER wavelengths as an adjunctive role in initial nonsurgical periodontal therapy, and the results showed improvement in clinical parameters compared to the group treated with only conventional methods. 13
A novel biomarker sTREM-1 is a cell surface receptor present on polymorphonuclear neutrophils, monocytes, and macrophages. Numerous authors like Su et al., Pregernig et al., Chang et al., and de Oliveira et al. have extensively studied the proinflammatory role of sTREM-1 in various systemic disease conditions contributing to disease progression. In the present study, sTREM-1 was found to be remarkably increased in chronic periodontitis patients with diabetes mellitus than those without diabetes mellitus. Providing nonsurgical periodontal therapy showed a significant reduction in sTREM-1 levels in both the groups and the subgroups with a p value <.001.14–18
IL-1β is a well-established biomarker in systemic diseases as well as in periodontal disease. A systematic review and meta-analysis by Arroyo et al. concluded that the IL-1β biomarker has the highest differential expression and the greatest potential for clinical applicability to differentiate periodontitis from gingivitis and periodontitis from healthy status. 19 The findings of our study also correlate with the literature, in which IL-1β was increased in systemic and periodontal disease, and the application of nonsurgical periodontal therapy was beneficial in reducing the inflammatory burden.
Balaji et al. evaluated sTREM-1, IL-1β, and MMP-8 in patients with stage III/IV periodontitis and type II diabetes mellitus. The study unveiled sTREM-1, IL-1β, and MMP-8 as potential diagnostic and prognostic biomarkers for periodontitis, and it also described the impact of diabetes mellitus on periodontal tissue destruction. 20
This is the first study to compare and evaluate the changes in salivary sTREM-1 and IL-1β levels and periodontal parameters following nonsurgical periodontal therapy in chronic periodontitis patients with and without type II diabetes mellitus. The study was specifically designed to use HA and diode LASER as adjuncts, which provided advantageous results compared to conventional root planing. The limitations of this study are a smaller sample size and a short-term follow-up. Further research with a larger sample size and long-term follow-up at multiple intervals will shed more light on changes in clinical parameters and salivary biomarker levels.
Conclusion
Therefore, within the study’s limitations, it can be concluded that adjuncts to nonsurgical periodontal therapy in chronic periodontitis had an additional improvement in all clinical parameters and salivary biomarkers over conventional root planing. Reduced inflammatory burden and clinical parameters in periodontitis patients with diabetes mellitus suggest the importance of periodontal health on systemic diseases.
Footnotes
Abbreviations
Acknowledgments
The authors would like to thank the Central Laboratory of Sri Ramachandra University, Chennai, for assisting with the ELISA tests.
Ethical Approval and Informed Consent
The ethical clearance has been obtained from the Institutional Ethics Committee (EC/NEWIINST/2023/TN/0321)
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
