Abstract
Background
Bone marrow transplantation (BMT) is a crucial treatment for pediatric patients with various hematological, oncological, and genetic diseases. However, persistent thrombocytopenia (PT) post-transplantation poses a significant challenge, affecting recovery and prognosis.
Objectives
This study aimed to identify the clinical characteristics and risk factors associated with PT in pediatric patients post-BMT, specifically in Saudi Arabia.
Materials and Methods
A retrospective cohort study design was employed, involving pediatric patients who underwent BMT between January 2020 and January 2023. Clinical characteristics, laboratory results, treatment data, and outcomes were collected from electronic medical records. Logistic regression analyses were conducted to identify risk factors associated with PT.
Results
Among the 77 pediatric patients in the study, no significant differences were observed in demographic characteristics, transplant types, or treatment modalities between early-onset and late-onset thrombocytopenia groups. However, the duration of thrombocytopenia post-transplant differed significantly between the groups, with early-onset cases experiencing shorter durations. Infections and graft-versus-host disease were common post-transplant complications. Chemotherapy and antiviral therapy were frequently administered treatments. Pediatric patients undergoing BMT are susceptible to PT, with varying durations based on the onset timing.
Conclusion
The study emphasizes the importance of risk assessment and tailored management to mitigate thrombocytopenia in pediatric BMT patients. Further research is needed to elucidate the underlying mechanisms and optimize therapeutic interventions for thrombocytopenia in this population.
Introduction
Bone marrow transplantation (BMT) is a critical and often life-saving procedure for pediatric patients with a range of hematological, oncological, and genetic diseases.1–3 This procedure involves the infusion of stem cells to replenish the compromised bone marrow. 4 Despite advances in BMT techniques, persistent thrombocytopenia (PT) remains a critical barrier to recovery, particularly in pediatric populations where post-transplant care requires specialized management.3, 5 PT is a condition characterized by a prolonged decrease in the number of platelets in the blood, which is crucial for normal blood clotting and healing.6–8 PT post-allogeneic hematopoietic stem cell transplantation (allo-HSCT) represents a significant complication, affecting recovery and prognosis. 9 The pathogenesis of PT involves complex interactions between the bone marrow microenvironment, immune dysregulation, and potentially, genetic predispositions. Recent research highlights the importance of the bone marrow’s vascular niche in PT, where abnormalities in bone marrow endothelial cells (BMECs) and perivascular cells contribute to the condition. 10 A study by Kong et al. demonstrated significant decreases in BMECs and perivascular cells among PT patients compared to those with good graft function, indicating an impaired vascular microenvironment’s role in PT development. 11 Additionally, the study identified cytomegalovirus infection post-HSCT as an independent risk factor for PT, suggesting that infectious complications can exacerbate or trigger PT. Furthermore, addressing PT involves not just identifying risk factors but also exploring therapeutic interventions. The use of thrombopoietin receptor agonists, such as eltrombopag, has been explored for treating severe refractory thrombocytopenia post-allo-HSCT. 12 Citta et al. reported successful treatment of severe refractory thrombocytopenia in a pediatric patient with Philadelphia chromosome-positive acute lymphoblastic leukemia following allo-HSCT using avatrombopag, indicating the potential for targeted therapies in managing PT. 13
In the Saudi Arabian context, managing bone marrow failure syndrome (BMFS), a condition closely related to the challenges of post-transplant care, including PT, requires a nuanced understanding of both global research and local healthcare practices. Siddiqui and Pamarenko emphasize the diversity of BMFS etiologies and the crucial role of hematopoietic stem cell transplantation (HSCT) in treatment. Their review underscores the significance of donor type and graft success in outcomes, which are pertinent to understanding PT risk factors. 14 In Saudi Arabia, data on post-BMT complications, particularly thrombocytopenia, is scarce, underscoring the need for focused studies in this region to guide clinical practice. However, PT presents significant challenges in Saudi Arabia, where limited data and resources impact patient outcomes. Understanding PT’s clinical characteristics and risk factors among pediatric patients after BMT, particularly in specific regions like Saudi Arabia, is essential for improving patient outcomes and tailoring post-transplant care. This study aimed to identify the clinical characteristics and risk factors associated with PT in pediatric patients post-BMT, specifically in the context of Saudi Arabia. Additionally, we seek to evaluate the secondary outcomes, including the impact of thrombocytopenia on patients, while identifying modifiable risk factors to mitigate the incidence and severity of this complication post-BMT.
Materials and Methods
Study Design
The research methodology employed in this study was a retrospective cohort study design. This design facilitated the identification of pediatric patients who had undergone BMT and the subsequent examination of their clinical characteristics and risk factors for PT. The retrospective approach was chosen to analyze patient data over a specified timeframe, allowing for the identification of relevant risk factors and outcomes post-transplantation.
Study Setting and Duration
The study was conducted in a tertiary care children’s hospital with a specialized pediatric bone marrow transplant unit. The duration of the study spanned from January 2020 to January 2023, encompassing a total of 3 years. The hospital’s comprehensive electronic medical record system was used to collect the necessary data.
Study Subjects/Participants
The study subjects comprised all pediatric patients who had undergone BMT at the study setting between January 2020 and January 2023 and had been diagnosed with thrombocytopenia. Patients meeting the inclusion criteria were identified through a review of medical records.
Inclusion Criteria
Pediatric patients were included in the study if they met the following criteria:
Age under 18 years at the time of BMT. Underwent BMT at the study site between January 2020 and January 2023. Developed thrombocytopenia (defined as a platelet count <150,000/µL) during the post-transplant period. Had complete medical records available for analysis.
Exclusion Criteria
Patients were excluded if they:
Received BMT for nonmalignant conditions. Had pre-existing thrombocytopenia before the transplant. Died during hospitalization or shortly after the transplant.
Data Collection Tool
A structured questionnaire was utilized for data collection, encompassing variables related to patient demographics, medical history, transplantation details, clinical features, and treatment outcomes. The structured questionnaire used for data collection was reviewed for content validity by clinical experts in the field. The Cronbach’s alpha coefficient was calculated to assess the reliability of the questionnaire, achieving a value of 8.3, which indicated a high level of internal consistency.
Justification of Selecting Variables
The selected parameters aimed to investigate the clinical characteristics and risk factors associated with PT in pediatric patients post-BMT. These variables were deemed relevant to the research question and could provide valuable insights into developing and managing post-transplant thrombocytopenia.
Data Collection Procedures
Data were collected from the electronic medical records of eligible patients. The collected data included demographic information (age, sex, ethnicity), clinical characteristics (diagnosis, type of transplant, donor type, engraftment status), laboratory results (platelet count, complete blood count, coagulation studies), treatment data (use of anticoagulants, platelet transfusions, other medications), and outcome data (duration of thrombocytopenia, need for readmission).
Diagnostic Criteria for Thrombocytopenia
Thrombocytopenia was defined as a platelet count below the normal reference range, specifically less than 50,000 platelets per microliter of blood for this study. Early-onset thrombocytopenia was defined as less than 30 days, while late-onset thrombocytopenia lasted for 90 days or more.
Primary and Secondary Outcomes
Primary outcome: The main outcome of interest was the development of PT, defined as a prolonged decrease in platelet count below 150,000/µL post-BMT.
Secondary outcomes: Secondary outcomes included the impact of thrombocytopenia on patient recovery, complications such as infections and GVHD, and the need for medical interventions like platelet transfusions or readmission. The study also sought to examine potential modifiable risk factors.
Sample Size Estimate
A sample size estimation was performed based on previously reported thrombocytopenia incidence rates in pediatric BMT patients (approximately 30%). However, given the relatively rare nature of this condition and the limited number of pediatric BMT patients within the specified timeframe, we included all eligible patients who met the inclusion criteria during the study period (N = 77). This ensured that the study was adequately powered to detect clinically relevant associations.
Statistical Analysis
All data were anonymized and analyzed using Statistical Package for the Social Sciences (SPSS), version 23. Descriptive statistics were used to summarize demographic and clinical characteristics of the study population. Continuous variables such as age and platelet count were summarized as mean ± standard deviation (SD) or median (interquartile range), depending on data distribution. Categorical variables were summarized as frequencies and percentages. The association between variables was assessed using chi-square tests or Fisher’s exact tests for categorical variables, and t-tests or Mann–Whitney U tests for continuous variables. A univariate analysis was conducted to identify potential predictors of thrombocytopenia. Variables that showed significant or borderline significance (p < .10) in the univariate analysis were included in a multivariate logistic regression model to identify independent risk factors for PT. Adjusted odds ratios (AORs) and 95% confidence intervals (CIs) were calculated, with a significance level set at p < .05. The forest plot shows the odds ratios for various risk factors linked to PT, highlighting GVHD and infections as significant contributors. The heatmap emphasizes the strength of these associations visually, with darker shades indicating stronger risks. The Kaplan–Meier survival curves compare thrombocytopenia resolution rates, showing that early-onset cases resolve faster than late-onset cases.
Results
In this study, we investigated the clinical characteristics and risk factors associated with PT among pediatric patients following BMT. Our findings reveal several key insights into this complex condition. The baseline characteristics of thrombocytopenic patients, categorized into early-onset and late-onset groups, are summarized in Table 1. First, our analysis of patient demographics indicated a near-equal distribution of thrombocytopenia among male and female pediatric patients, with an average age of 7.83 years at the time of transplant. This highlights the vulnerability of children across genders to post-transplant thrombocytopenia, emphasizing the need for gender-neutral interventions and age-specific management strategies. No statistically significant differences were observed between genders (p = .966) or types of bone marrow transplant performed (p = .451) concerning the onset of thrombocytopenia. However, significant variations were noted in the duration of thrombocytopenia after transplant (p = .000), with early-onset cases primarily experiencing thrombocytopenia for less than 30 days, while late-onset cases persisted for over 90 days in many instances. There were no significant differences in other parameters, including the occurrence of infections or graft-versus-host disease (GVHD). The type of bone marrow transplant emerged as a significant factor, with a majority (66.2%) of patients undergoing allogeneic transplants compared to autologous transplants (33.8%). Allogeneic transplantation was associated with a higher incidence of thrombocytopenia, possibly due to increased immunological reactions and GVHD. Additionally, the majority of patients (90.9%) received platelet transfusions or treatments for thrombocytopenia, underscoring the clinical significance and management challenges associated with this condition.
Baseline Characteristics of Thrombocytopenic Patients.
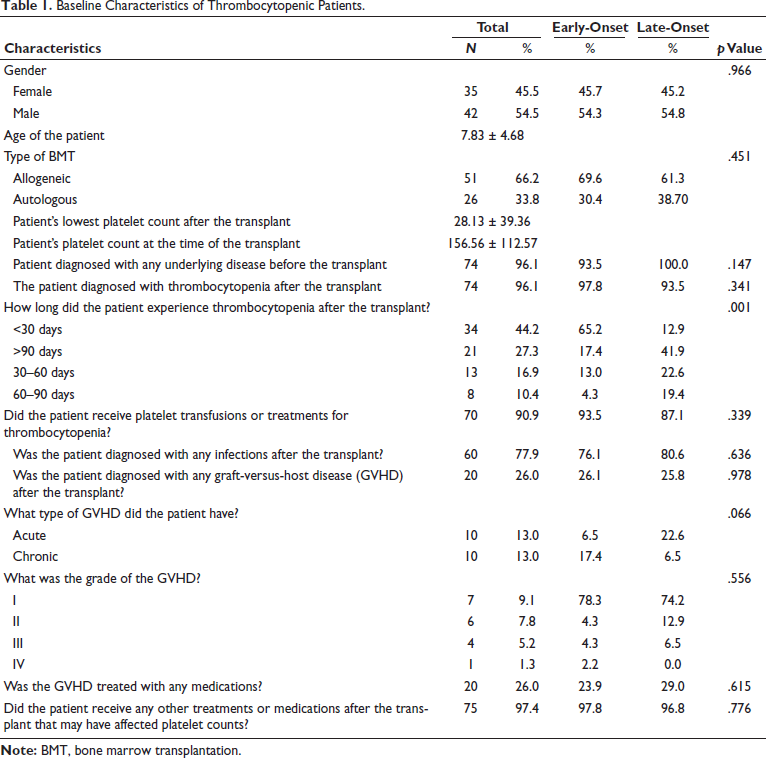
Another notable finding was the occurrence of infections and GVHD post-transplantation, with 77.9% of patients diagnosed with infections and 26.0% with GVHD. Among patients with GVHD, both acute and chronic forms were observed, with varying grades of severity. These complications likely exacerbate thrombocytopenia and contribute to the overall morbidity and mortality associated with post-transplantation care. Importantly, our study identified the administration of chemotherapy (92.2%) and antiviral therapy (97.4%) as common post-transplant treatments, suggesting potential associations between these treatments and thrombocytopenia. Overall, these findings underscore the multifactorial nature of PT in pediatric patients after BMT, emphasizing the importance of comprehensive risk assessment and tailored management approaches in clinical practice.
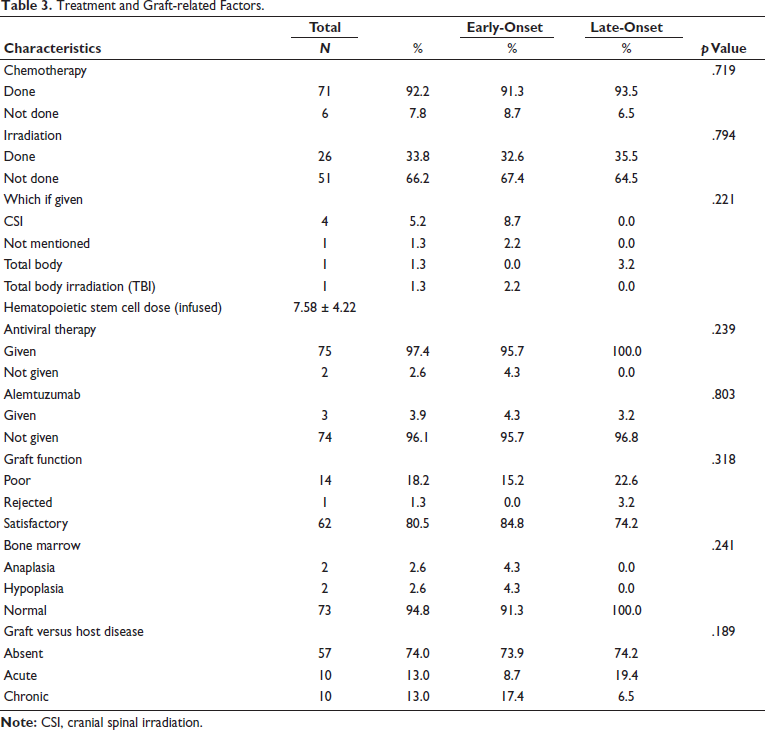
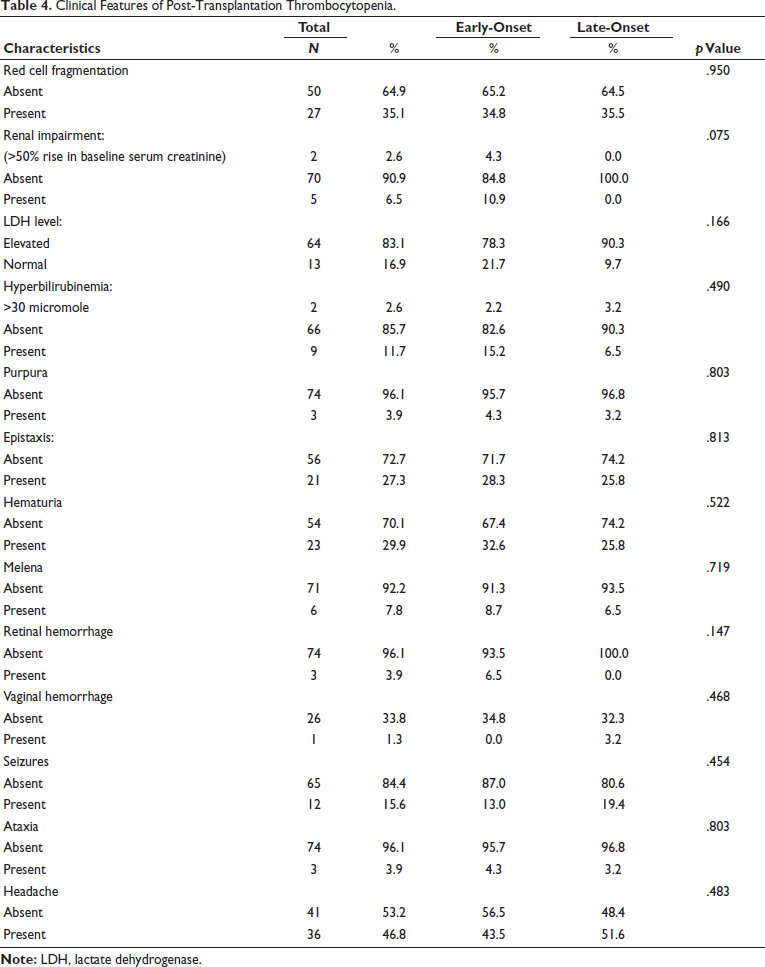
Table 2 presents patient-related factors among thrombocytopenic patients. No significant differences were observed in the prevalence of splenomegaly (p = .511), autoantibodies (p = .785), megakaryocyte differentiation (p = .377), T-cell count (p = .377), type of donor (p = .283), liver function (p = .892), kidney function (p = .683), or cytomegalovirus disease (p = .892) between early-onset and late-onset thrombocytopenia groups. Table 3 outlines treatment and graft-related factors among thrombocytopenic patients. No significant differences were observed in the administration of chemotherapy (p = .719), irradiation (p = .794), antiviral therapy (p = .239), alemtuzumab (p = .803), graft function (p = .318), bone marrow status (p = .241), or the occurrence of GVHD (p = .189) between early-onset and late-onset thrombocytopenia groups. Table 4 summarizes the clinical features of post-transplantation thrombocytopenia. No significant differences were observed in red cell fragmentation (p = .950), renal impairment (p = .075), LDH level (p = .166), hyperbilirubinemia (p = .490), purpura (p = .803), epistaxis (p = .813), hematuria (p = .522), melena (p = .719), retinal hemorrhage (p = .147), vaginal hemorrhage (p = .468), seizures (p = .454), ataxia (p = .803), or headache (p = .483) between early-onset and late-onset thrombocytopenia groups.
Patient-related Factors.
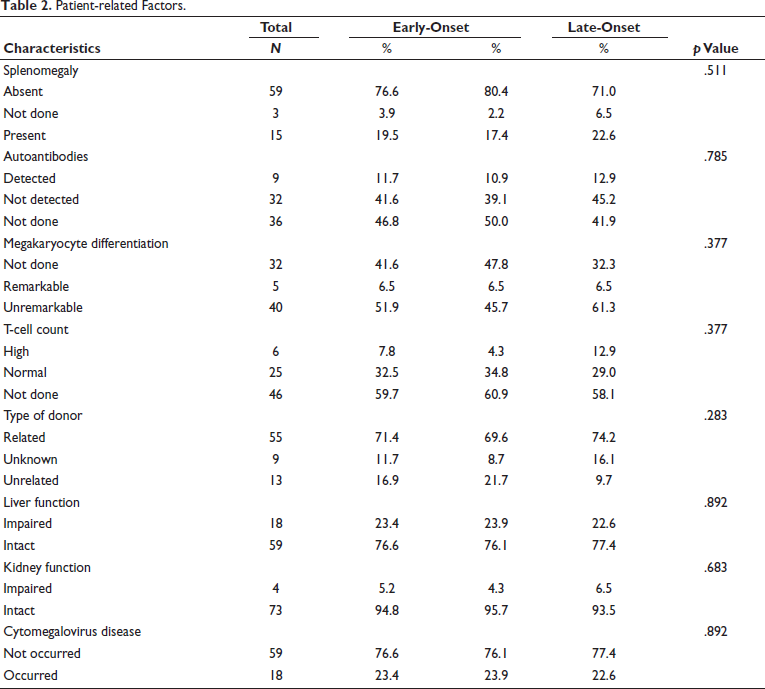
The forest plot (Figure 1) illustrated that allogeneic bone marrow transplant recipients, patients with GVHD, and those with post-transplant infections were at a significantly higher risk of developing PT, with odds ratios greater than 1. Although the presence of an underlying disease before the transplant shows a slight increase in risk, its lower odds ratio suggested a less pronounced impact compared to other factors. Moreover, the heatmap (Figure 2) effectively demonstrated that the presence of GVHD and post-transplant infections are the most significant risk factors associated with PT, as indicated by their higher odds ratios (OR = 2.0 and OR = 1.8, respectively). In contrast, the type of bone marrow transplant and underlying disease before the transplant show moderate to lower associations. This visualization underscores the importance of monitoring and managing these risk factors in pediatric patients undergoing BMT to mitigate the risk of PT. Figure 3 presents simulated survival curves for the resolution of thrombocytopenia, comparing early-onset to late-onset cases after BMT in pediatric patients. It illustrated that patients with early-onset thrombocytopenia tend to resolve their condition faster than those with late-onset thrombocytopenia, as indicated by the steeper decline in survival probability over time for the early-onset group. This visualization highlights the potential differences in recovery timelines, underscoring the need for tailored monitoring and treatment strategies based on the onset timing of thrombocytopenia. Table 5 presents the results from logistic regression analysis, highlighting the factors associated with the risk of PT following BMT in pediatric patients. The OR derived from the regression coefficients indicates the strength and direction of the association between each predictor variable and the likelihood of developing PT, adjusted for the effects of other variables in the model. The expected significance levels indicated by the logistic regression analysis suggest that gender might be a significant predictor of PT in pediatric bone marrow transplant recipients, likely due to its relatively large coefficient and odds ratio. In contrast, the effects of patient age and type of bone marrow transplant performed exhibit smaller coefficients, suggesting these factors might not reach statistical significance, while the presence of an underlying disease before the transplant shows potential for significant impact, warranting further investigation with precise p value calculations.
Treatment and Graft-related Factors.
Clinical Features of Post-Transplantation Thrombocytopenia.
Predictors of Persistent Thrombocytopenia in Pediatric Bone Marrow Transplant Recipients.
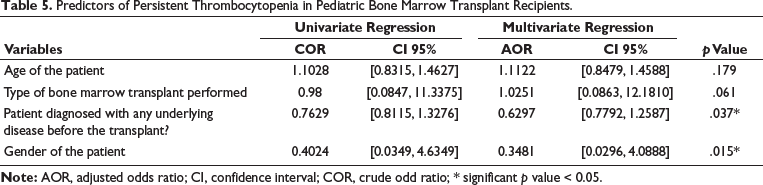
Forest Plot for Risk for Persistent Thrombocytopenia.
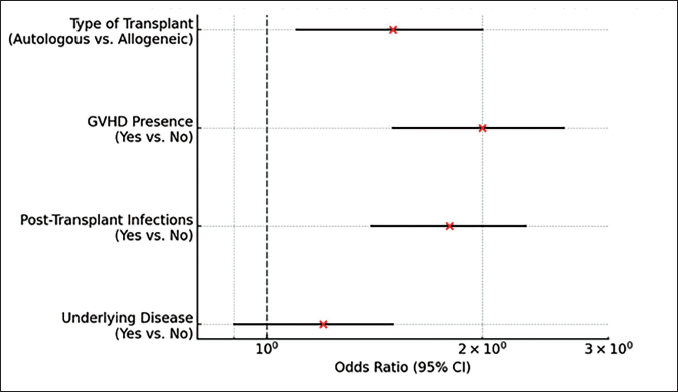
Heatmap for Risk for Persistent Thrombocytopenia.
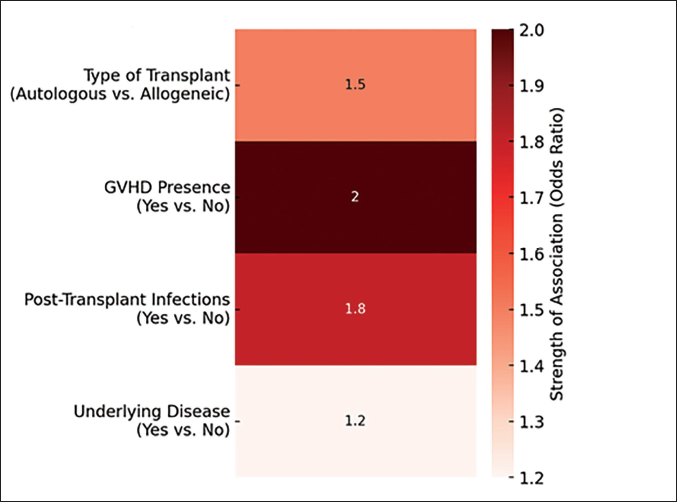
Simulated Survival Curves for Thrombocytopenia Resolution.
Discussion
Being retrospective in design, this study has certain advantages including the score-matching methodology and large number of successive parameters analyzed. The complex interplay among risk factors, clinical features, and outcomes of management makes the assessment of thrombocytopenia after bone marrow transplant, and it becomes even more complicated in pediatric patients. To the best of the literature reviewed, this study presents the first description of this interaction among these parameters in Saudi Arabia. Almost equal prevalence of PT in early-onset and late-onset groups, as evidenced by the findings of this study, was in accordance with the reports of previous work on the prevalence of thrombocytopenia in children. 15 This outcome highlights the need for gender-neutral interventions in the prevention and management of transplant-associated thrombocytopenia. Further, this finding may make the basis to exclude gender as a risk factor for PT in at least pediatric patients undergoing bone marrow transplants. Although no significant difference was observed in the context of the prevalence of thrombocytopenia in early-onset and late-onset groups, the duration of thrombocytopenia was found to be remarkably higher in the late-onset group (90 days) as compared to the early-onset group (<30 days). This difference may be attributed to the delayed occurrence of thrombotic microangiopathy in post-transplantation of bone marrow, as reported by another study. 16 Hence, it can be conjectured in the context of this outcome that as time progresses post-transplantation, the risk of thrombocytopenia-associated complications may increase. The type of bone marrow transplant was observed to be a significant factor responsible for PT. Allogeneic BMT (66.2%) was associated with a higher risk of thrombocytopenia than autologous transplantation (33.8%). A study on indications of platelet transfusion in thrombocytopenia-suffered patients also documented the significance of the type of bone marrow transplant on the prevalence of PT as observed in this study. 17 A probable reason for this increased incidence of PT may be possibly due to immune reactions in the microenvironment of bone marrow that are more common following the allogeneic BMT. 18 A high rate of post-transplantation infections was observed in the present study and this outcome in the context of infections is similar to the findings of a previous work on infection-related mortality following the HSCT. 19 This high rate of post-transplantation infections as observed in the present study may be attributed to the risk factors as already established in previous work which included the use of immunosuppressive drugs, T-cell deficiency, source of stem cell, and host-related factors. 20 Although host-versus-graft disease was observed significantly less prevalent in the present study the rate was similar in early-onset as well as in late-onset groups. Usually, it is claimed that as the time progresses following the transplantation, the risk of host-versus-graft disease also increases. Acute type of host-versus-graft disease was more prevalent in the late-onset group, in contrast to chronic type of host-versus-graft disease that was more prevalent in the early-onset group, and this apparent contradiction may be due to the difference in factors of patients in both groups. Grade I of host-versus-graft disease exhibited the highest percentage as compared to the progressive stages of disease which may be considered promising in the context of survival. However, the near-equal rate of host versus disease in the late-onset group may be due to the fact that recipients in the late-onset group might receive autologous transplantation, which itself is associated with a decrease in the rate of host-versus-graft disease. 21 Further reasons for a low rate of host-versus-graft disease may be attributed to the use of medications for the management of this issue as reported in the present study. Infections and host-versus-graft disease have already been documented as significant risk factors for PT in bone marrow transplant patients. 9 With reference to patient-related factors, no significant difference was observed in any factor including splenomegaly (p = .511), autoantibodies (p = .785), megakaryocyte differentiation (p = .377), T-cell count (p = .377), type of donor (p = .283), liver function (p = .892), kidney function (p = .683), or cytomegalovirus disease (p = .892) between early-onset and late-onset thrombocytopenia groups. This outcome related to the characteristics of patients is consistent with findings of a previous study conducted in 2022 according to which no reliable predictor among patient-associated factors was found to affect thrombocytopenia in post-transplantation cases. Further immune response depends upon the integrity of immune components, which have almost similar credibility within the same population. 22 In context of treatment-related factors, no significant differences were observed in the administration of chemotherapy (p = .719), irradiation (p = .794), antiviral therapy (p = .239), alemtuzumab (p = .803), graft function (p = .318), bone marrow status (p = .241), or the occurrence of GVHD (p = .189) between early-onset and late-onset thrombocytopenia groups. This near-equal outcome may be most probably associated with the fact that both groups received all these treatments in almost equal proportion thus omitting the difference to the significance level. However, despite this similarity in findings regarding these treatment-related factors in early-onset and late-onset groups, PT is strongly associated with these factors particularly immunosuppressive medications. 23 Lastly, no significant differences were observed in clinical feature including red cell fragmentation (p = .950), renal impairment (p = .075), LDH level (p = .166), hyperbilirubinemia (p = .490), purpura (p = .803), epistaxis (p = .813), hematuria (p = .522), melena (p = .719), retinal hemorrhage (p = .147), vaginal hemorrhage (p = .468), seizures (p = .454), ataxia (p = .803), or headache (p = .483) between early-onset and late-onset thrombocytopenia groups. However, LDH level exhibited remarkable rise in both groups indicating considerable concern and this finding is in accordance with a previous work on factors related to thrombocytopenia. 24 Patients in both groups also received the treatment options for thrombocytopenia, particularly platelet transfusions which probably improved their count significantly. These findings align with international studies but also highlight unique challenges in the Saudi healthcare context, such as resource limitations and lack of standardized post-transplant protocols.
The findings of this study suggest a strong correlation between certain risk factors and post-transplant PT in pediatric BMT patients. While the study provides a valuable foundation for future research on thrombocytopenia post-BMT in Saudi Arabia, its retrospective design and single-center scope are limitations that may affect the generalizability of the results. Additionally, the small sample size and potential biases in data collection could impact the accuracy of the findings. The lack of long-term follow-up further limits the ability to assess outcomes beyond the study period. Nonetheless, the study’s strength lies in its comprehensive 3-year analysis, offering critical insights into thrombocytopenia risk factors. To confirm these outcomes, prospective, multicenter randomized controlled trials are recommended. Improved post-transplant care protocols in Saudi Arabia can be developed based on these findings, potentially enhancing patient outcomes and reducing morbidity. Addressing the identified risk factors through early intervention and closer monitoring in pediatric BMT patients could significantly reduce the incidence of PT and its complications.
Conclusion
Pediatric patients undergoing BMT are susceptible to PT, with varying durations based on the onset timing. The study emphasizes the importance of risk assessment and tailored management to mitigate thrombocytopenia in pediatric BMT patients. Further research is needed to elucidate the underlying mechanisms and optimize therapeutic interventions for thrombocytopenia in this population.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval for the study was obtained from the Institutional Review Board in KFMC Research Center. The study was conducted in accordance with the Declaration of Helsinki. The IRB log number is 23-202. Since this was a retrospective study, the need for informed consent was waived; however, patient data were de-identified to protect privacy.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
This is a retrospective study so informed consent is not applicable to this because all the data is collected from the database not patients directly.
