Abstract
Background
Effectual management of Bladder Malignancies (BMs) is currently a global challenge due to their expensive therapeutic and diagnostic cost burden, together with augmented progression/recurrence rate.
Objectives
This review aimed to explore cutting-edge diagnostic and therapeutic techniques for meeting the current clinical requirements for effectively managing BMs encompassing dual muscle-invasive bladder tumors (MIBT) and nonmuscleinvasive bladder tumors (NMIBT).
Methodology
A comprehensive review of relevant literature (including data review from 2019 to 2024) was performed to collect valuable information regarding the utilization of advanced diagnostic and therapeutic techniques for the effective management of BMs.
Results
Studies investigating the potential applications and possible limitations of various methods, including UroVysion, continuous bladder irrigation (CBI), transurethral resection of bladder tumor (TURBT), laparoscopic, and robot-abetted extended pelvic lymph node dissection (PLND), 3D culture models and nerve spring technique (NST) in robotic intracorporeal Studer’s orthotopic neobladder (RISON) have been examined. Similarly, the role of Urothelial Piezo channels (Piezo1 and Piezo2) in urinary system modulation, human epidermal growth factor receptor (HER2)-selective therapy in metastatic urothelial carcinoma (MUC) patients, and diagnostic benefits of fluorescence in situ hybridization (FISH) in BMs have been analyzed. Current report enlisted the major clinical applications associated with the advanced diagnostic and therapeutic techniques with consequent management of lower urinary tract malfunctions (LUTMs), stress urinary incontinence, and BMs, including both MIBT, NMIBT, and MUC.
Conclusion
This review provided detailed information on numerous benefited outcomes attained from different approaches such as augmented bladder functioning, implementation of collective optical and cancer-specific imaging systems, utilization of tumor-specific biomarkers, precise tumor amputation, reduced urinary complications and hospital stay, postoperative comfort, limited invasiveness, limited blood loss, and ultimately reduced recurrence rates. BMs management at the clinical level needs to be effectively rationalized to enhance the patient’s quality of life and reduce the ultimate cost burden over bladder cancer patients together with appropriate management of associated limitations like urinary tract infections, bladder perforation, bladder infections, and carcinoma relapse in postoperative cases should effectively be encountered to further improve the therapeutic potential of advanced techniques in the near future.
Keywords
Introduction
Bladder malignancies (BMs) are currently a global challenge based on yearly impact on approximately 80,000 patients, with morbidity levels provoked to about 18,000 cases by the year 2020.1, 2 Based on the degree of tumor invasion, BMs are classified into muscle-invasive bladder tumor (MIBT) and nonmuscle-invasive bladder tumor (NMIBT), with MIBT diagnosed in about 25% of cases. In comparison, NMIBT is identified in approximately 80% of patients at the initial stages.1, 2
Augmented relapse or recurrence risk associated with the progression of bladder cancer to advanced/malignant phases eventually results in enhanced therapeutic or diagnostic cost burden over the bladder cancer patients. To overcome the limitations associated with cystoscopy (frequently employed diagnostic technique), non-invasive diagnostic approaches exhibiting superior specificity, sensitivity, or accuracy and utilization of urinary biomarkers such as UroVysion are employed that sort current issues to a greater extent with reduced risk of recurrence. 3
Dual therapeutic and diagnostic advancement is the ultimate requisite for effectively managing BMs. The current review briefly reviews numerous advanced approaches, including Chinese botulinum toxin A (CBT/A) has been explored to pay promising outcomes regarding the management of lower urinary tract infections (UTIs), bladder pain syndrome/interstitial cystitis, benign prostatic hyperplasia, chronic prostatic pain, detrusor-sphincter dyssynergia, idiopathic overactive bladder, neurogenic detrusor overactivity and improved bladder working due to reduced detrusor contractility and bladder hypersensitivity. 4 Similarly, implantation of the NuStim® device together with floor muscle exercises (FMEs) has been described to improve stress urinary incontinence (SUI).5, 6 Generally, combined intravesical instillation of chemotherapeutic and immunological components is used for NMIBC management that effectively eradicates residual cancer cells and prevents relapse risk after transurethral resection of bladder tumor (TURBT). 7 However, the current study also identified CBI’s role in the reduction of tumor progression/proliferation and relapse in NMIBC patients together with benefited outputs such as lowered morbidity rates, prevention of clot urinary retention, reduced harm/injury and continuous monitoring of critical events. 8 The current report also dealt with the construction of an orthotopic neobladder via utilization of nerve-sparing techniques capable of providing trifecta functional outcomes, that is, sexual functions restoration, urinary continence, and declination of reoccurrence rates. 9
Therapeutic advantages associated with the use of laparoscopic and robot-abetted extended pelvic lymph node dissection (PLND) include rapid retrieval, diminished blood loss, deep visualization, controlled invasiveness, and certain oncological benefits. 10 Identification of novel therapeutic drug components and use of targeted drug therapy based on designing of 3D culture models (such as bladder cancer organoids) to effectively introduce novel therapies is also part of the current review. 11 Piezo channels (Piezo1 and Piezo2) associated urinary functions modulation12, 13 together with the therapeutic role of HER2-selective treatment in MUC patents has also been identified. 14
Thus, effectual clinical management of Bladder cancer malignancies not only requires diagnostic advancement but also greatly depends on therapeutic modalities. The current report is designed to provide a comprehensive review of emerging diagnostic/therapeutic tactics, possibly exploring therapeutic innovations for the effective management of complex BMs (Figure 1).
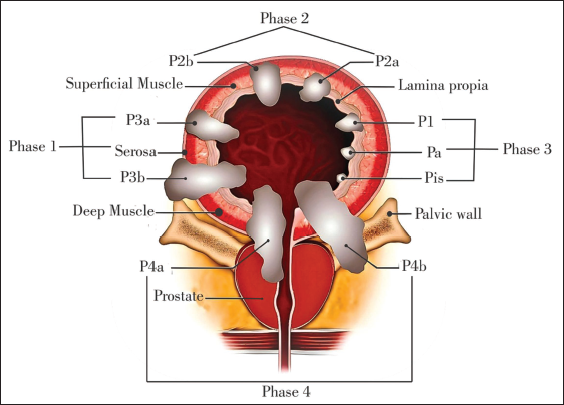
Various Phases of Bladder Cancer along with its Prime Location in the Bladder.
Therapeutic Functioning of CBT/A in Lower Urinary Tract Malfunctions (LUTMs) Management
Management of LUTMs using CBT/A is considered a viable option because of its safe, effective, and valuable outcomes comparable to other CBT/A products. 15 Effective management of neurogenic detrusor overactivity coupled with complete bladder emptying is another essential aspect of CBT/A therapy. 16 CBT/A clinical manifestations as per official recommendations include treatment of bladder pain syndrome or interstitial cystitis, benign prostatic hyperplasia, chronic prostatic pain, detrusor-sphincter dyssynergia, idiopathic overactive bladder, neurogenic detrusor overactivity, and UTIs. 4
Thus CBT/A, although considered to be an effective substitute for the management/treatment of LUTMs, may not be effectively utilized for the treatment of UTIs in all individuals.15, 17 Chief complications associated with CBT/A therapy include repeated injections for maintenance of optimum therapeutic level along with UTIs and partial bladder emptying necessitating clean intermittent catheterization. 4
Utilization of CBT/A injections for the effective management of neurogenic detrusor overactivity and idiopathic overactive bladder requires careful patient selection and clean intermittent catheterization due to partial bladder emptying. Before starting the procedure, obtaining complete patients consent is essential in the case of CBT/A therapy due to the need for its repeated dosing after every 6–9 months (for maintaining sustained therapeutic profile) and the occurrence of reduced autonomic dysreflexia, particularly in patients revealing spinal cord injury.18, 19
Therapeutic Functioning of Transdermally Implanted Wireless Microstimulator Device (NuStim®) Coupled to FMEs for Management of SUI
Surgical procedures, including adjustable male slings, adjustable balloon devices, artificial urinary sphincters, and fixed male slings, are considered the specific therapeutic options for SUI management in males.20, 21 Though current treatments present promising outcomes yet, incontinence reoccurrence in about 20–30% of cases provoked the need for latest interventions, including the inclusion of artificial urinary sphincters and urodynamics.22, 23 Research studies providing a deep understanding of complex SUI pathogenesis via interlinked molecular mechanisms involving conjugated functioning of different hormones, connective tissues, muscles, and nerves along with exosome differentiation and gene regulation reports revealing the pivotal functioning of specific genes (β-catenin) in SUI pathophysiology explored highly unique and efficacious therapeutic options for its management (Figure 2).
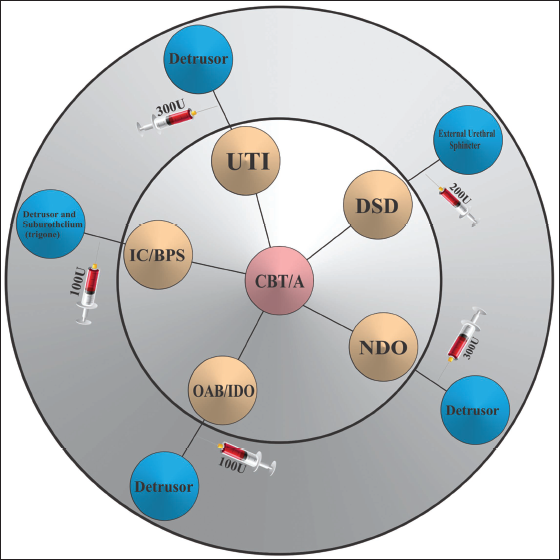
Therapeutic Applications of Chinese Botulinum Toxin A (CBT/A) at the Targeted Site in the Management of Lower Urinary Tract Malfunctions (LUTMs), Decreased Urethral Pressure (DSD), Improved Continence or Enhanced Bladder Capacity (NDO), Improved Micturition Frequency or Urgency (OAB/IDO), Reduced Bladder Pain and Urinary Frequency (IC/BPS), and UTI (Reduced UTIs in Patients with NDO).
SUI clinical manifestations/signs and symptoms have been significantly improved upon using combination therapy involving NuStim®: a transdermally inserted wireless microstimulator device alongside FMEs. Furthermore, the current device is reported as applicable in improving incontinence symptoms and found to be safe and effective upon adjunctive use with FMEs. 24 Similarly in nonpregnant females’ comparative analysis of pelvic floor muscle training with or without electrostimulation/biofeedback suggested improved SUI clinical manifestations related to improved pelvic floor muscle contraction.19, 25 Nevertheless, numerous tactics, including whole-body vibration, vaginal cones, and biofeedback training, have been reported to be efficacious in SUI management yet pelvic floor muscle training remained a mainstay due to its pivotal role in the enhancement of pelvic floor muscle functioning.26, 27
Involvement of Brain Disorders in LUTMs
Certain brain disorders/injuries of particular cranial areas may modulate neural pathways regulating bladder functioning. Detrusor over-activity most frequently occurs due to cranial dysfunctioning like stroke and dementia with Lewy bodies. In contrast, urge incontinence (indicated by abrupt voiding urges and urinary incontinence) is caused by damage to particular cranial areas with ultimate interruption of forebrain synchronized continence pathways.28, 29 Diagnostic analysis of detrusor sphincter dyssynergia patients presenting altered stimulation of the pontine region with consequent modulated brain-bladder control network compared to control/healthy subjects has been noticed in functional MRI reports.21, 30, 31 UTIs in neurological patients require a complete investigation of principal urological causes and are frequently more complicated in the sense that they preset potentially different microbiology and require specialized treatment in comparison to the general population. 32
Currently, numerous investigations have been conducted to analyze neural pathways that provoke urological dysfunctions in patients exhibiting brain disorders. The pivotal contribution of different mechanisms of a brain-bladder network, such as the anterior cingulate, prefrontal cortex, and insular cortex, towards bladder sensation/sphincter control has been stated. 28 Besides, greater emphasis is put on the need for individualized therapeutic design to effectively manage dual urinary dysfunction and cognitive disabilities in neurological patients based on the aforementioned complex interaction between urinary impairment and vascular cognitive disabilities.33, 34
Management of pelvic floor muscle disorders and LUTMs seems possible with the use of non-invasive brain stimulation approaches soon, thereby providing a path for innovative techniques in clinical practice.
Role of CBI in the management of cases displaying mild nonmuscle invasive bladder cancer (NMIBC) where single instillation of intravesical chemotherapeutic treatment following transurethral resection is considered unfeasible.
NMIBC management is carried out using numerous practices, including combined intravesical chemotherapeutic regimen, intravenous/intravesical immunotherapy, viral and bacterial intravesical therapies, and non-invasive urinary drug-disposing methods that encompass the employment of bioinert peptide for adequate provision of microtubule inhibitors to NMIBC.35, 36
In cases displaying mild NMIBC, the CBI technique plays a vital role in enhanced patient compliance or comfort associated with employing innovative self-improved CBI sensor devices. Reduced complications rate, improved patient compliance or comfort, reduced workload over nurses, and augmented disease-related information provoked its utilization as a promising tool for cases representing mild NMIBC considered unsuitable for single instillation of intravesical chemotherapeutic treatment.8, 37 The chemotherapeutic pattern of patients displaying mild NMIBC following transurethral resection exhibits remarkable variation between single instillation and CBI. 38
Latest Furtherance in TURBT Management
Upgraded non-muscle-invasive bladder cancer therapy combined with immunotherapy and TURBT-centered bladder preservation techniques are highly effective in enhancing particular patients’ quality of life and oncological consequences.27, 39
Innovative approaches, including robotic-assisted TURBT and high-intensity focused ultrasound (HIFU), indicated promising clinical outcomes such as precise tumor amputation from the bladder. Moreover, using MR-HIFU procedures resulted in augmented accuracy and clinical outcomes, particularly in subjects representing musculoskeletal lesions correlated to positioning limitations directed by MRI-assisted robotic systems.27, 40 HIFU is a non-invasive technique offering limited impairment of surrounding tissues due to its ability to induce coagulative necrosis in cancerous regions, thus ensuring anticancer, pain relief, and probably prolonged survival time.37, 41 In contrast to conventional treatments like radical prostatectomy and radiotherapy, HIFU is considered a practical treatment approach for the effective management of prostate cancer due to its restricted invasiveness and lower morbidity rate.6, 42 Thus, innovative TURBT-based conjugated therapeutic approaches might play a promising role or exhibit the potential to enhance patients’ quality of life or survival rates due to better accuracy and oncological effects (Figure 3).
Functional Trifecta Outcomes Related to the Utilization of Nerve Spring Technique (NST) in Robotic Intracorporeal Studer’s Orthotopic Neobladder (RISON) in Male Subjects
Detailed analysis of RISON in male subjects led to the establishment of the NST. Utilization of nerve-sparing techniques in surgical practices provoked a positive impact on urinary continence, retrieval of sexual functions, and improvement of postoperative outcomes, including earlier attainment of daytime continence with reduced nocturnal pad usage.9, 43, 44 Thus, following robotic intracorporeal orthotopic neobladder reformation with particular emphasis on nerve preservation and augmentation of functional aspects, NST is considered to participate well in comparison to nonnerve sparing techniques regarding optimization of patient’s quality of life and continence outcomes.43, 45 Numerous studies reported the successive involvement of nerve-sparing techniques in robotic intracorporeal surgeries for an orthotopic neobladder reformation with the capability of attaining earlier daylight continence. They reduced nightly pad utilization in postoperative patients, thus acquiring comparatively higher functional trifecta outcomes (like sexual function restoration, urinary continence, and reduced reoccurrence rates).43, 44, 46–48
Handling issues associated with the prevailing connector systems, complexity generation from the involvement of multiple electrodes with these tools, and constraints related to present nerve restoration systems. Utilization of magnetic connector systems for neuronal probes, assimilated multiple electrodes with short shanks for synchronized monitoring and introduction of tissue-engineered nerve supervision canals with advanced regenerative abilities somehow tackled aforementioned challenges based on improved connector utilization, augmented neurochemical observations and boosted nerve restoration results.46, 49 Overall, the implementation of NST or nerve-sparing techniques for orthotopic neobladder reconstruction following robotic intracorporeal surgery presents effectual benefits that indicate promising functional outcomes with ultimately improved patient quality of life.
Invasive Bladder Tumor Management via Laparoscopic and Robot-Abetted Extended PLND
Management of invasive bladder tumor via laparoscopic and robot-abetted extended PLNS is reported effective based on its unique therapeutic merits, including rapid recovery, high-class visualization, limited blood loss, and insignificant invasiveness in comparison to conventional open surgeries. 10 Clinical paybacks associated with the utilization of laparoscopic and robot-abetted extended PLND in numerous reports include its usage for inflammatory arthritis patients where it is suggested to improve physical body functioning, balanced body composition and cardiorespiratory fitness following 1-year postrehabilitation.50, 51 Further, in children undergoing appendectomy, the effective use of elective interval appendectomy is considered more appropriate than emergency appendectomy due to its potential cost-effective and reduced morbidity outcomes.34, 52 Continuous progression and improvement of surgical procedures are still required in urology despite numerous intervening disputes like process complexity, extended surgery time, and yet adherence to laparoscopic and robot-abetted prolonged PLND to open surgical techniques/anatomical margins.
Pivotal Role of Advanced 3D Culture Models in the Management of Bladder Tumor
3D culture models are currently employed to simulate the tumor microenvironment (TME) accurately with more precise identification of patient-specific mutations, ultimately directing individualized drug therapy and, consequently, effective bladder cancer treatment.11, 53 More specifically, personalized drug treatment and identification of novel therapeutic components take place due to the effective establishment of bladder cancer organoids. 54 3D culture models, including bladder cancer organoids, are designed to mimic the heterogeneity of patient tumors, test individual cases’ sensitivity towards a particular medication (like platinum-based therapeutic components and olaparib), and evaluate targeted therapy.55, 56 3D culture models like bladder cancer organoids exhibit the ability to more accurately simulate/represent TME, thereby identifying or testing subject sensitivity towards different therapies along with assessment of personalized therapeutic outcomes like immunotherapy.11, 53, 55, 57 Furthermore, current models’ involvement in identifying patient-specific mutations or their subtypes facilitates customized therapeutics capable of providing targeted drug therapy directed against particular carcinogenic pathways and improved drug screening.57, 58 Thus, the establishment of 3D culture models significantly enhanced the understanding and treatment of bladder tumor corresponding to more accurate TME understanding, enabling customized therapeutics, and improved drug discovery (Figure 4).
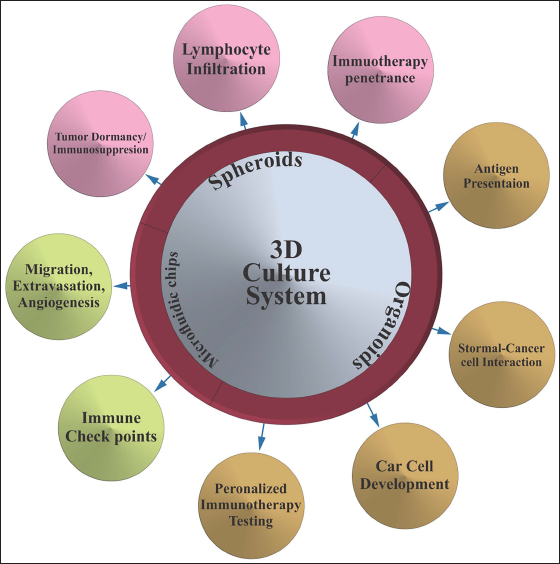
Major Clinical Applications of 3D Culture Models Indicating Effective Use of Spheroids, Organoids, and Microfluidic Chips.
In vitro 3D models focused preclinical trials suggested promising antitumor activity of STAT3 inhibitors against proliferative bladder tumor via selective targeting of particular signaling pathways.6, 59 Thus, the current encroachment successfully explored the effective participation of 3D culture models in advanced bladder tumor-based research or therapy coassociated with improved patient-related outcomes. Overall 3D culture models explored new insights and avenues for customized therapeutics. Efficient use of current models might help predict drug sensitivity, conduct gene profiling, identify novel therapeutic moieties and ascertain individualized therapeutic regimens with the ultimate improved efficacy of selected therapy.37, 57 Accordingly, the successful establishment of 3D culture models was correlated to the limitations brought about by conventional models incapable of effectively mimicking the in vivo TME of tumor cells. The effective cell-to-cell and cell-to-extracellular matrix interactions replicated by 3D culture models provided a more precise way of deeply understanding bladder tumor.27, 53 In contrast to conventional 2D cultures, the improved resistance against apoptosis and metabolic variations provoked by 3D culture models facilitates tumor characterization/therapy selection. Generally, meticulous therapeutic findings and bladder tumor understanding became possible with the effectual utilization of 3D culture models.31, 60 In spite of the aforementioned features 3D culture model usage or advancement also offers numerous challenging issues, including the requisite for a deep understanding of bladder tumor radio-sensitivity regarding various drug-delivering systems, labor-intensive and time-consuming factors related to organoids growth analysis, the requirement of upgraded software or algorithms for assessing the proficiency of drug screening systems along with shortcomings associated to accurate/precise mimicking of in vivo TME.11, 53, 57, 61
Bladder Functions Modulation via Expression of Piezo Channels
The pivotal participation of Piezo channels (Piezo1 and Piezo2) in bladder function maintenance and sensory perception has been described.13, 62–64 In the bladder, Piezo2 channel expression occurs within the neurons innervating the bladder and urothelial umbrella cells with consequent involvement in mechanosensation or sensory feedback.65, 66 Bladder function regulation via Piezo channels, particularly Piezo1 and Piezo2 takes place through mechanosensitive signaling regulation involving the conversion of mechanical signal, that is, bladder filling, into a biological one regulating void functions. Hence, normal functioning of urinary systems requires appropriate working of urothelial Piezo (Piezo1 and Piezo2) channels, including normal bladder voiding with its altered expression inducing remarkable implications including bladder hypoactivity and urinary incontinence, 12 bladder mechanical allodynia and hyperactivity and distorted responses affecting the bladder’s ability of urine filling/storage and micturition reflex upon mechanical stimuli.13, 62
Bladder malfunctioning and urinary incontinence perceived in Piezo1, Piezo2, or dual channels lacking knockout mice are correlated with the interruptions of urothelial responses to mechanical stimulation, ATP release, or voiding behavior. Similarly, Piezo2 channel is considered to effectively regulate micturition reflexes and bladder contractions based on its pivotal participation in recognizing bladder stretch responses upon sensory stimulation (via sensory neurons). Thus, in-depth analysis and understanding of Piezo channels ensues in the investigation of potential therapeutic targets exhibiting potential effectiveness in managing bladder dysfunctions like urinary incontinence and interstitial cystitis-related pain.13, 62 Correspondingly, in interstitial cells overexpression of piezo1 channels is recognized during bladder hyperactivity in cystitis. Here piezo1signaling pathway acts as a mechanosensory molecule regulating Ca2+ signaling and reacting to membrane tension. In the bladder interstitium, dual TRP and Piezo1 channels expression suggest its pivotal functioning in sensory stimulation associated with activation of mechanical or chemical stimuli.63, 67 Furthermore, the Piezo1 channel is also considered a promising drug target or therapeutic biomarker for the effective management of chronic cystitis-related bladder pain based on its conjugated interaction with other channels (NCX1) instigating bladder hyperactivity and Ca2+ overload with its site-specific influence over bladder contraction.67, 68 Moreover, variable expression of Piezo1 and Piezo 2 channels in bladder tumor also provoked its utilization as a therapeutic drug target for effective management of bladder tumor progression or prognosis using innovative biomarkers. 69
Profound analysis regarding the expression/functioning of Piezo channels in the regulation of sensory bladder functions also scouted its primary contribution to bladder system complications/disorders.70, 71 Numerous studies conducted on the lower urinary tract identified Piezo2 channels as an essential mechanosensitive transduction molecule significantly contributing to micturition reflexes or bladder sensitivity coupled to its potential role in regulating bladder functions via expression in bladder-innervating neurons and urothelial cells. Furthermore, upregulation of Piezo2 channels in afferent neurons during interstitial cystitis is associated with progressive bladder allodynia and hyperactivity.45, 72 Therapeutic benefits associated with the modulated expression or regulation of Piezo channels provoked its successful identification as a promising drug target or therapeutic biomarker for the effective management of various urinary system disorders, particularly bladder pain treatment.13, 62–65 All such findings urge a detailed analysis of the underlying molecular mechanism regulating the Piezo channel’s working in the urinary system, as well as its prospective clinical application in the physiology and pathophysiology of the urinary system. 73 Finally, successfully exploring drug targets anticipated for therapeutic management of mechano-sensation-related urinary/bladder disorders requires a comprehensive understanding of intricate interaction among Piezo channels and signaling pathways.
Advanced Bladder Replacement Therapies
Effective management of bladder cancer through advanced bladder replacement therapies involves the successful application of novel biomaterials, cell cultures, and innovative drug delivery systems.74, 75 Current techniques demand bladder repair or reformation devoid of intestinal tissue utilization, thus provoking a promising approach to treating patients requiring bladder replacement with improved clinical consequences.
Advanced bladder replacement therapies offered remarkable benefits over the previous methods, including the use of novel immunotherapeutic components like adoptive cell treatment, antibody-drug conjugates (ADCs), and immune checkpoint inhibitors, which probably upgraded bladder cancer management, offering innovative therapeutic options clinically.17, 74 Correspondingly, usage of a cycle treatment containing neoadjuvant dose-dense MVAC (methotrexate, vinblastine, adriamycin, and cisplatin) in cases suffering from muscle-invasive bladder cancer indicated benefited outcomes instead of four cycles of gemcitabine/cisplatin therapy.17, 35 Collective advancement of current options may result in value-added therapeutic options with ultimately improved clinical consequences in patients acquiring bladder replacement therapy (Table 1).
Advanced Therapeutic Factors Currently Employed for Management of Bladder Malfunctions with their Pivotal Advantages and Limitations.
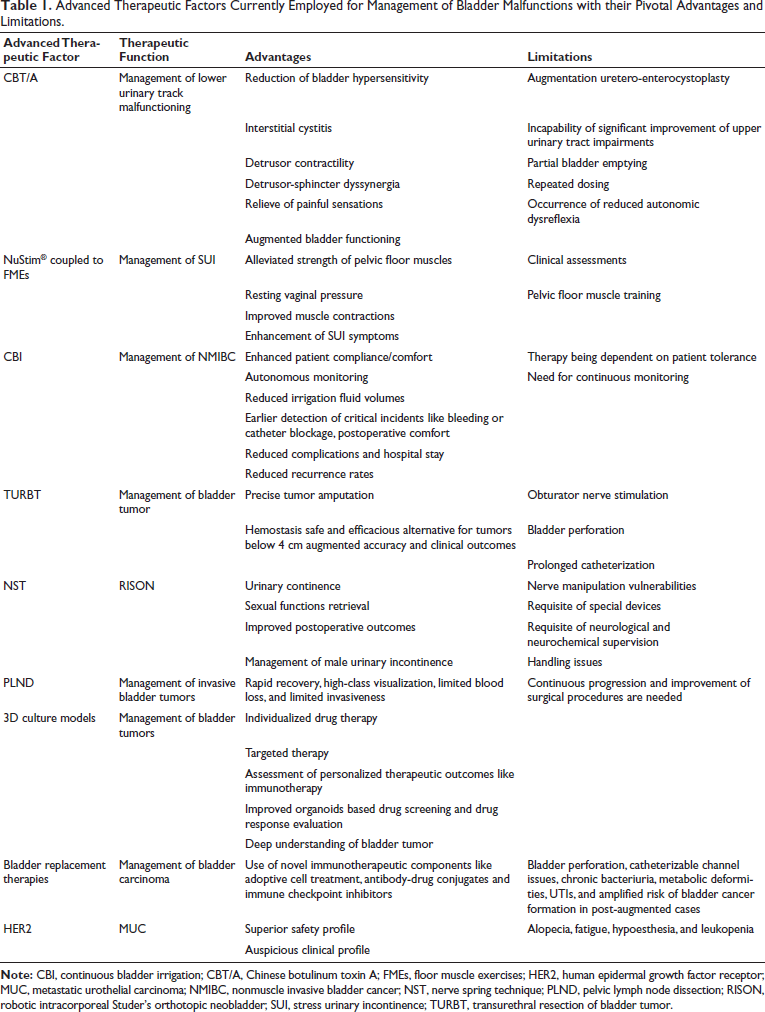
As mentioned earlier, bladder replacement therapies particularly bowel segment utilization for bladder reformation, are correlated to prolonged effects, including chronic infectious disorders, metabolic defects, stone formation, and malignancies with remarkable impact on a patient’s quality of life. 76 Similarly, utilization of the trial-and-error technique is another intervening issue associated with bladder pain syndrome based on its involvement in obscuring the disease progression, remission, and prevention identification.77, 78 Hence, profound knowledge regarding the potential risks/diverse obstacles encountered in bladder replacement technique is particularly important for earlier identification, prevention, and effective management of concerned risks.
Management of Metastatic Urothelial Carcinoma (MUC) via Advanced HER2-Selective Treatment
Assessment of Anti-HER2 RC48-ADC (disitamab vedotin) alone or in amalgamation with immunotherapy is currently focused on the management of MUC. RC48 alone or in combination with immunotherapy is found to be effective in cases indicating local or advanced stage, that is, MUC nevertheless of compromised renal functioning. 79 The pivotal role of HER2-selective treatment, particularly in MUC patients, indicating HER2 positivity or low expression80, 81 along with the instigation of modern anti-HER2 ADCs presenting advanced therapeutic potential and remarkable clinical activity in MUC patients has been expanded. 14
Similarly, expressively improved therapeutic efficacy resulted from RC48-ADC and EV (anti-HER2 agents) in HER2-positive MUC with objective comeback ratios extending from 38.9% to 51.2%.79, 82 Furthermore in individuals presenting HER2 expression as a predictor and prognostic marker, the selective incorporation of DV in HER2-selective treatment resulted in value-added survival of MUC patients. Regardless of the fabulous benefits, HER2-selective treatments are also associated with certain adverse effects like alopecia, fatigue, hypoesthesia, and leukopenia. Hypoesthesia and neutropenia are the reported grade 3 adverse effects occurring in certain individuals with no grades 4 or 5 adverse events noted for current therapy, suggesting its highly favorable safety profile. 83 Long-term side effects associated with current therapy are under investigation yet proliferation-free survival and upgraded overall survival response, even in cases presenting liver metastasis or prior immunotherapy response, have been proposed through current treatment. Current findings extremely upsurge the significance of targeted therapies, particularly HER2-selective treatment, in contrast to conventional chemotherapeutics for efficacious management of individuals presenting advanced or MUC. Fluorescence in situ hybridization (FISH) and bladder cancer diagnosis after primary resection, FISH represents efficacious performance with appropriate sensitivity or specificity (68% and 64%) regarding identifying remaining bladder cancer cells and the recurrence or relapse extrapolation in NMIBC. 84 Similarly, relapse prediction of NMIBC with approximate sensitivity and specificity levels of 60–82%, and 64–70%, respectively has been indicated in other similar findings. 85 Correspondingly, postoperative cancer progression from non-muscle-invasive bladder cancer to a muscle-invasive phase may effectively be predicted using UroVysion marking systems, thus provoking positive FISH findings as a potential tool or biomarker for identification of bladder carcinoma relapse following secondary electroresection. 3
A FISH institution with its significant contribution to the diagnosis and surveillance of bladder cancer (following secondary electroresection) became practicable due to the advancement of molecular biology/imaging techniques since FISH uses fluorescently labeled DNA probes for detecting genetic alterations with extreme specificity or sensitivity within urothelial cells.86, 87 Hence, the use of Fluorescent probes to identify specific nucleic acid sequences in a cell is the basic phenomenon of FISH working, while in the diagnosis of bladder cancer current technique provoked itself as a potential tool based on its capability of detecting chromosomal variations taking place in the carconigenic cells.88, 89 FISH usage for aneuploidy identification of chromosomes 3, 7, 17, and loss of the 9p21 locus in urine samples of bladder cancer individuals has been introduced after the successful approval of UroVysion Bladder Cancer Kit by the FDA in 2005. 89 Furthermore, continued monitoring of genetic alterations in bladder cancer cells with improved sensitivity/specificity became possible due to the concomitant utilization of optical imaging procedures with cancer-specific imaging components like fluorescently labeled CD47 antibodies. 90 Current developments thus provoke FISH as an auspicious tool for effective clinical management of bladder cancer.
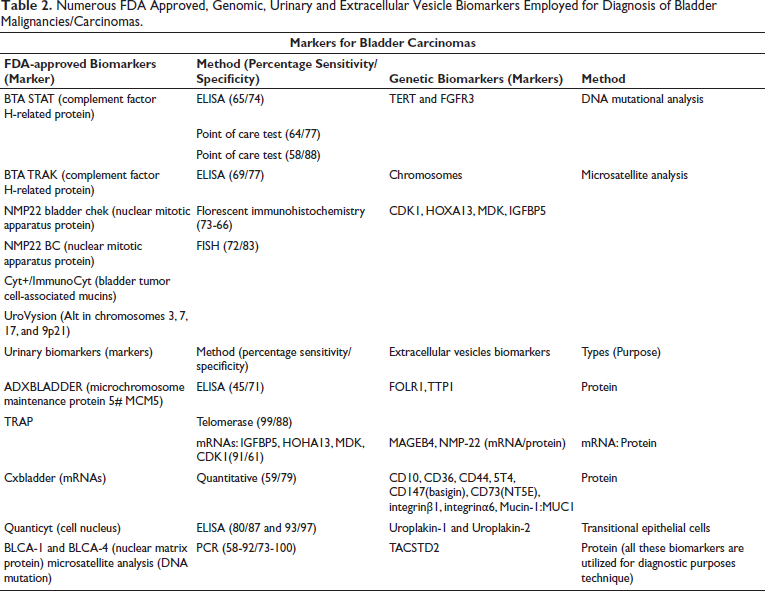
Despite the mentioned advantages, the current approach is also associated with certain limitations like variable sensitivity or specificity pattern attained from various FISH methods, along with the results being remarkably affected by the patient’s demographic variables like age, sex, smoking, and so on.84, 91–93 Similarly, FISH accuracy is also impacted by BCG due to its inability to predict tumor progression/recurrence before BCG perfusion (inability to detect flat lesions), thus acquiring complementary diagnostic techniques for effective management of bladder carcinoma.94, 95 Furthermore, genetic alterations occurring in chromosomes 3, 7, 17, and loss of 9p21locus and refined algorithm interpretation for obtaining accurate results might also be considered as the factors affecting FISH findings.84, 91–93 Hence effective diagnosis and treatment of bladder cancer requires the consideration of above-mentioned factors while relying on FISH for making clinical decisions regarding the diagnosis and management of bladder cancer (Table 2).
Numerous FDA Approved, Genomic, Urinary and Extracellular Vesicle Biomarkers Employed for Diagnosis of Bladder Malignancies/Carcinomas.
Conclusion
Clinical management protocols need to be effectively rationalized to improve the patient’s quality of life and reduce the cost burden on bladder cancer patients with ultimate effective management of BMs via the utilization of advanced technologies/therapeutics. Advanced diagnostic approaches involve non-invasive techniques presenting superior accuracy or precision with reduced risk of infection or injury. Bladder malignancy recognition at initial phases with better precision or accuracy has been assisted upon utilization of combinatorial techniques such as combined optical and cancer-specific imaging techniques, combinatorial use of TURBT, and artificial intelligence-based systems like CystoNet during cystoscopy, FISH-based earlier recognition of residual cancer cells with sensitivity or specificity level of 68% and 64% along with the employment of tumor specific biomarkers (UroVysion) for preventing tumor recurrence/relapse. Conjugated/cooperative working of researchers from multidisciplinary fields remarkably improved oncological assessments regarding the deep evaluation of underlying tumor mechanisms and possible diagnostic and therapeutic approaches that might successfully be employed for the effective management of BMs. The use of 3D culture models provides accurate simulation of TME with precise identification of patient-specific mutations, developing a pathway towards personalized/targeted drug therapy and effective treatment of bladder carcinomas. Similarly, UroVysion marking systems exhibit the capability of predicting postsurgical cancer progression from nonmuscle-invasive bladder cancer to a muscle-invasive phase, thus provoking positive FISH findings as a potential tool for determining relapse risk related to bladder carcinomas. On the other hand, conjugated regimens based on intravesical chemotherapy and immunotherapy, viral and bacterial intravesical therapies and non-invasive urinary drug-disposing techniques (use of bioinert peptide) have been employed for effective management of NMIBC. Furthermore, modulation of bladder functioning through Piezo channels (Piezo1 and Piezo2) regulation with ultimate regulation of bladder filling/voiding phenomenon via mechanosensitive signaling and effective management of MUC patients via HER2-selective therapy indicating auspicious outcomes like improved safety/clinical profiles associated with RC48-ADC treatment upon comparison to standardized treatment. Though multiple therapies and diagnostic tools are available for the efficacious management of BMs; however, selection of tools or treatments ultimately depends upon the patient’s willingness and requirement, which is why the utilization of combinatorial strategies is gaining preferences. Finally, dual diagnostic and therapeutic advancement involving conjugated use of multiple tools, devices, and treatments are considered to be a driving force for earlier and accurate/precise detection/prediction of bladder dysfunctions.
Footnotes
Abbreviations
ADCs: Antibody-drug conjugates;
Author’s Contribution
All three authors wrote the main manuscript text, prepared figures, and reviewed the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and Informed Consent
Not applicable.
Funding and Acknowledgments
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are thankful to the Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2024R73), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia. We extend our appreciation to the Deanship of Graduate Studies at Ajman University, Ajman, United Arab Emirates, for their support for publication charges.
