Abstract
Objectives
The current study aims to establish the pharmacognostic profile and evaluate the cytotoxic potential of Parmelia sulcata Talyor in the glioblastoma cell line.
Materials and Methods
In this experimental investigation, pharmacognostic evaluation has been conducted via standard procedures, and active fractions obtained through column chromatography, further identified by liquid chromatography–mass spectrometry (LC–MS). The total antioxidant capacity of the ethanolic extract of the lichen was obtained via the phosphomolybdate method, and a reducing power assay was conducted to determine its in vitro antioxidant potential. The glioblastoma U87-MG cell lines were cultivated in minimum essential medium supplemented with nonessential amino acids, and 10% fetal bovine serum was maintained under controlled conditions. 3-(4,5-Dimethyl thiazolyl-2)-2,5-diphenyltetrazolium bromide assay was used to test the cytotoxicity of ethanolic extract of lichen and its isolated active fraction.
Results
The pharmacognostic profile of P. sulcata inhabiting in lithophytic life form has been established. Morphological parameters aided in studying the physical attributes of lichen, whereas microscopic parameters helped to determine the histological characteristics. LC–MS analysis of purified fractions confirms the presence of phenolics in the lichens responsible for anticancer activity. Pharmacological data on U87-MG cell lines revealed the possible usage of this lichen in the treatment of glioblastoma and its associated pathological conditions. Lichen extract and its purified fraction (L16–L21) significantly (p < 0.05) inhibited the proliferation of U87 glioblastoma cells in a dose-dependent manner.
Conclusion
This study reported that the shield lichen has promising cytotoxic activities in U87-MG cell lines, which was marked by a reduction in cancer cell proliferation. Further pharmacognostic and phytochemical data confirmed its identity and facilitated its inclusion in various Pharmacopoeias.
Keywords
Introduction
Lichens have been a source of food, medicine, and dyeing since immemorial. 1 These symbiotic organisms inspired drug discovery due to their distinct types of chemical constituents. 2 The increasing prevalence of exploring natural products in managing several diseases would raise questions ascertaining its standardization, as adulteration is quite prominent in herbal products. To ensure the quality and purity of any herbal formulation, it must pass quality parameters as per Pharmacopoeias. Pharmacognostic studies of herbs, herbal products and herbal formulations will help ensure the identity and quality of any product/preparation. Evaluation via morphology, microscopy, and physico-chemical parameters is among the important facets of pharmacognostic evaluation. 3 Lichens from the Parmelia genus have been used in dyeing and food and explored ethnomedicinally and therapeutically. They exhibited significant pharmacological activities when tested in different in vitro and in vivo models. 4 The most common examples of therapeutically active Parmelia species include Parmelia saxatilis, Parmelia caperata, 5 Parmelia reticulata, 6 and Parmelia sulcata. 7 Considering their therapeutic applications, pharmacognostic standardization becomes the first and foremost criterion concerning its purity.
P. sulcata Taylor, belonging to the family Parmeliaceae, is a saxicolous lichen distributed among rocks and tree surfaces, generally has hammer-shaped and sharp lobes, and is widely spread across India, China, Thailand, and Malaysia.8, 9 It has been documented that due to the resemblance of the morphology of P. sulcata with the human brain, it has been used ethnobotanically in treating cranial disorders, which also justifies the principle “Doctrine of Signatures.”10–12 Glioblastoma is one of the devastating types of cancer of the brain and spinal cord affecting persons among all age groups irrespective of sexes. 13 The surgical procedure followed by radiation and chemotherapy for several months is the current therapeutic approach in glioblastoma. This therapy has reported some severe adverse effects, and also chances of remittance have been reported. 14 Natural products are generally considered safer alternatives in such situations, showing minimum side effects, but their efficacy should be tested clinically before usage. 15 The use of alternative therapeutic approaches in treating or controlling the symptoms associated with this disease would help treat patients suffering from this disease with minimal side effects. Parmelia lichen has also shown its ability to normalize and treat brain-related disorders. Lichen-derived constituents can cross the blood–brain barrier (BBB) and have profound applications in central nervous system (CNS)-related disorders. 16 Apart from this, P. sulcata commonly known as shield lichen is reported for antioxidant, antimicrobial, and cytotoxic activities. Several therapeutic constituents like salazinic acid, 17 phenolics, 18 atranorin, 19 volatile oils, and so on 17 have been reported from this lichen.
The phenolics present in the methanolic extract showed free radical scavenging activity involving the 2,2-diphenyl-1-picrylhydrazyl (DPPH) method. 20 It has also been observed that P. sulcata, Parmeliaceae, commonly known as hammered shield lichen, 21 is cytotoxic to PC3, Hep3B, and A549 cell lines. 7 Furthermore, the methanolic extract of shield lichen exhibited genotoxicity in MCF-7, MDA-MB-231 cell lines. 17 Silver nanoparticles synthesized from its extract showed significant apoptosis in NIH-3T3 and MCF-7 cell lines. 22 In a study, Lichen extracts (Hypogymnia physodes, Evernia prunastri, and P. sulcata) and their derived constituents were reported to inhibit the Wnt/β-Catenin pathway in glioblastoma (cancer associated with brain and spinal cord) and improve the anticancer activity involving Temozolomide. 23 Very few studies have been conducted concerning the standardization of shield lichen and investigating the therapeutic potential of its biologically active fractions and extracts in glioblastoma. Therefore, this present study will focus on unveiling its pharmacognostic profile and phytochemical analysis and also exploring its therapeutic usage concerning its ethnomedicinal relevance in brain-related disorders, particularly in glioblastoma.
Materials and Methods
Collection and Authentication
Shield lichen, inhabited as a lithophytic life form, was collected from the Kharota region of District Kangra, Himachal Pradesh, India. Lichen samples were photographed in their natural habitats, and the herbarium of the collected sample was prepared and authenticated by the Council for Scientific and Industrial Research-National Institute of Science Communication and Policy Research (CSIR-NIScPR) New Delhi, India, bearing authentication reference no. NIScPR/RHMD/Consult/2023/4429-30-2.
Scanning Electron Microscopy (SEM) Examination
The powdered sample of rock and lichen was subjected to SEM analysis. The sample stub was prepared by placing carbon tape on top of the stub, creating an adhesion surface area to fix the sample. The sample was then fixed on the stub and the excess powder was blown off using an air blower. The stub was then placed on the sample holder and taken for SEM analysis. The sample was visualized at different magnifications, and the images were recorded for analysis. To study its detailed anatomy, the lichen adhered to the rock was also magnified under SEM (27×–4,400×). 24
Characterization (Chemical Analysis) of Rock
Loss on ignition was determined in the sample to predict volatile impurities by calculating the loss in weight after ignition in a muffle furnace as per the standard procedure of the Bureau of Indian Standards (BIS) 1760 (Part 1): 1991. 25 For the determination of SiO2, gravimetric method has been used following BIS (IS) 1760 (Part 2): 1991. 26 Content of CaO, Fe2O3, and Al2O3 was evaluated by titrimetric methods prescribed in BIS IS 1760 (Part 3): 1991. 27
Microscopic Characters of P. sulcata
Histological studies on lichen thalli and powder have been conducted using an Olympus Magnus CH-20 i, Trinocular, microscope. Freshly collected lichen samples were selected for the study, and fine powder was used for microscopic examination.
Pharmacognostic Parameters
Standardization parameters, namely, moisture content determination, ash value, extractive value, and swelling index have been performed as per quality control methods on medicinal plant materials, WHO, 1998. 28
Phytochemical Screening
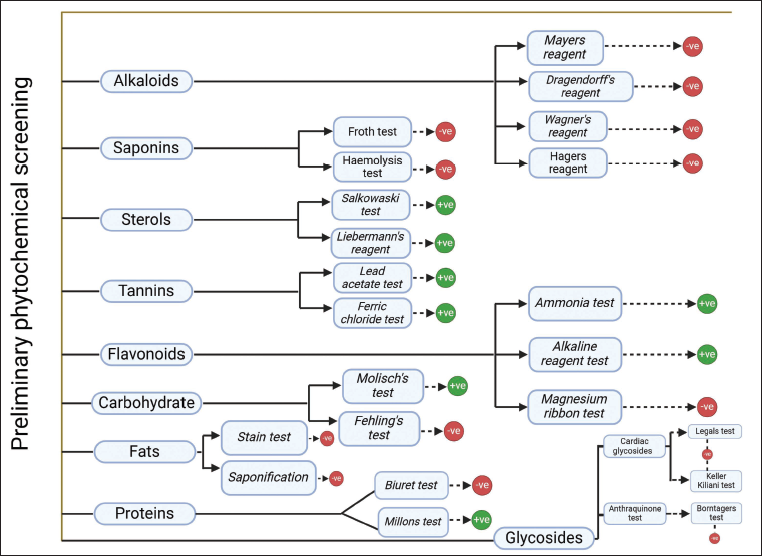
Preliminary phytochemical screening on the ethanolic extract of lichen has been conducted to identify the presence of primary and secondary metabolites. 29
Extraction and Isolation
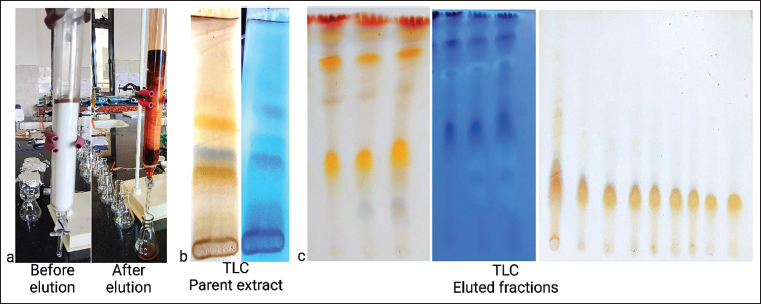
Fully dried lichen was extracted with ethanol via soxhlation. Purification was done using a sintered glass column, which has a length of 40 cm and a diameter of 30 mm (Figure 1a). The column was packed with the stationary phase (silica gel 100–200 mesh size) and eluted via gradient elution involving CHCl3:MeOH:H2O (8:2:0.01–8:8:0.8) as the mobile phase. Purified fractions were collected and further subjected to liquid chromatography–mass spectrometry (LC–MS) analysis. Thin layer chromatography (TLC) was performed on ethanolic extract (Figure 1b) as well as on eluted fractions (Figure 1c), and detection was done by 5% H2SO4 reagent and observation under ultraviolet (UV) light (366 nm).
Isolation of Lichen Constituents. a. Column Chromatography Set Up b. Thin Layer Chromatography (TLC) of Ethanolic Extract of Lichen c. TLC of Eluted Fractions.
LC–MS Analysis
Agilent high-performance liquid chromatography (HPLC) coupled with a UV-vis and a Waters MicroMass quattro microTM atmospheric pressure ionization (API) tandem quadrupole mass spectrometry (MS) (Waters Co., Milford, MA, USA) interface via electrospray ionization (ESI) source was used for qualitative analysis. The purified fraction (L16–L21) exhibiting anticancer activity was subjected to LC–MS analysis, and its chemical composition was determined. The separation was achieved on the Waters X-Bridge C18 column (50 × 4.6 mm, 3.5 µm). An optimized gradient mobile phase consists of 0.1% formic acid in water (solvent A) and acetonitrile (solvent B) delivered at 1.2 mL/min flow rate. The following gradient was applied from 15% to 75% B (0.1–6 min), 75% to 75% B (6–8 min), 75% to 15% B (8–11 min), and 15% to 15% B (11–15 min). The MS full scan was acquired on the triple quadrupole (QqQ) MS. The applied parameters for the electrospray (ES) source are as follows: capillary voltage 3.54 kv, cone potential 33 V, extractor 3 V, source temperature 110°C, desolvation temperature 350°C, and desolvation gas 750 L/h, cone gas 50/h, and scan time 0.2 s. Masslynx V4.1 software was used for the data acquisition and analysis.
Pharmacology
Total Antioxidant Capacity
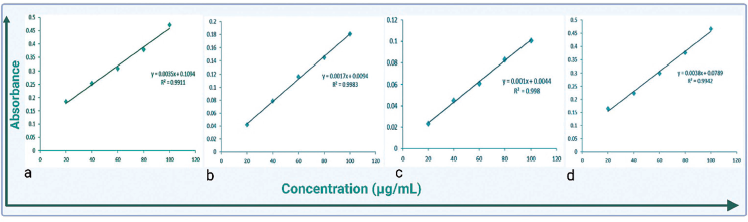
The total antioxidant capacity (TAC) of the ethanolic extract of P. sulcata was evaluated by the phosphomolybdenum method using ascorbic acid as a standard. The absorbance of the solution was measured at 695 nm, and the results were obtained in the form of a graph.30, 31
Reducing Power Assay
The reducing power of ethanolic extract of lichen was determined by ferric reducing antioxidant power assay following the methodology adopted by Aparadh et al. 32 and Philip et al. 33 The reduction of ferric ions to ferrous ions is brought about by the presence of reductants in the solution. An increase in absorbance with concentrations indicates lichen’s high reducing power. The absorbance was measured at 700 nm using a UV Systronics double-beam UV-vis spectrophotometer 2,201 and the results were obtained.32, 33
U87-MG Cell Lines
The human glioblastoma cell line U87-MG was procured from The National Centre for Cell Science, Pune, India, cultured in minimum essential medium (MEM) supplemented with nonessential amino acids and 10% fetal bovine serum (FBS). After attaining desirable mass, cells were further harvested with Dulbecco’s phosphate buffered saline (DPBS), 0.25% Trypsin-ethylenediaminetetraacetic acid (EDTA) solution and seeded in 96-well plates.
Cytotoxicity Assay
The cytotoxic assay was performed following the methodology adopted by Gerlier and Thomasset 34 and Khazaei et al. 35 and adhering to the instructions of ATCC, USA (American type culture collection) for 3-(4,5-dimethylthiazolyl-2)-2,5-diphenyltetrazolium bromide (MTT) assay. The percentage viability and concentration plots were observed, and visible colonies were photographed using a Binocular Microscope-XDFL series, which were counted using ImageJ (Fiji) software V1.53j. The same procedure was also performed on the purified fraction of P. sulcata (L16–L21) starting from 15.625 to 500 µg/mL concentrations to check its cytotoxic potential.34, 35
Acridine Orange and Propidium Iodide Staining
Acridine orange and propidium iodide (AO/PI) staining was carried out to confirm apoptosis in U87-MG cell lines by lichen fraction. U87-MG cells were seeded in six well plates at a density of 3 × 10 5 cells/2 mL and incubated in CO2 at 37°C for 24 h. The cells were treated with a selected concentration of purified fraction (L16–L21) in 2 mL of culture medium incubated for 24 h. Untreated and control groups were designated, and at the end, the cells were harvested into centrifuge tubes. This was followed by adequate washing with DPBS and centrifugation. The excess DPBS was discarded and the cells were finally incubated in an AO/PI staining solution for several minutes. The cells were photographed under a fluorescence microscope (XDFL series), and the results were recorded.36, 37
Statistical Analysis
Each experiment had three replicates, and data were shown as mean ± standard deviation (SD). To determine the anticancer activity of lichen, statistical analysis was conducted using a t-test/one way analysis of variance and nonlinear regression analysis through GraphPad Prism 7, and p < 0.05 indicated a statistically significant difference.
Results
Lichen Morphology
This foliose lichen is often inhabited in saxicolous habitats with an orbicular adnate thallus up to 4–8 inches in diameter. The thallus is usually covered with distinctive reticulate pseudocyphellae. Granular soredia (reproductive structures of lichens) are abundantly spread. The lichen has a furless white to gray-colored upper surface and a brownish to blackish wrinkled lower surface with a characteristic odor. The morphological characteristics are depicted in Figure 2A (a–c).
A. Morphology of Parmelia sulcata. a. Lithophytic Lichen b. Upper Surface c. Lower Surface. B. Scanning Electron Microscopy (SEM) Micrographs. a. Rock Sample b. Powdered Lichen c. Saxicolous Lichen (L—Lichen, R—Rock). C. Microscopy of Lichen. a–e. Powder Characteristics of Lichen Showing Fragmented Thalli, Crystals f. Medulla Under Magnification (FH—Fungal Hyphae).

SEM Examination
The microstructural study detected the fine-grained mica and microcrystalline quartz crystals in the foliated metamorphic rock sample as shown in Figure 2B (a). Fine lichen powder was detected the presence of medullar fragments as indicated in Figure 2B (b). Photomicrograph of lichen adhered on rocky substrate depicted in Figure 2B (c) in which complex networking of loosely arranged medulla (central region of thallus containing fungal hyphae) was observed under SEM.
Chemical Analysis of Rock
The elemental composition of the selected rock is chiefly composed of SiO2 (76.76%), Al2O3 (17.98%), Fe2O3 (2.06%), and CaO (0.58%) as determined by gravimetry and titrimetric methods suggested by BIS. Loss on ignition was found to be 3.15%, indicative of organic matter in the sample.
Microscopy of Lichen
In the fine powder of the lichen, fragmented thalli, fibers, and scattered crystals were visible at varying magnifications. Medulla (loosely oriented fungal hyphae) was observed in the thin section of saxicolous lichen. Detailed microscopic characters are represented in Figure 2C (a–f).
Pharmacognostic Parameters
Pharmacognostic data facilitates the framing of the standardization profile of shield lichen. The total ash value of ethanolic extract of lichen was found to be 1.27% w/w, representing inorganic content in the saxicolous lichen. Moisture content in the powdered sample was found to be 4.0% w/w. The alcohol and water-soluble extractive values of lichen were found to be 4.4% w/w and 13.5% w/w respectively, indicative of more water-soluble constituents in the sample than alcohol. The lichen is devoid of any mucilage and saponins, as confirmed by swelling index and foaming index values.
Phytochemical Screening
Preliminary phytochemical screening of the ethanolic extract of lichen revealed the presence of several secondary metabolites, namely, carbohydrates, proteins, sterols, tannins, and flavonoids, as indicated in Figure 3.
Extraction and Isolation
Shade-dried, powdered shield lichen was subjected to soxhlation using ethanol as solvent to obtain the desired extract. Purified fractions were obtained by column chromatography and identified by TLC. Composition of CHCl3:MeOH:H2O (8:2:0.2) seemed the appropriate mobile phase for ethanolic extract, which showed the presence of five distinct color spots at Rf 0.63, 0.52, 0.45, 0.34, 0.23 after detection with 5% H2SO4. Separation was carried out to screen chemical constituents present in the lichen, and around 40 fractions were collected and were mixed as per corresponding Rf values. Finally, the most purified fractions obtained from L16 to L21, having similar Rf were mixed and subjected to LC–MS analysis.
Assessment of Lichen Constituents via LC–MS
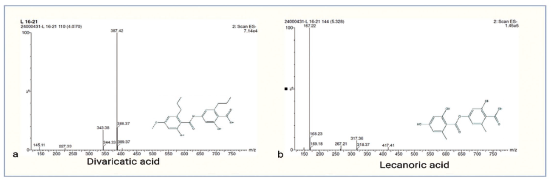
The purified fraction (L16–L21) exhibiting anticancer activity was subjected to LC–MS analysis, and its chemical composition was determined. Compound A, eluted at Rt 4.07, the molecular weight of 388 Da was determined based on m/z 387.4 [M-H]+ adduct formed under ESI negative ion mode (Figure 4a). The literature survey revealed that compound A had been previously reported from Parmelia pulla identified as divaricatic acid38, 39 and showed antitumor activity against melanoma cells. 40 Moreover, compound B eluted at Rt. 5.32, molecular weight 318 Da was determined based on m/z 317 [M-H]+, adducts formed in ESI negative ion mode (Figure 4b). The literature survey supported that compound B was commonly found as lecanoric acid in several Parmelia species like Parmelia flaventior, Parmelia subrudecta,41, 42 and was reported for its anti-proliferative activity. 43
Mass Spectra of L16–L21. a. MS1 Spectra of Compound A [Electrospray Ionization (ESI) Negative Ion Mode] b. MS1 Spectra of Compound B (ESI Negative Ion Mode).
Pharmacology
Total Antioxidant Capacity
TAC of shield lichen was analyzed via phosphomolybdenum assay. Ascorbic acid was used as a reference, and a calibration curve was obtained. Ethanolic extract of lichen showed absorption of 0.181 ± 0.014 at 100 µg/mL in comparison with ascorbic acid (standard), which showed more absorbance (0.472 ± 0.007) at the same concentration as shown in Figure 5(a–b).
Total Antioxidant Capacity (TAC) and Ferrous Reducing Power Assay (FRP) Data. a. TAC of Ascorbic Acid b. TAC of Ethanolic Extract of Lichen c. FRP of Ascorbic Acid d. FRP Ethanolic Extract of Lichen.
Reducing Power Assay
Ethanolic extract of shield lichen showed a dose-dependent increase in reducing power capacity marked in Figure 5(c–d), having an absorbance of 0.101 ± 0.014 at 100 µg/mL in comparison with ascorbic acid, which showed maximum absorbance of 0.467 ± 0.010 at the same concentration.
MTT Assay
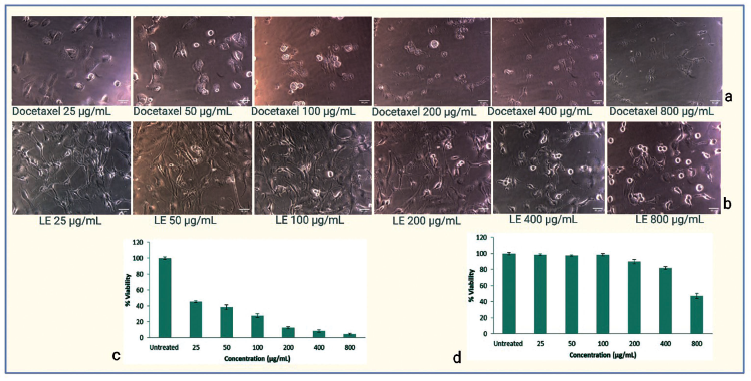
Ethanolic lichen extract was tested against U87-MG cell lines at different concentrations, namely, 25, 50, 100, 200, 400, and 800 µg/mL. The results were compared with the already established standard drug Docetaxel at the same concentration. P. sulcata exhibited cytotoxicity against U87-MG having an IC50 value of 797.43 µg/mL in comparison with Docetaxel, which has an IC50 value of 16.77 µg/mL. Lichen ethanolic extract significantly inhibited the proliferation of U87 glioblastoma cells in a dose-dependent manner. Detailed results are represented in Figure 6 (a–d).
3-(4,5-Dimethylthiazolyl-2)-2,5-Diphenyltetrazolium Bromide (MTT) Assay Data of Standard and Extract. a. Treatment of U87-MG Cell Lines with Docetaxel b. Treatment of U87-MG Cell Lines with Lichen Extract (LE) c. MTT Assay Displays the Percentage Viability of U87-MG Cells at Varying Concentrations of Docetaxel d. MTT Assay Displays the Percentage Viability of U87-MG Cells at Varying Concentrations of Ethanolic Extract of Lichen.
Purified fraction (L16–L21) was found to have IC50 of 458.48 µg/mL and showed significant cytotoxicity against U87-MG cell lines. The cell lines were tested over a range of 15.625–500 µg/mL, having an incubation time of 24 h, as shown in Figure 7(a–c) and compared with untreated ones. The purified fraction significantly (p < 0.05) inhibited the proliferation of U87-MG glioblastoma cells in a dose-dependent manner.
3-(4,5-Dimethylthiazolyl-2)-2,5-Diphenyltetrazolium Bromide (MTT) Assay Data of L16–L21 and Acridine Orange and Propidium Iodide (AO/PI) Staining. a. Treatment of U87-MG Cell Lines with Purified Fraction b. U87-MG Untreated Group. c. MTT Assay Displays the Percentage Viability of U87-MG Cells at the Specified Purified Lichen Extract (L16–L21) Concentrations d. Represents AO/PI Staining (Untreated, Standard, and L16–L21 Groups) Results.
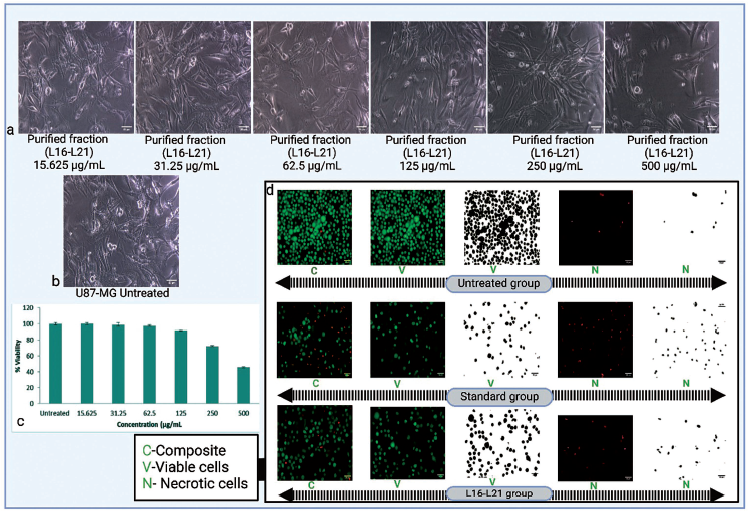
AO/PI Staining
To identify apoptosis in treated and untreated U87-MG cells, AO/PI fluorescence labeling was utilized, as demonstrated in Figure 7d. The purified fraction (L16–L21) significantly inhibited the proliferation of U87-MG glioblastoma cells, as evidenced by necrotic cells which appeared red in the staining process. The percentage of the live cell population declined in the treated group when compared with the untreated one. The results show that P. sulcata showed promising anti-proliferative effects in the U87-MG glioblastoma cell line.
Discussion
This research aimed to examine the cytotoxic potential of P. sulcata inhabited in lithophytic life forms in glioblastoma cell lines (U87-MG). Based on our findings, lichen-derived ethanolic extract and purified fraction can cause significant cytotoxicity in glioblastoma cell lines. Previous studies related to cell lines on this lichen indicated that this lichen has a potential source in cancer therapy, particularly in breast,8, 17 lung, and liver carcinomas. 7 Several constituents have also been reported from this lichen, namely, terpenes, 17 phenolics, 19 and flavonoids, 22 contributing towards its therapeutic potential. In the current study, the lithophytic life form of P. sulcata was identified and the samples were authenticated. Chemical analysis of rocky substrate and SEM studies on rock and lichen help in affirming the identity of the lithophytic form. The pharmacognostic profile has been established and the collected samples were subjected to extraction. U87-MG cancer cell lines were selected, and the ethanolic extract was tested involving MTT assay and its cytotoxicity was determined (IC50 797.43 µg/mL). TAC along with a reducing power assay on the crude extract was performed which showed its antioxidant potential. The crude extract was further fractionated via column chromatography to obtain the purified and several other fractions. LC–MS revealed the presence of phenolics like lecanoric acid and divaricatic acid in the purified fraction, and its cytotoxic potential was evaluated in selected cancer cell lines using an MTT assay. AO/PI staining method was also employed to check its cytotoxic behavior. It was found that the purified fraction at a higher dose (IC50 458.48 µg/mL) could cause significant cytotoxicity in U87-MG cell lines. However, more scientific studies should be conducted on this species to unveil its immense therapeutic potential.
Conclusion
Our investigations revealed that the ethanolic extract of P. sulcata and its active fraction showed prominent inhibition in the human U87-MG glioblastoma cells. Thus, more clinical research on this lichen should be encouraged as it may be a useful candidate source for creating a novel chemotherapeutic agent for glioblastoma.
Footnotes
Abbreviations
Acknowledgments
The authors acknowledge the continuous support and encouragement from Prof. Vivek Sharma, Govt. College of Pharmacy Rohru; Prof. Vinay Thakur, Govt. Pharmacy College Kangra at Nagrota Bagwan, Himachal Pradesh, India, and the Department of Pharmaceutical Sciences, CT University Ludhiana, Punjab, India. The authors highly appreciated the LC-MS and SEM facilities provided by Poornayu Research Labs and Dextrose Laboratories in Bangalore, India.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
