Abstract
Background
Oxidative stress is the primary factor responsible for the development of neuroinflammation and brain deficits. As aging occurs, the chances of oxidative stress increase due to lifestyle modification, environmental, and genetic factors. The brain is especially susceptible to oxidative damage because of its elevated oxygen consumption, lipid composition, and comparatively inadequate antioxidant defenses. Oxidative stress associated with aging results in DNA damage, mitochondrial dysfunction, and compromised repair mechanisms.
Objectives
To investigate the efficacy of Cordyceps militaris extract (CME) and alpha-lipoic acid (ALA) in mitigating oxidative stress and neuroinflammation in aged rats and to assess their potential therapeutic effects on cognitive performance, antioxidant enzyme activity, and oxidative stress markers.
Materials and Methods
This study involved using aged rats to evaluate the impact of CME and ALA on oxidative stress and neuroinflammation. The rats were administered CME and ALA, and various assessments were conducted to measure cognitive performance, antioxidant enzyme activity, and oxidative stress markers.
Results
The administration of CME and ALA to aged rats significantly improved cognitive performance. Additionally, there were an enhancement in antioxidant enzyme activity and a reduction in oxidative stress markers. These findings indicate that both CME and ALA positively mitigate oxidative stress and neuroinflammation.
Conclusion
CME extract and ALA exhibit therapeutic potential in treating aging-related oxidative stress and inflammation. Their use could be beneficial in combating brain disorders caused by aging, highlighting their importance in the field of geriatric healthcare.
Introduction
At some stage in our lives, we all experience the process of aging, and professionals need to understand its clinical and epidemiological importance. By 2050, there are expected to be 88.5 million people over the age of 65, which would increase demand for hospitals and healthcare services. 1 Senescence causes several body system changes, which need particular attention and control. This leads to the loss of function, physiological and biochemical harm, and the onset of certain age-related illnesses. 2 Among the different hypotheses related to aging, the theories concerning oxidative stressors and free radical accumulation are particularly noteworthy. 3 Progressive declines in memory and mobility are two of the most prevalent effects of neurodegenerative illnesses associated with aging, which include Alzheimer’s disease (AD), vascular dementia, Huntington’s disease, Parkinson’s disease, and amyotrophic lateral sclerosis. Oxidative stress has been shown to play a role in the development of impaired memory. 4 Many studies have examined the relationship between specific biomarker levels associated with oxidative stress and cognitive function as assessed by the cognitive impairment examination. A prior study found a correlation between more significant levels of inflammatory molecules and oxidative stress markers like glutathione peroxidase and malondialdehyde (MDA) with poor mental function in older institutional residents. 5 Reactive oxygen species (ROS) and glutathione (GSH) levels may rise due to poor glutathione peroxidase activity, as GSH is a substrate for the enzyme. 6
Cordyceps spp. mushrooms have been utilized as natural ingredients in Asian traditional medicine for many years because of their adaptogenic and tonic qualities, their capacity to alleviate fatigue, and their ability to boost the human immune system.
7
Of the roughly 500 Cordyceps species, Cordyceps sinensis has received the most scientific attention. This particular species has been listed in the Chinese Pharmacopoeia (2015) and is notably rich in adenosine, making it the key quality measure. However, because of a simpler and more sustainable culture, uniform quality, and higher quantity of medicinal substances like cordycepin, Cordyceps militaris is frequently favored over C. sinensis. It is also less costly than C. sinensis, which is rarer and more expensive.8, 9 Based on the studies, there are many biologically active compounds like cordycepin, ergothioneine,
Materials and Methods
Drugs and Chemicals
Quercetin, gallic acid, nitrogen blue tetrazolium, and 2,2-diphenyl-1-picrylhydrazyl (DPPH) were purchased from HiMedia, India. Folin–Ciocalteu’s reagent was purchased from Central Drug House, India, and alpha-lipoic acid (ALA) was purchased from Setu Nutrition Pvt. Ltd., India. All other chemicals used in the current study were of analytical grade.
Sample Collection
C. militaris used in this study were procured and authenticated from Indian Council of Agricultural Research (ICAR): Directorate of Mushroom Research, Solan, Himachal Pradesh, India [Accession Number: DMRO 1163]. Further mushrooms were cleaned with distilled water followed by air dried, grinded into coarse powder, and stored in an air-tight container for further use.
Extraction
The extraction was done with the help of a Soxhlet extraction apparatus. The hydroethanolic solvent was used (i.e., 50% ethanol and 50% water). A total of 1,000 g of fresh mushroom was obtained and then air-dried at normal room temperature until fully dried, and then grind by the help of electric grinder and a total 200 g of powder was obtained. The 200 g of powdered sample was loaded into the thimble. The extraction process was run for 8 h at 60–70℃. Filter the extract using Whatman filter paper and was evaporated through rota evaporator. The collected extract was then further stored in deep freezer at −20℃. 14
Animals and Treatments
Before the animal research began, the protocol for the experiment was approved by the Maharishi Markandeshwar (MM) College of Pharmacy’s Institutional Animals Ethics Committee (Protocol no. MMCP-IAEC-110, dated 20/11/2021) and carried out in accordance with the regulations on animal experimentation issued by the Committee for Control and Supervision of Experiments on Animals (CCSEA), Government of India. Forty-eight Wistar rats, young (4–6 months old, body weight 150–250 g) and aged rats (18–24 months, body weight = 300–350 g) were kept in typical laboratory settings with controlled humidity, temperature, and a natural light and dark cycle (between 22°C and 25°C and 50% relative humidity). Before conducting any behavioral tests, the rats were acclimatized to the laboratory environment. The eight groups listed below comprised the rats (n = 6).
Group I (young rats): 4–6-month-old rats received ad libitum diet.
Group II (aged rats): 18–24-month-old rats received ad libitum diet.
Group III (aged + CME 125): Cordyceps militaris extract (CME) was administered (100 mg/kg p.o.) daily to aged rats for 3 months.15, 16
Group IV (aged + CME 250): CME (250 mg/kg/day p.o.) was administered to aged rats daily for 3 months.15, 16
Group V (aged + CME 500): CME (500 mg/kg/day p.o.) was administered to aged rats daily for 3 months. 15
Group VI (aged + ALA 50): ALA (50 mg/kg/day p.o.) was administered to aged rats daily for 3 months. 17
Group VII (aged + ALA 100): ALA (100 mg/kg/day p.o.) was administered to aged rats daily for 3 months. 18
Group VIII (aged + CME 125 + ALA 50): CME (125 mg/kg/day) and ALA (50 mg/kg/day) were administered in combination daily to aged rats for 3 months.
After 3 months of treatment, all the behavioral parameters (assessment of depression, anxiety, memory, and pain) were performed, and animals were fasted overnight for 12 h and then blood was collected for biochemical and molecular estimations. At the end, all the rats were sacrificed and the brain was isolated for biochemical, molecular, and histopathological analysis.
Analysis of Behavioural Parameters
All behavioral parameters were analyzed using various different procedures. The Morris water maze was performed as earlier described by Vorhees and Williams19, 20 and passive avoidance test was performed as earlier described by Eagle et al. 21 to evaluate memory impairment. For measuring the anxiolytic behavior elevated plus maze test was performed as described by Walf and Frye. 22 To measure the immobility time in an unescapable environment, forced swim test was performed as earlier described by Porsolt et al. 23 The locomotor activity was measured by using a digital actophotometer described by Bhosale et al. 24 The motor coordination was assessed using the rotarod apparatus as described by Bansal and Chopra. 25 To assess the thermal nociception, a tail immersion test was performed as earlier described by Courteix et al. 26 and mechanical nociception via Randall–Selitto test by Santos-Nogueira et al. 27
Analysis of Biochemical and Molecular Estimations
Brain samples were washed with ice-cold saline solution containing 0.9% sodium chloride. These samples were homogenized in chilled phosphate buffer with a potential of hydrogen (pH) value of 7.4. The homogenized samples were used to measure various parameters including lipid peroxidation,25, 28 superoxide dismutase (SOD),25, 29 reduced GSH,25, 30 catalase,25, 31 acetylcholinesterase (AChE) activity,25, 32 and tumor necrosis factor-alpha (TNF-α) 25 were measured. The hippocampal and cortical regions of the brain were also used to measure necrosis factor kappa B 65 [nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB)] activity. 33
Histopathological Analysis
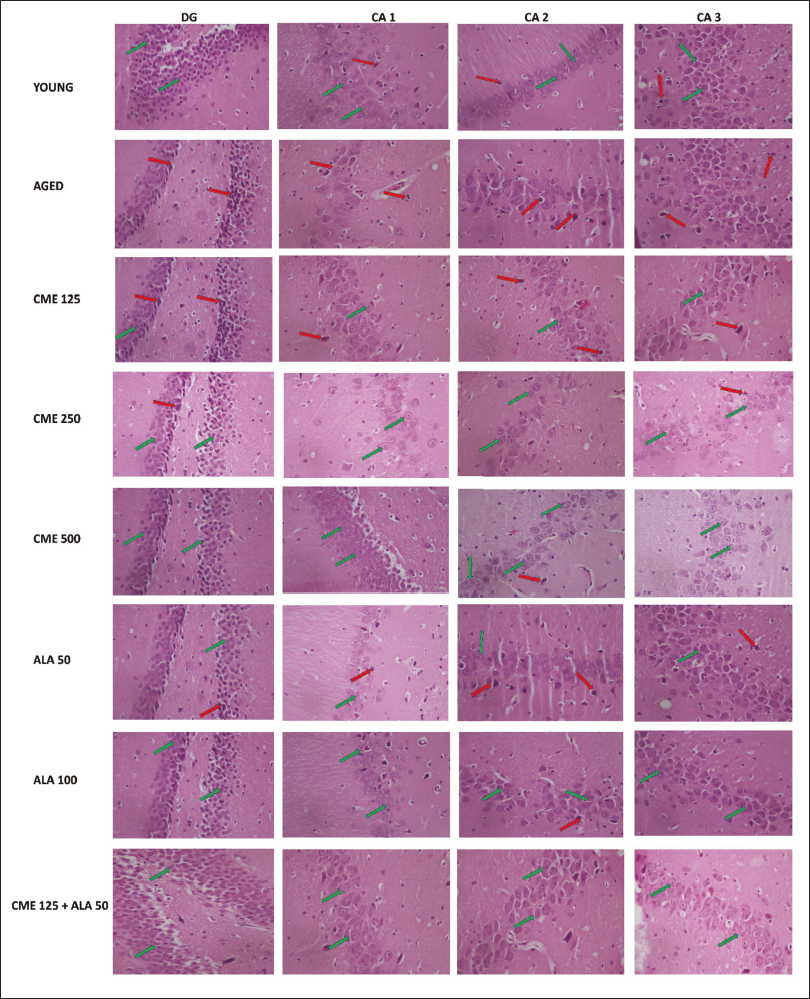
The histopathological analysis of the hippocampal region of the rat brain sample was performed by first fixing the brain tissue in formalin, embedding it in paraffin, and slicing it into thin sections. These sections were then stained with hematoxylin and eosin (H&E), enabling the visualization of cellular structures under a microscope and the assessment of neuronal morphology, tissue integrity, and pathological changes in the hippocampus. Additionally, the number of densely stained, irregularly shaped pyknotic neurons was counted at a 200 µm scale bar using a 40× objective. 34
Statistical Analysis
The results were shown as means ± standard error of the mean. The behavioral, biochemical, and histopathological evaluations were analyzed using a one-way analysis of variance (ANOVA), followed by Tukey’s post hoc test, using GraphPad Prism software version 5.01.
Results
Effect of CME on Behavioural Parameters
Morris Water Maze Test
In comparison to a younger group of rats, the aged rats on day 5 had substantially (p < .001) higher escape latencies (EL) and less time spent in the target quadrant (TSTQ), as seen in Figure 1. In comparison to the aged group rats, CME therapy (250 and 500 mg/kg) and ALA (100 mg/kg) significantly (p < .001) reduced EL and increased TSTQ on day 5. Furthermore, compared to the aged group rats, CME+ALA-treated rats showed a substantial decrease (1.7-fold) in EL and an increase (2.6-fold) in TSTQ (p < .001). Rats treated with CME (125 mg/kg) and ALA (50 mg/kg) showed a notable decrease in EL and an increase in TSTQ; however, the impact was not statistically significant.
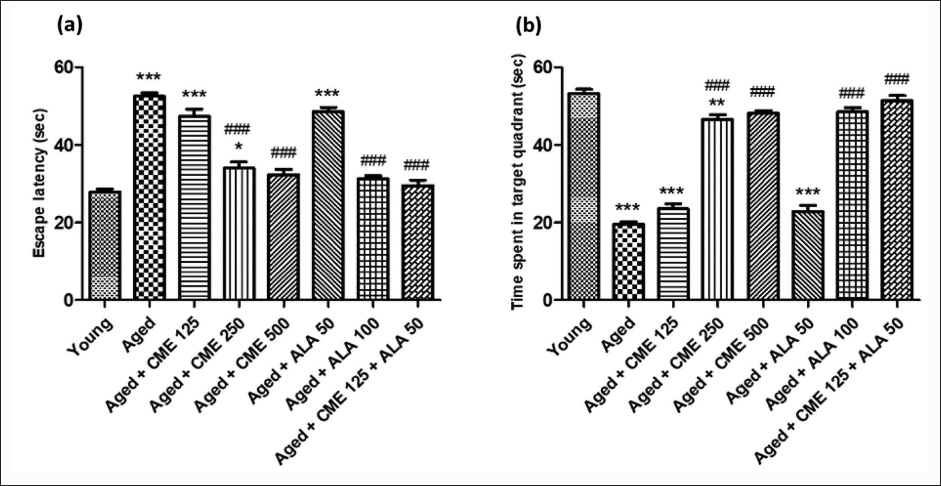
Passive Avoidance Test
During the passive avoidance test, step-down latency (SDL) was measured, representing the time the animal took to step down. The prolongation of SDL was taken as a parameter for learning. SDL significantly decreased (3-fold), while the number of errors significantly increased (4-fold) in aged rats in contrast to the young group of rats (Figure 2). Further, SDL was enhanced considerably (p < .001). At the same time, several errors were significantly attenuated (p < .001) in aged rats after treatment with CME (250 and 500 mg/kg) and ALA (100 mg/kg) for 3 months dose-dependently compared to the aged group rats. A marked increase in SDL and a decrease in the number of errors were observed in CME (125 mg/kg) and ALA (50 mg/kg) treated rats, but the effect was not at a significant level. However, when CME (125 mg/kg) and ALA (50 mg/kg) were given in combination, a substantial rise in SDL (2.9-fold) and a decrease in the number of errors (3.2-fold) were the key observations in these rats in contrast to the aged group of rats.
Effect of CME on anxiety (elevated plus maze) and depression (forced swim test) in aged rats.
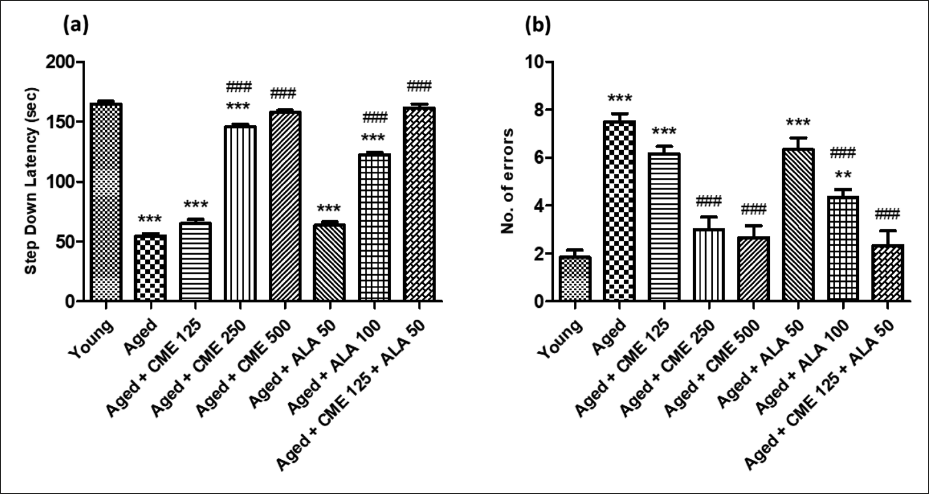
Anxiety and depression in aged rats were significantly increased, as indicated by decreased time spent in open arms (3.3-fold) and increased immobility time (2-fold) compared with the young rats’ group. The administration of CME (500 mg/kg) and ALA (100 mg/kg) led to a significant (p < .001) increase in the duration spent in the open arms. Additionally, CME (250 and 500 mg/kg) and ALA (100 mg/kg) resulted in a significant reduction in immobility time (p < .001) when compared with aged rats. After receiving CME (250 mg/kg) treatment, aged rats showed a significant increase (p < .01) in the time they spent in the open arms. A marked increase in time in the open arms and a decrease in immobility time were observed in CME (125 mg/kg) and ALA (50 mg/kg) treated rats, but the effect was insignificant. However, when CME (125 mg/kg) and ALA (50 mg/kg) were administered in combination, a significant increase in time spent in the open arms (3.2-fold) and a decrease in immobility time (1.7-fold) were observed in these rats in contrast to aged group rats (Figure 3).
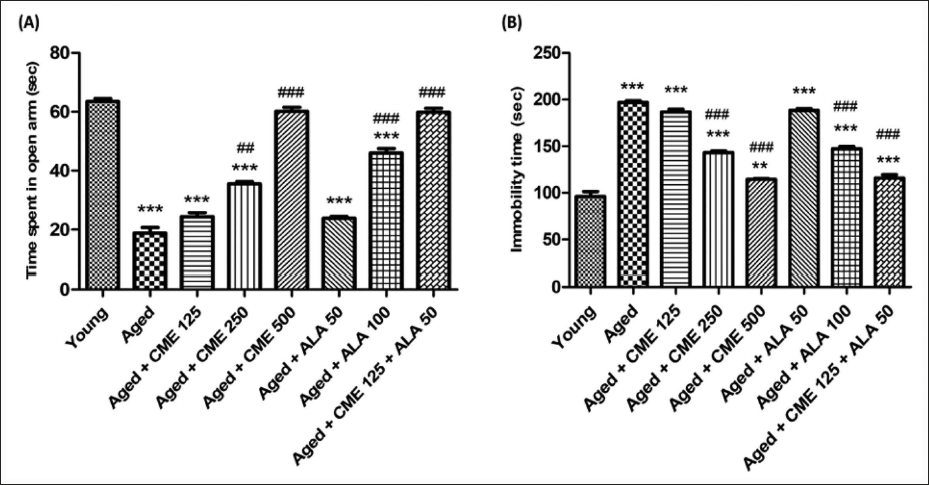
CME Effect on Locomotion (Actophotometer) and Motor Coordination (Rota Rod)
Locomotor activity and motor coordination were significantly decreased (2-fold and 3.6-fold) in aged rats, unlike the young group rats (Figure 4A and B). However, the number of counts and fall-off time were significantly increased (p < .001) in aged rats after treatment of CME (250 and 500 mg/kg) with ALA (100 mg/kg) for 3 months dose-dependently compared to aged rats. A mild increase in counts and fall-off time was observed in rats treated with CME (125 mg/kg) and ALA (50 mg/kg), but the effect was insignificant. However, CME (125 mg/kg) and ALA (50 mg/kg) were administered in combination, and a high rise in the number of counts (2-fold) and fall-off time (3.2-fold) was seen in these rats as contrary to aged group rats (Figure 4).
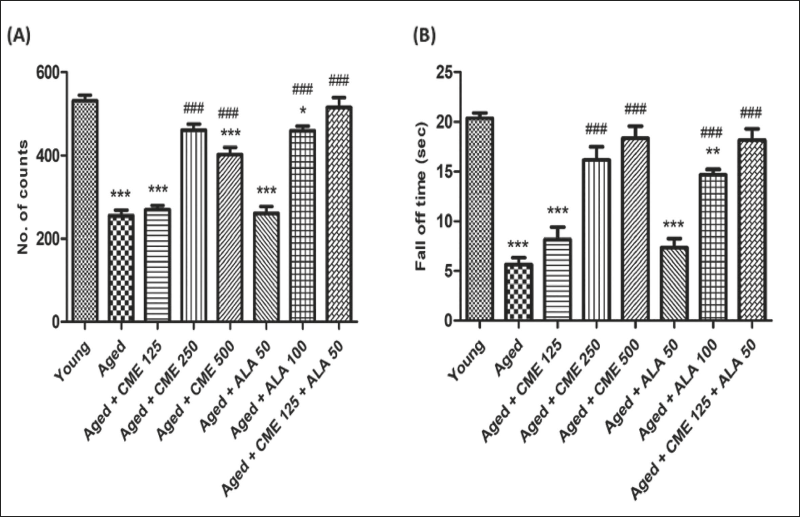
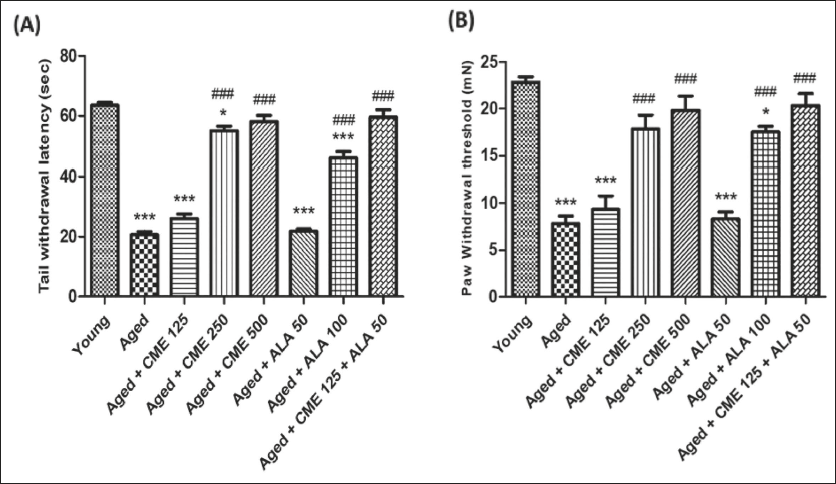
Analgesic Effect of CME by Tail Immersion and Randall–Selitto Test
Tail withdrawal and paw withdrawal threshold were significantly decreased (almost 3-fold) in aged rats in comparison to young group rats (Figure 5A and B). However, tail withdrawal latency and paw withdrawal threshold were significantly increased (p < .001) in the aged group of rats after treatment with CME (250 and 500 mg/kg) and ALA (100 mg/kg, p.o.) for 3 months, dose-dependently, compared to aged rats. Marked increases in tail withdrawal latency and paw withdrawal threshold were observed with CME (125 mg/kg) treated rats, but the impact was minimal. However, CME (125 mg/kg) and ALA (50 mg/kg) were administered in combination. Significantly increased tail withdrawal latency (2.8-fold) and paw withdrawal threshold (approx. 3-fold) were observed in these rats in comparison to aged group rats (Figure 5).
Effect of CME on Biochemical Parameters
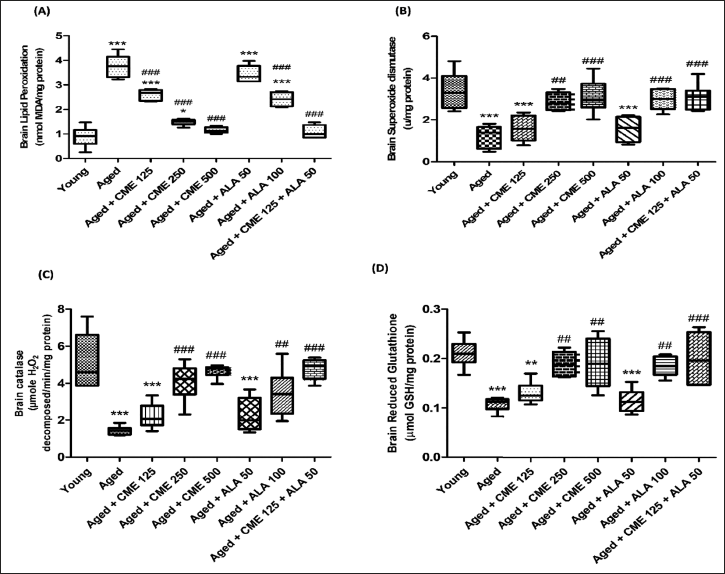
Effect of CME on Oxidative Stress Parameters
MDA levels noticeably rose (4.2-fold) and SOD, catalase, and GSH were significantly decreased (2.7, 3.6, and 2.1-fold) in aged rats, in contrary to young group rats (Figure 6A–D). However, the MDA level was significantly decreased (p < .001) in the aged group of rats after treatment with CME (125, 250, and 500 mg/kg) and ALA (100 mg/kg), and SOD, catalase, and GSH were significantly increased in aged rats after treatment with CME (250 and 500 mg/kg) and ALA (100 mg/kg) for 3 months dose-dependently compared to aged rats. However, CME (125 mg/kg) and ALA (50 mg/kg) were administered in combination, and a significant decrease in MDA levels (3.4-fold) and increased in SOD (2.5-fold), catalase (3.3-fold), and GSH (1.9-fold) in these rats in comparison with the aged group rats (Figure 6).
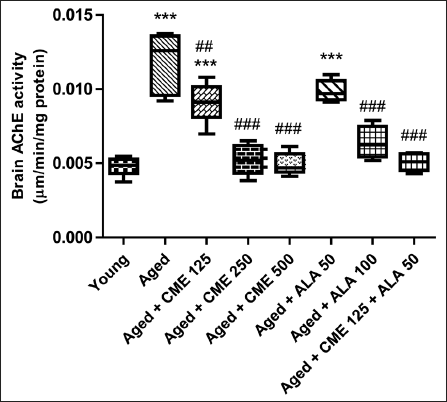
Effect of CME on AChE
The level of AChE showed a significant increase (p < .001) in aged rats compared to young rats (Figure 7). However, the AChE level was significantly decreased (p < .001) in the aged group of rats after treatment with CME (250 and 500 mg/kg) and ALA (100 mg/kg) for 3 months dose-dependently compared to aged rats. Marked decreases in AChE levels were observed in rats treated with CME (125 mg/kg) and ALA (50 mg/kg), but the effect was not significant. However, CME (125 mg/kg) and ALA (50 mg/kg) were administered in combination, and a considerable increase in AChE levels was observed in these rats (p < .001) in comparison with the aged group rats.
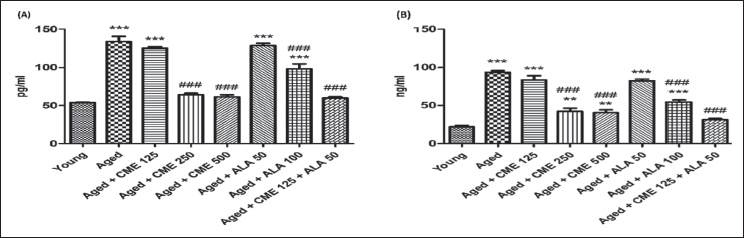
Effect of CME on Molecular Parameters
Effect of CME on Neuroinflammation
In aged rats, there was a significant increase (p < .001) in TNF-α and NF-κB compared to the young group rats (Figure 8A and B). Following treatment with CME (250 and 500 mg/kg) and ALA (100 mg/kg) for 3 months, TNF-α and NF-κB showed a significant decrease (p < .001) in aged rats in a dose-dependent manner compared to aged rats. A marked decrease in TNF-α and NF-κB was observed in CME (125 mg/kg) and ALA (50 mg/kg) treated rats, but the effect was insignificant. However, CME (125 mg/kg) and ALA (50 mg/kg) were given in combination. A substantial rise in TNF-α (2.2-fold) and NF-κB (almost 3-fold) was observed in these rats (2.2-fold) compared to the aged group rats (Figure 8).
Histopathological Parameters
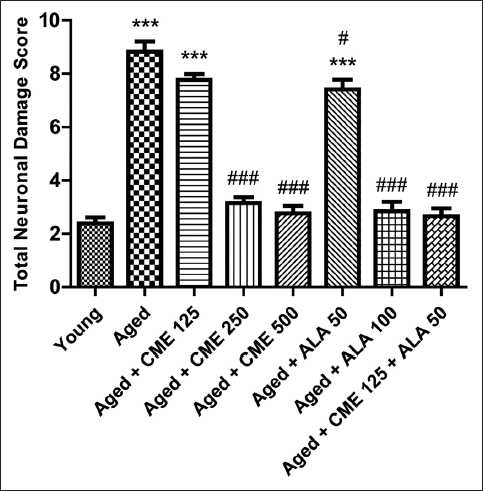
During the histopathological examination of rats’ brain hippocampal region, neuronal damage was noted in the dentate gyrus of the brains of elderly rats, compared with young rat’s brains, as indicated by large number of pyknotic and granule dead cells. Administration of CME (250 and 500 mg/kg) and ALA (50 and 100 mg/kg) dose-dependently modulates the neuronal damage by a reduction in pyknotic and granule dead cells, as shown in Figure 8. Further, administration of CME (125 mg/kg) in combination with ALA (50 mg/kg) markedly reduced the neuronal damage in the rat hippocampal region (Figures 9 and 10).
Discussion
Age is the leading risk factor for most neuronal impairments. 35 Free radicals are very reactive molecules or atoms with one or more unattached electrons in their outer shell, which can be created when oxygen reacts with certain compounds. 36 These radicals are generated within cells when a lone electron is either gained or lost, allowing them to act as either reducing agents or oxidizing agents. 37 Free radicals cause chronic oxidative damage that ages the brain and modifies neural connections, eventually deteriorating cognitive decline. 38 In addition, we found that elderly rats had significantly higher levels of inflammation, oxidative stress indicators, and impairments in memory, nociception, anxiety, and depression when compared to younger rats. These findings suggest that age-related neuronal deficiencies may cause neuronal impairments in old rats.
Compared to elderly rats, the administration of CME improves nociception, sadness, anxiety, memory, and learning. These outcomes were consistent with previous research published by Kim et al., which showed that the administration of C. militaris improves memory deficits caused by worldwide cerebral ischemia and scopolamine-induced memory deterioration. 39 It may also be used as a treatment option for neuroinflammatory illnesses and vascular dementia and to protect the hippocampus. Further, the study by Lin et al. 40 reported that the medicinal fungus known as C. militaris may be used to treat mood disorders such as anxiety and depression. 40 Furthermore, research conducted by Phull et al. 41 stated that the primary component in C. militaris, “cordycepin,” suppressed the expression of two genes linked to inflammation: inducible nitric oxide synthase (iNOS) and cyclooxygenase-2 (COX-2). 41 Administration of ALA improves memory, 42 depression, 43 anxiety, 44 and nociception. 45 In our study, we also made observations, which state that a combination of CME and ALA significantly improved memory, depression, anxiety, and nociception in aged rats.
The body commonly experiences oxidative stress. Enzyme systems involved in in vivo redox homeostasis typically keep intracellular levels of ROS low under normal conditions, in order to maintain physiological balance. Thus, it may be stated that an imbalance in the body between prooxidants and antioxidants is referred to as oxidative stress. 46 MDA, SOD, GSH, and catalase are examples of oxidative stress biomarkers that are crucial for determining the extent of disease and the advantages of antioxidants for general health. One of the main theories of aging is oxidative stress, and the first line of defense antioxidants are SOD, GSH, and catalase. 47 Reduced SOD expression increases the vulnerability of many species to oxidative stress. 48 Reduced GSH levels in the brain are linked to neurodegeneration or aging-related neuronal loss. 49 Several age-related degenerative disorders are related to the pathophysiology of catalase dysfunction or shortage. 50
A study by Olatunji et al. 51 stated that the C. militaris attenuates the levels of lipid peroxidation product (MDA), while enhancing the functioning of antioxidants (SOD and GSH) in 6-hydroxydopamine-treated cells. 51 Further, the study by Li et al. 52 found that C. militaris polysaccharide dramatically raised the performance of catalase, SOD, glutathione peroxidase, and antihydroxyl radicals in the liver of mice. 52 Moreover, a study by He et al. 53 demonstrated that C. militaris inhibited the activity of AChE in the brains of AD mice injected with Amyloidβ1-42. ALA exerts strong antioxidant properties, and when administered, it reduces MDA levels and AChE activity in the brains of elderly rats, 54 while also enhancing the activity of antioxidant enzymes in cases of lipopolysaccharide-induced kidney dysfunction. 55 Consequently, we also discovered that the combination of CME and ALA significantly lowers the generation of oxidative stress and raises the activities of antioxidant enzymes compared to aged rats.
The activation of oxidative stress can result in the activation of various transcription factors, which in turn leads to the differential expression of particular genes that are implicated in inflammatory processes. 56 For example, studies have shown that the production of TNF-α and the activation of NF-κB significantly impact the inflammatory process, leading to certain chronic disorders. 57 ROS can impact both synaptic and nonsynaptic neuronal communication in brain tissues, potentially leading to memory loss, neurodegeneration, and neuroinflammation. 58 The body might trigger several immune system cells if an inflammatory reaction occurs. The body releases different chemicals known as “inflammatory mediators,” including histamine and bradykinin, which cause the small blood vessels in the tissue to enlarge and allow more blood flow to the damaged area. Heat is felt in reddish-pink inflammatory areas. 59 Biomarkers such as ROS, cytokines [including TNF-α and interleukin-6 (IL-6)], chemokines, C-reactive proteins (CRPs), prostaglandins, NF-κB, and critical immune cell types have a significant impact on the pathophysiology of inflammation. 60 Considering that growing older is a significant cancer risk factor, systemic, sterile, age-related chronic inflammation—also known as inflammaging—is deemed to have a connection to the illness’s underlying etiology. 61 The CRP, along with other inflammatory markers, rises with age. As people age, their gut microbiota is assumed to play a part in systemic chronic inflammation, evolving and becoming more proinflammatory. 62
According to Jo et al. 63 CME suppressed the growth, survival, and migration of ovarian cancer cells, perhaps via coordinating NF-κB activation with TNF-α/tumor necrosis factor receptor 1 (TNFR1) signaling. 63 Further, a study by Park et al. 64 CME dramatically decreased the level of TNF-α. 64 In research conducted by Akbari et al., 65 the impacts of ALA supplementation on inflammatory markers in individuals with metabolic syndrome and related disorders were reported. 65 Furthermore, ALA suppresses NF-κB activation independently of its antioxidant action. 66 Consequently, our study also revealed that the CME and ALA combination significantly reduced aging-related inflammation.
Conclusion
The combination of CME and ALA demonstrated significant potential in improving cognitive function, reducing oxidative stress, and enhancing antioxidant enzyme activities in aging models. This study adds new evidence supporting their synergistic neuroprotective effects. However, the present findings are limited to preclinical models, and further clinical studies are necessary to confirm their efficacy in humans. These insights suggest that targeting oxidative stress and inflammation with CME and ALA could be promising for managing neurodegenerative diseases linked to aging.
Abbreviations
AChE: Acetylcholinesterase; ALA: Alpha lipoic acid; CME: Cordyceps militaris extract; COX-2: Cyclooxygenase-2; CRP: C-reactive protein; DPPH: 2,2-Diphenyl-1-picrylhydrazyl; EL: Escape latencies; GAE: Gallic acid equivalent; GSH: Glutathione; IC50: Half maximal inhibitory concentration; IL-6: Interleukin-6; iNOS: Inducible nitric oxide synthase; MDA: Malondialdehyde; NBT: Nitro blue tetrazolium; NF-κB: Nuclear factor kappa-light-chain-enhancer of activated B cells; p.o.: Per os (by mouth); QE: Quercetin equivalent; ROS: Reactive oxygen species; SD: Standard deviation; SDL: Step-down latency; SFZ: Shock-free zone; SOD: Superoxide dismutase; TBARS: Thiobarbituric acid reactive substances; TFC: Total flavonoid content; TNF-α: Tumor necrosis factor-alpha; TPC: Total phenolic content; TSTQ: Time spent in the target quadrant; UV: Ultraviolet.
Author Contributions
HK, SB, RC, SG, and SC conceived the study. HK, RC, SC, and SB involved in protocol writing, database search, screening of articles, and preparation of final draft. SB, SG, NG, KA, and SC were responsible for article screening till the writing of the final draft. SG, SC, and SB were consulted for the critical revision of the manuscript to resolve any discrepancy. Finally, all the authors read and approved the final manuscript.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Before the animal research began, the protocol for the experiment was approved by the Maharishi Markandeshwar (MM) College of Pharmacy’s Institutional Animals Ethics Committee (Protocol no. MMCP-IAEC-110, dated 20/11/2021) and carried out in accordance with the regulations on animal experimentation issued by the Committee for Control and Supervision of Experiments on Animals (CCSEA), Government of India.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
