Abstract
Background
Apremilast is a phosphodiesterase-4 (PDE-4) inhibitor approved by the US-Food and Drug Administration (US-FDA) for the management of adult patients with plaque psoriasis (who are candidates for phototherapy/systemic therapy), moderate-to-severely active psoriatic arthritis and Behcet’s disease associated with oral ulcers.
Objectives
This review aims to summarize the current evidence for the off-label use of apremilast in various dermatological disorders.
Methodology
A comprehensive search of relevant literature was conducted using databases like PubMed, MEDLINE and Embase. Studies investigating the use of apremilast for dermatological conditions beyond its approved indications were included.
Results
The use of apremilast has been outlined in a number of reports for inflammatory dermatoses. Randomized controlled trials showed promise for atopic dermatitis, hidradenitis suppurativa and vitiligo. Open-label trials suggested potential benefit in cutaneous sarcoidosis, discoid lupus erythematosus, rosacea and lichen planus.
Conclusion
Apremilast can be considered a valid therapeutic option for a range of inflammatory dermatological conditions beyond its approved uses. Further, owing to lack of immunosuppression, apremilast delineates an added advantage when compared to the usual immunosuppressive drugs used by dermatologists. However, stronger evidence from well-designed clinical trials is needed to establish its role in these aforementioned disorders.
Introduction
Apremilast is an oral phosphodiesterase-4 (PDE4) inhibitor that is currently approved by the US-Food and Drug Administration (FDA) for the treatment of adult patients with moderate-to-severe plaque psoriasis (who are candidates for phototherapy/systemic therapy), moderate-to-severely active psoriatic arthritis, and Behcet’s disease (BD) associated with oral ulcers. 1 Of late, apremilast’s utility as an off-label drug for many dermatoses has been reported with promising results. This review will explore the off-label applications of apremilast in dermatology.
Pharmacokinetics
Apremilast is administered orally, after which it gets rapidly absorbed in the body, reaching peak plasma concentrations after 2–3 hours. The bioavailability of apremilast is ~73% and its t1/2 ranges from 6 to 9 hours. Metabolism of apremilast occurs through cytochrome (CYP) 3A4-mediated oxidative metabolism, followed by glucuronidation, nonenzymatic hydrolysis, and a non-CYP3A4-mediated metabolism. Apremilast is eliminated mainly via the renal route, with some of it excreted through the feces. 2
Methodology
PubMed, MEDLINE, and Embase databases were searched for the period January 1, 2010 to March 15, 2023. Searched terms were “apremilast” in combination with the following: “atopic dermatitis,” “pityriasis rubra pilaris,” “folliculitis decalvans,” “cutaneous sarcoidosis,” “seborrheic dermatitis,” “calcinosis cutis,” “Anti p-200 pemphigoid,” “pemphigus vulgaris,” “granuloma annulare,” “Hailey-Hailey disease,” “hidradenitis suppurativa,” “erythema nodosum leprosum,” “dermatomyositis,” “discoid lupus erythematosus,” “vitiligo,” “chronic actinic dermatosis,” “orofacial granulomatosis,” “SAPHO syndrome,” “rosacea,” “acrodermatitis continua of Hallopeau,” “palmoplantar pustulosis,” “alopecia areata,” “erythema multiforme,” “lichen planus,” “lichen planopilaris,” “pyoderma gangrenosum,” “chronic hand eczema”. All original studies, including case reports, case series, and clinical trials, were included if they were full text, involved a dermatologic condition treated by apremilast, and were published in English. Articles were excluded if they were conference abstracts, nonclinical reports, or in vitro studies. All references were checked, and additional articles were included. A schematic representation of the search strategy is delineated in Figure 1.
Schematic Representation of the Search Strategy Followed for This Review.
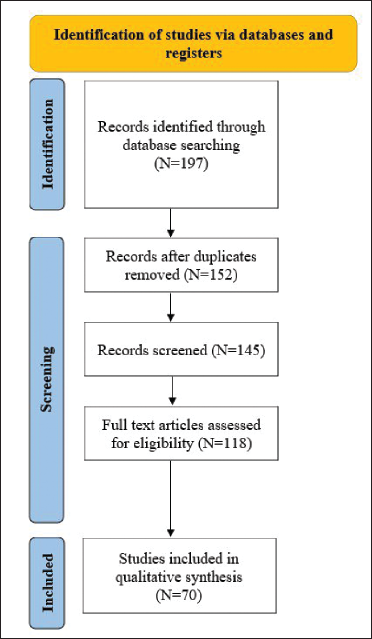
Results
Atopic Dermatitis (AD)
AD is a chronic relapsing eczematous disorder that, in moderate-to-severe proportions, cannot be managed by topical agents alone, necessitating the need for systemic therapy. Conventional systemic immunomodulatory drugs like cyclosporine, azathioprine, and methotrexate, though effective in AD, are commonly associated with adverse effects, as a result of which treatment often warrants discontinuation.
Apremilast, though not an approved drug for AD, has been reported to be beneficial for the same.3–8 Although the mechanism of action of apremilast in AD is not completely elucidated, its role in AD occurs owing to its anti-inflammatory effects, details of which are elaborated in Figure 2.9–12 Besides, apremilast indirectly inactivates BCL2-associated agonist of cell death (BAD), a well-known proapoptotic factor expressed in AD. By doing so, it prevents BAD from nullifying the effects of B-cell leukemia/lymphoma 2 protein (Bcl-2), an anti-apoptotic factor that has a protective role in AD.13–15 In addition, apremilast modifies the expression of the chemokines IL-12 and CC chemokine receptor 3 (CCR3), which are elevated in the lesional skin and blood of AD patients, further reducing cutaneous inflammation.16, 17
Mechanism of Action of Apremilast in Atopic Dermatitis.
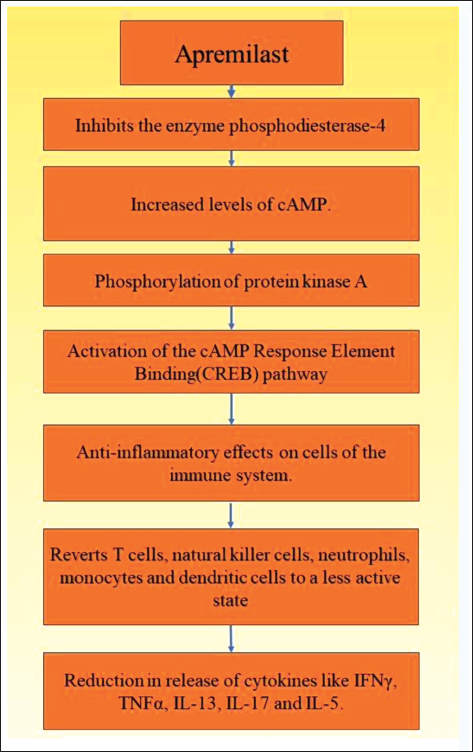
An open-label pilot study on 16 adult patients with AD demonstrated the beneficial effects of apremilast in two groups receiving the drug in two different dosages. The group that received 20 mg (twice daily (BID)) of apremilast (n = 6) displayed a significant reduction in pruritus (p = .002) and Dermatology Life Quality Index (DLQI) (p = .003) at 3 months from baseline, and the group receiving 30 mg (BID) of apremilast (n = 10) demonstrated notable reduction of the Eczema Area Severity Index (EASI) score at 3 months (p = .008) as well as 6 months (p = .002), along with a prominent reduction in the DLQI, both at 3 (p = .1) and 6 months (p = .003). Besides, treatment was well tolerated by all patients, except for one in the 20 mg group who withdrew from the study following an outbreak of herpes zoster. 3
In a phase 2 randomized controlled trial (RCT), two dosing schedules of apremilast were compared with placebo. Both groups, one receiving apremilast 30 mg (BID) and the other getting apremilast 40 mg (BID), demonstrated improvement in the EASI score at week 12, but it was not statistically significant in comparison to the placebo group. Further the Static Physicians Global Assessment (SPGA)-acute signs response at week 12 was achieved in 6.3% (placebo), 14.3% (apremilast 40 mg BID), and 3.1% (apremilast 30 mg BID) of patients, which again was not significant statistically. Although modest efficacy of apremilast was observed in the 40 mg (BID) group in treating moderate-to-severe AD, not much of a difference was seen when collated with the apremilast (30 mg BID) and placebo groups. Interestingly, in the apremilast 40 mg (BID) group, biomarker analysis delineated a reduction in Th17/Th22-related markers (IL-17, IL-19, IL-22, S100A12), which was not forthcoming in the other two groups. Treatment with apremilast was safe in all patients in this RCT, except for six patients in the apremilast (40 mg BID) group who developed cellulitis and treatment had to be discontinued. 4
Abroule et al., 5 delineated the rapid onset of improvement of chronic, severe, recalcitrant AD in their series of four patients. Of the four patients, two delineated >75% reduction of eczematous plaques/papules and pruritus, one demonstrated near clearance of cutaneous lesions, and the other portrayed a substantial decrease in erythema along with a significant reduction of pruritus. Besides, in two patients who continued apremilast therapy, sustained improvements were maintained, but there was worsening of AD after 6 weeks in the other two patients owing to treatment discontinuation.
In an individual report, apremilast (30 mg/day) was found to be effective in treating AD in an 8-year-old child who was refractory to prednisolone and omalizumab. Within 2 weeks of initiating apremilast, a satisfactory reduction of pruritus was reported, accounting for a substantial reduction in body excoriation. 6
However, in the publication by Volf et al., 7 worsening of 20% of the EASI score over 16 weeks of apremilast (20 mg BID) therapy was observed that resulted in discontinuation of treatment. Interestingly, it was noted that the AD patients in this trial had a worse EASI score from the onset.
Despite the propitious effects of apremilast in AD, as outlined by the above reports, more clinical trials are needed to firmly establish its role in this dermatosis.
Pityriasis Rubra Pilaris (PRP)
PRP is a papulosquamous disorder that has a wide clinical heterogenicity. Cardinal features characterizing PRP include hyperkeratotic follicular papules, salmon-to-orange colored scaling plaques, and palmoplantar keratoderma that may progress to erythroderma. Although the disease may heal spontaneously, many patients experience a disabling course. Given the absence of controlled clinical trials for the management of PRP, the therapeutic approach rests on anecdotal clinical observations, with methotrexate and acitretin being considered first-line systemic agents. 18 Unfortunately, in many patients, PRP fails to respond to conventional medications, warranting the need for newer and effective drugs for its management.
Currently, the use of apremilast in PRP has been expounded in four case reports as delineated in Table 1.19–22 However, RCTs are needed to strengthen the above observations.
Reports Elucidating the Efficacy of Apremilast in Pityriasis Rubra Pilaris.
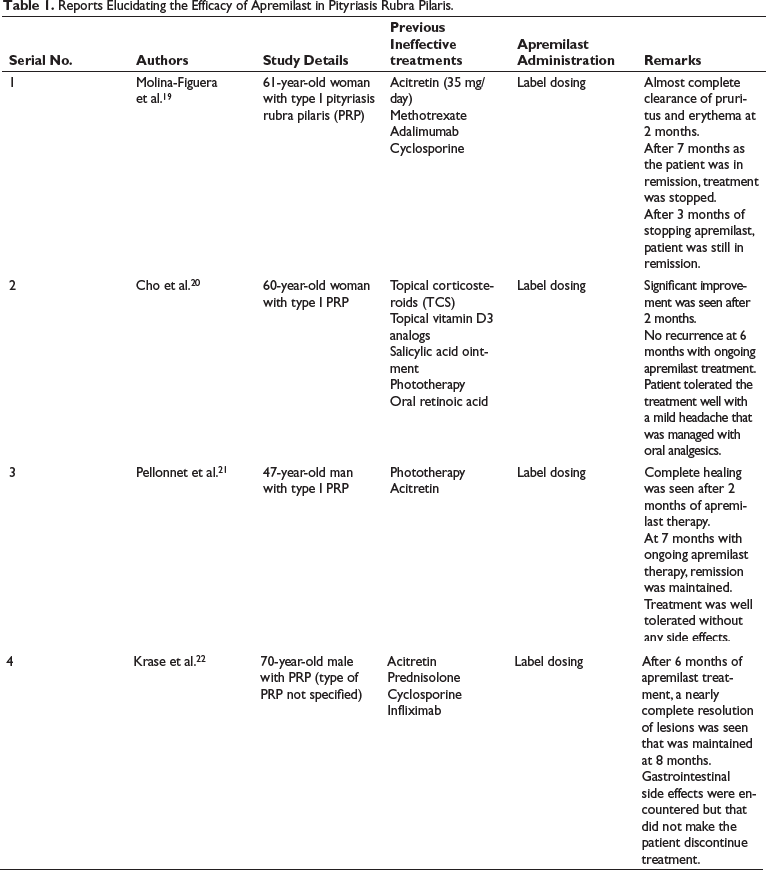
Apremilast’s therapeutic effects in PRP are considered to be brought about by its ability to block PDE4 and increase levels of cAMP that reduces expression of inflammatory cytokines like TNF-α, IL-23, and INF-γ, along with an elevation in the levels of the anti-inflammatory cytokine IL-10. Further, apremilast also suppresses the activation of NF-κB, a transcriptional factor responsible for inducing inflammation, and in this way, helps in ameliorating lesions of PRP. 23
Folliculitis Decalvans (FD)
FD is a rare destructive folliculitis involving the scalp and presenting as painful follicular papules, pustules, crusting, and hair tufting. Histologically, FD outlines a predominating neutrophilic infiltrate, admixed with lymphocytes, eosinophils, and plasma cells. Conventional drugs employed in treating FD include rifampicin, clindamycin, doxycycline, lymecycline, dapsone, and isotretinoin. 24
In an anecdotal report, apremilast was found to be effective in treating FD in a 28-year-old man who had the disease for 5 years and had been refractory to the usual therapeutic agents used in FD. Within 3 weeks of apremilast monotherapy supported by 2% chlorhexidine shampooing, nearly complete remission was obtained, with total cessation of pain and pruritus. 25
The mechanism of apremilast in FD is linked with its ability to suppress neutrophils, reduce neutrophilic extracellular traps, and plummet levels of reactive oxygen species. These anti-neutrophilic effects of apremilast may account for its beneficial effects in FD owing to the predominant neutrophilic inflammation that characterizes this disorder. Besides, downregulation of other pro-inflammatory cytokines (TNF-α, IFN-γ, IL-6, IL-8, IL-12, IL-17, and IL-23) and upregulation of the anti-inflammatory cytokine IL-10 further augment apremilast’s anti-inflammatory effects in FD.2, 26
Cutaneous Sarcoidosis
Sarcoidosis is a chronic granulomatous multisystem disorder mainly involving the lungs, mediastinal and peripheral lymph nodes, eyes, and skin. Although cutaneous sarcoidosis rarely causes significant morbidity or mortality, it may be grossly disfiguring and have a solid psychosocial impact. Systemic corticosteroids form the basis for treating systemic sarcoidosis. However, when significant visceral involvement is absent, the use of oral steroids cannot be justified. Besides, other options for cutaneous sarcoidosis are not completely effective, with therapeutic recommendations being supported by isolated case reports and short series. 27
The profitability of apremilast (20 mg BID) in cutaneous sarcoid was elaborated in an open-label trial on 15 patients. At the end of 12 weeks, the Sarcoidosis Activity and Severity Index significantly improved, and on analysis of paired before and after treatment photographs, notable clinical improvement was observed. Of the 15 patients, 2 patients withdrew due to nausea and jitteriness, and 3 patients who completed the study reported worsening of lesions following drug discontinuation. 28 As pro-inflammatory cytokines like TNF-α, INF-γ, IL-2, IL-12, and IL-23 are involved in the initiation and perpetuation of sarcoidosis; it is suggested that apremilast’s antagonistic effects on these cytokines may promote lesional regression in cutaneous sarcoid. 28 However, RCTs become compulsory to strengthen this observation further.
Seborrheic Dermatitis (SD)
SD is a common, relapsing dermatitis presenting as erythematous patches with superficial greasy scales. Commonly involved sites include the scalp, face, central chest, and anogenital areas. In those patients presenting with extensive and recalcitrant SD, management becomes a challenge. 29 Besides, with the limited available options, any addition to the therapeutic armamentarium of SD is always welcome. Cohen et al., 30 in a series of three patients (25- and 66-year-old males and a 47-year-old female) with extensive SD, reported apremilast (label dosing) to be effective in bringing about significant improvement in the disease after 3 months of treatment. In one patient, a complete clinical cure was obtained, whereas in the other two patients, mild unifocal disease persisted. Prior topical agents that had failed to show any clinical response included corticosteroids, calcineurin inhibitors, vitamin D analogs, crisaborole, coal tar, ketoconazole, sodium sulfacetamide, and sulfur. Besides, all patients tolerated apremilast well, with mild nausea being experienced by one patient. The role of apremilast in SD has been suggested due to its anti-inflammatory properties following the blockade of PDE4.
Apart from this publication, the use of apremilast in SD has not been reported. More studies evaluating the effects of apremilast in SD become essential to further establish its utility for this dermatosis.
Calcinosis Cutis (CC)
CC represents a form of dystrophic calcification wherein, despite normal levels of serum calcium, phosphate, and parathyroid hormone, deposits of hydroxyapatite and amorphous calcium phosphate form over damaged subcutaneous tissue. 31 Current treatment options for CC include calcium binders, calcium channel blockers, sodium thiosulfate (a calciomimetic drug), surgical excision, and lithotripsy. In a recent report, apremilast was found to be a useful adjuvant in treating CC, helping to soften and fragment the calcified plates deposited in the subcutaneous tissue. Within 2 months of apremilast therapy, improvement was visible. 32
The mechanism of action of apremilast in CC is unknown, but its capacity to downregulate pro-inflammatory cytokines forms the basis for its application in CC. Inflammatory cytokines like TNF-α, IL-6, and IL-1β are responsible for chronic tissue damage and/or vascular hypoxia, resulting in tissue fibrosis and an increased propensity for phosphate binding that forms a scaffold for calcification. Once these cytokines are antagonized, their contribution to the formation of calcium salts is halted and lesions begin to slowly regress.33, 34 However, in order to elucidate these mechanisms better, clinical trials with apremilast for treating extensive CC become essential.
Anti-laminin γ1 (p200) Pemphigoid
Anti-p200 pemphigoid is a distinct subepidermal blistering disorder that clinically resembles bullous pemphigoid but tends to affect younger individuals. The C-terminus of laminin γ1 has been recognized to be the immunodominant region in anti-p200 pemphigoid. Often, anti-p200 pemphigoid is associated with psoriasis. 35
In an individual report, apremilast was found to be helpful in treating anti-p200 pemphigoid in a 57-year-old man with concomitant psoriasis. Within four weeks of starting apremilast (60mg/day), improvement was witnessed in psoriatic plaques as well as bullous lesions. 36 In anti-p200 pemphigoid, matrix metalloproteinase (MMP)-1 initiates blister formation by degrading laminin and promoting the formation of anti-laminin γ1 antibodies against an acidic non-collagenous N-linked glycoprotein in the lower lamina lucida. 37 Besides, other pro-inflammatory cytokines like TNF-α, IL-1, and IFN-γ whose levels are elevated in blistering disorders upregulate the expression of MMP-1, setting into play a relentlessly progressive inflammatory cycle. Apremilast plummets the levels of these pro-inflammatory cytokines and indirectly reduces the expression of MMP-1, with consequent reduction in the production of anti-laminin γ1 antibodies and eventual blister formation. 38 In addition, apremilast may directly suppress autoimmunity by unknown mechanisms. 36 Despite this encouraging finding in this anecdotal report, more cases need to be studied to better assess the efficacy and safety of apremilast for anti-p200 pemphigoid.
Pemphigus Vulgaris (PV)
Meier et al. reported apremilast to be a valuable add-on drug for therapy-resistant PV. In their report, apremilast (30 mg BID) enabled dose reduction of prednisolone to a maintenance dose of 5 mg/day, without the reappearance of painful and severe oral lesions, that had earlier occurred when dose titration of prednisolone below 20 mg/day was attempted. Further, treatment with rituximab (lymphoma protocol) was ineffective. Following the addition of apremilast to the therapeutic regimen, the Autoimmune Bullous Skin Disorder Intensity Score (ABSIS) reduced from 38 to 0, along with suppression of anti-desmoglein (Dsg)1 and anti-Dsg3 autoantibody levels in serum. 39 Based on laboratory studies, the suggested mechanism of action of apremilast in PV has been described in Figure 3.40–43 Currently, apart from this report, no other documentation of the use of apremilast in PV is available.
Mechanism of Action of Apremilast in Pemphigus Vulgaris.
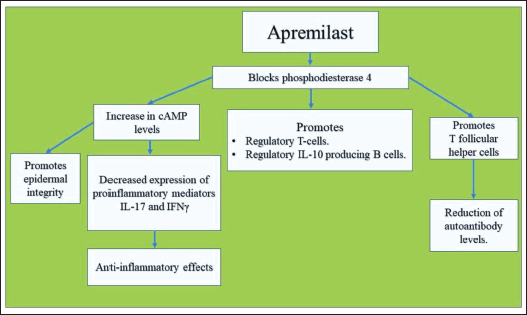
Granuloma Annulare (GA)
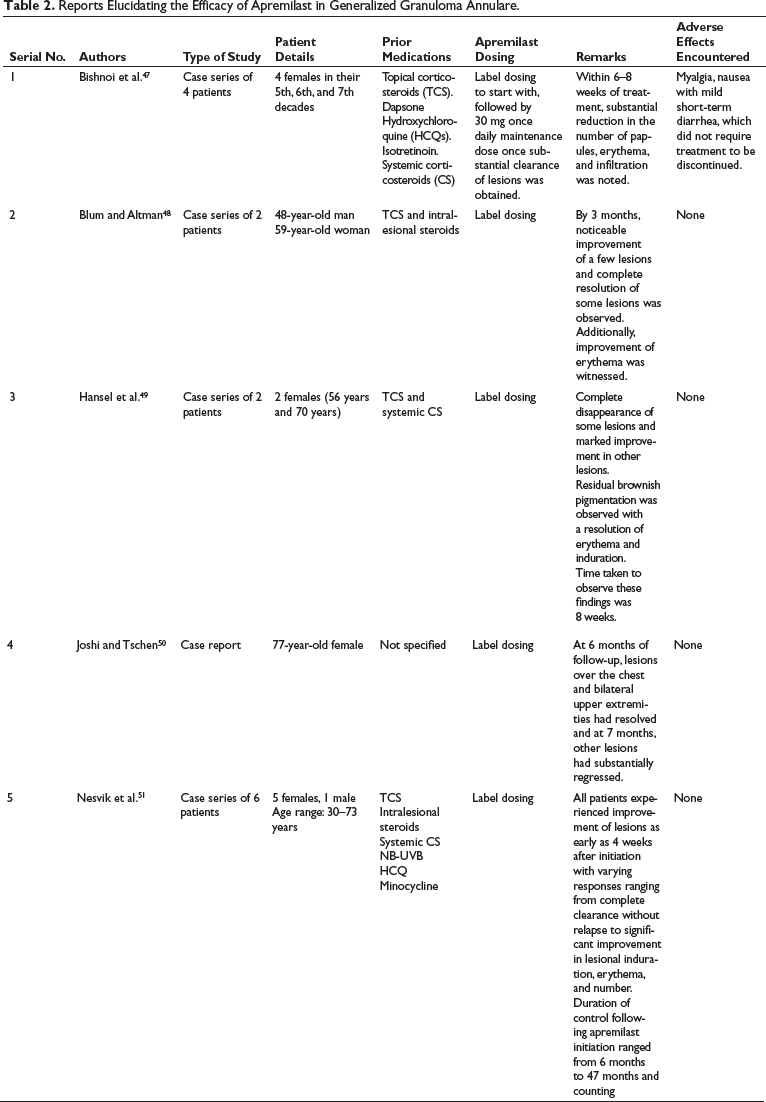
GA is a benign cutaneous necrobiotic disorder characterized by annular plaques composed of coalescing papules, which on histology outline a granulomatous inflammation. Although localized variants of GA are self-limited, generalized GA is refractory to most treatments with the possibility of persistence for years. 44 Although the pathomechanism of generalized GA has not been precisely elucidated; a Th1-mediated hypersensitivity reaction triggered by unknown antigens is suggested to be responsible for the same. Th1 cytokines (IL-2, TNF-α, IFN-γ) in turn stimulate macrophages to release MMP that induces collagen necrobiosis, granuloma formation, and mucin deposition. 45 Apremilast in GA acts by suppressing the production of Th1 pro-inflammatory cytokines, particularly IFN-γ, from macrophages and lymphocytes, and, in this way, promotes resolution of lesions. 46 Presently, there have been 15 patients with generalized GA in whom treatment with apremilast has been effective, details of which are highlighted in Table 2.47–51 Despite these promising reports, RCTs are mandated to strengthen the utility of apremilast in GA.
Reports Elucidating the Efficacy of Apremilast in Generalized Granuloma Annulare.
Hailey–Hailey Disease (HHD)
HHD is a rare genodermatosis presenting as recurrent blisters and erosions involving the intertriginous areas. Despite progression in our understanding of the molecular genetics of HHD, therapy remains suboptimal, and there is no known cure. Recently, the utility of apremilast has been explored for HHD in two publications with promising results.52, 53
In the first publication, apremilast (label dosing) was used to treat HHD in four patients (three women, one man, age range 50–60 years) who had treatment-resistant HHD for almost 30 years. Prior failed treatments included high-potency topical steroids, topical tacrolimus, oral antivirals/antibiotics, methotrexate, carbon dioxide laser, Erbium-Yag laser, and botulinum toxin injections. Following apremilast (label dosing) treatment, symptomatic improvement was seen after 1 month, and after 6 months, moderate-to-almost complete clearing of lesions was reported among the patients. However, after 6–10 months, in two patients, some amount of disease exacerbation was encountered, and apremilast treatment was stopped. Of the two patients, one developed a diffuse uncontrolled lesion, and apremilast was reintroduced, resulting in partial control of the disease. Besides, apart from gastrointestinal side effects, the drug was well tolerated by all patients. 52
The second report outlined the successful treatment of HHD in a 68-year-old woman who had been recalcitrant to treatment with topical high-potent steroids, topical and systemic antibiotics, methotrexate, and Erbium laser. Besides, remission with apremilast was maintained for 12 months. 53
Though the exact action of apremilast in HHD is not very clear, blockade of PDE4 with subsequent Th1/Th17 inhibition and reduction of C-X-C motif chemokine ligand (CXCL)10 release has been postulated to be the most likely mechanism. 54
Hidradenitis Suppurativa (HS)
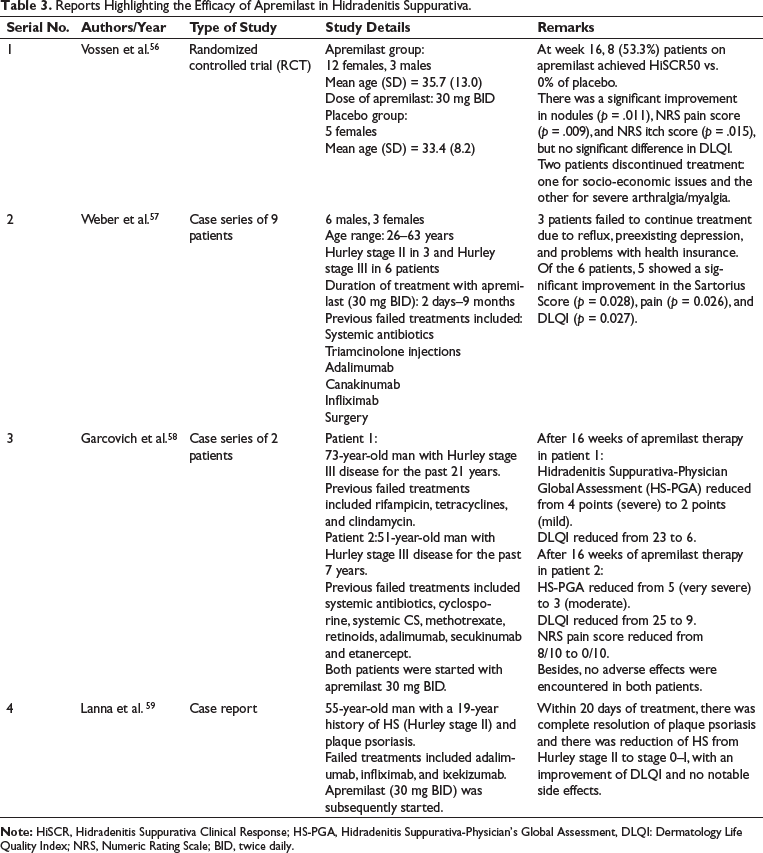
HS is an immune-mediated disorder of the follicular epithelium characterized by painful, deep-seated lesions with or without suppuration, predominantly affecting the intertriginous areas. Presently, oral antibiotics and biological drugs like adalimumab and secukinumab constitute the backbone of HS management. However, the emergence of bacterial resistance with continuous antibiotic use and the need for frequent laboratory monitoring with ongoing biological therapy pose restrictions in patient management. Besides, the formation of antidrug antibodies against TNF-α antagonists neutralizes the therapeutic effects of these drugs over time. 55 The need for newer and effective drugs without the constraints associated with conventional drugs therefore becomes essential, owing to the long duration of treatment for patients with moderate-to-severe HS. Recently, the benefits of apremilast in HS have been exemplified in one RCT, two case series, and one case report (Table 3).56–59
Reports Highlighting the Efficacy of Apremilast in Hidradenitis Suppurativa.
Although the exact mechanism of action of apremilast in HS is unknown, it has been postulated that by blocking PDE4, apremilast reduces levels of pro-inflammatory cytokines like TNF-α, IL-23, and IL-12 and increases levels of the anti-inflammatory mediator IL-10. Further, by regulating IL-8 and IFN-γ-induced protein 10 expression, apremilast suppresses the cellular mechanisms promoting inflammation, thereby preventing the expression of other inflammatory cytokines involved in HS.60, 61
Despite the few promising reports of apremilast in HS, more RCTs are required to strengthen its utility in the management of HS, both as monotherapy and/or combination therapy.
Erythema Nodosum Leprosum (ENL)
ENL is an immune-mediated complication of leprosy with a recurrent and chronic disposition. Further, if not promptly treated, it can severely affect patient life quality. Management of ENL is challenging, and no single drug is universally effective. While systemic steroids are commonly used in ENL management, their prolonged usage often heralds profound toxicities owing to the persistently high dosing schedules utilized. Besides, thalidomide, another highly effective crisis buster for ENL, often triggers peripheral neuropathy with long-term use. Other drugs like methotrexate, azathioprine, and cyclosporine, though utilized in ENL, often elaborate toxicity profiles serious enough to discontinue treatment. 62 The need for a safe and effective steroid-sparing drug therefore becomes a requirement while managing these patients.
The propitious effect of apremilast as a standalone steroid-sparing agent for ENL was first illustrated in two patients by Narang et al. 63 In both patients, significant clinical improvement of cutaneous lesions as well as subsidence of constitutional symptoms was observed within 2–4 weeks of initiating treatment with apremilast (label dosing). Besides, no adverse events were encountered, and none of the patients developed new episodes of ENL even after cessation of prednisolone therapy. 63
Similar findings were reiterated in a Spanish report on a 23-year-old woman with ENL. 64
However, in a pilot study by Patil et al., 65 out of the 10 patients, only 1 responded favorably with apremilast, as a standalone steroid-sparing drug. Of the other nine patients, four did not respond to apremilast, and in the remaining five patients, a reduction of 50–64.70% in the 16-item ENLIST ENL Severity Scale (EESS) score was ascertained, delineating a partial response following treatment with apremilast. This mixed response of apremilast in ENL can be accounted for by the heterogenicity involving the entire spectrum of ENL in relation to the elaboration of pro-inflammatory cytokines. Though apremilast may be effective in antagonizing the effects of TNF-α and IFN-γ, its efficacy against other lymphokines (IL1β, IL-2, IL-2R, IL-6, IL-12, and IL-17) may be negligible, and in those patients where these lymphokines are majorly expressed, a high likelihood of therapeutic failure with apremilast can be expected. Despite the mixed reports in relation to apremilast, it still would be worthwhile to dispense the drug along with systemic steroids owing to its better safety profile and studying the EESS score at each juncture of patient management, so that the therapeutic response can be adequately assessed. 65
Dermatomyositis (DM)
DM is an idiopathic inflammatory myopathy, presenting with a prominent cutaneous rash in ~80% of patients, with or without proximal muscle weakness. 66 Though not included in the current therapeutic armamentarium of DM, the efficacy of apremilast in treating DM has been elaborated in two reports.67, 68 In the first report (a lady in her 50s), the addition of apremilast (30 mg BID) to the existing therapeutic regimen (prednisolone 75 mg/day, diltiazem 120 mg daily, and yearly zolendronic acid) resulted in the resolution of heliotrope rash/facial erythema and highly satisfactory amelioration of scalp rash and pruritus within 3 months that had been unresponsive to azathioprine, methotrexate, MMF, IVIG, dapsone, and tacrolimus. 67 In the second report, three female patients (ages ranging from 57 to 64 years) with cutaneous DM unresponsive to oral steroids and multiple steroid-sparing agents benefited when apremilast (30 mg BID) was added to their treatment regimens, consisting of prednisolone and MMF in two patients and additional hydroxychloroquine in the third. Within 3 months of apremilast therapy, an 85% improvement in the Cutaneous Dermatomyositis Activity and Severity Index (CDASI) skin score was observed, allowing tapering of steroids and MMF and then complete discontinuation in two patients who were maintained on apremilast monotherapy. The third patient, on the other hand, encountered a disease flare after 9 months of maintaining disease stability with apremilast and was planned to receive IVIG. Besides, apremilast was tolerated by all patients, with transient gastrointestinal symptoms experienced by one. 68
Although the use of apremilast in DM has not been previously investigated, its role in improving cutaneous lesions of DM has been suggested secondary to its anti-inflammatory properties. Apart from antagonising Th1 cytokines (TNF-α, IFN-γ, IL-12), apremilast has also demonstrated inhibitory effects against Th2 responses by interfering with levels of IL-6 secreted by type 2 macrophages. As both Th1 and Th2 immune pathways play a fundamental role in DM, by interfering with both responses, apremilast can be a potential treatment for DM. Besides, as apremilast is not cytotoxic, it can be an added advantage for long-term therapy.69–71
Discoid Lupus Erythematosus (DLE)
DLE is a chronic inflammatory disorder characterized by erythematous plaques with adherent scales and mediated by Th1 cells. The therapeutic armamentarium of DLE comprises many drugs that include antimalarials, systemic steroids, gold, retinoids, sulfasalazine, cyclosporine, thalidomide, and clofazimine, to name a few. 72 De Souza et al. 73 demonstrated favorable responses following apremilast (20 mg BID) monotherapy in DLE patients in a single-arm open-label pilot study on eight patients. After 85 days, the Cutaneous Lupus Erythematosus Disease Area and Severity Index (CLASI) outlined significant improvement (p < .05). Besides, 50% of participants noted resolution of scalp lesions during the study, However, four patients withdrew early; two due to disease progression and two due to treatment-emergent adverse events (lichenoid reaction and neuropathy), both of which resolved with apremilast discontinuation. The exact mechanism of apremilast has not been clearly outlined, but its antagonistic effects toward Th1 cytokines have been postulated for the same. 2
Vitiligo
Vitiligo is a disorder of pigmentation characterized by immune-mediated melanocyte damage. The exact etiopathogenesis of vitiligo is still unclear and is believed to be secondary to cytokines like IFN-γ, TNF-α, IL-6, and IL-17 that recruit cytotoxic CD8+ T cells for melanocyte destruction. 74 Further, as vitiligo has a chronic course and a highly unpredictable progression, none of the available treatments can be considered full proof. Besides, systemic glucocorticoids and cytotoxic agents cannot be used for long periods owing to the toxicity profile, making it necessary for the introduction of safer and more effective drugs to serve the purpose. Apremilast appears to be a safe alternative and has been used as an off-label drug for vitiligo in one RCT, one randomized split pilot study, one case series, and two case reports.75–79
The RCT compared the efficacy of apremilast (30 mg BID) plus biweekly NBUVB (group 1, n = 40) with placebo plus biweekly NBUVB (group 2, n = 40). After 24 weeks, the mean Vitiligo Area Scoring Index (VASI) score decreased from 23.63 to 19.49 (p = .011) in group 1 and from 21.57 to 15.25 (p < .0001) in group 2, with no statistically significant difference between both groups (p = .18). Also, the Vitiligo Extent Score (VES), Vitiligo European Task Force (VETF) score, and DLQI score did not demonstrate any statistically significant difference in both groups. 75
The randomized split pilot study compared combined apremilast (30 mg BID) and NB-UVB (2–3 times per week) with NB-UVB monotherapy for vitiligo in Fitzpatrick skin types IV–VI. It was observed that patients receiving apremilast and NB-UVB had a significantly greater improvement in the primary endpoint of >50% re-pigmentation (p = .001) and Vitiligo Area Severity Index (VASI) score (p = –.0001) than those receiving NB-UVB monotherapy. 76
In Majid et al.’s 77 case series of 13 patients, apremilast (30 mg BID) was helpful in stabilizing the disease process by the end of 3 months in all patients. Furthermore, partial re-pigmentation was noted in eight (61.5%) patients with significant re-pigmentation even in those areas where topical tacrolimus (0.1%) was not applied. Additionally, the mean reduction in the VASI score among responders was 7.11%, which was statistically significant. 77
In both case reports, acceptable levels of re-pigmentation were noticed following 6–13 months of apremilast therapy.78, 79
Moreover, in all the above publications, apremilast was tolerated well by all patients except for a suicidal attempt in one patient in the report by Khemis et al. 75
Although the exact mechanism of apremilast in vitiligo has not been elucidated, its major contribution occurs because of its anti-inflammatory properties against the pro-inflammatory cytokine network dysregulated in vitiligo. In addition, by inhibiting PDE4 and increasing levels of cAMP, phosphorylation of the transcription factor cAMP response element binding (CREB) is induced, which in turn activates melanocyte inducing transcription factor (MITF), which is the key transcription factor for melanocytes and regulates their proliferation and melanogenesis by increasing the transcription of tyrosinase and dopachrome tautomerase enzymes. Furthermore, stimulation of the cAMP pathway also enhances UVB-induced re-pigmentation in vitiligo patients that accounts for the synergistic outcome obtained while combining both treatment modalities.80, 81
Though effective in bringing about re-pigmentation in vitiligo, monotherapy with apremilast may not be very practical. Combination therapies with various drugs can result in more effective re-pigmentation, for which RCTs are needed to clarify things better.
Chronic Actinic Dermatitis (CAD)
CAD is a photosensitive disorder comprising persistent eczematous skin lesions involving photo-exposed surfaces. 82 Successful use of apremilast in CAD was documented in a 36-year-old female who had the disease for 8 years, was unresponsive to steroid-sparing drugs (azathioprine, cyclosporine, and hydroxychloroquine), and had become steroid dependent, manifesting with toxicities like hypertension, peptic ulcers, and weight gain. Following 6 weeks of apremilast therapy, complete remission was obtained, allowing tapering and subsequent stoppage of steroids within the next 4 weeks, without any disease recurrence. 83 Although the exact role of apremilast in CAD is unknown, downregulation of TNF-α, IFN-γ, IL-2, IL-6, and IL-12 and upregulation of IL-10 by apremilast has been postulated for the same. Despite this promising case report, RCTs become essential to substantiate the efficacy of apremilast for this indication.
Orofacial Granulomatosis (OFG)
OFG is a relatively rare disorder presenting with recurrent episodes of labial swelling and/or oropharyngeal ulcer. 84 It may be associated with Crohn’s disease, and when fissured tongue and facial nerve palsy accompany OFG, it is termed as Melkersson–Rosenthal syndrome. OFG on its own, though not a serious disorder, is often associated with a chronic and disfiguring course, which, if not promptly treated, can affect the quality of life (QoL) of patients. The exact etiology of OFG still remains obscure, and elevated levels of Th1 cytokines following activation of the Th1 pathway have been implicated. 85
Kaushik et al. 86 evaluated the utility of apremilast (30 mg BID) in five patients with OFG. Prior failed therapies included systemic steroids, oral antibiotics (azithromycin and minocycline), and antihistamines. Following 4 weeks of apremilast therapy, >50% improvement in labial swelling and erythema was documented in three patients, which was maintained at 3 months of follow-up. In one patient, <25% improvement was noted after 12 weeks, and in the fifth patient no effect was observed.
Although the exact role of apremilast in OFG is unknown, it is suggested that as Th1 cytokines are putatively involved in granulomatous inflammation, apremilast may prove efficacious by antagonizing the effects of these inflammatory mediators. 86 With the observation of mixed results in this case series, further studies become imperative to ascertain the precise efficacy and mechanism of apremilast in OFG. Nonetheless, owing to apremilast’s favorable toxicity profile, it can be tried both as monotherapy and in combination with other drugs for OFG.
SAPHO Syndrome
SAPHO (Synovitis, Acne, Pustules, Hyperostosis and Osteitis) syndrome is a rare disorder presenting with inflammatory osteoarthritis and cutaneous involvement. Palmoplantar pustulosis (PPP) and acne are the dermatologic manifestations of SAPHO syndrome observed in up to 65% and 39% of cases, respectively. 87 The cytokine profile of SAPHO syndrome is similar to that of psoriasis with heightened expressions of TNF-α, IL-1, IL-8, IL-17, and IL-18. 88 Apremilast suppresses the above pro-inflammatory cytokines and consequently inhibits activation of T-cells and neutrophils by a pan-cytokine multicell immunomodulatory approach, and in this way helps in ameliorating symptoms in SAPHO syndrome. 89 The beneficial effects of apremilast for SAPHO syndrome were observed in a 24-year-old female who had been unresponsive to systemic steroids, nonsteroidal anti-inflammatory drugs, etanercept, ustekinumab and secukinumab. Following 6 months of apremilast (30 mg BID) therapy, long-lasting disease control was achieved without significant side effects. 90 Despite this favorable outcome, RCTs become essential to further strengthen this observation.
Rosacea
Rosacea is a chronic dermatological condition presenting as facial flushing, persistent erythema, telangiectasias, and inflammatory papules/pustules. In a pilot study of 10 patients (3 men and 7 women; age range 39–74 years), apremilast (20 mg BID) given for a period of 12 weeks demonstrated a statistically significant reduction in four rating scores; namely the Physician Global 7-point assessment (p = .02), erythemotelengiectatic rating (p = .005), Physician Overall Erythema Severity (POES) (p = .001), and non-transient erythema (NTE) (p = .04). Further after 1 month of follow-up, the POES and NTE scores maintained similar values. However, the primary endpoint of papule and pustule count did not reach statistical significance. This report concluded the utility of apremilast for erythemotelegiactatic rosacea (ETR) refractory to other treatments and associated with adverse effects with topical brimonidine tartarate. 91
Although the exact mechanism of apremilast’s utility in rosacea has not been elucidated, its role in modulating both pro- and anti-inflammatory cytokines, with resulting anti-inflammatory effects has been proposed. 91 Nevertheless, more studies are needed to establish the role of apremilast, not just for ETR but also for other variants. As this study utilized a lower dose of apremilast, assessing the therapeutic response with conventional apremilast dosage can be an avenue to embark upon.
Acrodermatitis Continua of Hallopeau (ACH)
ACH is a chronic sterile pustular eruption involving the tips of fingers and toes initially, but can slowly progress locally and rarely evolve into generalized pustular psoriasis. Currently, there are no therapeutic guidelines for ACH, with treatment being solely based on case reports. Besides, as ACH is often recalcitrant to various therapies, treating it becomes very challenging.
Currently, two individual reports have described the utility of apremilast in ACH. In the first report, apremilast (30 mg BID) was associated with marked improvement of ACH in a 75-year-old man within 1 month of therapy, which was maintained at 6 months of follow-up. Besides, improvement of onychodystrophy, joint symptoms, and QoL was also noted, without the occurrence of adverse events. 92 However, in the second report, ACH failed to respond to apremilast and was eventually managed successfully with secukinumab. 93
With the current limited data of apremilast regarding ACH, RCTs become mandatory to strengthen a positive association between the two.
Palmoplantar Pustulosis
PPP is a chronic inflammatory cutaneous eruption characterized by sterile pustular eruptions along with erythematous scaling involving the palms and soles. 94 Though earlier considered a part of psoriasis, it was proposed by the International Psoriasis Council to be considered a separate clinical entity rather than a palmoplantar variant of psoriasis. 95
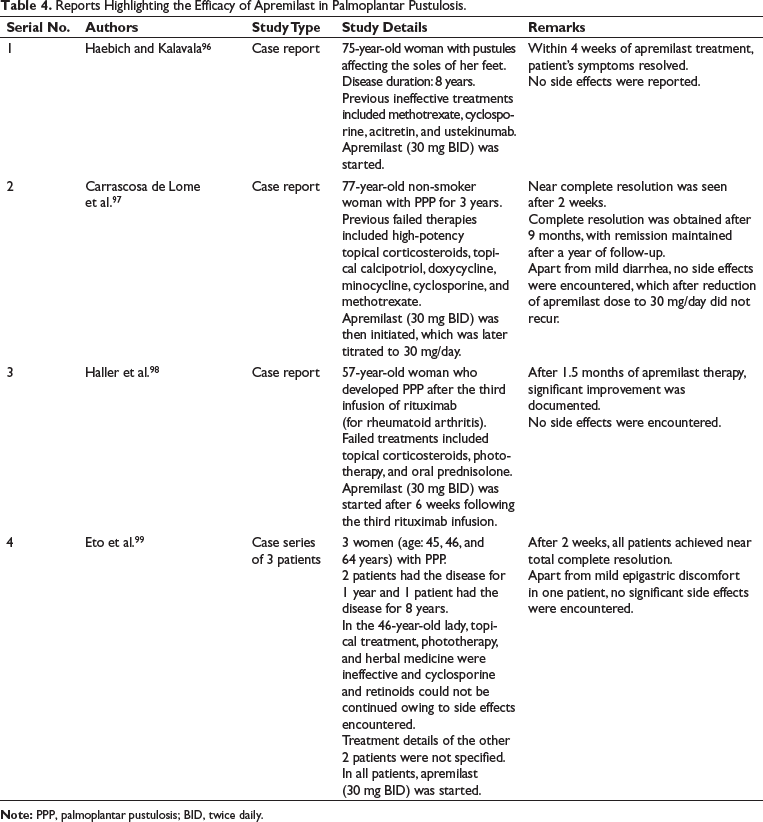
Till date, there are no RCTs assessing the efficacy of apremilast for PPP; data being confined to individual reports and case series as elaborated in Table 4.96–99
Apremilast’s activity against neutrophils has been suggested to be primarily responsible for its beneficial effects in PPP. It is observed that PDE4 is involved in the production of IL-8, leukotriene B4, and superoxide ions that facilitate neutrophilic degranulation and chemotaxis. Besides, PDE4 also mediates neutrophil adhesion to the vascular endothelium by inducing the expression of β2-integrin Mac-1. Once PDE4 is antagonized, the above mechanisms are thwarted, and disease remission is brought about.100, 101 Furthermore, apremilast is associated with higher drug survival rates than traditional systemic therapies, which again is an added advantage. 102 Even so, apremilast’s efficacy has been found to be superior than fumaric acid esters and methotrexate in PPP. 102
Reports Highlighting the Efficacy of Apremilast in Palmoplantar Pustulosis.
Alopecia Areata (AA)
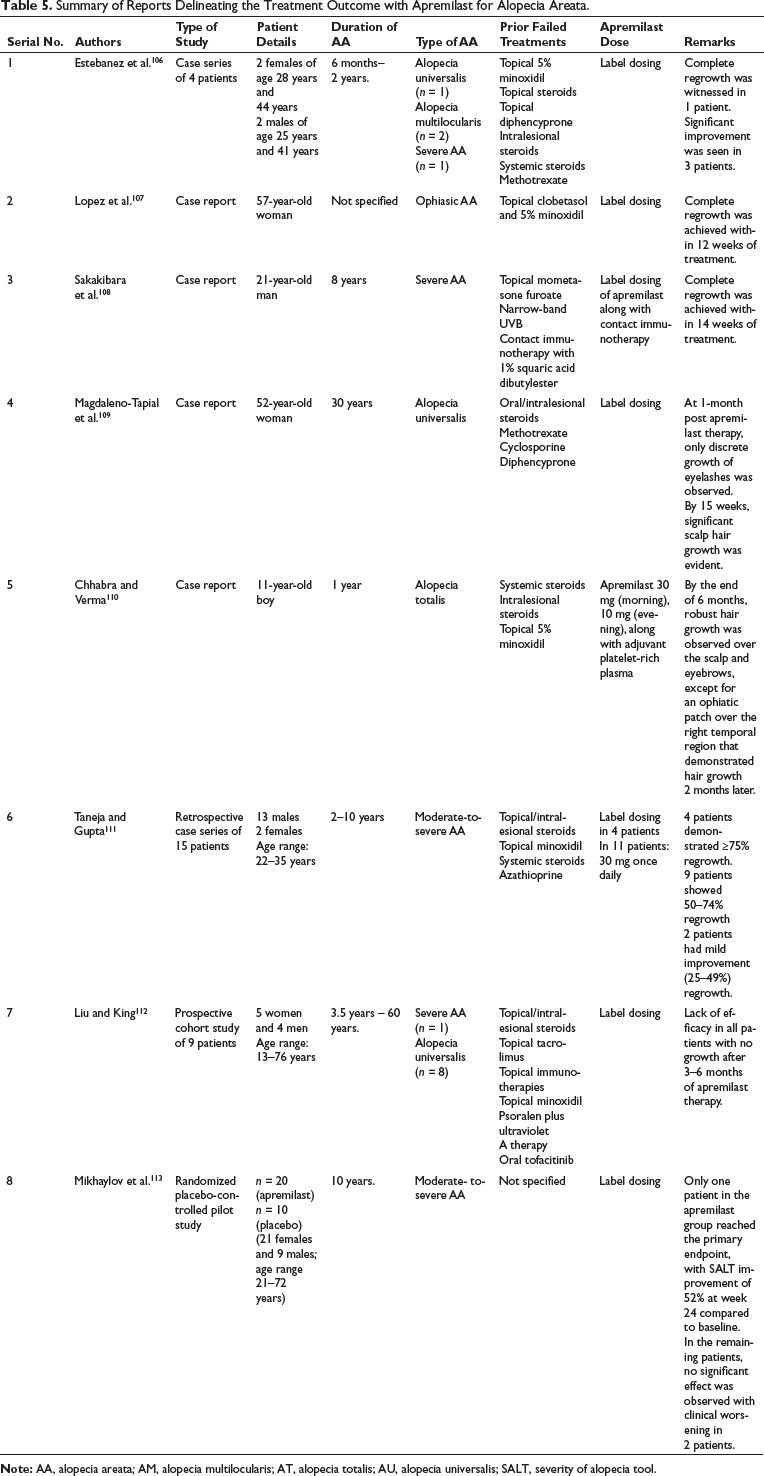
AA is an autoimmune disorder resulting in patchy, non-scarring hair loss, following CD4+ T cell-mediated damage of hair follicles. The exact pathogenic mechanism of AA still remains obscure, involving both Th1 and Th2 pathways. Recently, the role of PDE4 in the pathogenesis of AA has been propounded. 103 By blocking PDE4, apremilast results in the accumulation of cAMP, with subsequent reduction of pro-inflammatory cytokines, particularly IFN-γ.2, 104 In a humanized mouse model, oral apremilast prevented experimentally induced AA-like hair loss, with a significant reduction in inflammatory cytokines, while no effect was observed in the vehicle-treated controls. 105 However, in clinical reports, results obtained following apremilast treatment for AA have been mixed, details of which are elaborated in Table 5.106–113
Summary of Reports Delineating the Treatment Outcome with Apremilast for Alopecia Areata.
Erythema Multiforme (EM)
EM is best regarded as a self-limiting cytotoxic dermatitis resulting from cell-mediated hypersensitivity most commonly to drugs or infection. Owing to its recurrent nature, managing these patients often requires the introduction of more than one therapeutic agent. Currently, continuous antiviral therapy for 6 months is considered the most effective first-line therapy for recurrent EM regardless of an associated herpes simplex virus etiology. 114 However, if patients are unresponsive, immunosuppressive drugs, dapsone, thalidomide, and IVIG may need consideration and often precipitate unwanted effects. 115 Currently, the beneficial effects of apremilast have been delineated in a case series of four patients who had been unresponsive to antivirals as well as immunomodulatory drugs. Lesions cleared by 4 days to 2 weeks in three patients and by 2 months in another patient. Besides, no side effects were encountered. 116 Though, this appears to be promising, more studies are needed to substantiate the above findings.
Lichen Planus (LP)
LP is a chronic inflammatory disease primarily affecting the skin, mucous membranes, and nails. It is seen that 15–20% of patients with LP demonstrate a relapsing and remitting course often resistant to most conventional treatment modalities. 117 In such scenarios, the need for newer and more effective therapeutic alternatives becomes essential. Apremilast inhibits the production of pro-inflammatory cytokines like IFN-γ, TNF-α, IL-2, IL-5, IL-8, and IL-12, thereby preventing the activation of cytotoxic T-cells and averting apoptosis of basal keratinocytes, the underlying mechanism of LP. 118 Presently, there are no RCTs outlining the efficacy of apremilast in LP, with publications limited to a pilot study and three case reports.118–121
In the open-label pilot study on 10 biopsy-proven patients with cutaneous LP, apremilast (20 mg BID) demonstrated complete clearance of lesions in three patients and good clinical improvement in the remaining seven patients after 12 weeks of apremilast therapy. Besides, apremilast was well tolerated and patients who experienced nausea and headache did not require treatment discontinuation or dose alteration. 119
In oral LP, apremilast’s efficacy was highlighted in three case reports, with apremilast monotherapy resulting in complete resolution of lesions in one patient and marked improvement in another patient.118, 120 In the third report, along with apremilast, short courses of prednisolone had to be added to obtain complete remission. 121
Lichen Planopilaris (LPP)
LPP is an inflammatory scarring alopecia, whose management is very challenging owing to the lack of clarity in the natural course of this disease. 122 Apremilast’s use in LPP has been delineated in an individual publication comprising a series of four patients. Of the four patients, significant improvement was observed in two patients, with some improvements in the remaining two patients, with one having to discontinue apremilast owing to gastrointestinal side effects. 123 With this preliminary finding, nothing significant can be concluded regarding the use of apremilast in LPP, with the need for clinical trials to conclusively establish the favorable profile of apremilast for LPP.
Pyoderma Gangrenosum (PG)
PG is a neutrophilic ulcerative dermatosis characterized by a chronic and relapsing course. Generally, long-term treatment is required, often compounded with adverse effects with the existent therapeutic agents. Apremilast (label dosing) has been reported to be a valuable adjunct in the management of PG, helping in reducing the dose of immunosuppressive drugs and thereby alleviating the associated adverse effects. Responsiveness with apremilast is generally seen within 4–5 months of treatment.124, 125
Chronic Hand Eczema (CHE)
Apremilast led to complete clearance of CHE in a 65-year-old man after 1 month of therapy. Besides, there was a complete disappearance of hepatogenic pruritus in this patient. The exact role of apremilast in CHE is not clear but its ability to reduce pro-inflammatory cytokines (IL-2, IL-5, IL-13, IL-17, and TNF-α) and increase IL-10 (an anti-inflammatory cytokine) following blockade of PDE4 has been proposed. 126 In hepatogenic pruritus though, its mechanism needs elucidation.
The level of evidence for the use of apremilast for off-label dermatologic indications is represented in Table 6.
Level of Evidence for the Use of Apremilast in Off-label Dermatological Indications.

Adverse Effects
The most common adverse effects encountered with apremilast are diarrhea, nausea, and headache. Other side effects include upper respiratory tract infection, vomiting, nasopharyngitis, upper abdominal pain, dyspnea, cough, and skin rash. However, most of these side effects are mild-to-moderate in intensity with a self-limiting nature. As far as the laboratory parameters are concerned, no significant abnormalities have been encountered. Overall, apremilast is a safe drug with a favorable toxicity profile. 2
Future Directions
Although apremilast has been effective in many dermatologic disorders, its utility as a standalone therapeutic agent still remains debatable. However, combining it with systemic steroids and other immunosuppressive drugs can be considered avenues for further exploration; and as apremilast has a favorable safety profile, its addition to various regimens can prove to be advantageous, first by its own anti-inflammatory actions and second by enabling quicker reduction in the dose of immunosuppressive drugs, thereby avoiding the likelihood of adverse reactions occurring following the usage of these drugs for prolonged periods.
Conclusion
Though initially evaluated for treating plaque psoriasis and psoriatic arthropathy, apremilast has demonstrated to be a valuable adjuvant in a number of dermatological disorders. Further, owing to the lack of immunosuppression, apremilast delineates an added advantage when compared to the usual immunosuppressive drugs used by dermatologists, and with various reports outlining its efficacy in numerous dermatoses, its addition to various therapeutic regimens can be an approach to contemplate upon. Evidence for most off-label uses comes from open-label trials or case reports, with limited sample sizes and short-term follow-up. Robust clinical trials therefore become necessary to confirm the efficacy and safety of apremilast for these indications.
Abbreviations
Footnotes
Acknowledgments
None.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
