Abstract
Background and Objectives
Conflict of interest (COI) exists when a set of circumstances creates a risk that a secondary interest will unduly influence professional judgment or actions regarding a primary interest. This knowledge regarding ethics and rights is given to postgraduate students in their first year of residency through various training programs. In this training, one of the concepts taught is COI. There is no available literature regarding awareness of COI among postgraduate students who are going to be future researchers; hence we decided to conduct this study.
Materials and Methods
It was a cross-sectional, observational, single-center, questionnaire-based Study. The validated questionnaire was administered to the postgraduate students after obtaining their written informed consent. These questions were designed based on four domains: Awareness, perception, case scenarios, and application of COI. The responses were recorded as Yes/No & presented in descriptive statistics.
Results
The mean age of the participants was 28.67 years with a standard deviation (SD) of 1.92. Sixty-one percent of were female. More than 85% of postgraduate residents have an awareness of COI. Also, 70% of participants felt that increased protocol deviations and 91% reported research study outcomes are likely to be positive when there is COI in the study. Ninety-three percent of participants thought that education regarding COI was necessary.
Conclusion
The majority of postgraduate students had an awareness of COI. Postgraduate students perceived that the results of the study involving COI may not be credible, and formal education and training are required in this area.
Introduction
Conflict of interest (COI) is a set of conditions in which professional judgment concerning a primary interest (patient’s welfare or the validity of research) tends to be or appears to be unduly influenced by a secondary interest (financial or non‑financial gain). 1 COI in clinical research extends to all stakeholders, including the sponsor, principal investigator (PI), ethics committee (EC), etc. COI in science may be a source of prejudice and ultimately lead to serious distortions of research results. 2 The interference of such interests may strike at science’s internal cohesion and integrity. Distorted or even false results of such research undermine the integrity of science and weaken the trust that society has in it. 3 Although it is not possible to eliminate COI from science, as well as from other areas of human action, attempts are made to limit its negative effects.
Research is an integral part of ever-changing medical science. Carrying out medical research by postgraduate students is a part of their postgraduate curriculum. Students must submit their thesis before being awarded the Doctor of Medicine/Master of Surgery (MD/MS) degree. A study by Giri et al. showed that the lack of infrastructure is one of the barriers to carrying out research. 4 Another study carried out by Saeed et al. showed that lack of proper research training, lack of previous exposure, and time management are major hurdles to carrying out postgraduate research work. 5 One of the keys to carrying out the research is an effective literature search and identifying and weighing evidence if there is a disclosed COI. 6 The disclosed COI in a publication should encourage the postgraduate student to consider whether the study design was objective, whether the data were appropriately analyzed, and whether interpretations and conclusions accurately reflect the data. Postgraduate students need to complete their thesis work, which is their exposure to research methods. Hence, it is relevant for postgraduate students to understand the importance of COI while reviewing the published articles as a part of literature review. Thus, we decided to carry out the study in them. Hence, this indicates that the postgraduate student’s sensitization toward this aspect is extremely important. Thus, this study was planned to evaluate the postgraduate student’s awareness and knowledge regarding COI in published research articles.
Materials and Methods
Study Design
It was a cross-sectional, observational, single-center, questionnaire-based study conducted at a tertiary care teaching hospital. Approval from the Institutional Ethics Committee was obtained before conducting the study (EC/OA-87/2019).
Study Population
One hundred and forty postgraduate medical students from all the specialties of a tertiary care teaching hospital were approached. Participants who were unwilling to give consent were excluded from the study. Out of 140 students, 100 students consented to participate in the study and were included in the study. The participants of male or female gender and in the II or III year of postgraduate residency students were included in the study.
Questionnaire Development and Data Collection
The draft version of the questionnaire was developed by the research team following a comprehensive literature review.4, 5 The study team developed the questionnaire and validated it for face and content by eight experts. A validated questionnaire was administered to each of the participants, and a time of 20 minutes was given to each for completing the questionnaire. The questionnaire was framed under four domains—general awareness, perception, case scenarios, and application. For general understanding, five questions were asked. For assessing the perception, nine questions were asked, then there were two case scenarios given having several questions 7 and 3, respectively. Finally, three application-based questions were asked, the first having three sub-questions and the third having two sub-questions. All the responses were recorded as Yes/No. Details pertaining to the questionnaire are provided in the appendix.
Data Collection and Analysis
The data obtained from questionnaires was entered into Microsoft Excel and analyzed using descriptive statistics. The categorical data was expressed as proportions, and quantitative data was expressed as mean with standard deviation (SD). All the statistical tests were done using IBM SPSS software, version 26.
Results
The mean age of the participants was 28.66 ± 1.92 years. Sixty-one percent of females and 39% of males participated in the study. Sixty-one percent of second year and 39% of third-year postgraduate residents have participated in the study. All the participants responded with a response rate of 100%.
Domain 1: General Awareness About COI
One-hundred percent of the participants were aware of the term COI. Ninety-three percent of the participants have reported that the researchers should declare the COI in the published research article. Seventy percent of the participants felt that researchers with COI were tempted to disregard inclusion criteria or delay withdrawing subjects from the study. Ninety-one percent of participants reported that if COI is present in the study, the chance of reporting positive results will increase. Eighty-seven percent of the participants reported that the doctors should be educated about the COI (Table 1).
General Awareness Regarding COI.
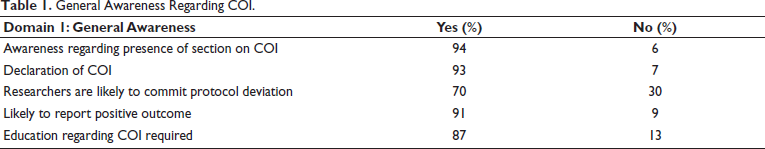
Domain 2: Perception About COI
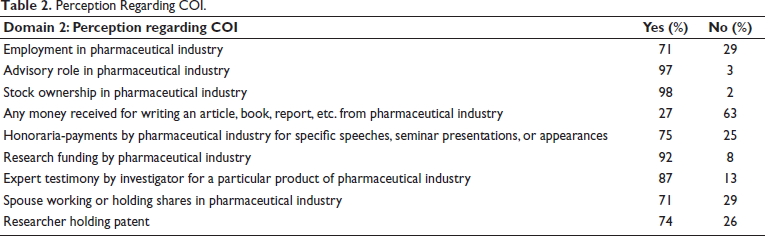
To know the perception, eight instances were given and asked about whether the investigator will have a COI or not. Seventy-one percent of participants said there will be employment in the pharmaceutical industry whose drug or device is being investigated. Ninety-seven percent participants reported an advisory role, 98% said getting stock ownership, and 92% said getting research funding in the pharmaceutical industry, whose drug or device is being investigated. Sixty-three percent participants reported money received for writing an article, book, or report, and 75% said about receiving honoraria-payments by the pharmaceutical industry whose drug or device is being investigated. Eighty-seven percent of participants reported expert testimony by investigators for products of the pharmaceutical industry whose drug or device is being investigated. Seventy-one percent said there will be COI if the spouse is working or holding shares in a pharmaceutical industry whose drug or device is being investigated. Seventy-four percent of participants reported having COI if researchers hold a patent on an intervention that is being investigated (Table 2).
Perception Regarding COI.
Seventy percent of the participants felt that the researchers were tempted to disregard the inclusion criteria or delay withdrawing subjects from the study. Ninety-one percent of participants reported that if COI is present in the study, the chance of reporting positive results will increase. When participants were asked regarding their perception of impact on the research when there is COI, 94% of the participants felt that there could be manipulation of data/findings, 12% felt that there could be pressure on participants to participate, and 92% felt that there could be tendency of study team members to not report/publish negative findings. When asked about probable reasons why there is reluctance to declare COI, 88% of participants felt that the validity of the findings may be questioned, and 86% felt that the investigator’s honesty and integrity will be questioned.
Domain 3: Case Scenarios About COI
In Case 1, where Dr. XYZ was an investigator of the research proposal and held shares in the company whose drug was being investigated, 72% of the participants stated that Dr. XYZ should not work on the project, 95% stated that Dr. XYZ should declare COI to the EC, 31% postgraduate residents reported that Dr. XYZ should declare his shares in the company to the research participants, whereas 92% said Dr. XYZ should declare having shares to conferences and publication house. Seventy-two percent participants reported believing the study results and only about 10% of participants reported changing their clinical practice according to the study results. Sixty-nine percent of the participants reported that it is necessary to know the amount of shares Dr. XYZ holds in the company (Tables 3 and 4).
Dr. XYZ is an investigator of a research proposal. He also holds shares in the company whose drug is being investigated. With respect to this scenario please answer the following questions.

Dr. ABC works in a reputed institute and has pressure to perform in his academic career by publishing articles and writing grants. He plans an ambitious study in his area of interest. Do you think the following scenario in this context amount to conflict of interest?

The second case was about Dr. ABC’s work in a pressurized environment to perform well in his academic career by publishing articles and writing grants, plans, and studies. For this case, all 100% of the participants have reported that there will be COI if Dr. ABC is a member of the EC that will review research protocol, if Dr. ABC applies for a research grant where he is on the advisory board providing research grants and if Dr. ABC sends the article for publication where he is part of the editorial board.
Domain 4: Application-Based Questions
When participants were asked about the impact on the research when there is COI, 97% of the participants stated there would be manipulation of the data, 72% of the postgraduate residents stated that there would be pressure on research participants to participate in a study, 96% of the participants stated that there will be pressure on the investigator to not to publish negative results. Ninety-six percent of the participants stated that if they have COI, they will report it while publishing the study. When participants were asked about the reluctance to declare COI in research 93% reported that the validity of the findings may be questioned and about 82% stated that investigators’ honesty and integrity will be questioned.
Discussion
The study found that postgraduate medical students are aware of COI and understand the importance of declaring the same. Most of the students agree that they must be educated regarding COI. Many postgraduates perceive unethical practices, for example, disregarding inclusion criteria as a possibility if the study has team members with COI. The participants identified probable reasons for reluctance to declare, which may be due to the questioning of the validity of the findings and the researcher’s honesty and integrity. The postgraduate students agreed that monetary interest (e.g., shares in the company) must be revealed to the EC and publication house. However, very few students felt that the same must be revealed to the participants of the research study. Most of the postgraduate students said they were not willing to implement the findings of studies with conflict in their clinical practice. All participants had knowledge of the obvious COI situations during research, for example, if the investigator was a part of the Ethics committee, was on an advisory board providing grants, or was on the editorial board of the journal. There was also good knowledge regarding what must be reported as COI except that the money received by the investigator as a part of medical writing from a pharmaceutical industry whose drug or device is being investigated was not considered as COI by most postgraduate students.
The knowledge regarding COI could be very important to readers considering that the COI, if it had become the primary interest of stakeholders then, may compromise research integrity and impend public trust in scientific research. There is evidence to suggest that industry sponsorship is associated with higher pro-industry results. 7 Also, Gajbhiye et al. study found a significant association between COI and reporting of a positive outcome. 8
We found that all postgraduate students were aware of the term COI in published articles. The students are exposed to a foundation course that includes research methodology. In India, according to National Medical Council (NMC) guidelines, students are required to complete the basic course in biomedical research, an online certification course during their postgraduate curriculum. This may be the reason for the increased awareness. The training of the students may impart a higher level of knowledge as most students in our study reported a need to educate regarding COI. Jahanfar and Molaei studied the training in two academic settings at the University of Kuala Lumpur. 9 The result suggests that students from the academic setting imparting training had a higher level of knowledge in publication ethics compared to students from the setting with no training. 9 Another study by Trigotra et al. showed that an educational intervention was given to postgraduate students. 10 It was found that the knowledge regarding COI was significantly increased following the educational intervention. 10
In a study conducted by Mashaqhbeh et al. on pharmacy students in Jordan, it was found that most participants agreed that COI should be disclosed by all authors (84.5%). 11 This finding is similar to our study in which 93% of participants agreed that the COI must be disclosed. However, a study conducted by Wager et al. which was conducted on more than 500 editors in chiefs found that they are concerned less about publication ethics and believe that misconduct in the form of undisclosed conflicts of author and reviewer severity is low (less than 1 on a scale of 0–3). 12 In a study conducted by Lacasse and Leo, the clinicians perceived lower credibility in the COI group than in the group with no COI, a difference of 31.42% on the credibility scale. 13 Similarly, in the study conducted by Mashaqhbeh et al. majority of participants (79.3%) agreed that COI could influence the integrity and the quality of research. 11 This finding is like our study, where 94% of participants felt that there could be manipulation of data doubting the credibility of data. Also, only 10% of postgraduate students said they were ready to change their practice based on the findings of the study. This could be due to the lower trust in the way the study was conducted and perceptions that secondary interests could influence the study team.
Revealing monetary interest to the EC and journal publishers was agreed upon by many students; however, a very few felt the need to disclose this information to research participants. However, a study conducted by Kim et al. found that 64–84% of potential participants felt financial COI should be disclosed as part of informed consent. 14 Also, when potential participants were asked, it was found that most people wanted to know about financial interests, but exactly how they would use the information in evaluating the risks and decision-making was less clear to them. 15 The policies of academic medical centers in the United States were reviewed to determine the level of disclosures to be made, and it was found that 48% mentioned disclosing conflicts to potential research participants. 16 Thus, though investigators think that the disclosure to participants may not be essential, participants think otherwise. Also, not all potential participants will make appropriate use of this information.
In a study conducted by Schroter et al. 2017, corresponding authors from 100 countries found that the perceived “substantial” knowledge about COI is approximately 60%, whereas 37% felt that they had “some” knowledge about COI. 17 In our study, though we did not ask for perceived knowledge, we found that the actual knowledge ranges from 27 to 100%. The Schroter et al. study reported that receiving speaking fees by the investigator was rated as less unethical than if a research grant was involved. 17 This finding is like our study, in which only 27% of postgraduate students thought that the money received for medical writing or speech from a pharmaceutical industry whose drug or device is being investigated can be considered COI.
The strength of our study is that there are no studies in India that have studied the knowledge, awareness, and perception of postgraduate students regarding COI in clinical research. The limitations of this study are that we conducted the study in a single center, convenience sampling was used, and study design was employed. Thus, most postgraduate students have an awareness of COI. Postgraduate students felt that formal education and training were required in this area. They also perceived that the results of the COI study may not be credible and that they are not likely to inculcate the findings of such studies in their clinical practice. Tertiary teaching hospitals can conduct more research methodology workshops and research-related conferences involving institutional EC for the postgraduate residents to implement strategies in teaching and learning to promote high-quality research, protection for research integrity, and public trust in medical research.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The ethical clearance has been granted from the Ethics Committee. Informed Consent has also been received from the participants.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
