Abstract
Background
Chronic myeloid leukaemia (CML) is a haematopoietic stem cell disease characterised by the proliferation of granulocytes and their immature myeloid precursors. The treatments recommended are oral treatments dispensed in the outpatient unit (OU) of the hospital’s pharmacy service (PS), where patient-centred pharmaceutical care (PC) is essential to enhance therapeutic adherence, detect drug interactions and create a pharmacist-patient relationship to educate the patient on the main potential toxicities derived from the treatment and warning signs or symptoms requiring immediate attention.
Methodology
We present the case of a 55-year-old woman, followed up in consultation by the Haematology Service (HS) since October 2021 for CML in the chronic phase.
Results
She started treatment on nilotinib 300 mg every 12 hours. The patient attended OU and was offered PC. After 12 months, nilotinib was well tolerated, but our patient experienced a loss of effectiveness, so HS decided to switch to dasatinib 100 mg daily. After one year of treatment, our patient was presented with symptoms of swelling and dyspnoea on moderate exertion. In January 2024, following a joint Haematology-Pharmacy session, a switch to bosutinib 400 mg daily was decided. After 15 days of treatment, the patient presented without an appointment due to a pruritic generalised rash on the torso, face, and extremities, being diagnosed with leukocytoclastic vasculitis.
Conclusion
In conclusion, we describe case reports of a patient who has a lack of effectiveness and ARs in several TKIs and the importance of identifying side effects, through early and close PC, in order for the patient’s evolution to be favourable.
Introduction
Chronic myeloid leukaemia (CML) is a haematopoietic stem cell disease characterised by the proliferation of granulocytes and their immature myeloid precursors. It is a common leukaemia, accounting for 15%–20% of all cases.
According to NCCN guidelines, CML manifests as:
Philadelphia chromosome-positive or BCR-ABL1 positive, encompassing chronic, accelerated and blast phases in this subgroup.
Philadelphia Chromosome-negative and BCR-ABL1 Negative
Chronic phase treatment is divided according to the risk index into low, intermediate and high risk. As stated by these guidelines, 1 for low-risk patients, the recommended treatment is first-generation tyrosine kinase inhibitors (TKI) (imatinib), followed by second-generation TKI (bosutinib, dasatinib or nilotinib) or clinical trial. For intermediate or high risk, the recommended initial treatment is second-generation TKI, as well as first-generation TKI or clinical trial.
TKIs inhibit the BCR/ABL protein by preventing its phosphorylation, so that it loses activity, causing leukaemic cells to enter apoptosis.
All the treatments indicated above are oral treatments dispensed in the outpatient unit (OU) of the Hospital’s Pharmacy Service (PS), where patient-centred pharmaceutical care (PC) is essential to enhance therapeutic adherence, detect drug interactions and create a pharmacist-patient relationship to educate the patient on the main potential toxicities derived from the treatment and warning signs or symptoms requiring immediate attention. The most common adverse reactions (ARs) of TKIs are respiratory tract infection, myelosuppression, headache, pleural effusion, diarrhoea, vomiting, nausea, rash and oedema.
Case Report
We present the case of a 55-year-old woman who has been followed up in consultation by the Haematology Service (HS) since October 2021 for CML in the chronic phase. At the time of diagnosis, she had BCR/ABL1 = 34.2% and a positive Philadelphia chromosome.
Prognostic indices were calculated: Sokal = 0.77 (low risk), Hasford = 1045 (intermediate risk) and Eutos = 37 (low risk). Our patient was classified as low-intermediate risk, and in October 2021, she started treatment on nilotinib 300 mg every 12 hours. The patient attended OU and was offered PC. It was stratified, according to the adaptation of the CMO model to onco-haematologic patients, 2 , into patients with priority. 3
Among the actions carried out, the following stand out: information on the dosage and form of administration of the new drug (without food), prevention and minimisation of ARs, written information regarding was provided, along with was made available the PS so that he could contact in case of any doubt. Furthermore, home treatment was reviewed for the detection of interactions with new TKI, but none were detected. During the various visits to the OU for medication collection, the main ARs requiring dose reduction and monitoring of the effectiveness and safety of nilotinib were monitored, as shown in Table 1.
Effectiveness and Safety of Nilotinib.
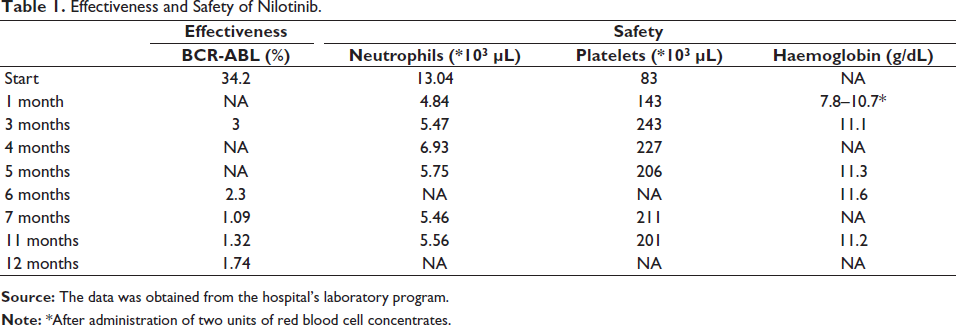
After 12 months of treatment, nilotinib is well tolerated. Anaemia was detected in the first month, which was reversed after administration of two units of red blood cell concentrates. Despite initial tolerability, our patient experienced loss of effectiveness with a BCR-ABL >1% at 12 months, prompting a joint decision with the HS to switch to TKI therapy due to lack of response.
It was decided to switch to dasatinib at a dosage of 100 mg daily. The patient was provided with information regarding the new treatment, common ARs, how to detect them, and the importance of contacting healthcare providers for any concerns. Monitoring of the effectiveness and safety of dasatinib is shown in Table 2.
Displays the Patient’s Follow-up on Dasatinib Therapy.
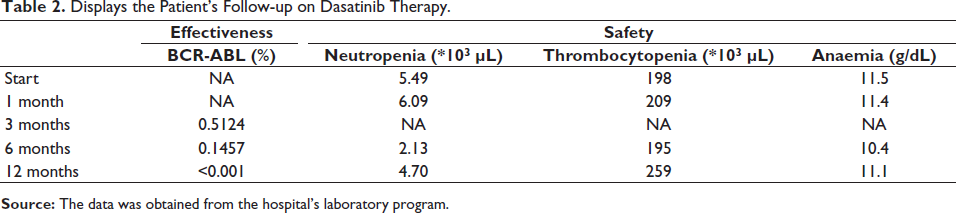
After one year of treatment, our patient presented with symptoms of swelling and dyspnoea on moderate exertion. Following consultation with HS, the diuretic dosage was increased, and the patient was promptly referred to the Cardiology Service. Subsequently, echocardiography revealed pleural effusion. HS decided to discontinue dasatinib and initiate diuretic therapy along with corticosteroid treatment until recovery.
Despite clinical improvement, it was decided to restart treatment with close follow-up. After 15 days, the patient experienced a progression of pleural effusion and dyspnoea on moderate exertion, leading to the definitive discontinuation of dasatinib. ARs were reported to the Spanish Pharmacovigilance System (SPS) by the PS.
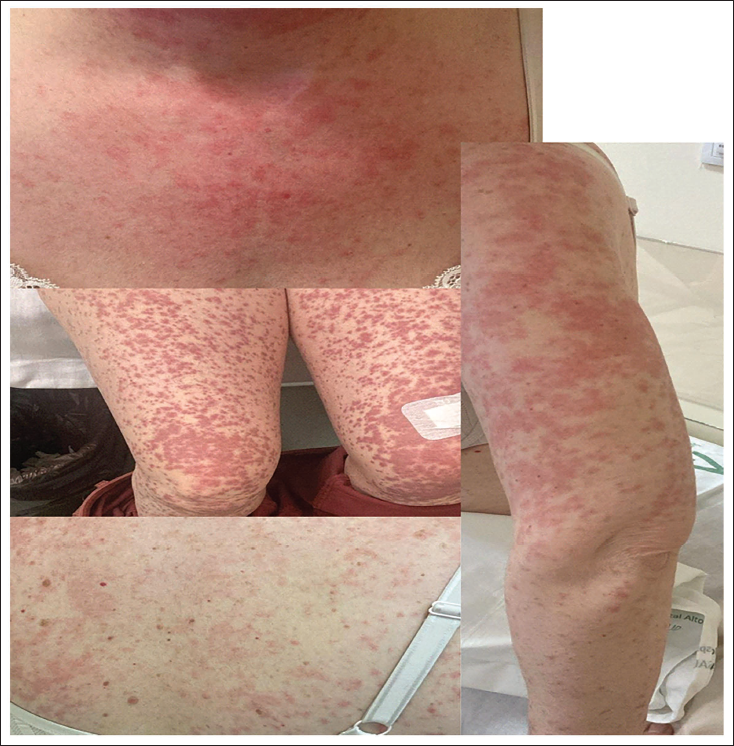
In January 2024, following a joint Haematology-Pharmacy session, a switch to bosutinib 400 mg daily was decided. Our patient returned to the OU for dispensing of the new treatment, being informed about the most frequent ARs, and emphasising the importance of contacting for any doubts or issues. After 15 days of treatment, the patient presented without an appointment due to a pruritic generalised rash on the torso, face, and extremities, as depicted in Figure 1. The HS decided to discontinue bosutinib and initiate treatment with corticosteroid. A consultation with the Dermatology Service was conducted, and a skin biopsy confirmed leukocytoclastic vasculitis. The PS reported the AR to the SPS. Following the resolution of cutaneous lesions, a multidisciplinary session decided to initiate asciminib at 40 mg every 12 hours without incidents to date.
Torso, Face and Extremities of our Patient.
Discussion
In the World Health Organisation’s Pharmacovigilance Database (VigiBase®), 3 4,335 cases of pleural effusion associated with dasatinib 3 have been documented, ARs described in the summary of product characteristics, along with two cases of vasculitis associated with the use of bosutinib 3 not described in the summary of product characteristics.4, 5 Additionally, articles related to pleural effusion associated with dasatinib have been found.6, 7
This case underscores the importance of PC, pharmacovigilance, and patient stratification in the active monitoring of their treatments, enabling the timely detection of ARs in the PS and lack of effectiveness. Furthermore, multidisciplinary teamwork plays a crucial role in contributing to patient safety.
The reporting of these side effects plays a key role in the expansion of the current pharmacovigilance database. The pharmacist has a big role in the Reporting of ARs and the PC of these patients with oral antineoplastic drugs. Literature has been found that points to the benefits of pharmacokinetic monitoring in patients being treated with dasatinib, with the possibility of reducing doses to minimise ARs without reducing efficacy. 8 The patient could have benefited from this information.
Conclusion
In conclusion, we describe case reports of a patient who has a lack of effectiveness and ARs in several TKIs and the importance of identifying side effects through early and close PC for the patient’s evolution to be favourable. The vasculitis described by our patient is not a typical reaction to bosutinib and the consequences can be dangerous. It is very important to report these types of rare ARs and to make them visible to other healthcare professionals.
Footnotes
Abbreviations
CML: Chronic myeloid leukaemia; OU: Outpatient unit; PS: Pharmacy Service; PC: Pharmaceutical care; HS: Haematology Service; TKI: Tyrosine kinase inhibitors; ARs: Adverse reactions; SPS: Spanish Pharmacovigilance System.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Not applicable.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed Consent
The consent form has been obtained from the participant.
