Abstract
Background
Diffuse pulmonary fibrosis is a progressively worsening lung disease that poses a serious threat to human health. Current treatment options are limited and generally ineffective.
Objectives
This study aimed to investigate the therapeutic effects of GubenYifei-Tang (GB) in a rat model of pneumoconiosis.
Materials and Methods
We established a chronic pneumoconiosis model in rats through intratracheal injection of silica dust. Subsequently, the rats were randomly assigned to various groups, including the model group (NC group), Hanfangji Masu tablet group (TET group), high-dose GB group (GB-H group), low-dose GB group (GB-L group), and a blank control group comprising 12 normal rats (NC group). We assessed blood biochemical, vascular endothelial function, immunoinflammatory factors, and oxidative stress indices in four rats from each group at the 1st, 3rd, and 6th months of administration.
Results
Throughout the 6-month administration period, rats in the GB-L and GB-H groups displayed no significant abnormalities in their general condition or body weight. Both high and low doses of GB significantly reduced the respiratory rate and increased the respiratory amplitude of the pneumoconiosis model rats (p < 0.05). Additionally, they significantly decreased the mean pulmonary arterial pressure of these rats (p < 0.05). Moreover, we observed noteworthy improvements in blood gas indices and hypoxemia symptoms, including increased pH, decreased arterial partial pressure of carbon dioxide, increased arterial oxygen partial pressure, and arterial oxygen saturation. Compared to the NC group, the model group rats exhibited significantly elevated concentrations of serum transforming growth factor-beta (TGF-β), tumor necrosis factor-alpha (TNF-α), and interleukin-1 (p < 0.01). High-dose GB significantly reduced serum levels of TGF-β, TNF-α, and interleukin-10 in rats, while low-dose GB partially improved serum inflammatory factor levels. Additionally, the lung mass index, malondialdehyde (MDA) levels, and hydroxyproline levels were significantly increased in the model group rats compared to the NC group (p < 0.01), accompanied by decreased superoxide dismutase (SOD) levels. Both high and low doses of GB demonstrated substantial improvements in lung mass index, MDA, SOD, and hydroxyproline levels in rats (p < 0.05).
Conclusion
In the rat model of pneumoconiosis, GB exerts a positive therapeutic impact on lung tissue lesions and respiratory function, with its effectiveness being both time- and dose-dependent.
Introduction
Diffuse pulmonary fibrosis is a systemic condition resulting from the long-term inhalation and retention of dust in the lungs, negatively impacting human health. 1 Immune dysfunction and the complex interplay of cytokines contribute to pneumoconiosis, a disease with a still poorly understood pathophysiology. The search for safe and effective preventive and therapeutic interventions, as well as the investigation of early pneumoconiosis pathophysiology, pose formidable challenges. 2 Currently available medical therapies offer limited effectiveness, addressing only pneumoconiosis symptoms. 3 Prolonged medication use can lead to drug resistance and adverse side effects such as immunosuppression, liver, and kidney damage. 4 GuBenYifei-Tang (GB), a popular traditional Chinese medicine remedy for lung ailments, has demonstrated positive impacts on lung health and immunity and possesses antioxidative and anti-inflammatory properties. The GB formula consists of Astragalus membranaceus, Codonopsis pilosula, Citrus aurantium, Angelica sinensis, Bupleurum chinense, Platycodon grandiflorum, Atractylodes macrocephala, Paeonia lactiflora, Salvia miltiorrhiza, Crataegus pinnatifida, and Glycyrrhiza uralensis. Patients with pneumoconiosis can benefit significantly from C. pilosula’s expectorant, antitussive, and bronchodilatory properties. A. membranaceus, A. sinensis, and other components nourish bones, replenish Qi, and fortify the spleen, enhancing patients’ physical well-being and resistance. Additionally, A. membranaceus, Panax ginseng, and other plants exhibit immunomodulatory actions that can bolster the immune system, offering valuable support in pneumoconiosis treatment. Despite its clinical applications in traditional Chinese medicine, rigorous scientific research is lacking to substantiate GB’s pharmacological mechanism and therapeutic effects in pneumoconiosis. Therefore, this study aims to establish a rat pneumoconiosis model to investigate GB’s pharmacological effects and therapeutic efficacy in treating pneumoconiosis. This research aims to contribute novel care techniques, a theoretical foundation for pneumoconiosis treatment, and scientific guidance for GB’s practical application. The ultimate objective is to provide safer and more efficient treatment options for pneumoconiosis patients, thus improving their quality of life and prospects for recovery.
Materials and Methods
Experimental Reagents
Tetrandrine tablets were purchased from Zhejiang Huarun Sanjiu Zhongyi Pharmaceutical Co. Ltd., with batch number H33022163. Quartz powder with a diameter <5 µm was purchased from Jiangsu Lianrui New Materials Co. Ltd. and prepared into a 50 mg/ml suspension with physiological saline for later use. Malondialdehyde (MDA), superoxide dismutase (SOD), nitric oxide (NO), transforming growth factor-beta (TGF-β), tumor necrosis factor-alpha (TNF-α), interleukin-6 (IL-6), endothelin (ET), and phosphatidylcholine (PC) enzyme-linked immunosorbent assay (ELISA) kits were purchased from Xinbosheng Biotechnology Co. Ltd.
Experimental Animals and Drug Dosage
Sixty male specific pathogen free (SPF)-grade Sprague-Dawley (SD) rats were purchased from Jiangsu Taiste Biological Technology Co. Ltd., (Production License: SCXK (SU) 2021-0010). The rats were housed at a temperature of (24 ± 2)°C, relative humidity of 60–80%, with an air exchange rate of 10–15 times per hour, and a light cycle of 12 hours (day)/12 hours (night). Each cage housed no more than five rats. For GB, the dosage for rats was calculated based on body surface area, resulting in 213.3 mg/kg. In this experiment, the dosages for rats were set at 400 mg/kg and 200 mg/kg, approximately 2 times and 1 time the clinical dosage, respectively. For tetrandrine tablets, the dosage for rats was calculated based on body surface area, resulting in 28.6 mg/kg. In this experiment, the dosage for rats was set at 30 mg/kg, approximately equivalent to the clinical dosage.
Animal Model
After satisfactory anesthesia of the rats, they were placed in a supine position. A neck incision was made to expose the trachea, and 1 ml of presuspended dust solution was slowly injected into the trachea with a 1 ml syringe over 10 seconds. After injection, the rats were placed upright and rotated left and right and massaged their lungs 10 times to ensure even distribution of the dust in the lungs. The incision in the neck was sutured, and the rats were marked and placed in separate cages for feeding. According to previous experiments and literature reports, significant lung tissue lesions appeared approximately 3 months after modeling using this method. Forty-eight of the modeled rats were randomly divided into the following groups based on body weight: (a) Model group: gavaged with an equivalent dose of physiological saline. (b) Tetrandrine tablets group (TET group). (c) GB high-dose group (GB-H group): gavaged with 400 mg/kg of GB. (d) GB low-dose group (GB-L group): gavaged with 200 mg/kg of GB. Each group consisted of 12 rats. Additionally, 12 normal rats were taken as the blank control group (NC group). The rats in each group were orally administered once a day, 6 days per week, for a continuous period of 6 months, with a dosage volume of 1 ml/kg.
Detection Indicators
General Observation
Before daily administration, observe the general condition of rats, including coat color, behavior, and movement. Focus on observing respiratory system-related symptoms, including difficulty breathing, abdominal breathing, wheezing, breathing pauses, cyanosis, rapid breathing, nasal discharge, etc. Weigh the rats once a week and adjust the dosage accordingly based on their body weight.
Respiratory Function Measurement
Respiratory function measurements should be conducted at 1, 2, 3, and 6 months after administration. At each time point, 6 rats from each group should be anesthetized with pentobarbital sodium (30 mg/kg), laid supine and fixed, and connected to a chest wall unit. The respiratory curve should be recorded using a biological function experimental system, with a 5 g calibration weight applied, to calculate the respiratory frequency and amplitude.
Pulmonary Artery Pressure Measurement
At 1, 3, and 6 months after administration, 6 rats from each group at each time point should be anesthetized with pentobarbital sodium (30 mg/kg). They should be laid supine and fixed, their right jugular vein should be isolated, and a heparinized PE tube should be inserted through the jugular vein into the right ventricle. The changes in right ventricular pressure should be measured using a BF-420S physiological recorder to examine the effects of the test drug on pulmonary artery pressure.
Blood Gas Analysis and Peripheral Blood Inflammatory Factors
Blood gas analysis should be conducted at 1, 3, and 6 months after administration. Arterial blood samples should be collected from the femoral artery of the rats, and a blood gas analyzer should be used to measure the pH, arterial partial pressure of carbon dioxide (PaCO2), arterial oxygen saturation (SaO2), and arterial oxygen partial pressure (PaO2). The levels of TGF-β, TNF-α, and interleukin-10 (IL-10) in the serum should be measured using ELISA kits.
Bronchoalveolar Lavage Fluid (BALF) Cell Classification and Composition
After blood collection, the rats should be euthanized, and the main bronchus should be punctured. Precooled phosphate-buffered saline (PBS) should be injected in four portions, with gentle lung massages to ensure complete recovery of the liquid. A cell counter should be used to determine the total white blood cell count in the BALF. The remaining lavage fluid should be centrifuged at 2,000 rpm for 10 minutes, and the precipitate should be used for cell preparation. The prepared smears should undergo Wright’s staining, and 200 cells should be counted under an oil microscope for white blood cell classification. Additionally, some of the BALF should be used to measure levels of ET, NO, and PC.
Lung Mass Coefficient, Oxidative Stress, and Hydroxyproline (HP) Level
At 1, 3, and 6 months after administration, after completion of bronchoalveolar lavage, the entire lung should be dissected, and the external appearance of lung tissue should be observed and recorded. The weight of the lung should be measured. The right lung tissue should be taken as much as possible removing the connective tissue in the hilum area and draining the fluid inside the lung. After labeling, the tissue should be frozen and stored. MDA, SOD, and HP levels should be measured using assay kits.
Statistical Analysis
The data were statistically analyzed using SPSS 25.0 software. The measurement data are all expressed in x ± s, and after analysis of variance (ANOVA) statistics, Dunnett’s t-test was performed between the two groups. p < 0.05 indicates a statistically significant difference.
Results
The General Condition of Rats in Each Group
The rats’ injuries were fully healed a week after the modeling session, and they were acting normally with no obvious signs of anomalous behavior. After modeling for one month, rats in the model group sporadically displayed cough symptoms. The number of atypical respiratory symptoms, such as belly breathing, nasal discharge, wheezing, coughing, and respiratory sounds, steadily grew over time. Most of the rats in the model group had filthy and untidy fur after three months of modeling. No notable abnormalities were noticed during the trial, and the body weight fluctuations in the rats in the model group were comparable to those in the normal group. Rats in the GB-L and GB-H groups had some behavioral and respiratory symptom relief in comparison to the rats in the model group.
Comparison of Lung Function Parameters in Each Group of Rats
One month after administration, rats in the model group showed significant respiratory abnormalities, including a significantly increased respiratory rate (p < 0.01), reduced respiratory amplitude, and increased pulmonary artery pressure. Compared to the model group, during the 6-month treatment period, both low-dose and high-dose GB treatment significantly reduced the respiratory rate and pulmonary artery pressure in rats (p < 0.05). As the duration of treatment increased, the improvement in respiratory rate by GB in rats gradually increased (Table 1).
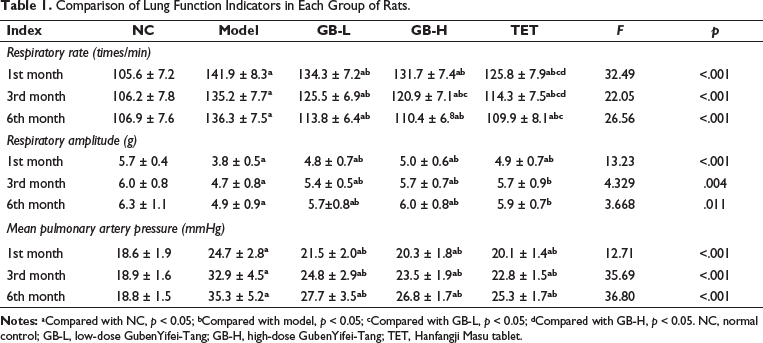
Comparison of Lung Function Indicators in Each Group of Rats.
Comparison of Blood Gas Composition Parameters in Each Group of Rats
Compared to the NC group, rats in the model group gradually developed symptoms of hypoxemia after one month of self-administration, including significantly decreased blood pH, PaO2, and SaO2, as well as significantly increased PaCO2 (p < 0.05). During the 6-month experimental period, both the GB-L group and GB-H group showed significant improvements in blood gas parameters compared to the model group (p < 0.05) (Table 2).
Blood Gas Composition Indicators of Rats in Each Group.
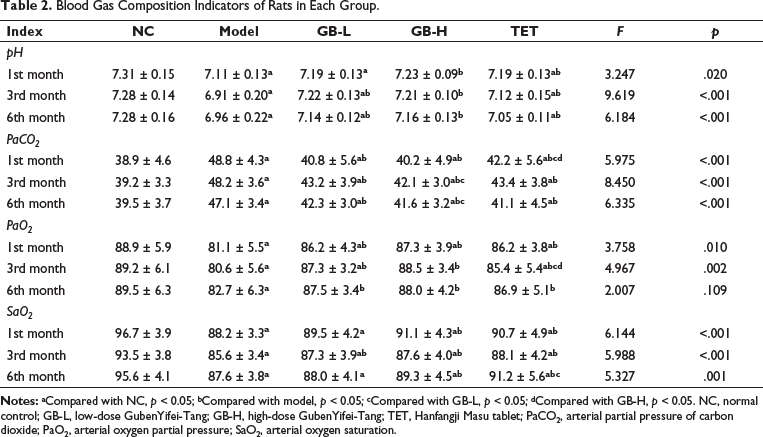
Comparison of Inflammatory Factor Levels in Peripheral Blood of Rats
Compared to the NC group, rats in the model group showed significant increases in serum levels of inflammatory factors TGF-β, TNF-α, and interleukin-1 (IL-1) (p < 0.01). High-dose GB significantly decreased serum levels of TGF-β, TNF-α, and IL-10 in rats, while low-dose GB also showed partial improvement in serum inflammatory factors (Table 3).
Comparison of Inflammatory Factor Levels in Peripheral Blood of Rats.
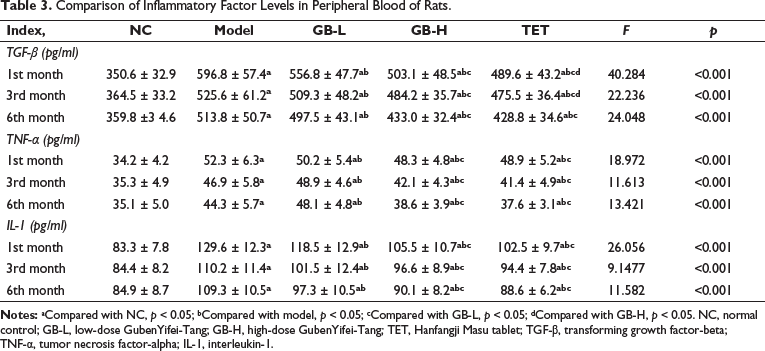
Classification and Composition of Cell Counts in BALF of Rats
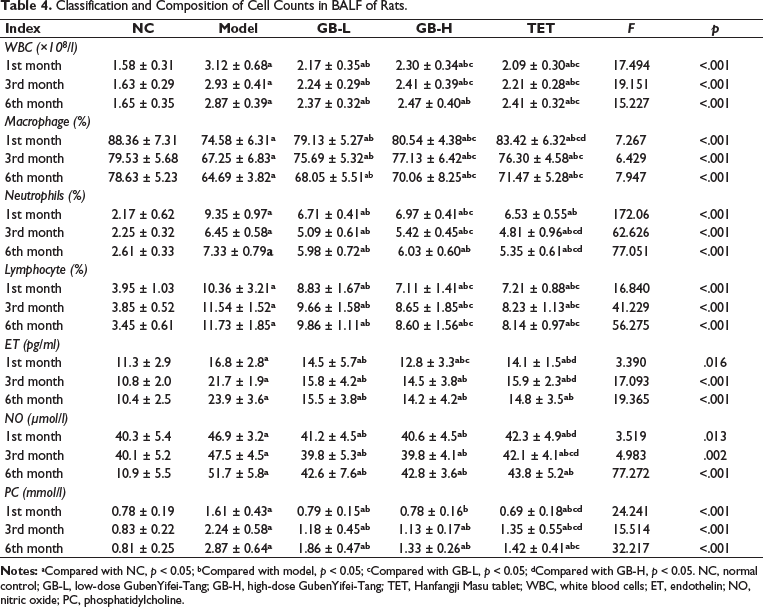
Compared to the NC group, rats in the model group showed a significant increase in the number of white blood cells in BALF (p < 0.01). The percentage of macrophages in the white blood cell count was significantly decreased (p < 0.01), while the percentage of neutrophils and lymphocytes was significantly increased (p < 0.01). Compared to the model group, high-dose GB significantly decreased the number of white blood cells in BALF of rats and showed significant modulation in white blood cell classification. Biochemical analysis of BALF showed a significant increase in ET and NO levels in rats of the model group starting from one month of self-induced modeling (p < 0.01), while PC levels gradually increased over time. Compared to the model group, high-dose GB showed significant improvement in ET, NO, and PC levels in BALF throughout the entire experimental period (Table 4).
Classification and Composition of Cell Counts in BALF of Rats.
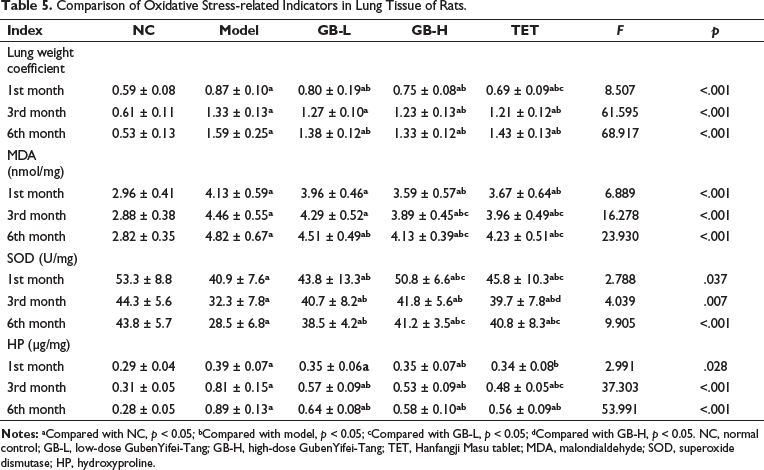
Comparison of Oxidative Stress-related Indicators in Lung Tissues of Rats
Compared to the NC group, rats in the model group showed a significant increase in lung weight coefficient, MDA levels, and HP levels after one month of administration (p < 0.01), and SOD levels were significantly decreased. As the modeling time increased, lung weight coefficient, MDA, and HP levels increased more significantly, while SOD levels decreased more significantly. High-dose and low-dose GB showed significant improvement in lung weight coefficient, MDA, SOD, and HP levels in rats (p < 0.05) (Table 5).
Comparison of Oxidative Stress-related Indicators in Lung Tissue of Rats.
Discussion
Patients with pneumoconiosis experience a range of cardiovascular changes, including alterations in lung tissue, chronic hypoxia, increased vascular resistance, myocardial remodeling, and decreased cardiac function. Furthermore, the stimulation of inflammatory factors may lead to reduced blood flow, blood clot formation, and increased blood viscosity, all of which diminish the oxygen-carrying capacity of the blood, exacerbating respiratory difficulties and hypoxia.
Respiratory function tests in the model group of rats revealed significant lung damage, characterized by a marked increase in respiratory rate, reduced respiratory amplitude, and elevated pulmonary artery pressure. This lung damage is a consequence of dust particle inhalation triggering an inflammatory response, resulting in lung tissue fibrosis and injury. This, in turn, reduces lung elasticity and imposes an increased respiratory rate to meet oxygen exchange demands. Additionally, pneumoconiosis-induced lung damage leads to a less compliant lung with reduced expansion capacity, limiting gas movement and causing decreased respiratory amplitude. 5 Moreover, lung fibrosis further diminishes alveolar elasticity and lung volume, compounding the impact on respiratory amplitude. Critically, inflammation and fibrosis in the lungs due to pneumoconiosis can result in pulmonary vasoconstriction, heightened pulmonary vascular resistance, and elevated pulmonary artery pressure. 6 However, following intervention with GB-H solution, the respiratory function of rats with pneumoconiosis significantly improved, confirming the therapeutic efficacy of GB-H solution. The specific mechanisms underlying these effects merit further investigation.
TGF-β, TNF-α, and IL-10 serve as pivotal inflammatory regulatory factors with essential roles in pneumoconiosis progression. 7 TGF-β, a versatile cytokine, triggers the proliferation of lung interstitial cells and fibroblasts, stimulating collagen and fibronectin production, ultimately leading to pulmonary tissue fibrosis.8, 9 Additionally, it inhibits alveolar epithelial cell proliferation and regeneration, thereby impacting lung reparative capacity. TNF-α, on the other hand, induces inflammatory cell infiltration and inflammatory responses, promoting lung tissue damage and fibrosis. It also activates lung interstitial cells and fibroblasts, resulting in collagen synthesis and pulmonary fibrosis.10, 11 IL-1, an inflammatory cytokine, promotes the production of inflammatory mediators and inflammatory cell infiltration. In crystalline silicosis, IL-1 overexpression significantly exacerbates inflammation and fibrosis, hindering lung repair. 12
GB exhibits anti-inflammatory and antifibrotic effects, conferring protective benefits against the progression of pneumoconiosis. These effects are attributed to the potent active components present in its herbal ingredients. For example, A. membranaceus, Ophiopogon japonicus, and Eucommia ulmoides are believed to possess anti-inflammatory and antioxidant properties, capable of mitigating lung inflammation and fibrosis. Pulmonary heart disease represents a severe complication of pneumoconiosis, often marked by recurrent acute exacerbations. 13 The condition deteriorates with the progressive decline in lung function, resulting in a generally unfavorable prognosis. Research indicates that pulmonary arterial hypertension stands as one of the primary factors directly contributing to pulmonary heart disease. Vasoactive substances, such as ET and NO in pulmonary tissue, closely correlate with endothelial dysfunction and play a pivotal role in the development of pulmonary arterial hypertension. 14 Concurrently, interstitial fibrosis in the lungs accompanies type II cell proliferation. 15 During this phase, the alveolar tissue’s surfactant protein C levels significantly increase. Hence, the measurement of PC levels in BALF can indirectly reflect the progression of lung tissue fibrosis. 16 This study observed that GB significantly reduced the number of white blood cells in BALF within the rat model. Additionally, it had a notably positive impact on the levels of ET, NO, and PC in the lavage fluid, suggesting that GB enhances vascular endothelial cell function and mitigates the progression of interstitial fibrosis in the lungs in the context of pneumoconiosis. Harmful substances present in dust particles, including heavy metals, organic compounds, and free radicals, can induce lipid peroxidation and cell damage by generating reactive oxygen species, leading to elevated MDA levels, an indicator of lipid peroxidation.17, 18 Dust particles can also disrupt the normal functioning of the antioxidant system, diminishing the activity of antioxidant enzymes like SOD and reducing the cell’s capacity to withstand oxidative stress. 19 GB exhibited a dose-dependent improvement in serum MDA and SOD levels in the rat model, underscoring its ability to regulate the oxidative/antioxidant imbalance in pneumoconiosis.
Conclusion
GB has demonstrated favorable effects in ameliorating inflammation, mitigating vascular endothelial damage, reducing oxidative stress, and attenuating lung tissue fibrosis in a pneumoconiosis rat model. These effects play a crucial role in the overall enhancement observed in the model. Leveraging the characteristics and mechanisms of action of GB in the pneumoconiosis model, further experimental research and clinical practice will be pursued to deepen our understanding and refine its therapeutic potential. This endeavor will significantly contribute to the advancement of pneumoconiosis prevention and treatment strategies.
Abbreviations
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All the experimental procedures were conducted in accordance with the Committee for Control and Supervision of Experiments on Animals (CCSEA), Organisation for Economic Co-operation and Development (OECD) guidelines.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: (a) Postgraduate Research & Practice Innovation Program of Jiangsu Province in China (KYCX22_3420). (b) Basic Research Program of Yancheng Science and Technology Bureau (YCBK202221).
