Abstract
Objectives
Delayed-type hypersensitivity (DTH) reactions represent a clinically important class of immune responses that have wide-ranging implications in various health and disease conditions. The existing anti-inflammatory drugs to treat DTH have several shortcomings including severe side-effects. Thus, a new plant-derived anti-inflammatory drug with no or less toxicity would be effective in treating DTH. The Cleome genus has been routinely used to cure various ailments including inflammation. This study investigates the anti-inflammatory properties of the ethanolic leaf extract of Cleome spinosa (CSE) in a DTH model for the first time.
Materials and Methods
DTH response was induced by 2, 4-di nitro-fluorobenzene in the mouse foot pad. Histological sectioning of the paw was carried out to find changes in the architecture of the tissue. Serum tumour necrosis factor-alpha (TNF-α) was estimated through enzyme linked immunosorbent assay (ELISA), and the bioactive compounds present in CSE were analysed through reversed phase-high-performance liquid chromatography.
Results
With CSE treatment, the oedema in the resensitized paw alleviated faster with no loss of digits compared to the controls. Histological study of the resensitized paw showed less dermal and sub-dermal thickening and less collagen deposition that regained normal tissue architecture during the healing process with CSE treatment. CSE treatment did not show any adverse haematological and histopathological changes in the visceral organs of mice. The level of serum TNF-α was found to be significantly lower with CSE treatment. CSE was found to be rich in phenol, flavonoids, tocopherol, carotene and phytosteroids.
Conclusion
The anti-inflammatory and immunomodulatory role of CSE against hapten-induced deleterious DTH inflammation suggests its therapeutic potential in treating inflammatory diseases and combating infections.
Introduction
Delayed-type hypersensitivity (DTH) is a major mechanism of defence against intracellular pathogens, including mycobacteria, fungi and certain parasites, and may also be involved in transplant rejection and tumour immunity.1, 2 However, DTH can also lead to pathological responses, such as granulomatous inflammation, calcification, caseation necrosis, and cavity formation. 3 Thus, anti-inflammatory therapy is suggested to be effective in curing tissue damage due to hypersensitivity reactions and towards early neoplastic progression and autoimmune disorders. 4
The currently available anti-inflammatory drugs such as corticosteroids (e.g., dexamethasone) and nonsteroidal anti-inflammatory drugs (NSAIDs, e.g., aspirin) are widely used to treat inflammatory symptoms. 5 However prolonged use of NSAIDs is associated with a number of side effects leading to organ damage. 6 Therefore, the research focusing on discovering new anti-inflammatory therapies remains essential in pharmacology where traditional medicine could be a robust source for the prevention and treatment of inflammatory diseases without any adverse side effects.
Cleome spinosa (Jacq.) is a plant species that is native to tropical America. The species was first reported as a wild species of West Bengal, India by Sivarajan. 7 It is an annual herb occurring in patches near wet sites, usually along roadsides, streams or abandoned areas across the northern side of the state of West Bengal. Genus Cleome has more than 200 species with abundant remedial and conventional applications.8, 9 Among the various species of the genus Cleome, most of the work has been carried out with C. viscosa, C. ruditosperma and C. gynandra to confirm its medicinal properties.8–11 However, few studies have been carried out to examine the anti-oxidant and anti-microbial efficacy of Cleome spinosa (CSE).12, 13 Several research studies including our previous work have shown that an individual isolated biochemical compound is often insufficient to show potent therapeutic capability; however, plant extract containing all its constituents together or a formulation containing multiple herbs shows better efficacy14–17 and this is also the focus of the Indian Ayurvedic system. Thus, the present investigation assessed the total content of the ethanolic extract of CSE in the DTH model.
Materials and Methods
Cleome spinosa Leaf Extract Preparation
Fresh leaves of CSE were collected, washed with sterilized water, shade dried and grinded into powdered form. 10 g of dried leaf powder was then soaked in 100 mL of ethyl alcohol (80%v/v) in 1:10 ratio and kept on a magnetic stirrer for 3 days. The leaf extract was then filtered through Whatman filter paper (grade 2) followed by 0.22 µm cellulose acetate syringe filtration (E4780-1223, Eppendorf) and used for experimentation.14, 15 The plant specimen was identified by the Department of Botany, University of North Bengal, West Bengal, India (Accession number 09874). 1 mL of ethanolic leaf extract of CSE contained 5.8408 ± 0.045 mg of dry weight, assessed by evaporating to dryness under reduced pressure (Rotary vacuum, Buchi).
DTH Assay
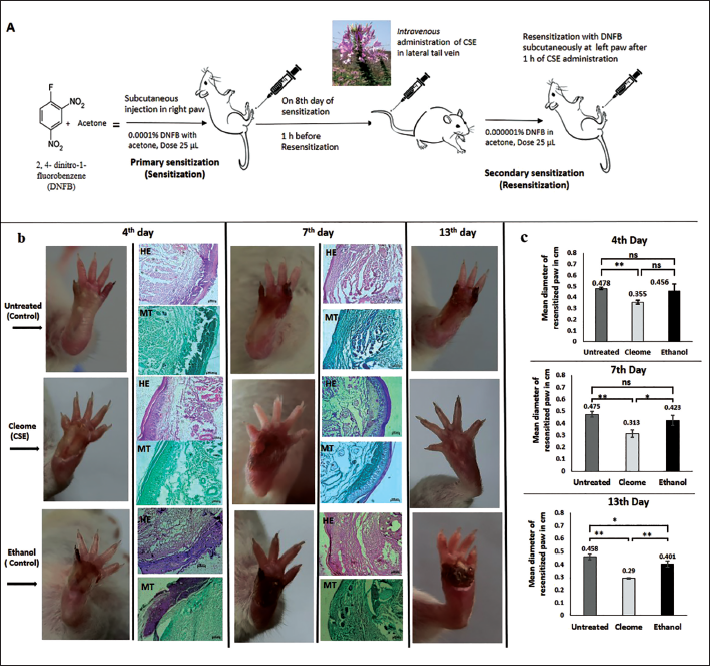
Swiss albino mice of both sexes, 12–14 weeks of age were used for the experimentation. Mice were acclimated for a week in a light and temperature-controlled room with a 12-light and dark cycle and maintained with food and water ad libitum in the animal house. All experiments were conducted at least thrice with a minimum of 10 mice per group. For inducing DTH reaction mice were sensitized by subcutaneous application of 2, 4-di nitro-fluorobenzene (DNFB; Loba Chemie) made in acetone. Primary sensitization was carried out by injecting 0.0001% DNFB (25 µL) in the right foot pad subcutaneously; and on the 8th day, resensitization (secondary sensitization) was done with 0.000001% DNFB (25 µL) injected subcutaneously on the left foot pad (Figure 1a). 14 Different doses of CSE were adjusted as µg per gram of body weight (b.w.) of the mouse. Thus, 1.82 µg/g b.w, 3.65 µg/g b.w, 7.3 µg/g b.w, 14.6 µg/g b.w and 29.2 µg/g b.w doses of CSE were administered intravenously (i.v.) 1 h before resensitization in different set of mice to ascertain the effective dose. Two control groups were considered: first group received no treatment (untreated) and for second group, 25 µL of ethanol was injected intravenously 1 h before resensitization as respective CSE doses were dissolved in ethanol (25 µL). The day of resensitization was considered as day 0 for the presentation of the data. The experimental protocols were approved by the Institutional Animal Ethics Committee (CBPBU/IAEC/ZOO/010/2018) as per the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA) guidelines (F.No.25/250/2012-AWD, dated 26 February 2014) and mice were obtained from an authorized supplier (Regd. No. 1443/PO/BT/s/11/CPCSEA). The size of the reaction spot was measured at right angles by a slide calliper as an index for the reaction.
(a) DTH was Induced in vivo by Challenging DNFB Subcutaneously in the Right and Left Mouse Foot Pad in Two Steps: Sensitization and Resensitization; (b) Micrographs showing the DTH reaction of resensitized paw in untreated (control), CSE and ethanol (control) treated mice in course of 13 days after resensitization. The photographs are representation of one set of experimental mice. c) Inflammation of resensitized paw on 4th, 7th and 13th day has been represented in cm, where day of resensitization is indicated by 0. Results are expressed as mean ± SD, ** p < 0.01 and *p < 0.05; ns: non-significant. The skin sections were stained with haematoxylin and eosin (H&E) and Masson’s trichrome (MT).
Histological Studies of the Resensitized Paw of DTH-bearing Mice
Skin biopsies were collected on fourth and seventh days after resensitization, fixed in 4% paraformaldehyde and cut into 5-µm-thick sections using cryomicrotome (Leica, CM3050S). The skin sections were stained with haematoxylin and eosin (H&E) and Masson’s trichrome (MT). 18
Toxicity Study of CSE
The experiment was conducted to study the effects of in vivo administration of CSE in normal mice. The effective dose of CSE was injected intravenously in the tail vein and after 7 and 14 days, mice were sacrificed and blood and different organs were collected. Haematological analysis was performed by Councell Penta 2.0 (Coral Clinical System) and H&E staining was done for the examination of pathological alterations.
Estimation of Serum TNF-α
Serum tumour necrosis factor-alpha (TNF-α) in DTH mice was measured via sandwich enzyme linked immunosorbent assay (ELISA) Kit (E0117Mo, BT LAB) as per the instruction manual provided. Briefly, mouse TNF-α was assayed on microplates precoated with affinity-purified polyclonal antibody specific for mouse TNF-α. Serum samples (collected on fourth and seventh days of resensitization), anti-TNF-α antibody and streptavidin-horseradish peroxidase (HRP) were added and incubated for 60 min at 37°C as per manufacturer’s instructions. After washing, substrates were added and kept in the dark for 10 min at 37°C. At the end, the reaction was terminated by adding 50 µL of stop solution and absorbance was measured at 450 nm in a Multimode Multiplate Reader (Synergy LX, BioTek).
Reversed Phase-HPLC (RP-HPLC) Analysis of CSE
Chromatographic analysis was performed on a high-performance liquid chromatography (HPLC) system (model no: 2695, Waters), multi-dimensional liquid chromatography (MDLC) equipped with a Dual λ Absorbance Detector and Dual Binary Pump (Model no. 515, Waters). The system was controlled by Empower-2 Software, Waters. Reserve Phase C18 (4.5 mm × 250 mm, 5 µm) column from Agilent Technologies was used for the chromatographic separation. Standard Stock Solutions of ascorbic acid, gallic acid, chlorogenic, tannic acid, vanillic acid, P-coumaric acid, trans-cinnamic acid, sinapic acid, ferulic acid, myricetin, quercetin, kaempferol, carotene, tocopherol and β-sitosterol was prepared in methanol and sonicated (1 mg/mL). The sample solution was prepared by dissolving CSE (5.84 mg/mL) with a mobile phase. The mobile phase was prepared with 0.5% phosphoric acid in water and acetonitrile for phenol and flavonoid isolation; methanol and acetonitrile (90:10, v/v) were used for β-Carotenes; methanol and water (90:10, v/v) was used for tocopherol, β-sitosterol and stigma sterol isolation. The responses were measured as peak areas versus concentration. The flow rate was 1 mL/min and the injection volume was 100 µL. Reversed phase-HPLC (RP-HPLC) analysis was carried out at CSIR-Indian Institute of Chemical Biology, Kolkata, India.
Statistical Analysis
All data were obtained and prepared as the mean ± standard deviation (SD) from three independent experiments. Statistical analyses were performed by one-way and two-way analysis of variance (ANOVA) using Microsoft Excel versions 2013 (64 bit) and the significance was set at *p < 0.05 and **p < 0.01.
Results
Inhibition of DTH Response with CSE Treatment
The DTH reaction in skin is biphasic with an early non-specific infiltration of cells occurring in every individual, followed by a second specific response happening only in previously sensitized individuals. 3 The successful establishment of the model causes erythema and induration at the site of antigen injection in immunized animals usually within 24–72 h, as shown in Supplementary Figure 1.
Among the different doses of CSE such as 1.82, 3.65, 7.3, 14.6 and 29.2 µg/g b.w., the dose 7.3 µg/g b.w. was found to be the most effective dose, which significantly alleviated the DTH inflammation compared to other doses (Supplementary Table 1) with no loss of digits. Hence, further experiments were carried out with 7.3 µg/g b.w. dose of CSE.
Haematological Parameters After 7th and 14th Days of CSE (7.3 µg/g b.w) Treatment in Normal Mice.
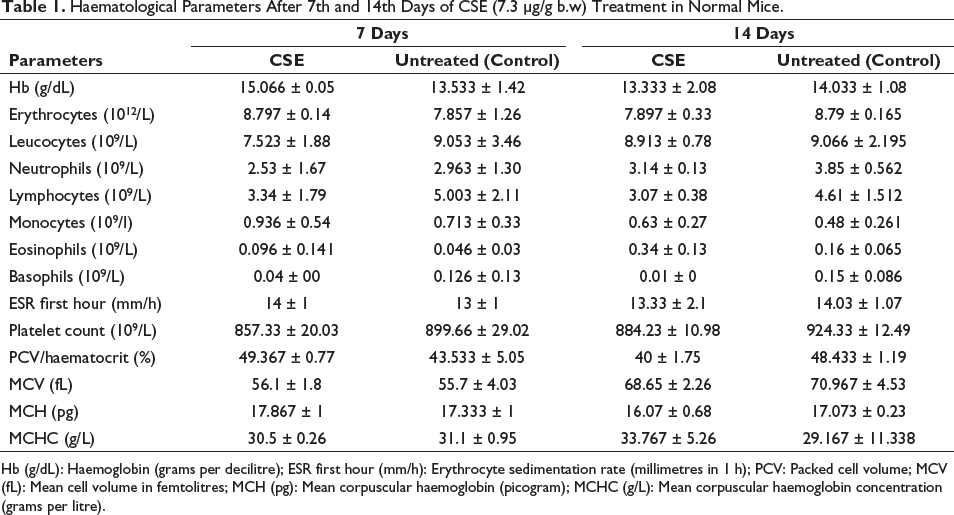
Hb (g/dL): Haemoglobin (grams per decilitre); ESR first hour (mm/h): Erythrocyte sedimentation rate (millimetres in 1 h); PCV: Packed cell volume; MCV (fL): Mean cell volume in femtolitres; MCH (pg): Mean corpuscular haemoglobin (picogram); MCHC (g/L): Mean corpuscular haemoglobin concentration (grams per litre).
DTH induration reached a maximum peak within 48 h of resensitization, which gradually declined and attained normal paw (0.29 ± 0.005 cm) size within 13 days with 7.3 µg/g b.w. dose of CSE (Figure 1c). In the control groups, on the contrary, inflammation did not subside so early and was 0.458 ± 0.022 cm (untreated) and 0.401 ± 0.024 cm (ethanol), respectively on the 13th day (Figure 1c and Supplementary Table 1). CSE significantly reduced DTH inflammation compared to both control groups (untreated and ethanol-treated). The untreated mice had swelling all through the study period and both the control groups showed loss of digit and foot pad (Figure 1b).
Effect of CSE on Skin Tissue Architecture Of Resensitized Paw
Histological study of the resensitized paw showed spongiosis, epidermal and dermal thickening as well as lymphocytic infiltrations, characteristics of DTH reaction. H&E-stained skin sections of resensitized paw from CSE (7.3 µg/g b.w.) treated DTH mice appeared to have more compact tissue architecture in comparison to the untreated and ethanol-treated control mice (Figure 1b) on fourth day after resensitization. In both control groups, the dermal and the sub-dermal layers were disrupted with the formation of sinuses and can be correlated to more swollen resensitized paw in control mice compared to CSE-treated mice, as expressed by the mean diameter of the paw (Figure 1c and Supplementary Table 1). MT-stained sections of the same tissue showed extensive layers of collagen in control groups and more confined layers in CSE-treated mice (Figure 1b) and corroborates with observations from H&E-stained sections.
Both H&E and MT-stained sections of skin biopsies collected on the seventh day after resensitization showed a thick, cutaneous epidermis layer in resensitized paw from both control groups and a much thinner and consistent epidermis in CSE-treated mice (Figure 1b). MT-stained sections also showed more greenish dermal and hypodermal layers in control groups indicative of more collagen deposition than the CSE treatment groups. In fact, for CSE treatment collagen level seems to be lesser on the seventh day compared to that on the fourth day.
Haematological and Histological Observation After CSE Treatment
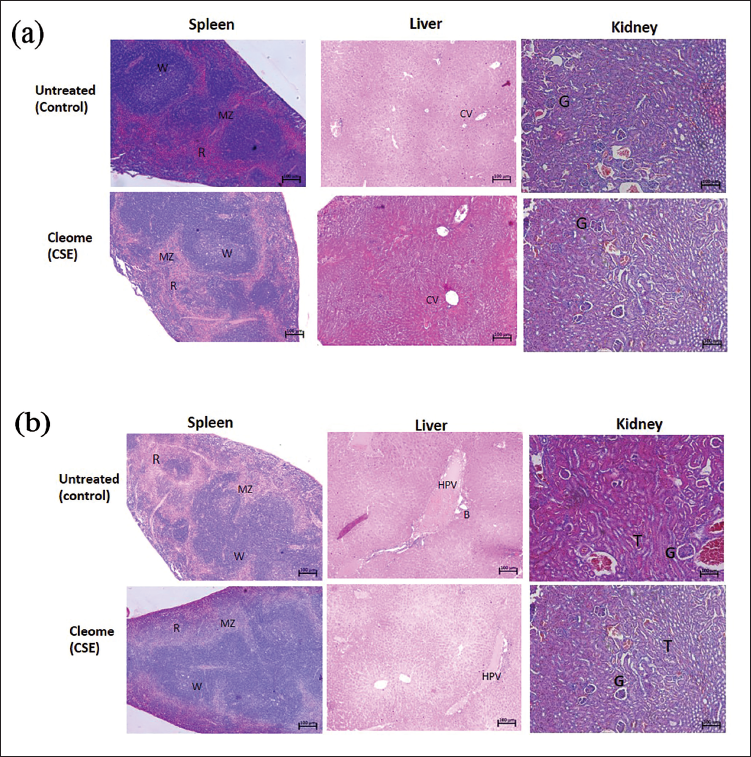
This study showed that the i.v. application of CSE (7.3 µg/g b.w.) in normal mice did not induce any apparent changes in the haematological as well as the histological architecture of spleen, liver and kidney, and was consistent with those of untreated ones (control) after 7 and 14 days. Table 1 shows no marked pathological difference in the haematological parameters of CSE-treated mice compared to the untreated mice. Supplementary Table 2 shows the weight and Supplementary Figure 2 shows photographs of the spleen, liver and kidney of both treatment and control group with no marked differences after 7 days. The spleen of CSE-treated mice had distinct white and red pulps with clear margins and no vascular expansion. Similarly, the hallmark events of hepatotoxicity like distorted lobule, cholestasis, glycogen and lipid deposition as found in drug-induced liver injury 19 were not seen in the liver of CSE-treated mice (Figure 2a and 2b). Kidneys from CSE-treated mice were also comparable with those of untreated ones (Figure 2a and 2b) and were devoid of any nephrotoxic signs like necrosis, diffused and tenuous Bowman’s space.
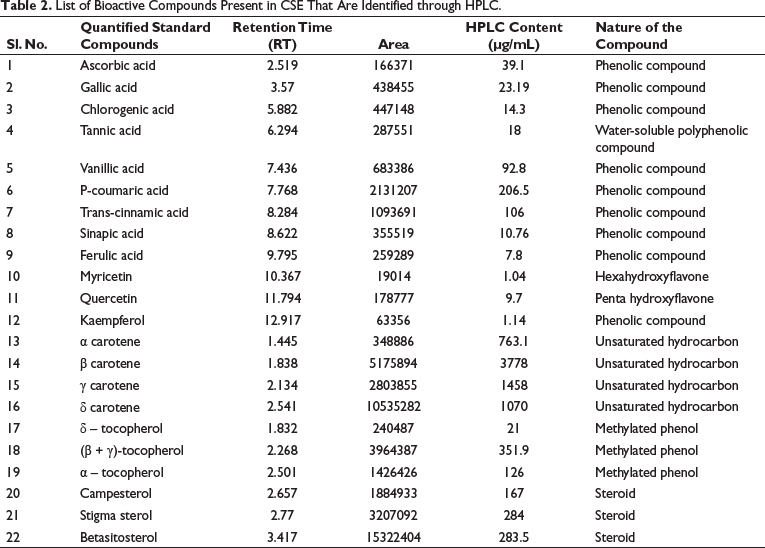
List of Bioactive Compounds Present in CSE That Are Identified through HPLC.
Inhibition of Serum TNF-α in DTH-induced Mice with CSE Treatment
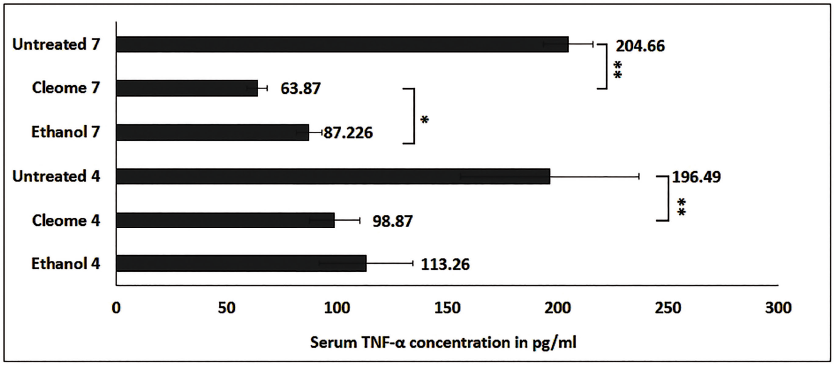
One of the key molecules mediating inflammatory processes is TNF-α, belonging to the family of pro-inflammatory cytokines that contributes directly and indirectly to the occurrence of oedema. Thus, to look at the production level of TNF-α in DTH mice under the influence of CSE (7.3 µg/g b.w.), we performed ELISA. The level of serum TNF-α was found to be significantly lower with CSE treatment compared to the control groups on the fourth and seventh days of resensitization. The TNF-α concentrations were 98.87 pg/mL and 63.87 pg/mL with CSE treatment on the fourth and seventh days of resensitization, whereas in control groups the TNF-α concentration was 113.26 pg/mL and 87.226 pg/mL with ethanol treatment and in untreated mice, it was 196.49 pg/mL and 204.66 pg/ml on fourth and seventh days of resensitization respectively (Figure 3). The results suggest inhibition of TNF-α production with CSE treatment in DTH mice which corroborated well with the rate of inhibition of paw oedema on the respective days (Figure 1b and 1c and Supplementary Table 1), further confirming its anti-inflammatory and immunomodulatory properties.
Inhibition of Serum TNF-α Level in pg/mL with CSE Treatment and Its Respective Control Groups (Ethanol and Untreated) on 4th and 7th Days of Resensitization. CSE treatment showed significant inhibition of serum TNF-α production over the control groups. Results are expressed as mean ± SD, ** p < 0.01 and *p < 0.05.
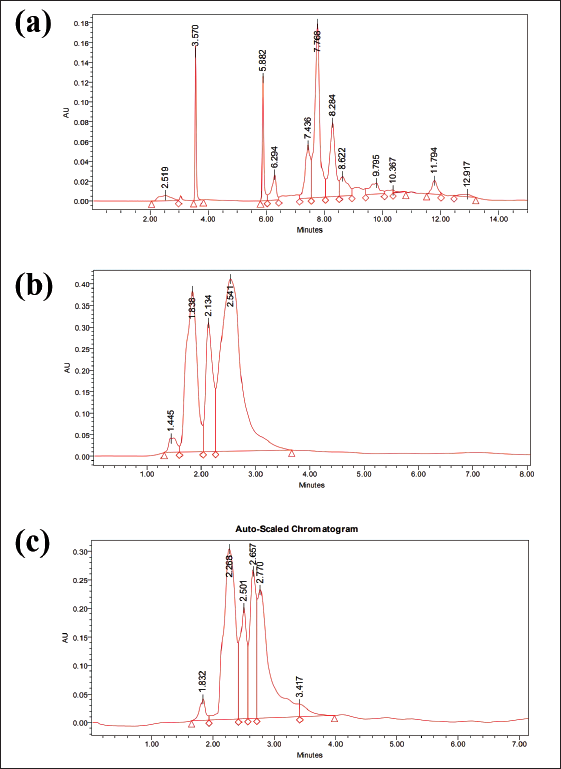
Determination of Bioactive Compounds by RP-HPLC From CSE
RP-HPLC analysis detected different bioactive compounds present in the ethanolic leaf extract of CSE (Figure 4 and Table 2). The chromatogram of the different bioactive compounds is shown in Figure 4. CSE extract contained several phenolic compounds, flavonoids, unsaturated hydrocarbons as well as steroids. Among the 10 phenolic compounds identified p-coumaric acid, trans-cinnamic acid, vanillic acid, ascorbic acid and gallic acid were in high concentration. Four methylated phenols, α, β, γ, and δ-tocopherol which are natural forms of vitamin E were also present. The extracts contained four unsaturated hydrocarbons: α-carotene, β-carotene, γ-carotene, δ-carotene. HPLC analysis suggested that P-coumaric acid, α-carotene, β-carotene, γ-carotene, δ-carotene, (β+ γ)-tocopherol were present in the extract in very high amounts. CSE also contained three phytosterols, stigmasterol, β-sitosterol and campesterol in good amounts.
RP-HPLC chromatogram of CSE.
Discussion
This study documents for the first time that the ethanolic leaf extract of CSE acts as an inhibitor of DTH reaction induced in mouse paw by DNFB and also healed the wound faster compared to control when applied intravenously. Histological observations of skin biopsies from resensitized paw with HE and MT staining were consistent with the efficacy of CSE to reduce DTH-induced mouse paw oedema (Figure 1b and 1c) as the plant extract could restrict dermal and sub-dermal sinuses in the inflamed tissues and regaining the normal tissue architecture during healing (Figure 1b – HE and MT). CSE also seems to inhibit collagen deposition at the DTH site compared to the control. CSE at a dose of 7.3 µg/g b.w. in mice did not have any inclement effects on the spleen, liver and kidney and possibly not leading to any pathophysiological consequences and the haematological parameters also remained similar with the untreated control group after 7th and 14th day of treatment. Thus, 7.3 µg/g b.w. dose of CSE was found non-toxic to the mice.
To understand the mechanism behind the DTH inhibition by CSE, we carried out TNF-α estimation in the serum of DTH mice. TNF-α is a strong pro-inflammatory Th-1 cytokine which participates in vasodilation, oedema formation, leukocyte adhesion and contributes to oxidative stress in sites of inflammation 20 . TNF-α acts to increase vascular permeability and blood flow resulting in the erythema and induration at the site of inflammation as well as increasing expression of various adhesion molecules. 21 . TNF-α contributes to oedema by disrupting the structure of endothelium of the vessel structure and hence, augments permeability.20, 21 CSE treatment showed significant inhibition of TNF-α production and this possibly contributed to the alleviation of mouse paw oedema. On the 7th day when the peak of induration was least with CSE treatment compared to control, the TNF-α level was also inhibited maximum by CSE. Both on the fourth and seventh days of resensitization the TNF-α level was in agreement with the reduction in paw oedema in almost all the groups. This suggests possible involvement of TNF-α in DTH inhibition with CSE treatment. Thus, the present investigation suggests CSE could significantly inhibit TNF-α production in DTH mice and by inhibiting a pro-inflammatory cytokine, possibly contribute towards wound healing of the mouse paw oedema. CSE also showed the capability to scavenge free radical generation and thus behaved as an antioxidant.12, 22 Knowing that ROS generation leads to tissue damage 23 , this free radical scavenging property of CSE could also possibly play some role in faster wound healing.
RP-HPLC analysis of CSE revealed that the leaf extract constituted mainly of phenols, unsaturated hydrocarbons, phytosteroids, and flavones. Most of the phenols found in CSE such as p-coumaric acid, trans-cinnamic acid, sinapic acid, vanillic acid, ascorbic acid, gallic acid and chlorogenic acid have antioxidant properties 24 . Derivatives of cinnamic acid such as p-coumaric acid, trans-cinnamic acid and sinapic acid which were present in high content in CSE are well-known antioxidants and have several health benefits due to their strong free radical scavenging properties as well as anti-bacterial, anti-fungal and anti-inflammatory properties 25 . It has also been found that p-coumaric acid can decrease the expression of TNF-α in adjuvant-induced arthritic rats 26 . This possibly could be one of the reasons for the inhibition of TNF-α with CSE treatment in our DTH model. Four methylated phenols, α, β, γ, and δ-tocopherol which are natural forms of vitamin E are present in CSE, and are also known for their strong anti-oxidant and anti-inflammatory properties. 27 Carotenes which are polyunsaturated hydrocarbons were found abundantly in CSE. α-carotene, β-carotene, γ-carotene and δ-carotene were in very high concentrations as examined through HPLC. Among them, β-carotene exhibits several protective roles which include provitamin-A activity, antioxidant properties, immunomodulation, and enzymatic activation of procarcinogens. 28 Phytosterols are biologically active plant-derived steroids that are molecules with multiple health applications such as anti-inflammatory, anti-oxidant, chemopreventive, anti-diabetic and anti-atherosclerotic. 29 Stigmasterol found in CSE is one of the most common phytosterols. It is an unsaturated phytosterol belonging to the class of tetracyclic triterpenes. In vitro and in vivo assays and molecular docking suggested stigmasterol as an anti-fungal, anti-bacterial, antioxidant, anti-inflammatory and immunomodulatory compound. 30 Thus, it is clearly established that the plant CSE contains several bioactive compounds which exhibit anti-oxidant and anti-inflammatory activities and probably that is the reason it could efficiently exert an inhibitory effect against hapten-induced deleterious DTH reaction by reducing the paw inflammation in this study. These preliminary studies suggest the potential therapeutic use of CSE to treat DTH-driven pathological disorders. Further research to explore the underlying molecular mechanism in inhibiting DTH response by ethanolic leaf extract of CSE may shed more light on its immunotherapeutic applications.
Conclusion
The ethanolic leaf extract of CSE can act as a potent anti-inflammatory agent as it can efficiently inhibit hapten-induced deleterious DTH reaction in murine model possibly through downregulation of TNF-α production.
Abbreviations
Footnotes
Acknowledgments
The authors dedicate this paper to their mentor Late Prof. Ashim K Chakravarty. The authors acknowledge Dr. Ramdhan Majhi, IICB, Kolkata for his support in RP-HPLC analysis.
Author Contribution
TR: Investigation, writing-original draft, writing – review & editing, and formal analysis. TM: Conceptualization, methodology, investigation, writing – original draft, writing – review & editing, and formal analysis. PS and TR: Investigation, writing – review & editing, and formal analysis. TCN: Conceptualization, methodology, resources, writing – review & editing, and validation. SB and SCD: Conceptualization, methodology, writing – review & editing, and validation. HY: Conceptualization, investigation, writing – original draft, writing – review & editing, methodology, project administration, resources, funding acquisition, and supervision. All authors read and approved the submitted version.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
The experimental protocols were approved by the Institutional Animal Ethics Committee (CBPBU/IAEC/ZOO/010/2018) as per the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA) guidelines.
Funding
This work was supported by grants from Science & Technology and Biotechnology, Govt. of West Bengal, India (Memo No. 342 (sanc.)-ST/P/S&T/16 G-27/2018).
Informed Consent
All experiments were done in mouse model, no human subjects were included, hence the study doesn’t need consent approval statement.
Supplementary Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
