Abstract
Background
The future of smart factories and pharmaceutical industries has evolved significantly since the 19th century. Computers have been used in the pharmaceutical field since the 1980s with the emergence of Artificial Intelligence (AI). In addition, automation and robotics are used in the pharmaceutical industry to improve the efficiency of pharmaceutical development and production.
Purpose
The present review article covers the future roles of AI, automation and robotics in pharmaceutical industries.
Methodology
The current review article employed a comprehensive search strategy across relevant databases, including PubMed, Scopus and Web of Science, utilizing keywords such as AI, Automation, Robotics and Pharmaceutical Industries. The articles considered a focus on recent advancements and emerging trends in the intersection of AI, automation and robotics within pharmaceutical sectors.
Results
AI, incorporating predictive machine learning and reasoning techniques, aids in the preclinical identification of molecules and forecasting potential lead compounds before conducting clinical trials. automation offers significant benefits in monitoring and predictive maintenance of production lines, power distribution and control machines. Robotic process automation improves efficiency by connecting computer terminals to handle various manufacturing process elements.
Conclusion
Artificial intelligence, automation and Robotics have sparked innovations in the healthcare business, benefiting the global ecosystem and healthcare delivery. Incorporating these advanced tools in pharmaceutical industries from raw material selection to final product development could improve the quality and safety of the pharmaceutical products and reduce the time and cost.
Introduction
The pharmaceutical and biotechnological industries have evolved significantly since the 19th century, with the Middle Age’s apothecaries and pharmacies being the origins. The modern form emerged in the latter half of the 19th century, with a combination of rationalism and experimentation during the 17th century and the Industrial Revolution in the late 18th century. Merck, founded in 1668, transformed from a pharmacy into an industrial enterprise in 1827. Beecham entered medicine production in the mid-19th century, focusing on patented drugs in 1842 and opening the first dedicated factory in 1859. 1
Computers have been used in the pharmaceutical field since the 1980s for data collection, clinical research, drug design, education, and clinical pharmacy. 2 Back in 1995, a pioneering AI program was developed by Newell and Simon, and during this time, John McCarthy, widely acknowledged as the forefather of artificial intelligence (AI), coined the term “artificial intelligence.” AI encompasses the creation of computer systems or software capable of executing tasks traditionally reliant on human intelligence. These systems assimilate knowledge from data, adjust to novel inputs, and demonstrate behaviors analogous to human cognitive processes. AI finds widespread utility across diverse scientific domains, including but not limited to healthcare, finance, autonomous vehicles, recommendation systems, virtual assistance, and climate modeling. 3 Machine intelligence or AI is often confused with robotics and automation. AI focuses on exhibiting human-like behavior or intelligence by computers or machines. Robots were not designed to possess these intelligent capabilities, but they can carry out difficult repetitive tasks and move or carry objects independently using a designed program and surface sensors in automation. 4
In relation to AI, automation is also used widely in the pharmaceutical industry. Automation denotes the utilization of technology to execute tasks or procedures with minimal human involvement, with the overarching goal of optimizing operations, enhancing efficiency, and diminishing manual labor. The synergy between automation and AI further amplifies their effectiveness. 5 Automation is the practice of using machines or equipment that dates back to the 11th century. The arrival of modern automation during the 1800s brought about a significant transformation known as the Industrial Revolution. This era witnessed the utilization of mechanized methods and advanced tools to improve the productivity of factories. Moreover, the introduction of electricity in the 1920s served as a catalyst, further expediting manufacturing processes, and feedback controllers in the 1930s and 1940s further advanced automation. By the 1980s, various sectors, including manufacturing, retail, pharmaceuticals, and consumer goods, had adopted automation technology to enhance their productivity. 6
Regulatory issues arose due to worries regarding the management of systems, automation of production, and generation of data. To address these uncertainties, a collaboration was formed between end users, vendors of automation, and consultants to establish and standardize protocols. The Parenteral Drug Association and Good Automated Manufacturing Practice (GAMP) forum responded to this by publishing technical reports in 1994 and 1995. Furthermore, the GAMP forum released “The GAMP Guide for Validation of Automated Systems in Pharmaceutical Manufacture” to tackle these concerns. 7
The pharmaceutical industry experienced significant profitability and political power in the late 1980s and 1990s. In these industries, labor-saving devices were needed for drug discovery. Projects with 100,000 or more compounds were considered small, and testing these compounds within a reasonable timeframe was necessary. Miniaturizing assays and automating them was crucial as technology was already in use in medical diagnostics. 8 Hence, robots first made their appearance in solutions provided by original equipment manufacturers, such as cartooning and filling machines, immediately capturing the interest of drug manufacturers. They later became crucial in intralogistics and auxiliary tasks. Today, the technology is mature enough for mass adoption of drug manufacturing, and the degree of automation is expected to increase at record rates in the coming years. 9
A thorough literature survey was carried out for this study using AI, automation, robotics, and pharmaceuticals as keywords in scientific databases such as Google Scholar, PubMed, Scopus, and Web of Science. The present review article covers the future roles of AI, automation, and robotics in the pharmaceutical industry. It includes the history and role of these advanced technologies, the evaluation of labs, their advantages and disadvantages, and the future scope of the systems in the pharmaceutical field.
Advancements in Future Labs and Their Types
Due to the advancements and technological developments, pharmaceutical laboratories are experiencing transformation through digital and automation technologies, evolving over three horizons. While most have not fully achieved full transformation, they aim for one of these futuristic technological evolutions. Future labs can be classified as digitally enabled labs, automated labs, and distributed quality control labs. 10
Digitally Enabled Labs
The horizon of digitalization involves transforming manual data transcription to automatic data transcription between the equipment and the laboratory information management system (LIMS). This improves visibility, reducing risk and enabling targeted investments to improve input quality, redundant testing, and material release. These labs utilize real-time data analytics for process verification, scheduling optimization, and capacity management. Digital tools such as smart glasses and digital twins can predict impacts before physical changes occur. Digitalization can reduce costs by 25%–55% for chemical quality control labs and 15%–35% for microbiology labs. Productivity improvements result from eliminating manual documentation work and optimizing planning and scheduling. 11
Automated Labs
Pharma companies are utilizing robots and cutting-edge technologies like automation to execute repetitive tasks such as sample preparation and delivery. These labs can deliver greater value and save costs by 10%–20% in microbiology labs and 10%–20% in chemical labs. By automation, up to 80% of sample-taking and delivery tasks and 50% of sample preparation tasks can be automated. Through remote monitoring, we can reduce equipment maintenance costs and prevent failures. It can also reduce sampling and logistics tasks, saving up to 25% of lab costs for microbiology labs and 8% for chemical labs. Remote monitoring and predictive maintenance capabilities can decrease downtime and reduce the use of expensive devices. Shifting to instantaneous microbial detection can reduce lab lead time by 40%–75%. 12
Technologies from healthcare, research, and manufacturing can be implemented in pharma quality control labs, offering solutions such as sample-distribution systems, online and instantaneous microbial-testing systems, automated sample preparation stations, workflow optimization, and remote equipment monitoring. Next-generation analytical testing technology, such as ultra-performance liquid chromatography (UPLC) and Raman spectroscopy, offers industrialized designs with additional automation features. In 2018, additional technologies such as remote equipment monitoring and improved methods for testing sterility and analyzing data were developed to enhance the speed and efficiency of laboratory investigations. 13
Distributed Quality Control
The third horizon represents a significant shift from traditional methods of ensuring quality, as most regular product testing takes place directly on the production line, allowing for real-time release testing (RTRT). The integration of process analytical technology and RTRT in the pharmaceutical manufacturing sector has been slow due to complex regulations. To smoothly transition to online testing, operational teams need to collaborate with research and development during the early stages of development to establish an optimal approach to quality control and regulatory submissions, particularly for new products and manufacturing facilities. Quality control responsibilities are spread out, with equipment and robots equipped with integrated AI, while specialized and stability testing still takes place in laboratory settings. This testing can be conducted on-site or remotely, even within a centralized and highly automated facility. 14
Distributed quality control facilities reduce the need for a physical space and lower expenses connected with traditional laboratories, thereby speeding up product release. However, existing facilities that have steady or declining production levels may not find enough reasons to embrace distributed quality control in the short to medium term. On the other hand, rapidly growing facilities or those currently being built can benefit significantly by reducing the costs of constructing or expanding traditional quality control labs. By implementing distributed quality control, implementing real-time release practices, and utilizing certificates of analysis provided by suppliers, genuine continuous manufacturing processes can be achieved. 15
Pharma companies can pull in relevant technologies from adjacent spaces, such as advanced process control platforms and AI systems, to automate tasks previously performed by highly trained, expert employees. The outbreak of the COVID-19 pandemic brought new restrictions, such as maintaining social distance in crowded laboratory environments, limitations on the number of on-site analysts, and the need to keep records of close personal interactions for contact tracing purposes. By adopting a smart approach to quality controls, it is possible to devise effective strategies for conducting tests with reduced physical presence of analysts in laboratories, thus allowing a significant portion of the work to be carried out remotely. 16 Table 1 describes the different quality control labs.
Role of Artificial Intelligence
Use of AI in Pharmacovigilance
Drug safety is the primary risk involved in new medicine when introduced in the market. During clinical trials and post-marketing surveillance (PMS), the major threats are unpredicted toxicities, which may lead to morbidity and mortality. Key pharmacovigilance (PV) tasks include adverse drug reaction (ADR) detection, reporting, coding adverse events (AE) in technical terms, individual case safety report (ICSR) preparation, seriousness assessment, and interaction with the suspected medication. These are time consuming and depend on human interference; thus, detection and analysis of ADRs require new technology. 20
AI-enabled automation in AE case processing could impact PV cost drivers. AI is needed to help increase ICSR volume and complexity, which can be handled by drug safety professionals with the help of AI. Acquiring new skills and abilities is necessary for understanding and applying AI effectively. 21
Electronic health records (EHRs) play a crucial role in providing essential external data for patients’ health. These records provide detailed accounts of the patient’s clinical history, including indications, the current state of the illness, its intensity, and any possible factors that may affect the accuracy of the diagnosis. AI solutions, such as VigiFlow, VigiBase, VigiAccess, and VigiLyze, can automate various aspects of PV, such as case processing and risk tracking, reducing processing costs and time. Other tools, such as VigiGrade, VigiMatch, and VigiRank, are utilized for case report analysis.22, 23
Use of AI in Drug Design and Clinical Research
AI has a crucial impact on drug design, drug development, decision-making assistance, and clinical data management, enabling customized treatment methods, such as personalized medications for each patient. This enhances the product’s entire life cycle, ensuring a predictable and effective pharmaceutical industry. AI can improve drug development by identifying lead compounds, accelerating target validation, and enhancing structure design. 24
Traditional medicine development takes 10 years and costs 2.8 billion dollars. AI, incorporating predictive machine learning (ML) and reasoning techniques, aids in the preclinical identification of molecules and forecasts potential lead compounds before conducting clinical trials. By utilizing this methodology, the speedy identification of lead compounds with a higher probability of successfully advancing through clinical trials becomes feasible. This technology selects diseased populations for clinical trials, boosting adherence by 25% in schizophrenia patients using mobile software and ensuring successful trial completion. 25
AI Use in Medical Affairs
Medical affairs play a crucial role in exchanging external medical interactions with healthcare professionals and key customers. To embrace digital transformation, medical affairs must learn from other sectors and push for greater regulatory flexibility for digital data exchange. Reducing barriers between commercial teams in the pharmaceutical industry and medical teams is essential. 26
AI in the Marketing of the Pharmaceutical Industry
Market positioning is the process of establishing the distinctiveness of a product to attract customers. AI can assist in predicting market trends by using business-intelligent smart scale prediction analysis, which combines time series forecasting with real-time applications. This methodology helps in forecasting future product sales. 27
Role of Automation
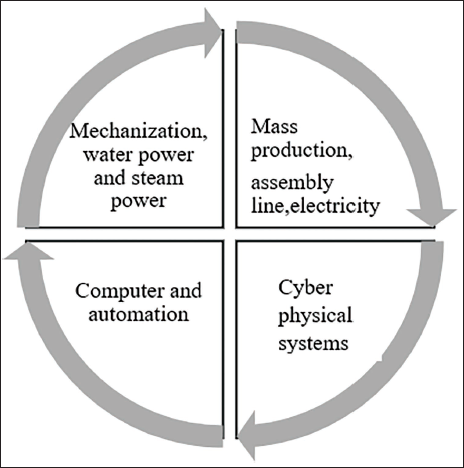
Automation refers to the implementation of machines to carry out monotonous and important duties within the pharmaceutical field. This encompasses both physical and intellectual activities in the manufacturing process, thereby eliminating the necessity for human intervention (Figure 1). The inclination to incorporate cutting-edge technology to replace the human workforce has become prevalent in different sectors worldwide, including the swiftly progressing pharmaceutical industry (Figure 2). 28
Automation Used in Pharmaceutical Industries: Four Stages of Manufacturing.
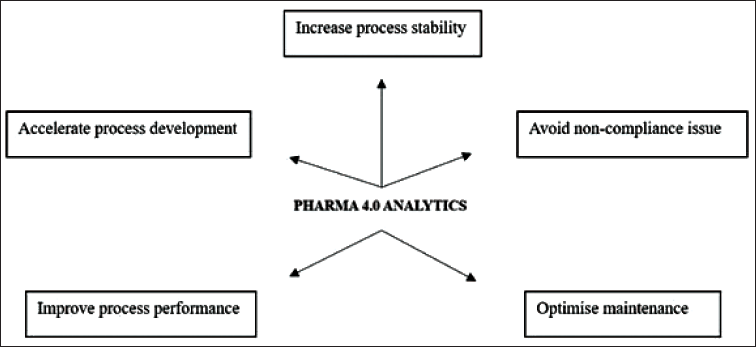
Pharma 4.0 Analytics in Industries.
Filling, Inspection, and Packing
The packing system uses various tools for animation, wrapping, labeling, shrinking, closure, cases, trays, assembly, chilling, drying, feeding, pouring, picking, installation, cleaning, disinfection, and diagnostic testing. During the manufacturing, it must be ensured that the correct amount of medication with the exact composition of drugs and its excipients is weighed and packed precisely into its appropriate containers. Some pharmaceutical packing systems use automation for bottle orientation, capping, labeling, and collation systems. It can be used for various processes such as vial, capsule, and container filling, ensuring consistent product quality and inspection. These automated processes are monitored by a centralized computer, which effectively monitors all critical parameters to ensure product quality and reject specific products that are not appropriately filled. 29
Making Personalized Medicines a Reality
Although there are countless genetic variations among individuals, people are treated with the “one-size-fits-all” approach. Personalized medications have gained recognition in the medical field due to their effectiveness in treating ailments. To make this a reality, automation is necessary. Various modern computational tools are installed to run multiple tests to identify the perfect combination of drugs. Automation advancements can greatly benefit in making personalized medicine a reality. Automated equipment efficiently regulates process parameters to manufacture the required concentration of drugs. 30
Continuous, Uninterrupted Manufacturing
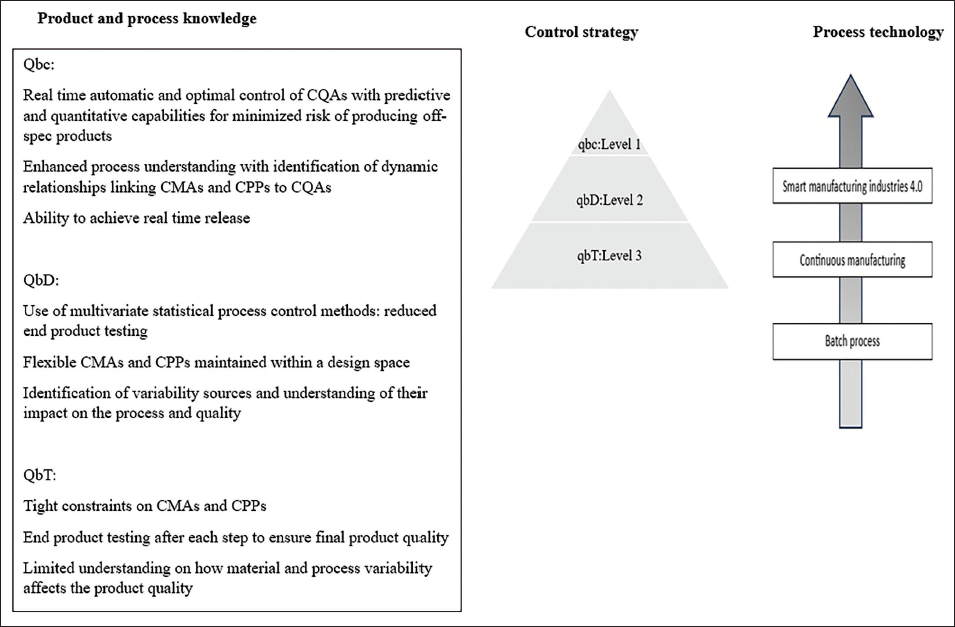
Continuous manufacturing, according to the FDA, can be defined as “an integrated process that consists of a series of two or more-unit operations” where “the input material(s) are continuously fed into and transformed within the process and the processed output material(s) is continuously removed from the system.” Pharmaceutical continuous manufacturing is an emerging technology with a framework in development (ICH Q13) (Figure 3). 31
Control Levels, Related Process Technologies, and Anti-counterfeit Technologies.
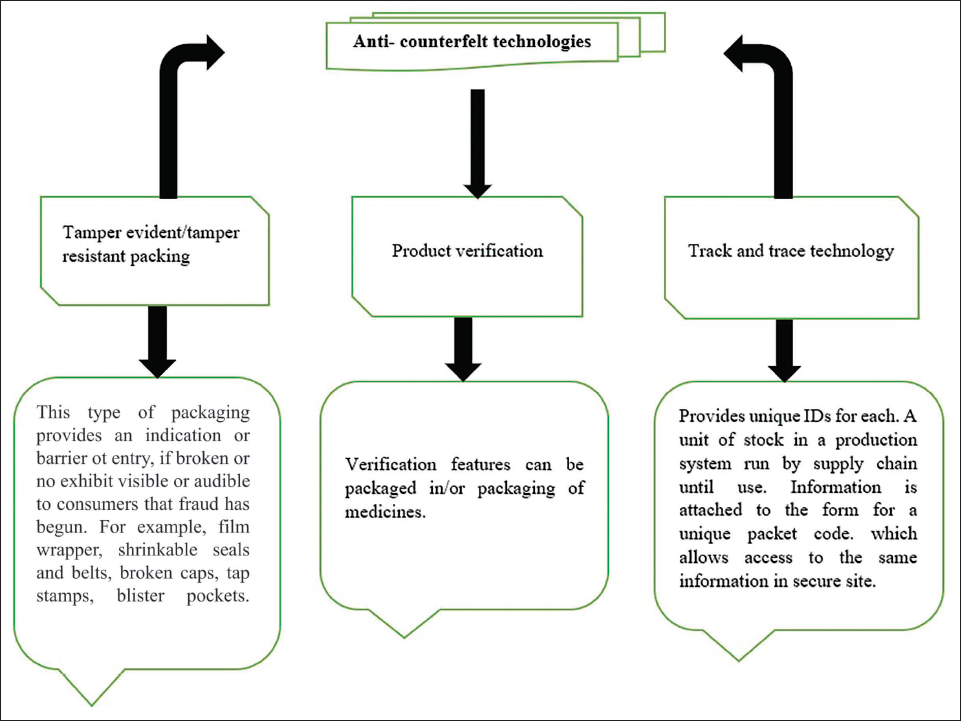
This technology can be used as tamper-evident or tamper-resistant packing, which oversees broken film wrappers, belts, caps, tape stamps, blister pockets, and shrinkable seals. It acts as a barrier to fraud entry. Product verification features can be packaged in packaging materials or in the packaging of medicines. Track and trace technology is essential for packaging medicines and other products. These technologies assign individual IDs to each unit of stock, allowing secure access to data through packet codes (Figure 4). 32
Anti-counterfeit Technologies.
Role of Robotics in Laboratories
The pharmaceutical sector is increasingly using robotics to automate various processes in manufacturing, such as production, packaging quality control, research and development, including drug screening and anti-counterfeiting tasks. Robotics is defined as “a re-programmable multifunctional manipulator designed to move material parts, tools, or specialized devices through various programmed motions for the performance of a variety of tasks.” 33
Production
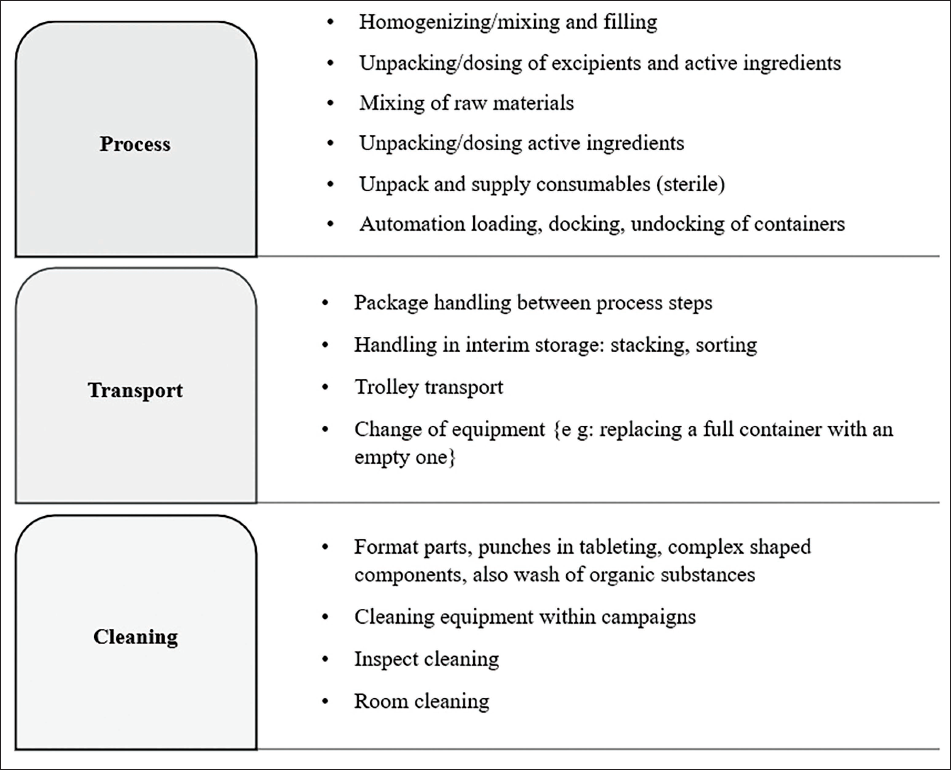
The main physical activities for the people working in production are changeover, cleaning, and preparation of materials. These tasks necessitate an elevated degree of flexibility, documentation, etc.; thus, robotics is employed in these sectors to increase productivity and reduce errors. International Society for Pharmaceutical Engineering’s (ISPE) Special Interest Group (SIG) Robots are used in pharmaceutical industries for automating specific processes in production, such as the production core process, transportation, and cleaning (Figure 5). There are extra responsibilities in the manufacturing sector that include administering solid substances, sampling for quality control, maintaining the cleanliness of standardized manufacturing machinery, unloading from pallets, and reloading onto pallets. These duties involve tasks such as selecting orders, transitioning from one kind of pallet to another (for example, switching from wooden to aluminum), and various other actions. 34
Selection of Needed Applications for Production by SIG Robotics.
Packaging
Classic packaging systems are established for large-volume standard packaging. They require a lot of space for their operation, which is carried out in different parts of the line and requires high costs in construction and system integration. Thus, robotics is used to replace monotonous and ergonomically questionable tasks such as packaging. These are installed for the use of palletization at packaging lines and simple pick-and-place applications such as from belt to box. Further, it is used in complex packaging, such as combining various components in one package and labeling the carton finally. SIG robots play a vital role in packaging. 35
Quality Control/Research and Development Laboratories
Quality control (QC) responsibilities generally consist of regular tasks, and human intervention remains essential for material flow and sample preparation. 36 Numerous analytical instruments can be mechanized, simplifying time-intensive processes. This advancement has significantly lessened the burden on the QC division. In situations where production is extensive and the analytical department confronts timing obstacles, automated systems can offer support. Traditional laboratory automation is suitable for high throughput and low sample and process variance, and it is suitable for applications such as sampling from containers, microbiological laboratory preparation, and sample separation and dosing for various analysis needs. 37
Robotics in the Laboratories
With the help of robotic arms, we can carry out processes such as nuclear magnetic resonance and high-performance liquid chromatography (HPLC). Undoubtedly, automation can significantly enhance the study of protein structures by utilizing both NMR and X-ray crystallography methods. A cutting-edge micropipette machine, operating automatically, holds immense potential to produce a vast assortment of crystals. This enables the concurrent examination of numerous structures using X-ray crystallography. Likewise, an automated HPLC system offers the advantage of streamlining the collection and analysis of samples, instantly transmitting the results to a central computer. The utilization of advanced industrial robots further amplifies efficiency, but we should ensure uninterrupted operation, consistent power supply, and regular maintenance. Consequently, these factors contribute to increased productivity and precision in protein analysis processes. 38
Other Pharmaceutical Applications
Automation, robotics, and AI offer a wide array of advantages in pharmaceutical industries, encompassing specific uses in processing systems, such as powder blending, fluid bed granulation, high sheer/wet granulation, hot melt extrusion, liquid dosing (used in tablet manufacturing), tablet compression and coating, encapsulation (solid and liquid dosages), drying, milling, micronization (dry granulation), radio frequency identification (RFID), labeling (product movement monitoring and quality management), filling, inspection and packing, drug delivery systems, conveyor and sortation systems (high-speed bottle filling, inspection, sorting and grouping, rejection, and transferring), tracking, and traceability counterfeit prevention. 39 These advancements often necessitate a substantial initial investment but bring forth benefits such as adaptability for batch production, reprogramming options for different product styles, and the capacity to modify operational sequences. Despite these merits, there are obstacles to overcome, such as the requirement for reprogramming and setup alterations between batches, potentially lower production rates in comparison to fixed automation, and the need for skilled personnel. Nevertheless, the implementation of these technologies can result in efficient utilization of equipment and manpower, heightened effectiveness, improved customer experiences, and bolstered cybersecurity measures. Installing these new technologies can result in high investment in programmable equipment, lower production rates than fixed automation, flexibility to deal with variations and changes in product configuration, most suitability for batch production, change of physical setup and past programs between batches, efficient use of equipment and manpower, increased effectiveness, lower cost and high return on investment, a scam-less customer experience, and enhanced cyber security. 40
Advantages of AI, Automation, and Robotics
Reduce Cost
Automation offers a significant cost reduction in manufacturing by reducing the need for multiple supervisors to monitor an operation, which can be replaced with robots. Although the initial investment may be high, it has long-term benefits. We should only monitor maintenance, repairs, and energy expenses. AI and data analytics have a crucial impact on reducing production expenses by providing valuable insights and information that steer informed decision-making throughout the manufacturing process. 14
Increase Productivity
Robotic process automation improves efficiency by connecting computer terminals to handle various elements of the manufacturing process. These automated systems can be customized to handle raw materials, clean the equipment, and operate high-pressure systems. These technologies can emulate a diverse array of human behaviors, efficiently streamlining operations and enhancing effectiveness within manufacturing settings. 41
Enhance Quality
Industrial automation improves output quality and reduces error rates from 1% to 1.15% in manual processes. Automated devices in the manufacturing sector demonstrate an incredibly minimal mistake percentage, frequently plunging to as low as 0.00001%. The implementation of adaptive control and monitoring technologies additionally diminishes the scope of inaccuracies within manufacturing procedures. This methodology ensures a remarkable degree of uniformity in the excellence of outcomes throughout production operations. 42
Industrial Safety
Automation significantly improves workplace safety by reducing accidents and ensuring workers do not get too close to the assembly line. Robots are used for material loading and unloading and transferring huge machine parts. This has reduced the risk of accidents. while thermal sensors monitor production temperature, alerting workers to any spikes and implementing immediate precautions to ensure everyone’s safety. 43
Accurate Results
Data automation relies on accurate data integration and connectivity, ensuring precise results in production processes. AI and ML solutions provide detailed data analysis, while deep learning algorithms are utilized for self-healing digital grids and intelligent energy forecasting. Machine learning apps are also utilized for quality control systems in assembly lines. These scalable and self-learning solutions guarantee accurate results without fail, ensuring the success of automated systems. 44
Better Working Conditions and Value Addition
Industrial automation ensures consistent production and results by connecting computers, robots, and automated machines at a steady pace. This allows for better control of production rates and quality. A flexible manufacturing system connects tools, processing machines, and material-handling robots, ensuring continuous production without any drop in pace or results. This allows for the customization of machines to suit different product measurements or new products. Traditional production processes can take days or weeks to train employees, and it can be difficult for workers to adapt to new processes, leading to production delays or quality issues. Reprogramming machines or rebooting them is easier and takes less time, and after a few trials, machines are ready for full production. Automation frees employees from tedious tasks, allowing them to focus on value-addition, research, and process development. Workers can also effectively use robotic tools and machines to deliver faster and quicker results, and employees experience a positive work environment. 3
Industrial Communication
Industrial communication is crucial for industrial automation, enabling monitoring, operation, and control of production lines, power distribution, and control machines. Popular protocols include Fieldbus, Profibus, ether cat, and ethernet/IP, enabling faster data analysis and real-time decision-making. 45
Monitoring and Predictive Maintenance
Industrial automation offers significant benefits for monitoring and predictive maintenance. Sensors track various parameters, such as temperature, acoustics, time-frequency, and oil pressure, allowing for continuous monitoring of production lines and floors. If changes are detected, they send alerts to technicians, who can identify the cause. This helps to identify potential equipment problems and prevents downtime by enabling immediate services or repairs. Automation helps prevent major issues from escalating into major problems, ultimately enhancing overall efficiency and productivity. 46
Equipment Monitoring
Using sensors, cameras, and networks helps in monitoring the working conditions of manufacturing units. This system diagnoses issues and problems and performs necessary repairs and services. It is suitable for production plants and manufacturing units. The system enhances safety, reduces operator numbers, and improves machine performance and life span. 47
Disadvantages of AI, Automation, and Robotics
Worker Displacement
Automation has a significant disadvantage in displacing humans, as it can execute tasks faster and with greater accuracy than humans. 48
Needs Large Capital Expenditure
These newer technologies have become a popular industry staple, but manufacturers must consider the potential consequences, such as increased capital expenditure for maintenance and service and increased vulnerability to cyberattacks. Proper infrastructure protection can ensure a company’s durability and resilience in the face of cyber threats. These technologies can be a convenient solution for various problems, but they can be redundant when changes require modification. These changes increase the company’s workload and may cost time and resources. Although the overall benefits of these technologies have been proven, there are still certain tasks that require human intervention. 49
Ethical and Societal Challenges Posed by Emerging Technologies
The rapid progression of technology prompts inquiries into the impartiality, fairness, and societal ramifications of automation. Automation offers advantages such as heightened productivity, reduced waste, and the potential for generating automated or online reports promptly. Consequently, the adoption of automated processes can enhance efficiency while diminishing labor input. However, concerns persist regarding potential job displacement resulting from automation and AI-driven mechanisms. This underscores the responsibility incumbent upon both organizations and governments to facilitate the smooth transition of workers into automated workflows. Moreover, the proliferation of autonomous systems necessitates a critical examination of liability and accountability in instances of accidents or errors. Thus, ensuring that AI systems prioritize safety and ethical considerations is imperative to forestall unintended adverse outcomes and safeguard the welfare of individuals and society.50, 51
Conclusion and Future Advancements
The revolution of AI, automation, and robotics is expected to be even more disruptive than the previous revolutions. The palletizing application is the only known off-the-shelf solution in pharmaceutical production, while all other robotics applications are custom built. AI adoption is crucial for pharmaceutical companies to gain a strategic advantage and compete in the industry. As productivity increases and new jobs are created, it is essential to equip displaced workers with the necessary skills. Technologies such as digitization, autonomous systems, robotics, and computing advancements can transform pharmaceutical manufacturing and logistics platforms. The pharmaceutical supply chain, production processes, distribution, and inventory frameworks could see significant improvements. The smart factory of the future will take on autonomous features, enabling more production flexibility and agility.
However, human intervention is necessary for successful deployment, development, and operation, reducing unemployment concerns. Advances in ML, robotic process automation, and AI have sparked innovations in the healthcare business, benefiting the global ecosystem and healthcare delivery. The long-term value of this new manufacturing paradigm should make the business case for adopting new technologies, such as more control, fewer errors, more responsiveness, and fewer drug shortages. In the future, more accessible and real-time product quality information will be generated, making it more transparent to purchasers, payers, healthcare providers, and patients.
Footnotes
Acknowledgments
The authors would like to thank the JSS Academy of Higher Education & Research, Mysuru, and the JSS College of Pharmacy, Ooty, for supporting their research work by providing research facilities. The authors would like to thank the Department of Science and Technology—Fund for Improvement of Science and Technology Infrastructure (DST-FIST), New Delhi, Govt. of India, for providing funding for the instruments LC-MS/MS (Shimadzu-8030) and Preparative HPLC (Waters-2998).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The Department of Science and Technology—Fund for Improvement of Science and Technology Infrastructure (DST-FIST), New Delhi, Govt. of India, provided the funding for the instruments LC-MS/MS (Shimadzu-8030) and Preparative HPLC (Waters-2998).

