Abstract
Background
Head and Neck Cancers (HNC) account for 5% of all malignant tumours, but 80% of patients experience pain. As per the WHO analgesic ladder, analgesics are a cornerstone for the management of pain in HNC patients.
Purpose
Our study aims to analyse the prescribing pattern of analgesic drugs in patients with HNC pain.
Materials and Methods
A prospective, continuous, observational study was conducted among patients with HNC pain. Demographics, detailed history of pain (type, duration, location and Numerical Rating Scale [NRS] score), details of drugs prescribed and any adverse drug reactions (ADRs) were recorded at baseline visit and at first, second and sixth month follow-up. Descriptive statistics were used to analyse the above parameters. The mean reduction in NRS was analysed by the Z test.
Results
Out of 145 patients, 118 (81.37%) were male and 27 (18.62%) were female. The mean age of patients was 51.89 + 7.07 years. Tramadol (81.37%) followed by morphine (62.75%) were the commonly prescribed analgesics. Amitriptyline (20%) followed by pregabalin (19.31%) were commonly prescribed adjuvant analgesics. A statistically significant reduction in mean NRS was found at each follow-up visit (p < .05). A total of 50 ADRs were observed. Constipation (38%) and drowsiness (24%) were the most frequently reported ADRs, with morphine and tramadol being the most common suspected causal drugs.
Conclusion
Tramadol and morphine were frequently prescribed analgesics in patients with HNC pain in accordance with the WHO analgesic ladder. The majority of patients with HNC pain achieved a significant reduction in NRS pain scores at the end of six months.
Introduction
Pain is the most prevalent and distressing symptom reported by patients with cancer. 1 Head and Neck Cancers (HNCs) account for approximately 5% of all malignant tumours, but up to 80% of HNC patients experience pain. Recent advances in HNC reconstructive techniques have improved results, but unbearable pain leads to poor quality of life. 2
Pain in HNC patients may have multifactorial origin involving direct tumour involvement (ulceration, early metastasis, tumour invasion of bone and distension of pain-sensitive structures in soft tissues), tumour compression or infiltration of nerve root trunks or plexuses and lastly treatment modalities (surgery, chemotherapy and radiotherapy). 2
The gold standard for pain management must begin with pain assessment with patients’ self-report, which includes assessment of location, type, temporal profile and severity of pain. 3 Cancer pain can be classified as nociceptive, neuropathic, or a combination of both. 4 Sequential measurement of pain severity using a validated severity pain assessment scale such as a Visual Analogue Scale (VAS), Numerical Rating Scale (NRS) or Verbal Rating Scale (VRS) and multidimensional tools such as Brief Pain Inventory (BPI), McGill Pain Questionnaire (MPQ) and Edmonton Symptom Assessment Scale (ESAS) indicates the changing intensity of the pain over time and also guides analgesic management. 3
Pharmacotherapy with analgesics is the foundation for cancer pain management worldwide. In 1988, the World Health Organization (WHO) first promoted the analgesic three-step ladder. The analgesic ladder is simple to use and well validated and provides a clinically useful strategy for cancer pain management including HNC. If the pain is mild (1–3 out of 10 on the NRS), an analgesic can be chosen from Step 1, which comprises acetaminophen and nonsteroidal anti-inflammatory drugs (NSAIDs) including acetylsalicylic acid (ASA). If pain is moderate (4–6 out of 10 on the NRS), starting with an analgesic from Step 2, which constitutes weak opioids such as tramadol, oxycodone hydromorphone etc. in combination with acetaminophen and NSAIDs, is recommended. If pain is severe (7–10 out of 10 on the NRS), treatment with strong opioids such as morphine, fentanyl, meperidine, methadone, etc. from Step 3 is recommended. 5
The fourth step has also been recommended, which involves the administration of opioid analgesics through parenteral routes and neuroablative procedures like ganglionic blockade etc. and also non-pharmacological measures such as acupuncture meditation etc. 3
At any step, adjuvant analgesics can be added to optimize pain control. Adjuvant analgesics are used to enhance the analgesic efficacy of opioids, treat concurrent symptoms that exacerbate pain and/or provide independent analgesia for specific types of pain. Neuropathic pain is rarely controlled with opioids alone. Chemotherapy-induced neuropathic pain is of increasing clinical importance and prevalence, particularly due to the increased use of platinum-derived agents, taxanes, vinca alkaloids and proteasome inhibitors for the treatment of solid tumours.6, 7
Though pain in HNC patients is a prevalent and severe problem, up to 80% of all patients with cancer suffer inadequate pain management. Moreover, very few studies have been conducted evaluating the pharmacotherapy of pain and its outcome in HNC patients in our hospital. Hence, this prospective study was conducted to evaluate the prescribing pattern of drugs and to analyse the change in drug therapy during a six-month period in HNC patients suffering from pain.
Materials and Methods
This was an observational, continuous, prospective study conducted at Ahmedabad, Gujarat, India, for a duration of 24 months from November 2020 to November 2022.
Prior permission from the Head of the Department of Palliative Medicine was obtained. The study protocol was approved by the Institutional Ethics Committee (EC permission number-IRC/25/2020). The investigator visited the outpatient department of palliative medicine twice a week. Patients with HNC pain attending the OPD and meeting the inclusion and exclusion criteria were enrolled in the study (Figure 1).
Flow Chart of Study Design.

Patients with HNC pain attending the outpatient department of palliative medicine for the first time, of either gender, between the age of 18 and 65 years and who had given written informed consent to participate in the study were included, while patients with HNC pain suffering from acute/emergency conditions requiring indoor hospitalization were excluded.
At the time of enrolment, baseline data such as demographic details, history of cancer treatment, addiction history, family history, chief presenting complaints with duration, type of pain and its location, and NRS were recorded in a predefined case record form (CRF). Each patient was followed up for the next six months at the end of the first, second and six months. At each follow-up visit, the NRS, any change in treatment and ADRs were recorded. The NRS for pain was assessed using a 10-point scale, where 0 = absent, 1–3 = mild pain, 4–6 = moderate pain and 7–10 = severe pain.
Data Analysis
The sample size was calculated based on the prevalence of HNC pain and the duration of the study. Recorded information of patients was entered in Microsoft Excel worksheet version 2019. Descriptive statistics (mean, standard deviation, median and ratio) were used to describe continuous variables. Frequency statistics (number and percentage) were used to describe categorical variables. Z test was used to determine the statistical significance of NRS scores at baseline and at first, second and sixth month follow-up visits. p < .05 was considered statistically significant. Statistical analysis was done using Microsoft Excel® 2019.
Results
A total of 150 patients with HNC pain attending the palliative medicine outpatient department were included in the study. However, 145 patients completed the study and were analysed, while 5 patients were lost to follow-up and hence excluded from analysis. The mean age of patients was 51.89 ± 7.07 years, and the majority of patients (62; 42.75%) were in the age group of 51–60 years. The male-to-female ratio of the patients was 4.5:1.
Tobacco chewing was the most commonly observed addiction in 61 (42.08%) patients, while a family history of HNC was present in 12 (8.27%) patients. Tongue and buccal mucosal cancers were the most commonly observed cancers in 46 (31.72 %) and 26 (17.93%) patients, respectively (Table 1).
Diagnosis of Head and Neck Cancer Among the Study Population (N = 145).

Out of 145 patients, head and neck TNM cancer staging of 137 (94.82%) patients was available; among the 137 patients, a majority (61; 44.52%) had stage III cancer followed by 47 (34.30%, N =145), 20 (14.59%) and 9 (6.56%) patients with stage II, stage IVA and stage IV B, respectively, at baseline visit. The majority of patients (81.38%) had received more than one type of cancer therapy, while only 18.62% of patients had received a single type. A history of both surgery and radiotherapy was present in 30.44% of patients, while 26.20% of patients had received both radiotherapy and chemotherapy.
A total of 74 (51.03%) patients had mixed types of pain followed by nociceptive (43.44%) and neuropathic (5.51%) pain. The NRS scores and mean NRS at baseline, first, second and six month in patients with HNC pain are shown in Table 2.
Numerical Rating Scale at Baseline, First, Second and Six Month in Patients with Head and Neck Cancer Pain (N = 145).
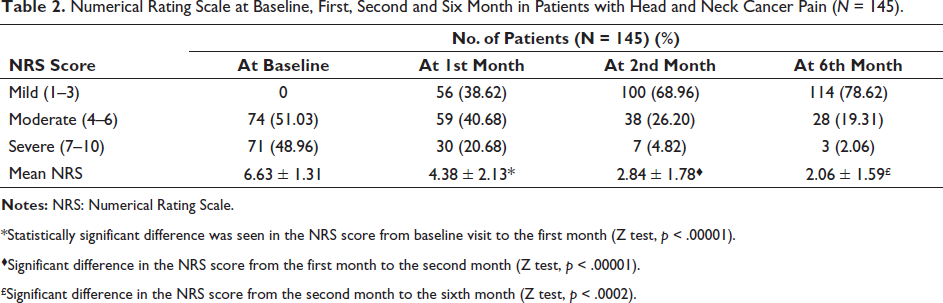
*Statistically significant difference was seen in the NRS score from baseline visit to the first month (Z test, p < .00001).
♦Significant difference in the NRS score from the first month to the second month (Z test, p < .00001).
£Significant difference in the NRS score from the second month to the sixth month (Z test, p < .0002).
Out of 145 patients, 92 (63.44%) experienced breakthrough pain, while the remaining 53 (36.55%) did not experience the same. 95 (65.52%) patients experienced palliative symptoms other than cancer pain such as difficulty in swallowing, loss of appetite, anxiety, depression, tiredness, nausea, etc., while 50 (34.48%) patients did not experience any other palliative symptoms. Difficulty in swallowing was the most commonly observed other palliative symptom in 89 (61.37%) patients. The NRS score (mean ± SD) in patients experiencing palliative symptoms was 6.84 ± 1.43, whereas in patients without palliative symptoms, the NRS score was 6.24 ± 0.96. The mean number of analgesic drugs prescribed per patient was 2.94 ± 0.94 (mean ± SD). The mean number of concurrent drugs prescribed was 3.82 ± 1.06 (mean ± SD)
Opioids as a monotherapy were prescribed in 96 (66.20%) and 84 (57.93%) patients at baseline visit and in the sixth month, respectively. Combination therapy of opioids and NSAIDs was given to 47 (32.41%) and 13 (8.96%) patients at baseline visit and sixth month, respectively. At the sixth month follow-up, 12 patients (8.27%) whose NRS score was 1 and who had clinically significant relief from pain were prescribed neither opioids nor NSAIDs and only paracetamol was prescribed as and when required by the patients.
Paracetamol was prescribed to all patients throughout the six-month period. Tramadol, morphine and fentanyl were prescribed in 74 (51.33%), 56 (38.66%) and 13 (8.67%) patients, respectively, at baseline visit and in 55 (37.93%), 31 (21.37%) and 11 (7.58%) patients, respectively, at six months. Ibuprofen, diclofenac and aceclofenac were prescribed in 19 (12.67%), 19 (12.67%) and 11 (7.33%) patients, respectively, at baseline visit and in 28 (19.31%), 10 (6.89%) and 11 (7.58%) patients, respectively, at six months (Figure 2).
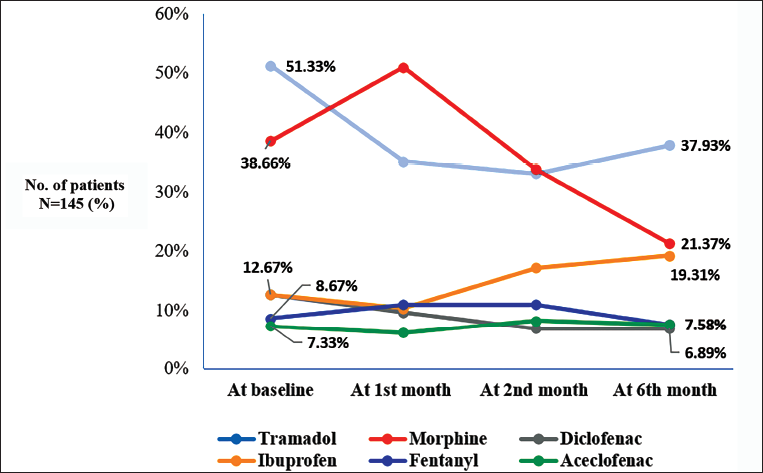
Out of 145 patients, tramadol, morphine and fentanyl were prescribed to 118 (81.37%), 91 (62.75%) and 18 (12.41%) patients, respectively, during the six-month interval. NSAIDs and paracetamol were given to 76 (52.41%) and 145 (100%) patients, respectively, during the six-month interval.
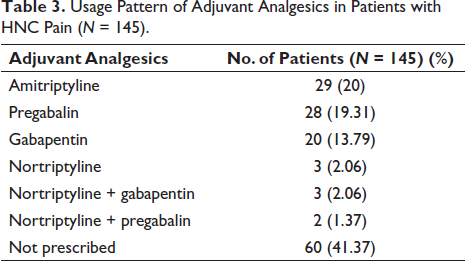
The prescription pattern of adjuvant analgesics in patients with HNC pain is shown in Table 3.
Usage Pattern of Adjuvant Analgesics in Patients with HNC Pain (N = 145).
Lactulose and pantoprazole were the most concurrent medications given to 137 (94.48%) and 103 (71.03%) patients, respectively. All (100%) analgesic drugs were given orally, while 18 (12.41%) patients received analgesics through the topical route and 15 (10%) patients received analgesics (morphine) through the intravenous route during the study period. All concurrent medications were given orally. Out of 91 patients who received morphine, 9 (9.89%) patients were prescribed sustained-release tablets and the rest 82 (90.10%) patients received simple oral tablets. Fentanyl was prescribed as a transdermal patch to all 18 (12.41%) patients. Other analgesic drugs were prescribed as oral tablets. In concurrent medications, lactulose as a syrup to 137 (94.48%) patients, betadine as a gargle to 53 (36.55%) patients and metronidazole as a suspension to 12 (8.28%) patients were prescribed, while other remaining concurrent medications were prescribed as tablets.
The mean duration (mean ± SE) of use of tramadol was 67.35 ± 4.53 days, while that of morphine and fentanyl were 69.51 ± 5.51 and 143.63 ± 11.25 days, respectively. NSAIDs were prescribed with a mean duration of use of 100 ± 6.35 days.
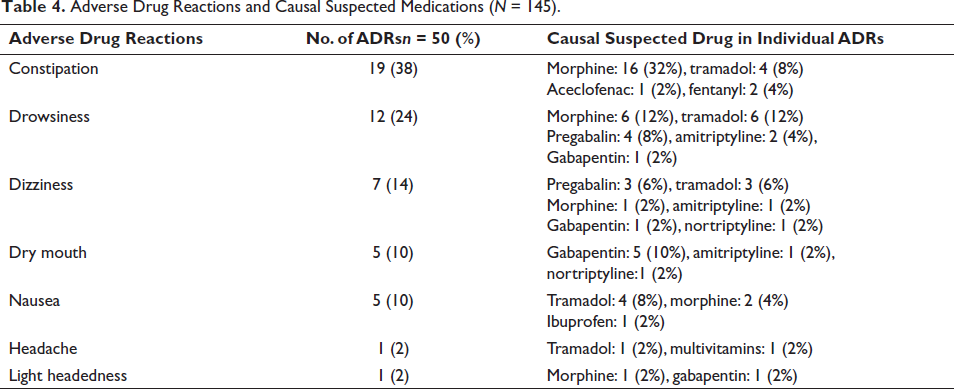
ADRs were observed among 22.06% of patients with HNC pain. The total ADRs in these patients were 50. Constipation (38%) and drowsiness (24%) were the most commonly observed ADRs. Morphine and tramadol were the common causal suspected drugs for ADRs (Table 4). All 50 ADRs had a possible causal association as per the WHO-UMC causality assessment scale. 52% of ADRs were mild in severity and 48% of ADRs were moderate in severity as per the Modified Hartwig’s and Siegel Severity Scale. 48% of ADRs were preventable, while 52% of ADRs were non-preventable as per the Modified Schumock and Thornton’s Scale. Lactulose was prescribed for the treatment of constipation. All patients were recovered from ADRs.
Adverse Drug Reactions and Causal Suspected Medications (N = 145).
Out of 145 patients, 3 (2.06%) patients had increased NRS scores at the end of 6 months as compared to baseline visits. 14 (9.65%) patients had increased NRS scores in the first month as compared to the baseline, while 5 (3.44%) patients had increased NRS scores in the second month as compared to the baseline.
Discussion
The present study focused on the prescribing pattern of drugs in patients with HNC pain. The mean age of patients was 51.89 ± 7.07 years, in contrast to the Chaplin et al. 8 study and Schaller et al. 9 study, where the mean age of patients was 61.3 and 65 years, respectively. Discrepancies in the mean age in the above-mentioned studies might be due to differences in the sample size between these studies and also due to variable prevalence of HNC respective to different study sites.
There was male preponderance in the present study, which is similar to Grond et al.’s 10 study. The probable reason for the male preponderance in HNC pain is that tobacco consumption, which is the greatest risk factor for oral cavity and oropharyngeal cancers, is more in males as compared to females.
A statistically significant difference was observed in the NRS scores from baseline to the first-month follow-up visit (p = .00001), from the first- to the second-month follow-up visit (p = .00001) and from the second- to the sixth-month follow-up visit (p = .0002). In the present study, 63.44% patients experienced breakthrough pain, which is similar to that reported in Caraceni et al.’s 11 study, where 64.8% patients experienced breakthrough pain.
Difficulty in swallowing was the most frequently observed symptom in 61.37% patients, which is similar to a study done by Grond et al. (61%). The mean NRS score in patients experiencing palliative symptoms was 6.84 ± 1.43, which suggests that the severity of pain was more in patients experiencing palliative symptoms, which is similar to the study by Grond et al. 10
In the present study, opioid prescriptions were decreased by 8.27% during the six-month follow-up period. Moreover, there was an increase of 23.45% in the consumption of NSAIDs during the six months. The combination therapy of NSAIDs and opioids was decreased by 23.45% during the six months. This change in prescription pattern indicates that patients with moderate and severe pain intensity were shifted towards mild pain as 78.62% patients had mild NRS (score of 1–3 out of 10) in the sixth month.
In the present study, tramadol, morphine and fentanyl were prescribed to 81.37%, 62.75% and 12.41% patients with HNC pain, respectively, for a total of six months. In Thota et al.’s 12 studies also, a large number of patients (42.5%) received tramadol during the study period.
In the present study, amitriptyline and pregabalin were the commonly prescribed adjuvant analgesics in 20% and 19.31% patients, respectively. However, in Menezes et al.’s 13 study, pregabalin was prescribed in 17.92% patients and amitriptyline in 10.35% patients.
In the present study, a statistically significant difference was observed in mean NRS scores at baseline and in the sixth month in patients started on morphine, tramadol and fentanyl with a p value of <.05 in all three groups. Morphine had a maximum reduction in NRS (5.86 + 0.81) during the six months followed by fentanyl (5.53 + 1.48) and tramadol (3.48 + 0.94). Similar results were shown by Nosek et al.’s 14 studies, where morphine, fentanyl, buprenorphine and oxycodone caused a statistically significant difference in the reduction of the mean value of NRS from baseline to 28 days in patients with cancer pain.
Increased pain intensity was observed with patients at stage IV as compared to stages II and III. Tramadol was the most prescribed in stages II and III and morphine in stage IV. 2.06% patients were treatment refractive at the end of six months. So, adequate pain relief was obtained in 97.94% patients. In 95.86% patients, analgesics were prescribed as per the WHO analgesic step ladder.
ADRs were observed in 22.06% patients in our study. A total 50 ADRs were observed in these patients. Constipation (38%) and drowsiness (24%) were the most commonly observed ADRs. However, Nosek et al.’s 14 studies reported nausea and drowsiness as frequent ADR. Morphine and tramadol were the most common causal suspected drugs for constipation in our study. In Tokoro et al.’s 15 study, 50% patients with cancer pain developed opioid-induced constipation (OIC) within two weeks of initiating strong opioid therapy. The same study also showed that the use of prophylactic treatment was associated with a reduction in the incidence of OIC (cumulative incidence of OIC: 48% among patients using prophylactic laxatives; 66% among patients not using prophylactic laxatives). Nonetheless, in this study, a substantial proportion of patients receiving opioid analgesics experienced constipation despite prophylactic therapy.
The present study suggests that a fentanyl patch causes fewer ADRs and so is better tolerated as compared to morphine and tramadol. A systematic review and meta-analysis comparing efficacy and adverse effects of morphine and fentanyl also showed that morphine and fentanyl have similar efficacy but the latter has fewer adverse effects as compared to the former. 16
All 50 ADRs had possible causal association as per WHO-UMC causality assessment scale 16 while 44 (86.27%) ADRs had possible causal association as per Naranjo’s Algorithm. 17 Total 6 (13.73%) ADRs had probable causal association. 52% ADRs were mild in severity while 48% were moderate in severity as per Modified Hartwig and Siegel Severity Scale. 18 48% ADRs were definitely preventable whereas 52% ADRs were non preventable in nature as per Modified Schumock and Thornton’s Scale. 19 Regarding the outcome of ADRs, all HNC pain patients were recovered from ADRs.
Accurate dose estimation could not be done in the present study as the data of breakthrough pain episodes with their frequency was not obtained at follow-up visits. Moreover, we could not measure serum levels of prescribed analgesics as therapeutic drug monitoring was not available in our setup. There is insufficient evidence from the literature regarding the management of pain in HNC patients despite the high prevalence of pain in these patients. Also, we have not evaluated long-term pain management and its effect on the quality of life of patients with HNC pain. However, we have assessed the prescribing pattern of drugs at various follow-up visits up to six months and also in different types of HNCs and different stages of cancer.
Conclusion
The present study concluded that tramadol and morphine were the most commonly prescribed analgesic drugs for patients with HNC pain following the WHO analgesic ladder pattern. This study highlights the need for long-term prescribing pattern studies to evaluate the long-term efficacy and safety of analgesic drugs in patients suffering from HNC pain. Moreover, guidelines for early assessment and change in analgesics are required for the treatment of refractory patients suffering from HNC pain.
Abbreviations
Footnotes
Acknowledgments
The authors would like to extend regard to the Department of Palliative Medicine, Gujarat Cancer Research Institute, for supporting the research.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study protocol was approved by the Institutional Ethics Committee (EC permission number-IRC/25/2020).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
The participant has consented to the submission of the article to the journal.
