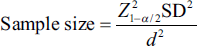Abstract
Background
Palliative care is the care of patients who have progressive, life-threatening illnesses and who are facing death in the foreseeable future. Since there is a growing need for pharmacist’s intervention in palliative care and there is a gap in the education of palliative care for pharmacy students, this study aims to identify roles and services that have to be performed by palliative care pharmacists in medication-related areas and also to formulate an add-on course to train pharmacy graduates.
Materials and Methods
A prospective interventional study was done in the palliative care centre in Perinthalmanna, Kerala, India. Relevant data were pooled and analysed for drug-related problems. Necessary interventions were made. Results were interpreted and an add-on course for the training of pharmacy graduates was formulated.
Results
A total of 88 patients were included in the study. The most prevalent conditions in our palliative setting were kidney disease, liver disease, cancer and stroke. Notably, 38 and 10 cases were found to have drug interactions and drug duplications, respectively. Four patients required dosage adjustments. Fifty cases involved pharmacist intervention which improved the outcome by 80%. At the end of the study, a curriculum was formulated to implement a 3-month add-on course entitled ‘PALLIATIVE PHARMACY’ under the guidance of an Expert committee for training pharmacists and pharmacy students.
Conclusion
The study identified that there are various roles a pharmacist must undertake in a palliative setup. The involvement of pharmacists in patients’ treatment helps to prevent drug-related problems to a greater extent. The addition of an integrated course for the management of palliative care patients will help to improve the learning as well as practical skills of pharmacists. Incorporating basic and clinical sciences in the therapeutic course is an effective learning plan.
Introduction
Palliative care is a healthcare specialty that is both a philosophy of care and an organised, highly structured system for delivering care to persons with life-threatening or debilitating illnesses from diagnosis to death and then into bereavement care for the family. The World Health Organization (WHO) annotates palliative care as:
Palliative care is an approach that improves the quality of life of patients and their families facing the problems associated with life-threatening illness, through the prevention and relief of suffering using early identification and impeccable assessment and treatment of pain and other problems, physical, psychosocial and spiritual.
1
Hospice care is comfort care without curative intent whereas palliative care is comfort care with or without curative intent. Here the goals of both hospice and palliative care tend to be different but the objective of both is pain and symptom relief. In India, the earliest facilities to deliver palliative care were established in the late 1980s and the early 1990s. Since then, hospice and palliative care services have been developed through the efforts of committed individuals including Indian health professionals as well as volunteers in collaboration with international organisations and individuals from other countries. It is estimated that in India the total number of people who need palliative care is likely to be 5.4 million people a year. 1 The coverage of palliative care services is extremely patchy in India, with services being concentrated in large cities and regional cancer centres except Kerala where this study is conducted and services are more widespread here. Also, in India, there is a growing need for palliative care pharmacists and a gap in the education of palliative care for pharmacy students. To address both, pharmacy courses must be developed and disseminate palliative care-focused experiences, including interactive skills and experiences. 2 The significance of palliative care in the undergraduate syllabus of a health professional has been well accepted in the Western world. Hence, this study focuses on developing an add-on course titled ‘Palliative Pharmacy’ to train pharmacy graduates and to identify the role of clinical pharmacists in palliative care. Augmentation of a palliative care pharmacy course allows pharmacists to improve their knowledge about palliative care attitudes towards patients, enabling a palliative care clinical pharmacist to improve patient health outcomes through the rational use of medications. As palliative patients typically have complex medication regimens requiring frequent adjustment and monitoring, hence, a clinical pharmacist is considered a highly desired team member. 3
As palliative patients typically have complex medication regimens requiring frequent adjustment and monitoring, hence, a clinical pharmacist is considered a highly desired team member. 4 In India, home-based service is the most common setting for the provision of palliative care, a common problem encountered here is the inability of patients/carers to manage medications, which can lead to misadventure and hospitalisation. This can be averted through the detection and resolution of drug-related problems (DRPs) by a pharmacist; however, they are rarely included as members of the palliative care team. 5 So, the main objective of the study was to pilot a model of care that evaluates the roles and responsibilities of a clinical pharmacist in a palliative setup. It also focuses on introducing a palliative care course to improve the palliative care knowledge, attitudes and behaviour of the pharmacist. This project empowers us to create a perfect opportunity to educate patients and caregivers about palliative care early and often throughout the treatment. Patient outcomes can be achieved through the rational use of medication.
Materials and Methods
This project is a prospective interventional study that was conducted on the patients of a palliative care centre in Perinthalmanna, Kerala, India. Pain and palliative care in Perinthalmanna was started to integrate palliative care into comprehensive cancer and provide continuity in care to cancer patients. It also provides home care services in nearby towns. The study was carried out over 6 months, from December 2021 to May 2022 by visiting the patients in their homes.
The study protocol was accepted by the institutional ethical committee gathered on 30 March 2022 as per letter no: KAS:ADM: IEC: 054:2, and an official informed consent was prepared for conducting the study. The study included patients with kidney disease, liver disease, cancer and stroke, as these were the most prevalent conditions in our centre. This study excluded accident cases, psychiatric cases and patients below 60 years old. The patients were selected for the study as per these inclusion and exclusion criteria, and the selected patients received an informed consent form before participation in the study. The study is conducted in the Pain and Palliative Care Clinic, Perinthalmanna, which is a widely accepted palliative care clinic in Kerala. Here, the study setting is a home visit and external review pharmacists interacted with the patients in person, took the medical, medication, social and family history and reviewed the medication chart during the home visit. Here, while reviewing the medication chart, we mainly focused on drug interaction, duplication, dosage adjustment based on creatinine and hepatic clearance and other medication errors. For the collection of patient medication details, a well-structured data collection form was designed, which includes patients’ age, sex, the reason for admission, past medication and medical history, comorbidities, allergies, medication chart (generic name of medicine, dosage form, dose, frequency and indication) and drug-related problems. The Informed Consent Form and Data Collection Form were approved by the Palliative Committee team of Perinthalmanna, which is composed of doctors, nurses and healthcare volunteers. Data were collected through regular visits to the patient’s home along with the palliative team, and follow-ups were done through homecare visits and were recorded in the data collection form. As there is a lack of pharmacists during palliative care home visits, we as external pharmacists obtained informed consent from all the patients whom we visited and drug-related problems like drug interactions, drug duplication and so on were identified using Up-to-date and MEDSCAPE. Drug interactions were categorised into mild, major, moderate and severe by checking through the Medscape drug interaction checker and Stockley’s drug interaction book. Medication errors were also identified and reported in the appropriate form.
All the modifications to be done in the treatment chart were discussed with other healthcare professionals and clinical pharmacists’ interventions were done in the patient’s medication chart. The major areas that require close monitoring were identified and interpreted. These areas were then categorised as modules of the curriculum that were designed for training pharmacists in palliative care. As most patients in palliative care have kidney disease, cancer, stroke and liver problems, these were the major modules in our curriculum that are needed to train a pharmacy graduate. The curriculum also comprised Basic Life Support, waste management in a palliative setting, general pharmacology and dosage adjustments in various conditions. For the implementation of the curriculum, during the clerkship period, an Expert Committee was formed on 7 May 2022, comprising physicians and other healthcare professionals. The designed model of the curriculum was brought for scrutiny under the Expert Committee. A second meeting was conducted on 10 June 2022 to review the curriculum, and relevant changes were made. All these changes were taken into consideration, and a final draft of the curriculum was designed to implement it as an add-on course from the very next academic year onwards. Statistical procedures were done using Statistical Package for Social Sciences (SPSS) 20.0. The sample size was calculated using the following formula:
where Z1−a/2 is the standard normal variable, SD is standard deviation of the variable and d is the absolute error or precision.
A sample size of 88 was obtained from the above equation. Calculations for power (80%) of the study were completed before the commencement of the study. All quantitative variables were expressed in mean and standard deviation. Qualitative variables were expressed in percentages. The chi-square test was used for the association between variables. A probability value of p < .05 will be considered statistically significant.
Results
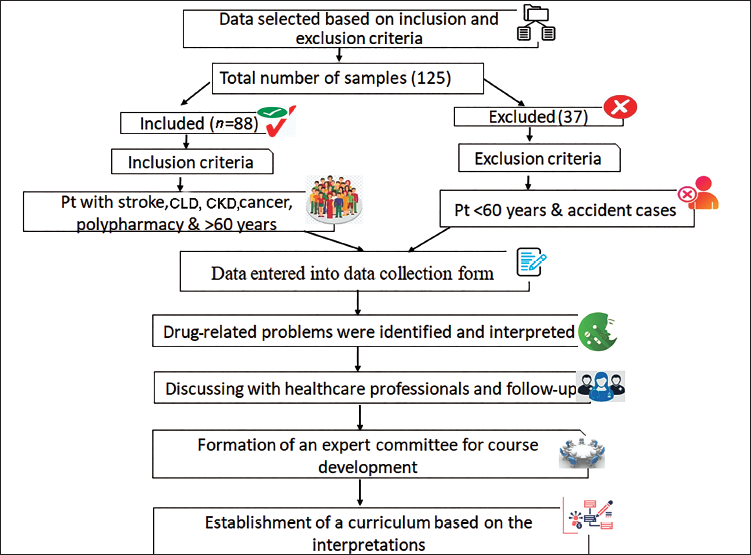
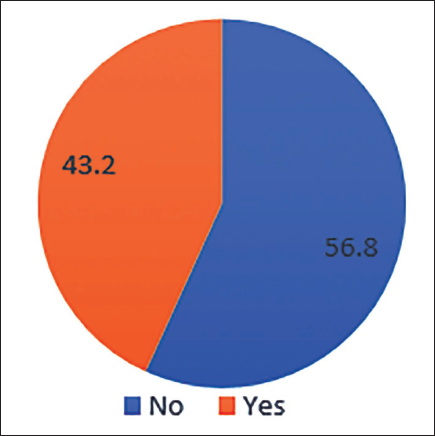
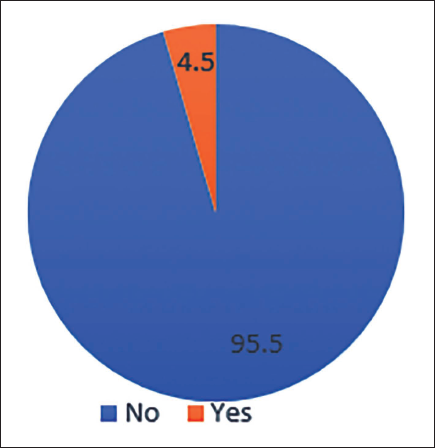
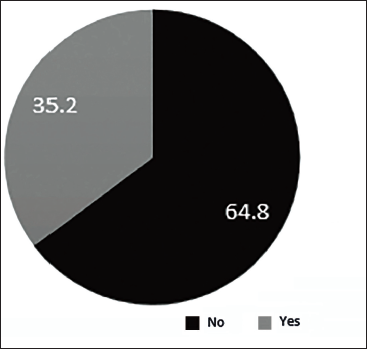
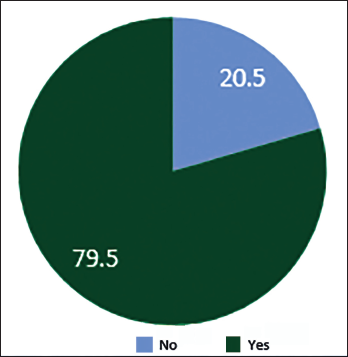
A total of 88 patients were included in the study. Notably, 37.5% of females were enrolled in the study, most of whom were 70 years old. About 62.5% of the total population were males, most of whom were around the age of 68 years. Among the study population, the most common condition was CKD (36.4%, n = 32) followed by cancer (27.3%, n = 24), stroke (27.3%, n = 24) and CLD (11.4%, n = 10). Out of n = 88, kidney disease patients who participated in the study were 32 (n = 32), chronic liver disease (n = 10), stroke and cancer (n = 24). Most of the patients had some other comorbidities, among which was found to be the most prevalent comorbidity (77.3%) (n = 68), followed by diabetes (50%) (n = 44). Nine (10.2%) dyslipidaemia cases were studied in the project. The majority of the patients in the palliative were Catheterised (37.5%) and Bedridden (35.2%) (n = 31). Out of 88 samples, 38 cases had drug interactions, 10 involved drug duplications, 4 required dosage adjustments and 6 samples had medication errors. From the total sample size, pharmacist intervention was found in about 56.8% of cases (n = 50); p < 0.001 is found to be statistically highly significant by chi-square test (Figure 1). A total of 56.8% of cases required active pharmacist intervention, and there was an overall improvement of 79.5% in the outcome of patient’s health and a p-value of <.001 was obtained, which is highly statistically significant. p-value < .05 is statistically significant; p-value < .001 is statistically highly significant by chi-square t-test (Figure 2). Drug interactions were reported in 43.2% (n = 38) of total cases (n = 88), as depicted in Figure 3. Notably, 4.5% (n = 4) out of total samples (n = 88) required dosage adjustment, as shown in Figure 4. Dosage adjustment was necessary in 4.5% of cases, and 95.5% (n = 84) cases did not require any adjustments in their doses. Most dosage adjustments were seen in patients with chronic kidney disease. Patients undergoing dialysis were found to be 35.2%, and most of them fall under stages 4 and 5 (Figure 5). There was an overall improvement of 79.5% (n = 50) in the outcome of patient health, due to pharmacist intervention. About n = 70 of the cases showed significant improvement in their health outcome. A p-value of <.05 was obtained, which is statistically significant (Figure 6). The most prevalent stage of chronic kidney disease (CKD) was stage 5 (62.5%), out of which 64.8% underwent dialysis. Drug interactions were reported in 38 out of 88 cases. Drug duplication was found in 11.4% of total cases. Four cases required dosage adjustment. More than 72% of the total population (n = 88) used multiple medications simultaneously. From this, pharmacist intervention was required in about 56% of cases.
Pharmacist Intervention.
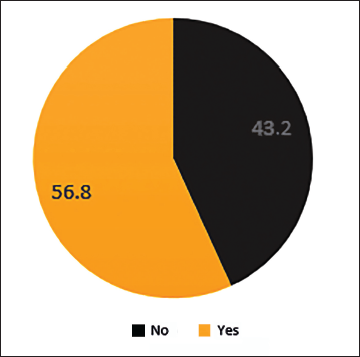
Relationship Between Pharmacist Intervention and Improvement in Outcome.
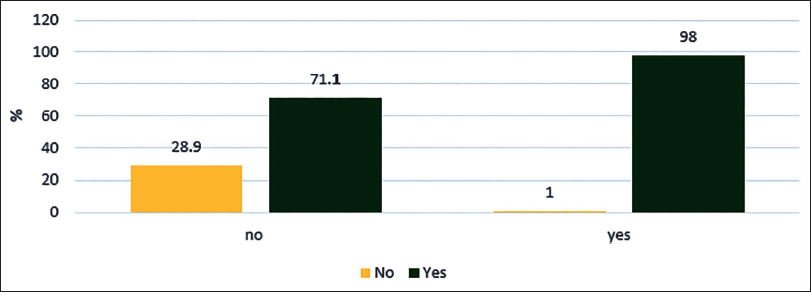
Drug Interactions.
Dosage Adjustment.
Patient on Dialysis.
Improvement in Outcome.
All statistical procedures were calculated using SPSS 20.0. A p-value of <.05 is found to be statistically significant, and a p-value <.001 was obtained by chi-square t-test, implying highly statistically significant.
Discussion
Pharmacists can play a vital role in palliative care in several ways, including the timely provision of medications, assessment of medication plans and counselling of the palliative care team. Palliative care patients often need to take multiple medications simultaneously and, as a result, have an increased risk of drug interactions and drug-related problems. While each health professional in the palliative care team should have basic knowledge about the appropriate use of medications, it is the responsibility of the pharmacist to assess the appropriateness of medications that have been prescribed for patients and do patient counselling. The study objectives were to assess the effectiveness of the clinical services provided in palliative settings, to identify hurdles and overcome and develop a strategy for improving the delivery of palliative care services. This study comprehensively assessed pharmacists’ knowledge, attitude and involvement in palliative care settings. However, it was found that many pharmacists had inadequate knowledge about palliative care, which complies with the study of Pruskowski et al. conducted at the University of Pittsburgh, Pennsylvania, which marks the significance of an add-on course for pharmacist training in palliative care setup. 6 More than 90% of pharmacists identified the goal of palliative care, and at the same time, all of them recognised the fact that pharmacist involvement can decrease the need for medical emergencies.
A prospective interventional study was carried out in the Palliative care centre, Perinthalmanna, Malappuram, Kerala, for 6 months. A bunch of 88 samples were enrolled in the survey to evaluate the need for pharmacists in a palliative setting. The study was conducted in six phases, which commenced with data collection and ended with the formation of a curriculum. Data collection was combined with a literature search and identification of pharmacist interventions. The modified changes were brought to the attention of the healthcare professionals in the palliative centre and made accordingly. Based on the changes made, a new curriculum for palliative pharmacy has been developed. About 50 cases required active pharmacist intervention and greatly impacted the outcome of patients’ health. This was consistent with a study carried out by Geum et al. about the interprofessional collaboration between a mutidisciplinary palliative care team and thereby reviewing the role of pharmacist in palliative care. 7 The factors that influence the role of pharmacists include comorbidities, drug interactions, drug duplication, medication errors and dosage adjustments. The association of these factors was assessed statistically by using the chi-square test. Out of the total population, 33 were catheterised, and there was a need for proper care to reduce complications associated with catheterisation. This result was consistent with a study conducted by Poudel et al. that evaluated the clinical pharmacy services offered for palliative care patients. 5 Most of the cancer patients had undergone chemotherapy and required clinical pharmacy assessments and services for pain management as per the study carried out by Patel et al. that focused on Oncology palliative medicine clinics conducted at Janssen Research and Development, USA. 8 Clinical pharmacist interventions like reporting medication errors, drug interactions, drug duplication and dosage adjustment based on creatinine clearance were done during palliative care services. This was consistent with a study conducted by Atayee et al. in Australia, which compared the role of pharmacist in palliative settings worldwide. 9
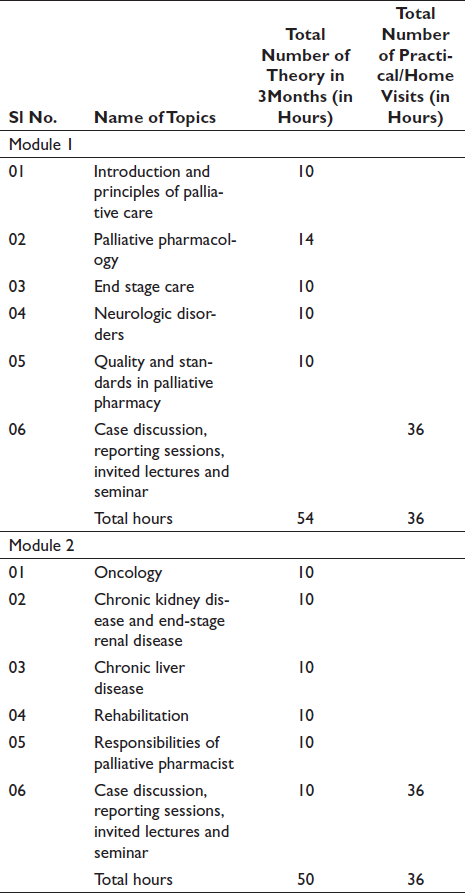
Palliative care pharmacists evaluated patients for symptom management, prevented medication-related problems and initiated and modified treatment regimens that comply with a prospective study conducted by Atayee et al. 9 in the United States. 10 Pharmacists with special training or experience in palliative care bring added value to the healthcare services to monitor therapeutic responses. 11 There were various areas where a pharmacist needed to be actively involved, like answering prescription questions, communicating with other healthcare professionals, providing in-service programs to staff, managing patients’ alternative therapy and so on. In palliative care, pharmacists were able to improve patient outcomes through the rational use of medications. This complies with the study conducted by Pruskowski at the University of Pittsburgh in 2017. 12 The study by Pruskowski et al. conducted at Pennsylvania concluded that there should be a palliative care content in the Doctor of Pharmacy [Pharm D] curriculum to learn the development and validation of skills-based palliative care measurement tool to quantify the impact of focused experiences and their long-term effects. A curriculum was formulated for pharmacy graduates to specialise in palliative training under an Expert committee’s guidance and advice. This committee included various healthcare professionals participating in a palliative team to manage the disease. Hence, a course titled ‘PALLIATIVE PHARMACY’ was developed and implemented. The content of the syllabus is divided into two modules, Modules 1 and 2, which are listed below.
There were a few constraints in the study. The study was carried out only for a period of 6 months in a single palliative care centre. The number of study subjects included in the study was minimal, which led to inefficient comparison with other palliative settings. Another restriction study was the lack of results regarding patient outcomes to evaluate constipation. Adverse drug reactions were not monitored and reported. Accident cases and psychiatric cases were excluded from the study. Patients below 60 years were also not included in the survey, as an age group of more than 60 years was found to be most prevalent in the palliative care centre. Cancer, stroke, kidney disease and liver disease were only included in the study.
Conclusion
As pharmacists can significantly contribute to palliative care, it is essential to encourage the benchmarking of practice across various clinical settings and countries to promote a consistent and equitable approach. This study showed that the inclusion of clinical pharmacists in a multi-disciplinary palliative care team has great benefits for the team, patients and carers. There are various roles that pharmacists can undertake, which leads to an increase in medication-related knowledge and improves the medication and symptom management of patients. The addition of pharmacists to the palliative team plays a vital role in minimising medication-related problems like drug duplication, drug interactions, dosage adjustments, medication errors and so on. A statistically significant change in the outcome was observed with pharmacists’ intervention in this study. This is important for all patients, but particularly so for those who require palliative care because the medications play a key role in increasing the quantity and quality of life for palliative care patients. Pharmacists, in general, emphasise their educational and research roles more than clinical work. In India, there is a critical lack of pharmacists in the palliative setup. Many pharmacists are unaware of their duties in a palliative centre. This marks the need for a curriculum for pharmacists in palliative training. Hence, this study focuses on the augmentation of an add-on course that helps pharmacy graduates learn palliative care skills. The future of sustaining pharmacists’ success in palliative care depends upon reimbursement of services, standardisation of pharmacists’ skills and increased acceptance of a pharmacist’s role by national palliative care professional organisations.
Summary
This study showed that the inclusion of a clinical pharmacist in a palliative care team has great benefits for the patients as well as carers. Therefore, this study was utilised to improve patient’s quality of life through the rational use of medications by employing effective and valuable patient care service, research and clinical education. There was an overall improvement in the patient’s quality of life with the inclusion of a clinical pharmacist. Hence, this leads to the development of an add-on course for the training of pharmacy graduates in a palliative setup.
Abbreviations
Footnotes
Acknowledgments
The authors thank Pain and Palliative Unit, Perinthalmanna, for extending all support towards this research project.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
The study protocol was accepted by the institutional ethical committee gathered on 30 March 2022 as per letter no: KAS:ADM: IEC: 054:2.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
The participant has consented to the submission of the article to the journal.