Abstract
Background
The term “gout” refers to a broad clinical spectrum of diseases, including common and complex forms of arthritis, that affect multiple joints in a patient due to an elevated serum urate concentration.
Purpose
The study aims to compare the efficacy and safety of selective cyclooxygenase-2 (COX-2) inhibitors and non-selective nonsteroidal anti-inflammatory drugs (NSAIDs) in the treatment of acute gout, as well as to conduct a meta-analysis on safety.
Methodology
As of December 2021, the literature search was conducted using authorised electronic databases such as PubMed, Embase, Web of Science, and the Cochrane Library.
Results
There were seven investigations included in this study’s findings. Three COX-2 inhibitors, etoricoxib, celecoxib, and meloxicam, were reportedly compared to indomethacin or diclofenac. Four hours after the initial dose, 120 mg of etoricoxib reduces pain and inflammation by diminishing erythema. At higher dosages, celecoxib is more effective than at lower doses. Meloxicam has the same efficacy as NSAIDs. The subgroup analysis revealed that the risk of adverse events was 8.0% lower in the COX-2 inhibitors group than in the non-selective NSAIDs group (risk ratio = 0.92, 95% confidence interval = 0.60 to 0.40, p-value = 0.5).
Conclusion
Etoricoxib, a COX-2 inhibitor, has a more potent effect and may be more effective than non-selective NSAIDs. COX-2 inhibitors are more well tolerated and reduce the risk of adverse gastrointestinal events more effectively than non-selective NSAIDs. In the treatment of gout, non-selective NSAIDs may be a suitable alternative to COX-2 inhibitors.
Introduction
Gout manifests as hyperuricemia with plasma or serum urate concentrations greater than 6.8 mg/dL and is characterized by the deposition of monosodium urate crystals in the synovial membrane of the joints. The ionized form of uric acid is urate in the organism. In humans, purine degradation results in the production of uric acid. Uric acid is considered a waste product because it does not have any known physiological function. At room temperature, blood uric acid is saturated between 6.4 and 6.8 mg/dL. In patients with gout, the accumulation of urate is multiplied by several orders of magnitude. Excess accumulation may be a consequence of excessive uric acid production or inadequate excretion.1–3
The lower limb joints, especially the big toe, are frequently affected, followed by the mid-tarsal, ankle, knee, and upper limb joints; subsequent acute flares tend to be more prolonged and involve multiple joints, particularly the upper limb joints like the wrist or elbow. 4
The global burden of gout is increasing both in developed countries and in developing countries due to an aging population and increasing obesity rates. According to a study carried out in 2020, the prevalence and incidence of gout ranged from 0.1% to 0.3% worldwide. Approximately 3:1–10:1 more males than females suffer from gout. The incidence and prevalence of gout increased with each decade of life, reaching 11%–13% and 0.4%, respectively, in those aged >80 years. Approximately, 10% of the patients with hyperuricemia develop gout. Around 80%–90% of patients with gout are hyperuricemic.5, 6
The management of gout can be divided into two categories: treatment of acute gout attacks and prevention of gout flares. Treatment of acute arthritis attacks aims to reduce pain and inflammation.
Non-steroidal anti-inflammatory drugs (NSAIDs), such as selective cyclooxygenase-2 (COX-2) inhibitors,7–9 are frequently utilized in the management of inflammatory conditions. As outlined in published recommendations, they are to be employed for the treatment of acute attacks, administered at maximum dosages, but for a limited duration.10–12 These guidelines assert that all NSAIDs possess equivalent efficacy.
NSAIDs, corticosteroids, and colchicine are usually administered orally or intravenously. Prophylactic treatment of gout flare-ups involves lowering serum urate levels by decreasing uric acid production or increasing uric acid excretion from the body.13, 14
In accordance with the 2006 and 2016 recommendations of the European League Against Rheumatism 15 and the American College of Rheumatology (ACR), NSAIDs have been considered the first-line treatment for acute gout in recent decades. 16
NSAIDs are mediators that inhibit the cyclooxygenase (COX) enzyme. There are two types of COX: 1 and 2. COX-1 is an essential enzyme that is widely distributed throughout the body. It contributes to the maintenance of gastrointestinal (GI) mucosa, kidney function, and platelet aggregation. However, the expression of COX-2 increases only during inflammatory reactions. Therefore, NSAIDs can be categorized according to the COX enzyme that inhibits them. Non-selective NSAIDs inhibit both COX-1 and COX-2, whereas selective COX-2 inhibitors inhibit only COX-2.
In 2014, van Durme CMPG 17 conducted a systematic review to evaluate the benefits and drawbacks of NSAIDs when used in the management of acute gout. It is of utmost importance to re-examine this review to integrate any relevant recent findings.
Because traditional NSAIDs inhibit prostaglandin secretion, they increase the risk of GI haemorrhage and renal impairment, despite the importance of COX-1. 1
Given the increasing use of COX-2 inhibitors and the relatively large number of ongoing studies, a comparative assessment of the efficacy of different COX-2 inhibitors is crucial from the point of view of clinical efficacy and safety. In clinical practice, etoricoxib, celecoxib, and meloxicam are used as COX-2 inhibitors. Numerous studies on COX-2 inhibitors for the treatment of gout have also been published in recent years. We conducted a systematic review and meta-analysis on safety in patients with acute gout to provide an updated picture of the comparative clinical efficacy and safety of traditional non-selective NSAIDs and COX-2 inhibitors.
Methodology
According to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines, this systematic review was conducted.
Bibliographical Research
As of December 2021, the data sources were collected from published original articles from authorized electronic databases, such as PubMed, Embase, Web of Science, and the Cochrane Library.
Article Search Technique
COX-2 inhibitors, acute gout, gouty arthritis, celecoxib, etoricoxib, meloxicam, safety, tolerability, NSAIDs, indomethacin, diclofenac, ibuprofen, NSAIDs, efficacy, safety, tolerability, adverse events, and effectiveness were the keywords used to search for articles.
Inclusion Criteria
We only included studies that confirmed a diagnosis of acute gout according to the diagnostic criteria established by the American Rheumatology Association. We included only English-language, full-text studies of randomized controlled trials conducted on adult patients (aged >18 years) and conducted in the English language. Only studies that compared COX-2 inhibitors to conventional non-selective NSAIDs were included.
Exclusion Criteria
We excluded studies that reported acute gout in conjunction with other diseases. We excluded studies that compared the effects of non-selective, non-traditional NSAIDs. We excluded preclinical studies, non-English studies, and studies without full-text accessibility.
Statistical Analysis
Using a meta-analysis, the safety profile was evaluated by combining the relative risks from multiple studies. Using I2 and Cochran’s Q test, heterogeneity across the studies was evaluated. A fixed-effects model was used to aggregate the studies because heterogeneity between studies was less than 50%. RevMan Software (V.5.3; Cochrane Collaboration) was used to conduct the meta-analysis. Pooled relative risks and their respective confidence intervals (CIs) at 95% were reported.
Results
A preliminary literature search revealed 266 relevant articles; of which 259 were excluded due to duplication (n = 58), a review of the abstract and title (n = 181), and a review of the full text (n = 17), as presented in Figure 1.
Schematic Representation of the Selection of Studies.
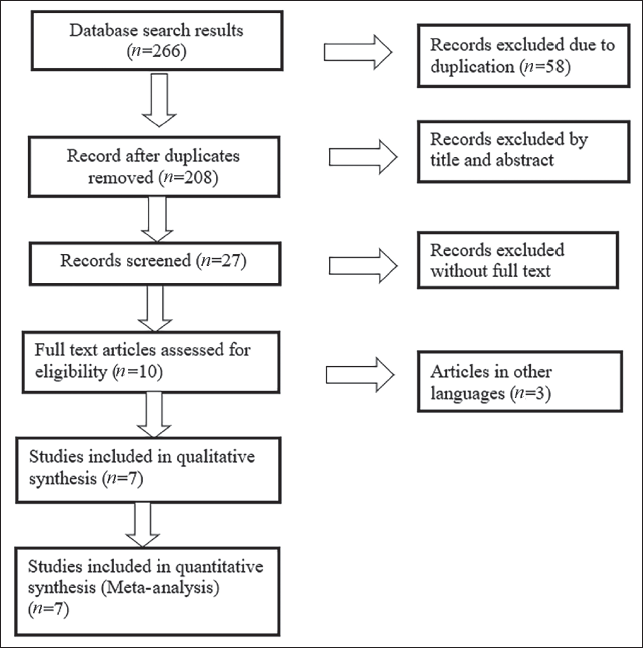
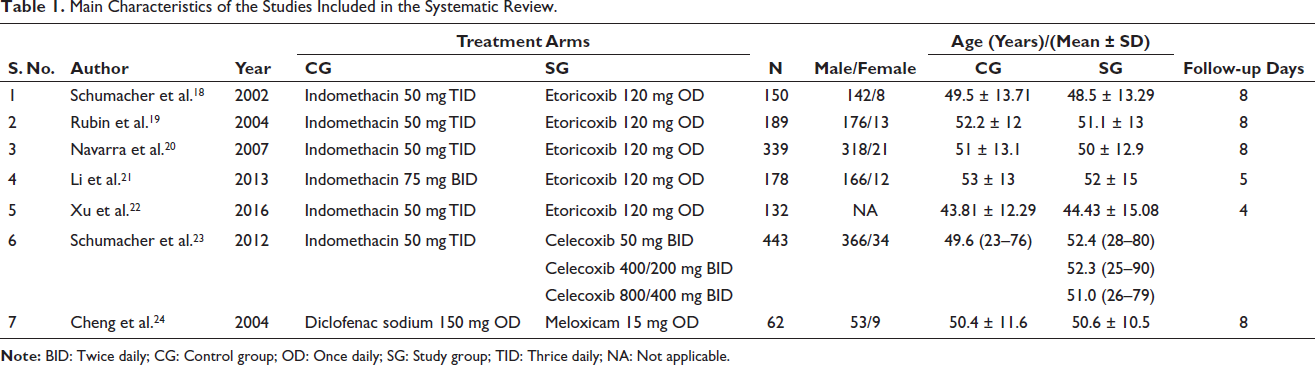
Table 1 presents the information from seven studies that may be eligible for inclusion. One thousand five hundred and eleven patients were administered COX-2 inhibitors. Three COX-2 inhibitors were compared with a comparator. Five studies had 8 days of follow-up, whereas two studies had 4 and 5 days of follow-up. In five studies, etoricoxib was compared with placebo. One study compared celecoxib with a placebo and meloxicam with a placebo. In six studies, indomethacin was administered to the control group, while in one study, diclofenac was administered to the control group. The sample population ranged from 62 to 443 participants, with males outnumbering females in every study. Almost all reported studies included age-standardized studies and control groups.
Main Characteristics of the Studies Included in the Systematic Review.
Efficacy of Etoricoxib
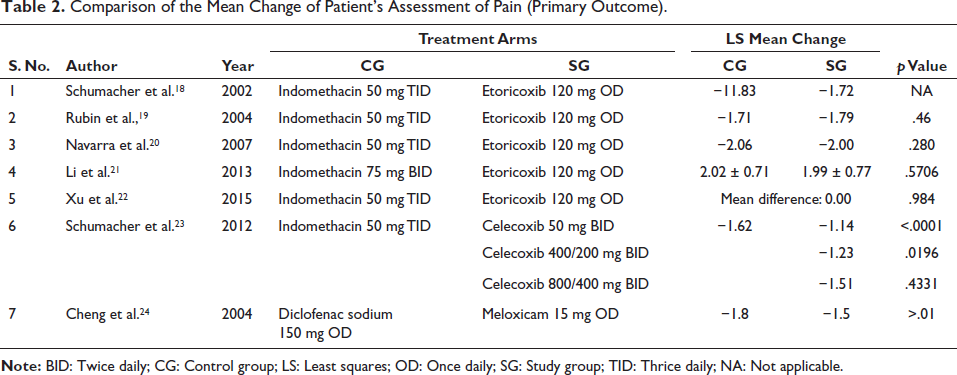
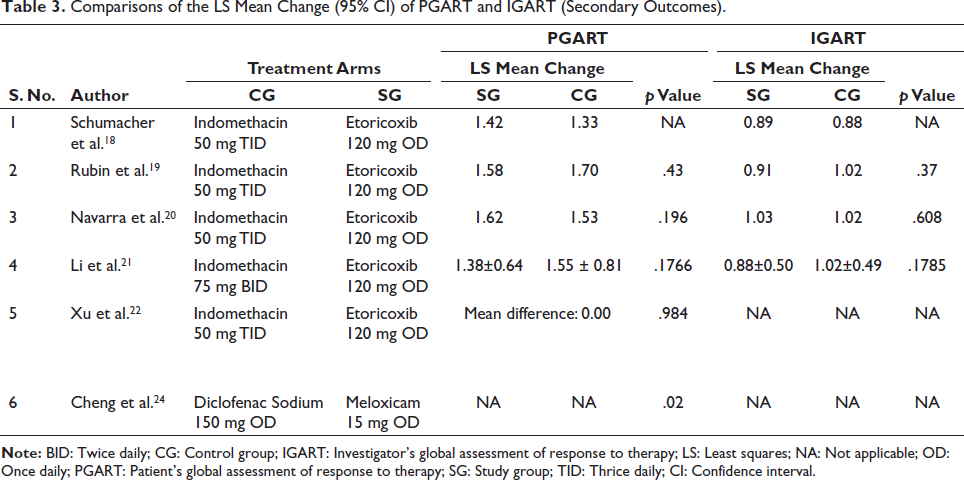
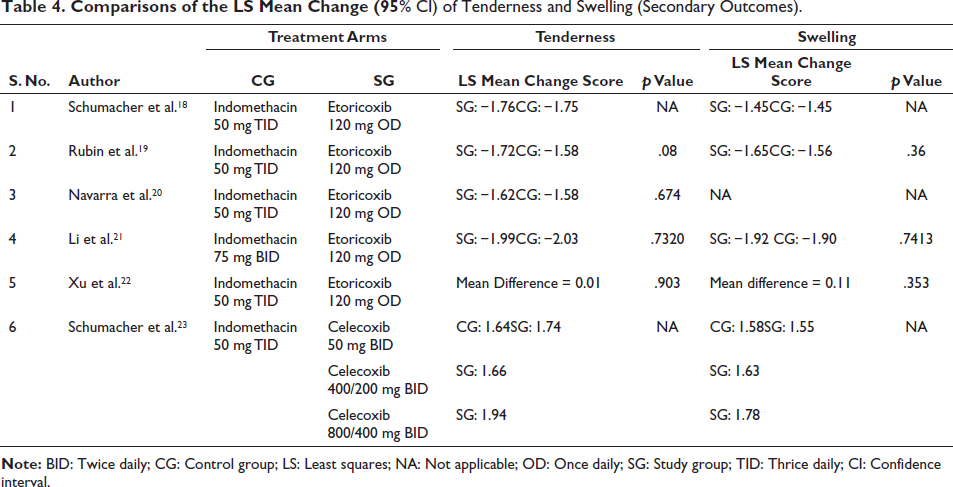
The study compared a study group with a control group in five studies.18–21 The patients received etoricoxib or indomethacin. Controls in one study received 75 mg twice daily (BID), while controls in four other studies received 50 mg thrice daily (TID). Eligible patients were at least 18 years old and had developed gout with accompanying pain within 48 h. The primary endpoint in all five investigations was the patient’s assessment of pain in the primary joint (Table 2). Pain was assessed at baseline and 4 h after the first dose in four studies. One study did not specify the time of measurement. Pain decreased from baseline to days 2–5 or days 2–4 in four studies. Two studies found that etoricoxib and indomethacin were equally effective in reducing pain. Etoricoxib and indomethacin were compared over the entire treatment period in two studies with similar results. Treatment differences were not statistically significant. Secondary endpoints (Tables 3 and 4) included the patient’s global assessment of response to therapy (PGART), the physician’s assessment of tenderness and edema, and the investigator’s global assessment of response to therapy (IGART). Etoricoxib and indomethacin demonstrated comparable efficacy in controlling pain based on secondary endpoints.21, 22 One participant in the indomethacin group withdrew from the study due to a lack of efficacy. Etoricoxib was more effective than indomethacin in controlling erythema. Approximately 70% of subjects experienced severe or extreme pain at baseline. After 4 h, 32% of patients treated with etoricoxib and 23% of patients treated with indomethacin reported no or minimal pain. Etoricoxib may have a slightly faster onset of action than indomethacin.
Comparison of the Mean Change of Patient’s Assessment of Pain (Primary Outcome).
Comparisons of the LS Mean Change (95% CI) of PGART and IGART (Secondary Outcomes).
Comparisons of the LS Mean Change (95% CI) of Tenderness and Swelling (Secondary Outcomes).
Efficacy of Celecoxib
In a study, the effectiveness of celecoxib was compared to that of indomethacin. 23 Three comparisons were made: celecoxib 800/400 mg BID, celecoxib 400/200 mg BID, and celecoxib 50 mg BID with indomethacin 50 mg TID. Of the 402 patients, 400 received treatment; 98, 99, 101, and 102 patients, respectively, were assigned celecoxib 800/400 mg, 400/200 mg, celecoxib 50 mg, and indomethacin 50 mg. Eligible patients were at least 18 years old, had acute gout pain within 48 h, and experienced moderate to extreme pain. Patients with gout affecting more than four joints were excluded.
The primary endpoint was the change in pain intensity from baseline to day 2 (Table 2). All treatment groups showed a decrease in discomfort compared to baseline. On day 2, high-dose celecoxib resulted in a greater reduction in pain intensity compared to low-dose celecoxib but similar effectiveness compared to indomethacin 50 mg TID. There was no significant difference between celecoxib 400/200 mg and low-dose celecoxib, and the indomethacin group was significantly less effective. Secondary endpoints included the physician’s pain assessment, the patient’s 24 h assessment of pain intensity, and changes in the patient’s assessments of pain intensity (Tables 3 and 4). The physician’s evaluation of the index joint showed comparable results between the celecoxib and indomethacin groups. No significant differences were observed in erythema and index joint warmth.
Patients receiving high-dose celecoxib reported a greater reduction in pain intensity compared to those receiving low-dose celecoxib. No significant differences were observed between high-dose celecoxib and indomethacin regimens. Patients in the celecoxib group reported greater pain relief compared to the indomethacin group on day 1. The perception of pain differed significantly between high-dose celecoxib and low-dose celecoxib, but no significant difference was observed between the indomethacin and celecoxib groups.
Efficacy of Meloxicam
In one study, the effectiveness of meloxicam, rofecoxib, and diclofenac was compared. 24 Sixty-two participants were enrolled in the 8-day study. Meloxicam, rofecoxib, and diclofenac sodium SR were compared. Patients with acute gout attacks and a pain score greater than 5 were eligible. Polyarticular joints and recent GI haemorrhages were excluded. The efficacy was evaluated on days 3 and 8. The response to therapy was measured. Tenderness, swelling, and function restriction were assessed. Patients rated their pain intensity at different time points. No significant difference was found between the treatment groups on day 8. Meloxicam and diclofenac showed similar efficacy on day 3. Pain scores were reduced by meloxicam and diclofenac in the first 2 h. All three treatment groups showed significant anti-inflammatory effects on days 3 and 8. The differences between treatments were not statistically significant.
The Safety Profile of Etoricoxib, Celecoxib, and Meloxicam
Table 5 summarizes adverse event evaluations during safety investigations. Four out of five studies showed a clear safety profile. One study was excluded due to a lack of safety data. All four studies assessed adverse events, with three assessing drug-related and serious events. Table 6 shows the distribution of adverse events. Vertigo, edema, headache, and fever were reported in one study evaluating GI events. Etoricoxib had a lower incidence of non-serious adverse events compared to indomethacin. Two studies showed serious side effects, one of which was renal failure. One patient in the etoricoxib group and one in the indomethacin group withdrew due to adverse events. The etoricoxib withdrawal was not drug-related. The most common side effects were headaches and the worsening of gout symptoms. Celecoxib had a lower frequency of adverse reactions compared to indomethacin. Indomethacin was discontinued more often than celecoxib. No serious adverse events were reported in any cohort. No participants withdrew from the study. Edema and abdominal pain were the most frequently reported adverse reactions, likely related to treatment.
Comparison of Prespecified AEs Reported in the Studies.
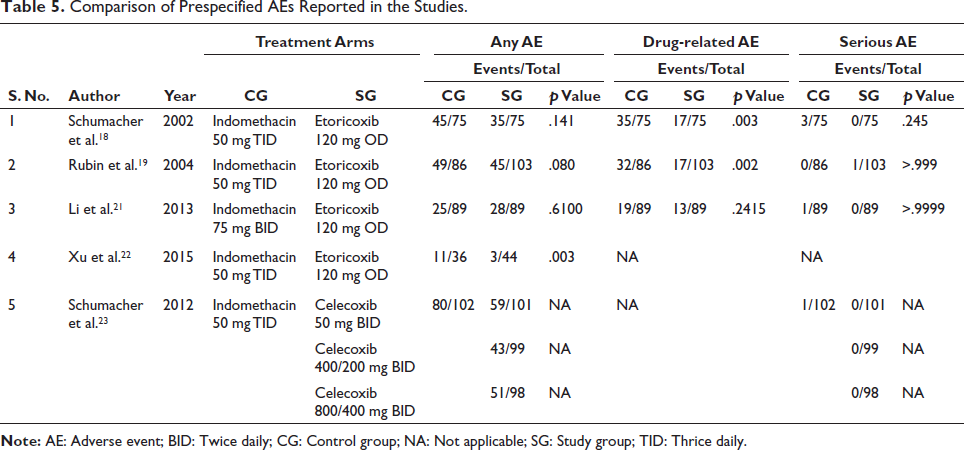
Distribution of Adverse Events Reported in the Studies.
Evaluation of the Bias Risk in the Included Studies
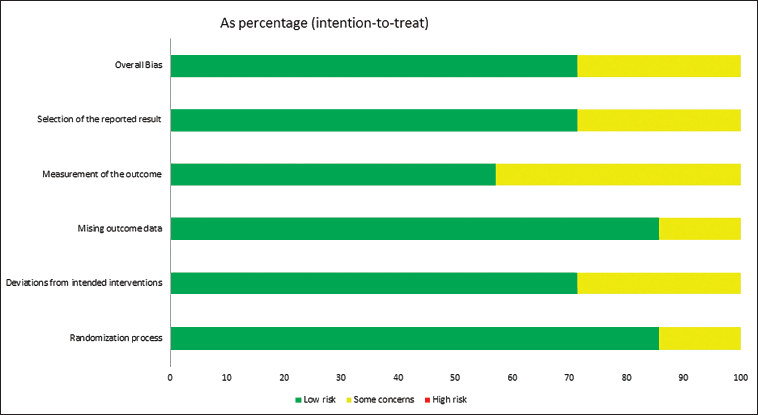
Six (85%) of the seven articles included in our investigation were randomized. It was unclear from the remaining articles whether randomization was employed. In six (85%) of the seven studies, there were no missing outcome data, and it was unclear in one study. Only five of the seven included studies (85%) have not deviated from the intended studies, and there is no information regarding the other two studies. Three studies have some concerns regarding the measurement of outcomes, whereas only four articles (55%) have low measurement risk. Approximately 70% of the included studies described blinding of participants and personnel; one study described blinding of personnel only, and one study was not blinded. In two articles, the selection of outcomes reported was ambiguous, whereas the remaining five articles (70%) had a low risk of bias. No additional bias was identified. Figure 2 indicates that the overall bias appears to be 70% for minimal risk and 30% for some concerns.
Risk of Bias Graph of Included Studies About Each Risk of Bias.
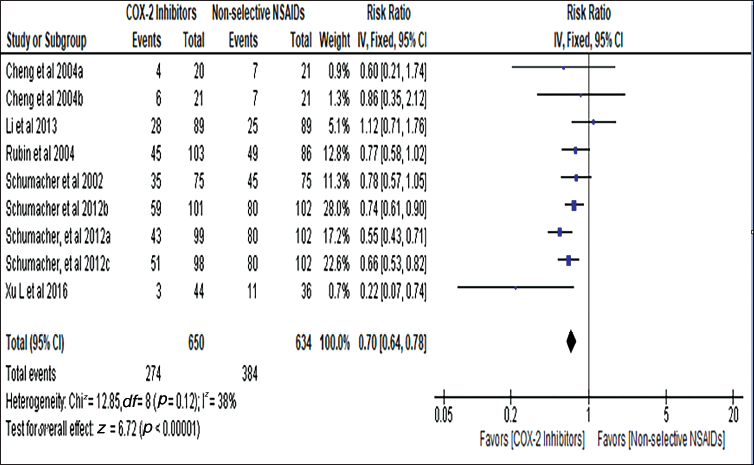
Meta-analysis
The safety profile of the included studies was analysed through a meta-analysis. In the selected studies, the COX-2 inhibitor and non-selective NSAID categories contained 650 and 634 patients, respectively. The studies were pooled using a fixed-effects model because heterogeneity was less than 50% (I2 = 38.0%, p = .21). Figure 3 shows that the risk of adverse events was reduced by 30.0% in the COX-2 inhibitors group compared to the non-selective NSAIDs group (R2 = 0.70, 95% CI: 0.64–0.78, p = .001).
Subgroup Evaluation
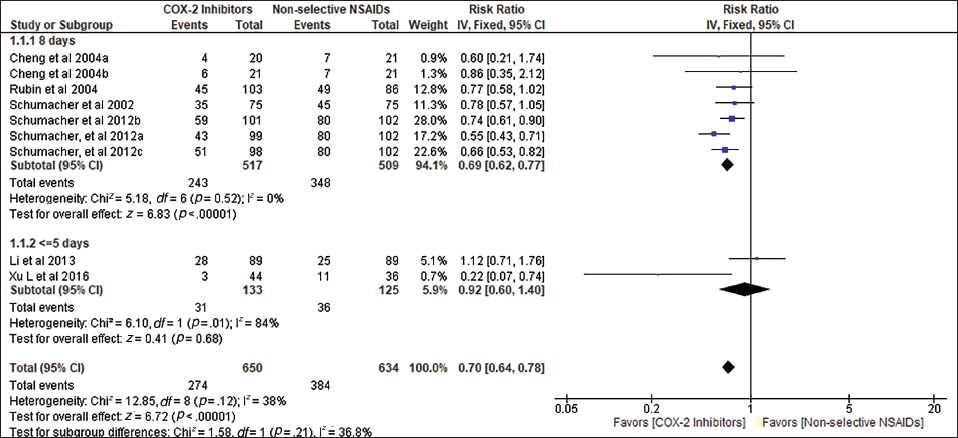
A subgroup analysis was conducted according to the number of follow-up days. Figure 4 depicts two subgroups: studies with 5 days of follow-up and 8 days of follow-up. There were only two studies in the 5-day follow-up group, whereas the remaining studies had a follow-up period of 8 days. The subgroup analysis revealed that the risk of adverse events was only 8% lower in the COX-2 inhibitors group than in the non-selective NSAIDs group in studies with only a 5-day follow-up (RR = 0.92, 95% CI: 0.60–0.40, p = .5). This was due to the Li et al. 21 study, in which the risk was 1.12 times higher in the COX-2 inhibitors group than in the non-selective NSAIDs group but was statistically insignificant. However, the studies with 8 days of follow-up provided a pooled relative risk of 0.69 (95% CI: 0.62–0.77, p = .001), which was also statistically significant. This indicated that the risk was 31.0% lower in the COX-2 inhibitors group than in the non-selective NSAIDs group among the studies with more than 8 days of follow-up and was closer to the overall aggregated effect estimate.
Discussion
In this investigation, the efficacy and safety of COX-2 inhibitors for treating acute gout pain are systematically evaluated. After analysing the results of numerous studies, it was determined that COX-2 inhibitors have comparable efficacy to NSAIDs for treating gout, as measured by Likert’s score and Visual Analog Scale (VAS) scores. COX-2 inhibitors had comparable effects on primary endpoints and secondary endpoints.
Comparatively, selective NSAIDs had a greater impact on secondary outcomes than non-selective NSAIDs. Because of the inhibition of the COX-1 enzyme, non-selective NSAIDs are associated with severe GI effects. Because selective COX-2 inhibitors do not inhibit COX-1 enzymes, they have fewer GI side effects than NSAIDs. Various studies have evaluated the efficacy and safety of COX-2 inhibitors in inflammatory conditions such as rheumatoid arthritis (RA) and osteoarthritis.8, 25 The 2012 ACR guidelines recommend the single administration of NSAIDs, systemic corticosteroids, and oral colchicine, but do not prioritize one substance over another. 26 However, because the therapeutic dose and the toxic dose of colchicine are so close, adverse effects are common. Consequently, there must be a distinct understanding of the drug of choice for treating conditions such as gout and RA.
Etoricoxib 120 mg provides pain relief as early as 4 h after the first dose and over days 2–5 when administered within 48 h of the onset of pain; it also reduces inflammation by alleviating erythema. For treating pain, a high dose (800/400) of celecoxib is more effective than a low dose (400/200) of celecoxib. Meloxicam (15 mg) provides comparable efficacy to NSAIDs.
Compared with traditional non-selective NSAIDs, the risk of GI adverse events is significantly lower with selective COX-2 inhibitors. In patients with previous myocardial infarctions, even short-term treatment with most NSAIDs (except naproxen) was associated with an increased risk of mortality and recurrent myocardial infarction, according to a review of a Danish database. They reported that non-selective NSAIDs are associated with a higher incidence of cardiovascular events than COX-2 inhibitors. 27
In most studies, subjects tolerated both selective COX-2 inhibitors and non-selective NSAIDs well. Significantly fewer subjects withdrew, and the withdrawals were unrelated to drug effects.
Minor adverse events, such as headache, vertigo, vomiting, and edema, are associated with both NSAIDs and COX-2 inhibitors, but the incidence of non-selective NSAIDs is higher.
Conclusion
This systematic review and meta-analysis has uncovered that COX-2 inhibitors demonstrate similar effectiveness to non-selective conventional NSAIDs in the treatment of gout-related pain. Etoricoxib, a specific inhibitor of COX-2, exhibits enhanced effectiveness in alleviating both pain and inflammation through the reduction of erythema. Celecoxib, on the other hand, proves more potent at higher dosages. Meloxicam exhibits equivalent efficacy to NSAIDs, including NSAIDs and other COX-2 inhibitors. Remarkably, COX-2 inhibitors present a significantly reduced risk of GI adverse effects in comparison to non-selective NSAIDs. Consequently, COX-2 inhibitors emerge as a viable alternative to non-selective NSAIDs for the treatment of gout.
Limitations
This research has several limitations. Very few articles were available for this study, and many studies were available only in languages other than English. Studies comparing the efficacy and safety of meloxicam in the treatment of acute gout are very limited. The studies currently available suffer from a restricted sample size. Acute gout is the main indication reviewed in the studies and the effect of COX-2 inhibitors in chronic gout needs to be assessed. Because some COX-2 inhibitors (lumiracoxib, rofecoxib, valdecoxib, and parecoxib) were withdrawn from the market, the study was limited to currently available pharmaceuticals such as etoricoxib, celecoxib, and meloxicam.
Summary
COX-2 inhibitors have a lower risk of GI side effects.
COX-2 inhibitors are as effective as non-selective NSAIDs for gout pain.
Etoricoxib is more effective than non-selective NSAIDs for pain treatment.
Abbreviations
Authors’ Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis, and interpretation, or all these areas; took part in drafting, revising, or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
