Abstract
Objectives
The prescription errors and prescribing fault analysis was assessed, the rationality of the prescriptions was checked, and the medication error was categorized according to the NCC MERP Index.
Materials and Methods
A cross-sectional, observational study was designed as per STROBE guidelines and conducted for 2 months in the pharmacy stores after approval of the Institutional Review Board. Patients’ written informed consent was taken before getting their prescriptions, and each of the prescriptions procured in this way was photographed for record. The completeness of 320 prescriptions of outpatients of all age groups regarding the details about the doctor and the patient and clinical diagnosis/indication was analyzed. The rationality of prescription was based on WHO core drug use indicators. Descriptive analysis was done by using Microsoft Excel.
Results
A total of 320 prescriptions were analyzed from eight departments. Information about patients and prescribers was mentioned in 100% of prescriptions. The diagnosis (40%), an indication was written in 195 prescriptions. Instructions for dispensing drugs (89%), instructions to patients (90%), duration of treatment (100%), follow-up visits (19%), and non-pharmacological instructions (13%) were mentioned. In total, 82% of prescriptions were legible. In a total of 1004 drugs, 92% of drugs were prescribed with a generic name, 100% from the essential drug list. The route and frequency of drug administration were mentioned for all drugs. According to NCCMERP, the category of medication errors falls under category B.
Conclusion
To reduce medication errors, we can implement an electronic system, involve clinical pharmacologists, utilize prescription charts, and organize nationwide workshops on rational prescription writing. We should encourage regular prescription audits and reporting to improve the healthcare system in the country.
Introduction
A prescription is a written document by the doctor for a pharmacist or nurse to take a patient’s health care. If this document is misinterpreted at any level, it can lead to wrong treatment, aggravation of the disease, health hazards, and economic burden on the patients’ healthcare system. These prescription faults and prescription errors lead to medication errors. 1 US National Coordinating Council for Medication Error Reporting and Prevention (NCCMERP) defines medication errors as “any preventable event that may cause or lead to inappropriate medication use or patient harm while the medication is in the control of the healthcare professional, patient, or consumer.” 2 Prescription errors are those related to the act of writing a prescription, whereas prescribing faults enclose irrational prescribing, inappropriate prescribing, under-prescribing, overprescribing, and ineffective prescribing arising from inaccurate medical judgment or decisions concerning treatment or treatment monitoring. 3 Error can arise from any step of prescribing such as the choice of drug, dose, dosage, route of administration, and wrong duration of treatment. Inaccuracy and poor legibility of writing or incomplete prescriptions may leave scope for misinterpretation, thus leading to errors in dispensing medication. Errors of omission and errors of commission are the two main types of prescription errors. Errors of omission are where a prescription is incomplete in some way, whereas errors of commission contain incorrect information. 4
Prescription errors and prescribing faults lead to 70% of total medication errors. 1 A mean value of prescribing errors with the potential for adverse effects in patients of about 4 in 1000 prescriptions was recorded in a teaching hospital. 5 Studies suggest that prescribing errors are common and can affect from 4.2% to 82% of prescriptions. 6
The error can arise from any step of prescribing such as the choice of drug, dose, dosage, route of administration, and wrong duration of treatment. Inaccuracy and poor legibility of writing or incomplete prescriptions may leave scope for misinterpretation, thus leading to errors in dispensing medication. These errors can be detected by a prescription audit. An “audit” is defined as “the review and the evaluation of the healthcare procedures and documentation to compare the quality of care which is provided, with the accepted standards”. 7 Prescription audit is a part of the audit which seeks to monitor, evaluate, and if necessary, suggest modifications in the prescribing practices of medical practitioners. 8
Prescription auditing is an important tool to avoid drug misuse and improve the rational use of drugs. If regularly done, prescription audits can aid in improving the prescription quality and thus enable the patient to receive high standard and best quality care and to reduce adverse drug events, hospital stay, and morbidity and mortality, and thus, overall burden to the healthcare system and community is decreased. 9
World Health Organization (WHO) collaborated with the International Network for Rational Use of Drugs (INRUD) to develop a set of “core drug use indicators.” These prescribing indicators evaluate the practice of prescribers in five key areas: percentage of drugs prescribed by generic name, the average number of drugs per prescription, percentage of prescriptions containing antibiotics, percentage of prescriptions containing injectable drugs, and percentage of drugs prescribed from the latest edition of National Essential Drug Lists (EDL) or formulary. 10 These indicators are useful for the assessment of prescribing patterns for the rational use of drugs.
Irrational design of prescription is one of the important causes of medication error leading to the reduction in efficacy or enhancement in toxicity. Prescription errors can be preventable forms of medication errors, and there is much scope for improvement in this area. 1 The NCC MERP Index was developed to know the severity level of medication errors. 11 Data are scarce on such studies on medication errors in hospital settings in India and at the local level.
After consideration of all the above points, this study was planned to evaluate medical error by prescription audit at our tertiary care teaching hospital, Gujarat, India, to generate more data regarding it and to provide guidelines to the healthcare professionals in policy making and to improve the healthcare system.
The objectives of the present study were to assess the prescription errors and prescribing fault analysis, document the information on the core prescribing indicators proposed by the WHO, check the rationality of the prescription, and categorize the medication error according to the NCC MERP Index. 11
Materials and Methods
It was a cross-sectional, observational, and non-interventional study designed as per STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines and conducted for 2 months in the pharmacy stores of the tertiary care teaching hospital of South Gujarat after ethical clearance from the Institutional Ethics Committee (Human Research Ethics Committee). Patients were approached for their prescriptions at the pharmacy store. Patients were informed about the purpose of the study, and their written informed consent was taken before getting their prescription.
Each of the prescriptions procured in this way was photographed to record the contained information. Only freshly registered outpatients with prescriptions of all age groups and irrespective of gender from the clinical department were included in the study. Patients who were not willing to give consent for using their prescription were excluded from the study. A total of 320 prescriptions were selected for the analysis.
Analysis was done by using the following parameters:
Completeness of prescription in terms of the format as compared against the Standard prevailing in the country including:
Details about the Doctor: Name, Qualification, Designation. Details about Patient: Full name, Age, Sex, Weight, Address, Contact No. Clinical Diagnosis/Indication. Details about prescribing Standards: Name of the Drug, Dosage form, Dosing Information, Route of administration, Duration of treatment, Non-pharmacological advice, Follow-up advice. Legibility of prescription. Rationality of prescription based on Core Prescribing Indicators proposed by WHO:
8
These indicators measure the performance of the doctors who appropriately use the drugs.
The core prescribing indicators are as follows:
Average number of drugs per prescription (encounter). Percentage of drugs prescribed by generic name. Percentage of encounters with an antibiotic prescribed. Percentage of encounters with an injection prescribed. Percentage of drugs prescribed from essential drugs list or formulary.
Health facility indicators
Availability of copy of EDL in all OPDs. Availability of key drugs.
A cross-sectional, observational study was designed as per STROBE guidelines and was started after approval from the Institutional Review Board.
Conventional data collection from pharmacy store for 2 months.
Prescription analysis was done as per WHO core prescribing indicators, and descriptive statistical analysis was done by using Microsoft Excel.
NCC MERP Index for Categorizing Medication Errors: 11
Category A: Circumstances or events that can cause an error.
Category B: An error occurred but the error did not reach the patient (An “error of omission” does reach the patient).
Data Analysis: Descriptive analysis was done by using Microsoft Excel. All data were expressed in numbers and percentages.
Results
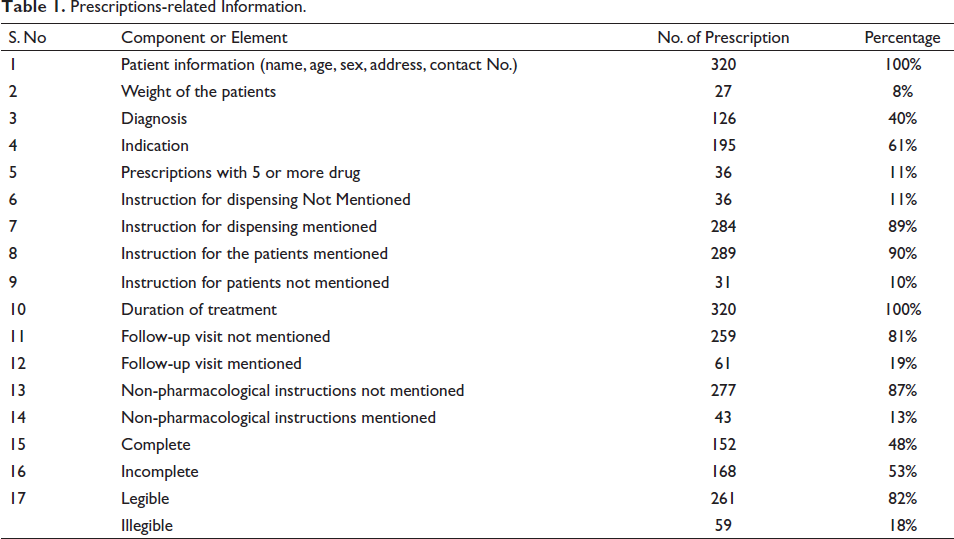
A total of 320 prescriptions were analyzed in this study, and all prescriptions contained demographic details of patients and prescriber’s information like name, qualification, and designation. The date and place were written in all prescriptions, and the weight of the patients was written in 27 prescriptions. The diagnosis was written in 126 (40%) prescriptions and an indication of drug usage was written in 195 prescriptions. Of prescriptions, 5 or more drugs were written in 36 (11%). In total, 284 (89%) prescriptions contain instructions for dispensing drugs, and instructions to patients were mentioned in 289 (90%) prescriptions. The duration of treatment was mentioned in all prescriptions. A follow-up visit was mentioned in 61 (19%) prescriptions, and non-pharmacological instructions were mentioned in 43 (13%) prescriptions. Of 320 prescriptions, 152 (48%) were complete, and 261 (82%) were legible (Table 1 and Figure 1).
Prescriptions-related Information.
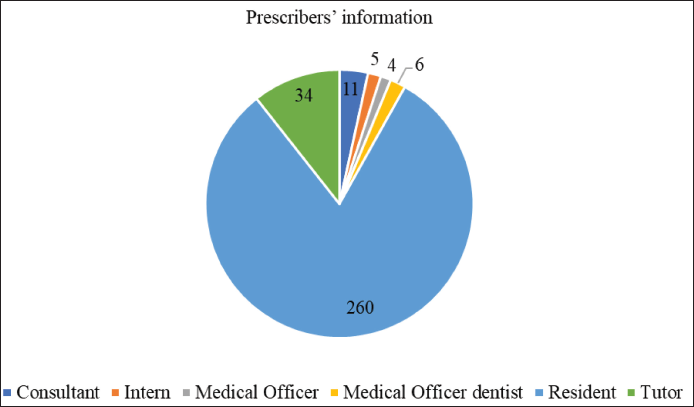
Prescriber’s Information.
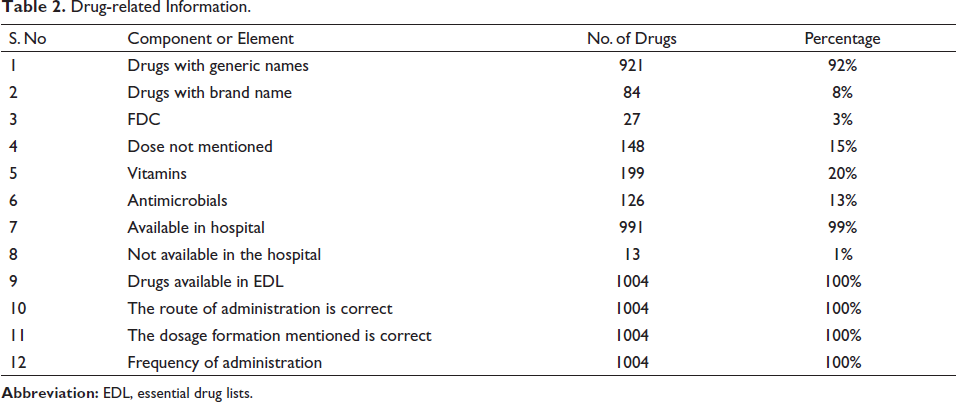
A total of 1004 drugs were prescribed in 320 prescriptions. An average of three drugs per prescription was prescribed. Out of 1004 drugs, 921 (92%) drugs were prescribed with a generic name and 27 (3%) fixed dose combinations were used. Various formulations of medications like tablets, capsules, creams, and injections were used. Out of 1004 drugs, doses of 148 drugs were not mentioned in the prescriptions. Most of the drugs (99%) were available in hospital formulary and all drugs were from the EDL; route and frequency of drug administration were mentioned for all drugs (Table 2 and Figure 2).
Drug-related Information.
Formulation of Medication.
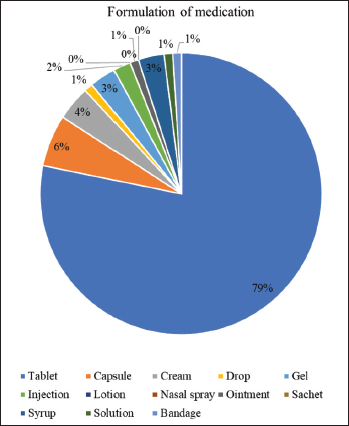
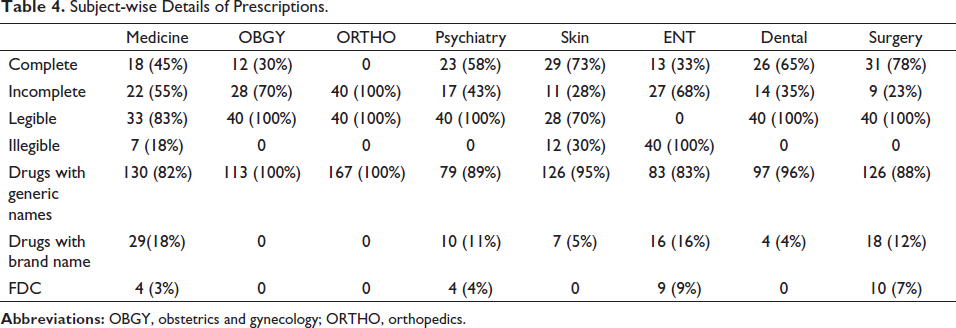
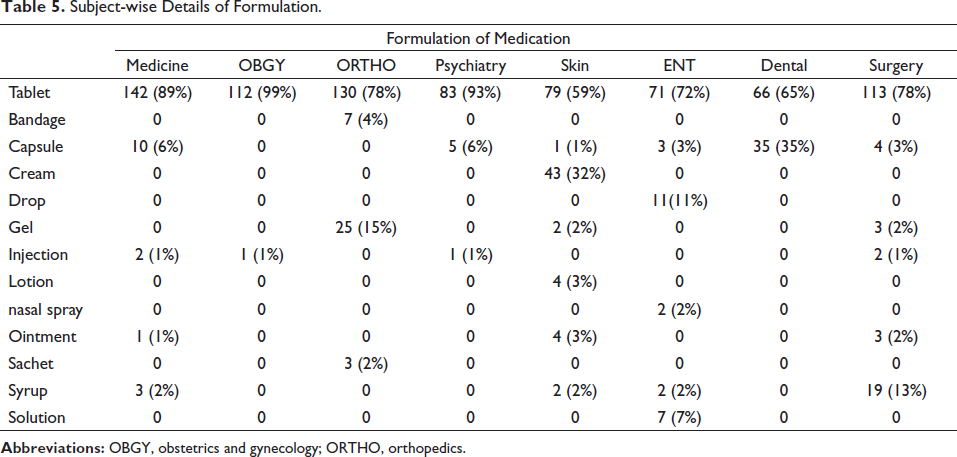
Table 3 shows subject-wise details of prescriptions. A total of 320 prescriptions were selected out of those 40 prescriptions from each department like medicine, obstetrics and gynecology (OBGY), psychiatry, skin, orthopedic, ENT, dental, and surgery. Patient information was mentioned in the prescriptions of all departments. Weight of the patients mentioned in prescriptions of 65% of patients in the OBGY department and only 3% of patients in the psychiatry department. Prescribers’ information was mentioned in all the prescriptions. Totally, 5 or more than 5 drugs were prescribed in prescriptions of medicine (30%), orthopedic (28%), skin (8%), and surgery (25%). The diagnosis was mentioned in 98% of prescriptions in the skin department, while in the dental department, diagnosis was not mentioned in prescriptions. In total, 100% of indications of the drug were mentioned in the skin, ENT, and dental departments, while in orthopedic, 0% of prescriptions were mentioned. Instruction for dispensing was not mentioned in prescriptions of OBGY (10%), orthopedic (18%), skin (25%), ENT (13%), and surgery (25%). Instruction for patients was not mentioned in prescriptions for OBGY, orthopedic (10%), skin (28%), ENT (5%), and surgery (25%). Follow-up visits were not mentioned in prescriptions of medicine (90%), OBGY (68%), orthopedic (100%), psychiatry (75%), skin (100%), ENT (100%), dental (33%), and surgery (83%). Non-pharmacological instruction was mentioned only in prescriptions of medicine (43%), psychiatry (8%), and surgery (58%). In total, 78% of prescriptions from the surgery department were complete followed by 73% from skin, while 100% were incomplete prescriptions from the orthopedic department. In OBGY, orthopedic, psychiatry, dental, and surgery departments, 100% of prescriptions were legible, and 100% of prescriptions were illegible only to the ENT department. In total, 100% of drugs were prescribed by generic name in OBGY and orthopedic departments, while 18% of drugs were prescribed only by brand name in medicine. The tablet formulation is prescribed in all the departments, 99% in OBGY, and 93% in psychiatry. Moreover, 99% of vitamins were prescribed in OBGY and 54% of antimicrobials were prescribed in the dental department. Drugs prescribed in all the departments were available in EDL. The route of administration of drugs, dosage formation, frequency of administration, and duration of treatment were 100% correct (Tables 3–5).
Subject-wise Details of Prescriptions.
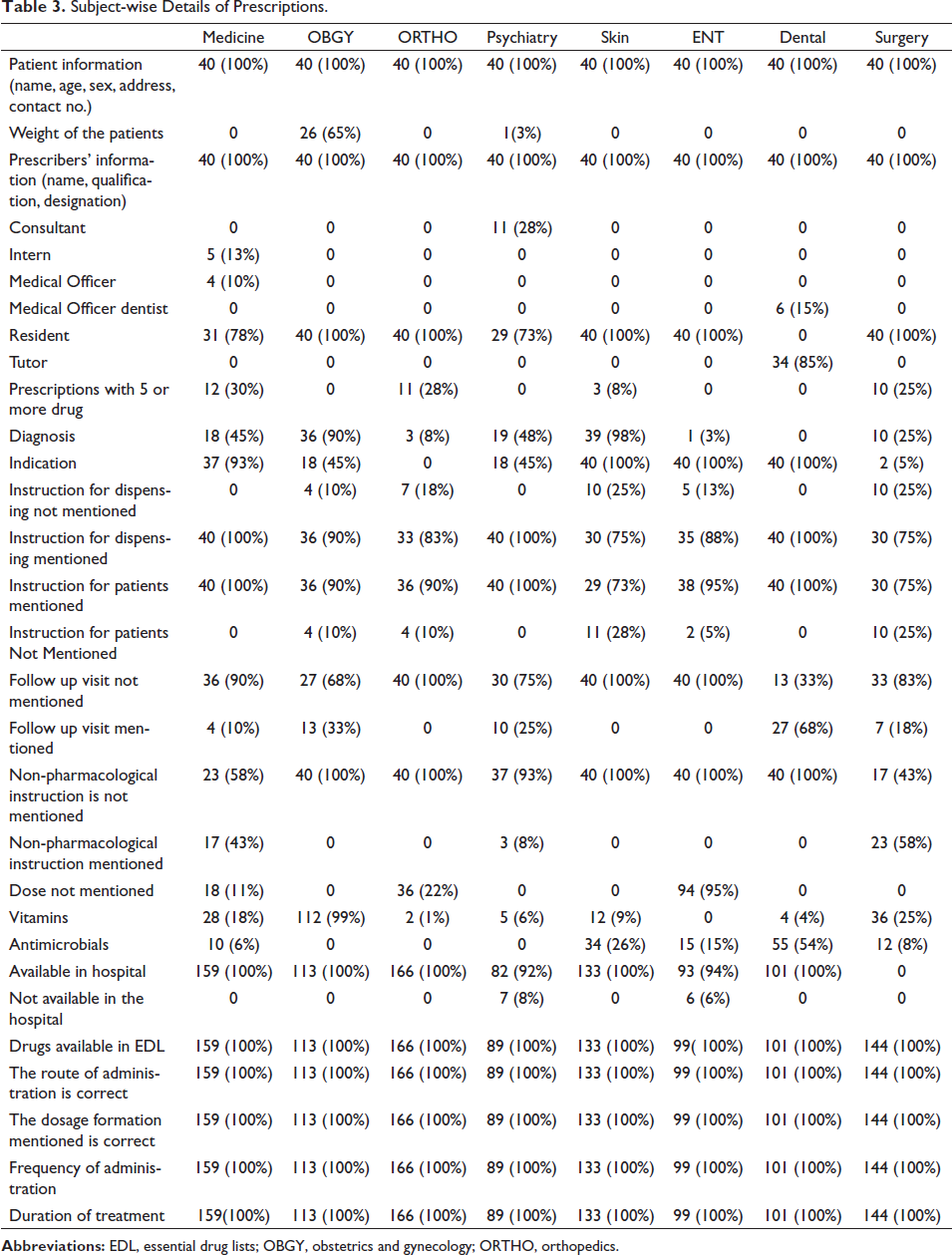
Subject-wise Details of Prescriptions.
Subject-wise Details of Formulation.
In the category of the medication error according to the NCC MERP Index, an error (“error of omission”) occurred but no harm (impairment of the physical, emotional, or psychological function or structure of the body and/or pain resulting therefrom) to the patient, which belongs to category B.
Discussion
The ability to deliver the right medicines to the right patients is the most essential function of the healthcare system. Prescriptions are an important intervention for the physician, and it is an ethical and legal duty of the practitioner to write complete and legible prescriptions. 12 By enhancing standards of medical treatment, quality of life can be improved, and prescription audit is important for this because it is documented evidence to support diagnosis, treatment, and utilization of hospital facilities. 13
This study was conducted in a tertiary care hospital to assess prescription errors and prescribing faults, analyze and document the information on the core prescribing indicators proposed by the WHO, and check the rationality of the prescriptions.
In the current study, a total of 320 prescriptions were analyzed from eight departments. Analysis was done on core elements of prescription like prescriber’s information (Name, Qualification, and Designation) and patient’s information (Name, Age, Sex, Weight, Address, and Contact No.). The details of date of the prescription, diagnosis, legibility of prescription, instruction for dispensing of a drug, instruction for the patient, incorporation of essential non-pharmacological measures in the prescription, and follow-up visit were also analyzed.
Drug-related information includes completeness in terms of drug with a generic name or brand name, formulation of the medication, fixed drug combination, route, frequency and duration of treatment, WHO indicators for rationality―percentage of drug prescribed by generic name, percentage of antimicrobials, injectables, and drug from essential drug list.
All prescriptions contain demographic details of patients and prescriber’s information like name, qualification, and designation. These results were better than the study conducted by Ansari et al. in which 85.4% of prescriptions missing the prescriber’s information. 4 Date and place were written in all prescriptions with the use of computer software. The date is essential to clarify the treatment plan duration and to avoid unnecessary filling and future misuse of medicines. The weight of the patients was written in 27 prescriptions; out of those, 26 prescriptions were from the OBGY department. For the calculation of the dose of the drug, weight is important for pediatric patients.
In the present study, from the majority of prescriptions, 260 (81%) were written by residents followed by tutors in the dental department 34 (11%) and consultants 11 (3%). The diagnosis was mentioned in 126 (40%) prescriptions, and the indication for drug usage was mentioned in 195 (61%) prescriptions. In comparison with these studies conducted by Debjit Chakraborty et al. and E. Yousif et al., 33.3% and 94% of prescriptions were missing diagnosis (Figure 1).14, 15
An average of three drugs per prescription was prescribed. In total, 11% of prescriptions contain more than five drugs. Polypharmacy increases the risk of hospitalization and medication errors. 16 It also increases the cost burden on the healthcare system, decreases adherence of the patients, and increases morbidity and mortality. 17
Instructions for dispensing of drugs were mentioned in 89% of prescriptions and the duration of treatment was mentioned in all prescriptions. Instructions for patients were mentioned in 90% of prescriptions and follow-up visit was mentioned in 19% of prescriptions, whereas in contrast, instructions to the patient was mentioned in 33.3% and follow-up advice was mentioned in 24.2% of prescriptions in a study conducted by Singh et al.; non-pharmacological instructions were not mentioned in 87% of prescriptions. 18
In our study, 82% of prescriptions were legible, for OBGY, orthopedic, psychiatry, dental, and surgery; 100% of prescriptions were legible except ENT department; and 0% of prescriptions were legible. Only 13% of prescriptions were illegible. Compared to the study conducted by Patel P. et al., 75% of prescriptions were legible. 1 In a study conducted by Natarajan et al., 92% of prescriptions were legible. 19 In a study conducted by Y.M. Irshaid et al., 64.3% of prescriptions were illegible. 20 Studies conducted by Einstein showed that 6.6% of prescriptions were illegible. 21 Illegible prescriptions lead to misunderstanding which results in an error in the dispensing or administration of medications by pharmacists or nurses, respectively. Hence, clear handwriting in prescriptions is important to prevent such types of medication errors.
In the present study, out of 1005 medicines, 92% were written with generic names. In a study conducted by Balbir K. et al., only 4.16% of drugs were prescribed by generic name, and 2.81% of drugs were in generic name in a study done by Patel P. et al. In total, 8% of medicines were written with the brand name in this study.1, 22 Drugs prescribed by brand name may be more expensive than the generic name. Non-availability of all drugs or lack of knowledge creates misunderstanding among the pharmacists.
In our study, 27 drugs were given as a fixed dose combination which was less than the study conducted by Balbir et al. (97.91%) and Chakrabarti et al. (59%).22, 23 Using fixed-dose combinations has some benefits like increased efficacy, reduced adverse effects, and improved patient compliance.
Dosage forms used were mostly tablets 79% followed by capsules 6%, cream 4%, and injections only 2% . Overuse of injections, when oral medication can be more appropriate, is irrational as the cost is higher than that of oral therapy. Moreover, blood-borne diseases such as hepatitis and HIV/AIDS can be transmitted by the use of non-sterile injections. Out of 1005 medication, dose was not mentioned in 148 drugs.
Out of 1005 medicines, 13% medicines were antimicrobials. In a study done by Rai S., 55.4% of prescriptions contain antimicrobials. 24 In a study conducted by Kaur B., 11.84% of antibiotics were prescribed. 22 Appropriate use of antibiotics after culture sensitivity testing is necessary to prevent the emergence of drug resistance. Most of the acute respiratory and acute gastroenteritis cases are viral in nature and may not need antibiotics. An antibiotic policy should be formulated so that clinicians can use them judiciously according to patients’ needs. In our study, 99% of drugs were available in hospitals and all drugs were from the EDL 2022, and when compared to a study conducted by Balbir K. et al., 53.25% of drugs were from EDL. 22
In a similar study on topical corticosteroids, Sharma et al. reported that the frequency of application was recorded in 93% of prescriptions. 12 In a study conducted by Dhamodharan et al. which is similar to this study, at an outpatient tertiary care teaching hospital, dose and dosage schedule were mentioned in 98% of prescriptions, and route of drug administration was present in 98.8% of prescriptions. 25 In the present study, dosage schedule, route of administration, and frequency were mentioned in 100% of prescriptions. This may reduce underdosing or overdosing, subsequent treatment failure, and drug-drug interactions. 25 In Joshi et al.’s study, 61.34% of prescriptions were complete, while in the present study, 48% of prescriptions were complete. 26
Prescriptions of surgery and orthopedic department have an error of omission like the weight of the patient was not mentioned in a single prescription, and instructions to the patient were not mentioned (43% of prescriptions) in contrast to our study by Subramaniam et al. where prescription error in surgery ward 97.75% and orthopedic ward 97.53%. 27 Errors of omission like diagnosis not mentioned in the prescription present in 60.63% of total prescriptions. 28
In a study by Subramaniam, et al., prescription errors were fall under category C, and 35 errors were in category C (87.5%) in the study by Sheik et al. In contrast to these studies, prescriptions of medicine, OBGY, psychiatry, skin, orthopedic, ENT, dental, and surgery departments were containing an error of omission which falls under category B according to the NCCMERP.27, 29
Polypharmacy, limited generic prescribing, prescribing of irrational FDCs, and drugs not from EDLs are areas of concern. The assessment of these prescribing errors on a basis of regular monitoring using WHO core drug use indicators which may reduce medication errors was required. A well-formulated action plan across the country are encouraging the physician to use rational FDCs, prescribing generic names in capital letters, adhering to standard treatment guidelines and antibiotic stewardship, and so on. 26
The automated computerized prescribing system is an effective tool to decrease medication errors, but electronic systems are not available at each level of health care, are expensive, and require training. The use of uniform medication charts including all the relevant clinical information along with prescriptions can be used. This approach has been a relatively simple, validated alternative to electronic systems. Also, training can be given to non-prescribing staff and pharmacists that can lead to a reduction in medication errors other than prescription errors. 3 Review of prescribing error ensures how prescriber and other staff screen drug-drug interactions by using various information resources. Electronic prescribing systems and community pharmacy patient medication record systems may provide alerts of interactions. 30
The reporting process in health facilities should be easy and encourage and support the staff for reporting. Some health facility implements a “Good Catch” program where physicians are rewarded with acknowledgment and prize. 31 Solanki ND et al. showed that compliance with the prescription audit rate was increased, and non-compliance with the prescription audit rate was reduced due to the use of Statistical Process Control (SPC). 8 The regular reporting of prescribing errors and regularly conducting training and learning may reduce medication errors. Prescription errors are avoidable and can be reduced if prescriptions are assessed by clinical pharmacologists. Clinical pharmacologists have deep knowledge of therapeutics, drug-drug interactions, safety, and properties of medicine. If the clinical pharmacologists, physicians, nurses, and administrative personnel will be working collectively, then the aim of minimizing medication errors can be achieved. 3
Conclusion
In this study, all the demographic details of patients and doctors were recorded using the computerized system. In drug details, dose, route, and frequency of drugs were mentioned in 100% of prescriptions. Instructions to patients, non-pharmacological instructions, and instructions for the dispensing of drugs were not mentioned in some of the prescriptions. In our study, according to the NCC MERP category, medication errors fall under category B. To reduce medication errors, we can implement an electronic system, involve clinical pharmacologists, utilize prescription charts, and organize nationwide workshops on rational prescription writing. We should encourage regular prescription audits and reporting to improve the healthcare system in the country.
Abbreviations
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was conducted after approval of the institutional ethical committee.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
Informed Consent
The participant has consented to the submission of the article to the journal.
